Hippocampal replay during wakefulness
The mammalian brain continuously processes, updates, and stores representations of environmental stimuli and internal states. Central to this capacity is the phenomenon of hippocampal replay, a neurophysiological process in which sequences of neuronal firing that occurred during active behavior are spontaneously reactivated in a temporally compressed format during periods of rest. Originally identified during non-rapid eye movement (NREM) sleep and theorized to serve as the primary mechanism for offline systems memory consolidation, replay is now recognized as a ubiquitous feature of the awake, idling brain. Waking replay occurs during brief pauses in behavior, quiet wakefulness, and consummatory actions, operating on a timescale significantly faster than the original experience 123.
The identification of waking replay challenges the classical dichotomy that partitions memory encoding strictly into active waking states and memory consolidation exclusively into sleep states. Instead, waking replay suggests a highly dynamic, real-time consolidation mechanism that not only stabilizes newly acquired memory traces but also retrieves stored representations to evaluate past outcomes and simulate future trajectories. Through highly coordinated interactions with the prefrontal cortex and other distributed neocortical networks, awake hippocampal replay serves as a fundamental computational substrate bridging immediate memory consolidation, spatial navigation, and prospective decision-making.
Generation of Sharp-Wave Ripples
Hippocampal replay is overwhelmingly associated with a specific electrophysiological biomarker known as the sharp-wave ripple (SWR) complex. SWRs are among the most synchronous spontaneous neuronal population events in the mammalian brain, observable across species from rodents and macaques to humans 45. These events occur most frequently during behavioral immobility, quiet wakefulness, and NREM sleep, when the dominant hippocampal theta rhythm (typically 4 - 10 Hz in rodents), which characterizes active exploration, subsides.
The generation of an SWR is a multifaceted process rooted in the intrinsic microcircuitry of the hippocampal formation. The event originates with a population burst of excitatory pyramidal cells in the CA3 subfield. The CA3 region is characterized by dense, recurrent collateral connections, which allow for the auto-associative completion of firing patterns from partial cues 567. A gradual build-up of excitatory postsynaptic currents in CA3 eventually reaches a threshold, triggering a massive, largely stochastic depolarization event across the CA3 network 7. This synchronous burst is transmitted via the Schaffer collateral pathway to the apical dendrites of pyramidal cells in the CA1 region, specifically in the stratum radiatum. The extracellular consequence of this massive dendritic depolarization is recorded as a large-amplitude negative deflection in the local field potential (LFP) - the "sharp wave" 57.
Coincident with this sharp wave, the depolarization of CA1 pyramidal cells recruits a network of local inhibitory interneurons, most notably the fast-spiking, parvalbumin-expressing (PV+) basket cells and chandelier cells 89. These interneurons exert powerful, rhythmic perisomatic and axoaxonic inhibition on the CA1 pyramidal cells. The interplay between the strong excitatory drive from CA3 and the precisely timed, rhythmic inhibition from the local interneuron network generates a high-frequency extracellular oscillation in the pyramidal cell layer (stratum pyramidale) 57. This oscillation, occurring at frequencies typically between 150 and 250 Hz (and up to 600 Hz in fast ripples), constitutes the "ripple" component of the SWR.
Within the extremely narrow time windows (approximately 5 milliseconds) between the peaks of interneuronal inhibition, specific subsets of CA1 pyramidal cells are released from inhibition and allowed to fire action potentials 5. The cells that fire during these narrow temporal windows are not random; they are organized into highly structured sequences that correspond to the sequential activation of place cells observed during prior spatial navigation or learning episodes 10. Because the entire SWR event lasts only 30 to 100 milliseconds, the firing sequences are dramatically compressed in time compared to the behavioral timescale, operating at approximately 20 times the speed of the physical experience 15.
Rigid and Plastic Neuronal Subnetworks
Recent electrophysiological and computational studies have further delineated the CA1 neuronal populations participating in these events into distinct "rigid" and "plastic" subnetworks. This distinction is critical for understanding how the hippocampus balances the need to reliably generate SWRs with the necessity of encoding highly variable, novel episodic information. Rigid neurons exhibit high firing probabilities during ripples regardless of recent experience, forming a stable computational backbone for the SWR event 7. Their activity exhibits limited change between pre-learning sleep, waking rest, and post-learning sleep, suggesting they maintain the basic oscillatory architecture of the ripple.
Plastic neurons, conversely, are highly responsive to environmental changes and learning. These cells alter their firing rates and participation within SWRs based on recent sensory experiences, carrying the novel, experience-specific information that constitutes the actual memory trace being replayed 7. This division of labor allows the hippocampal circuit to continuously generate SWRs while simultaneously accommodating the continuous influx of novel episodic information without overwriting existing structural frameworks.
| Neuronal Subnetwork | Firing Characteristics During SWRs | Response to Novel Experience | Primary Functional Role |
|---|---|---|---|
| Rigid Pathway | High, consistent firing probability across multiple SWRs and brain states. | Minimal change in firing rates between pre- and post-learning periods. | Provides a stable oscillatory backbone and generates the fundamental timing structure of the ripple. |
| Plastic Pathway | Variable firing probability; tightly linked to specific trajectories or items. | Highly modifiable; firing rates and sequence participation change drastically after learning. | Carries novel episodic information; constitutes the specific memory trace being actively replayed. |
Anatomical Pathways and Interregional Coordination
The computational utility of hippocampal replay relies on the successful transmission of these compressed representations to distributed neocortical sites. The prefrontal cortex (PFC), particularly the medial prefrontal cortex (mPFC), is the primary target for these operations. The interaction between the hippocampus and the mPFC is critical for executive functions, working memory, spatial navigation, and the extraction of generalized schemas from specific episodic instances.
The Hippocampal-Prefrontal Circuit
The anatomical basis for this interregional communication involves direct, monosynaptic projections from the hippocampal CA1 region and the subiculum to the mPFC 811. In rodents, detailed anterograde transsynaptic tracing reveals that the dorsal-caudal hippocampus (dcHPC) sends robust projections to ventral subregions of the mPFC, including the prelimbic cortex, anterior cingulate cortex, and the dorsal peduncular cortex 811. Notably, this pathway exerts a strong inhibitory influence on the ventral mPFC by directly targeting a substantial proportion of local GABAergic inhibitory neurons, including PV+ interneurons 11.
While direct return projections from the PFC back to the hippocampus are relatively sparse, complex indirect routes establish a closed-loop system for bidirectional communication. The nucleus reuniens of the midline thalamus serves as a critical anatomical relay linking the mPFC back to the hippocampus, specifically targeting the CA1 region and the entorhinal cortex 8. Additionally, the medial entorhinal cortex (MEC) provides cortical inputs that modulate CA1 ripples during waking states, suggesting a mechanism by which top-down attention and ongoing sensory processing can influence the content of waking replay 8.
Cell-Type Specific Modulation
During waking behavior, the occurrence of an SWR in the hippocampus drives profound and highly structured state changes in the mPFC. When an awake SWR occurs, a large proportion of mPFC neurons show rapid, spike-time modulation 1213. Crucially, unlike the broadly synchronized excitation observed within the hippocampus itself, the response in the PFC is heterogeneous and cell-type specific. Subsets of PFC neurons are strongly excited during the SWR window, while other distinct subsets are simultaneously inhibited 1213.
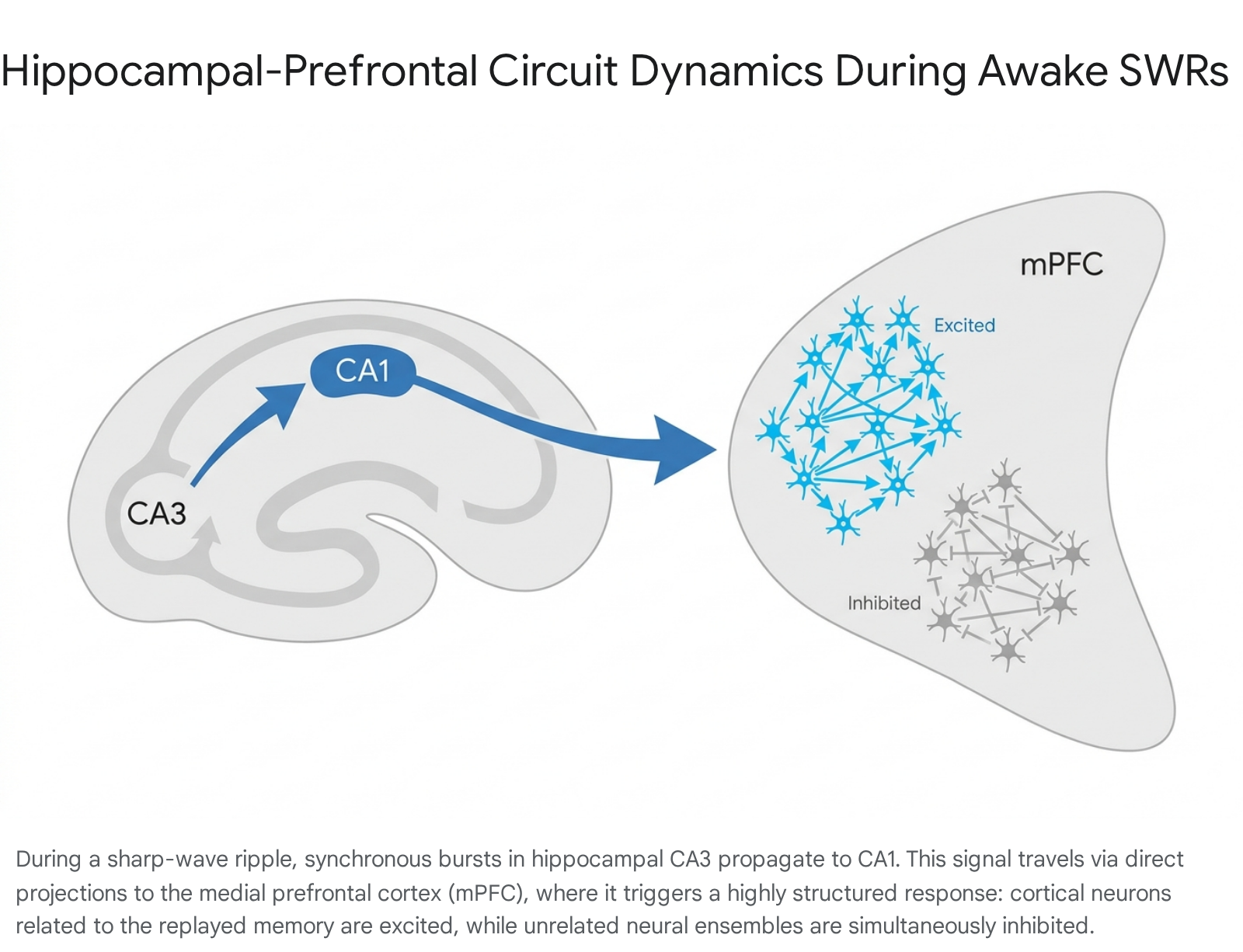
This coordinated excitation and inhibition serves a precise functional routing purpose. Electrophysiological recordings in animals learning spatial tasks indicate that during individual SWRs, the PFC cells that are excited tend to hold representations relevant to the specific spatial trajectory being concurrently replayed by the hippocampus. Conversely, PFC cells with spatial representations unrelated to the replayed trajectory are actively suppressed via inhibition 12. This selective routing mechanism indicates that awake SWRs do not simply broadcast a generic excitatory pulse to the cortex. Rather, they mark moments of highly structured, content-specific information transfer. The concurrent excitation of relevant cortical ensembles and suppression of irrelevant ones creates an optimal computational window for synaptic plasticity, allowing the neocortex to integrate the replayed hippocampal memory with ongoing cognitive demands and pre-existing knowledge frameworks.
Cross-Species Structural Variations
While the basic circuitry governing SWRs and replay is highly conserved across mammalian species, translational research comparing rodent and human hippocampi reveals critical structural variations that affect memory capacity and computational power. Recent analyses of intact human hippocampal tissue obtained from neurosurgical patients demonstrate that the human CA3 network is not simply a scaled-up version of the rodent CA3 1415.
In the human CA3, synaptic connectivity between pyramidal neurons is significantly sparser than in mice. However, the individual synapses that are present exhibit a much higher degree of reliability and precision in signal transmission 15. Computational modeling based on these physiological parameters suggests that this human-specific combination of sparse connectivity and high synaptic reliability drastically increases the overall memory capacity of the CA3 auto-associative network. This structural divergence implies that while human waking replay likely relies on the same fundamental SWR mechanisms mapped in rodents, the human hippocampal network is optimized to store and replay a vastly larger repertoire of highly detailed, interference-resistant episodic patterns.
Directionality and Temporal Dynamics of Replay
The sequential reactivation of place cells during replay is not restricted to a simple, chronological recapitulation of past events. Replay events possess distinct temporal structures and directional biases that correspond to differing cognitive functions. Depending on the current behavioral demands, the hippocampus can flexibly generate sequences that run forward in time, backward in time, or even construct sequences that the animal has never physically experienced. These diverse dynamics are theorized to be governed by continuous attractor networks (CANs) within the hippocampus, where neuronal fatigue and local inhibition prevent immediate self-repetition and naturally push the sequence along connected spatial representations 1616.
Reverse Replay and Reward Evaluation
Reverse replay occurs when a sequence of place cells fires in the exact opposite order of the sequence experienced during actual behavior 1018. For instance, if an animal traverses a linear track through spatial zones A, B, C, and D, and then stops at D to consume a reward, an immediately following SWR may contain the firing sequence D, C, B, A.
This retrospective sequencing is most prominently observed immediately following the completion of a trajectory, particularly when a reward is encountered or when an unexpected outcome occurs (a reward prediction error) 1019. The unique retrograde structure of reverse replay makes it an ideal biological mechanism for solving the "credit assignment problem" in reinforcement learning. By rapidly rewinding the chain of neural events that led to a specific outcome, the brain can effectively link the rewarding (or punishing) consequence back to the sequence of preceding actions and locations 1017. Consequently, reverse replay is heavily implicated in the immediate, real-time consolidation of value-based memories, allowing the organism to adjust the subjective value of specific paths based on recent outcomes.
Forward Replay and Prospective Planning
In contrast to the retrospective nature of reverse replay, forward replay entails the sequential activation of neurons in the same chronological order as the physical experience (e.g., A, B, C, D) 1018. Forward replay is frequently observed during brief pauses in behavior prior to the initiation of a movement - such as an animal pausing at the stem of a T-maze before choosing which arm to explore 1018.
The content of forward replay during these decision-making junctures often projects ahead of the animal's current location, effectively sweeping along potential future paths. Research indicates that the specific trajectory delineated during a forward replay event can reliably predict the physical path the animal will subsequently choose to take 1018. This prospective mapping strongly aligns forward replay with the psychological concepts of planning, mental simulation, and spatial working memory. By rapidly "testing" potential routes within the internal cognitive map before committing to physical action, the organism can utilize stored memories to guide future, goal-directed behavior 1923. This phenomenon is often behaviorally correlated with "Vicarious Trial and Error" (VTE), a state where an animal physically hesitates and looks back and forth between options while internal forward replay simulates the outcomes of those choices 24.
The Preplay Controversy
A highly debated extension of forward replay is the phenomenon termed "preplay." Preplay refers to the sequential firing of place cells that outlines a spatially coherent trajectory through an environment the organism has not yet explored 1720. First reported when animals were allowed to view, but not physically enter, a novel arm of a maze, preplay sequences were observed during rest periods prior to exploration, and these specific sequences were later recruited to represent the novel arm once the animal was finally allowed to traverse it 1721.
Proponents of preplay argue that the hippocampus contains pre-configured, latent network assemblies - a fundamental, innate grammar of space - that are readily available to rapidly map novel experiences 2122. Critics contend that putative preplay events may simply reflect the generalization of previously learned sequences from similar environments, or methodological artifacts inherent in decoding spatial maps from large neural ensembles using linear fitting algorithms 2021. While its exact nature remains controversial, the concept of preplay suggests that the hippocampal network possesses an inherent generative capacity, capable of synthesizing novel abstract sequences independent of direct sensory experience.
| Replay Category | Temporal Direction | Behavioral Context | Primary Cognitive Function | Evidence Consensus |
|---|---|---|---|---|
| Reverse Replay | Retrograde (e.g., D → C → B → A) | Immediately following trajectory completion; heavily modulated by reward delivery or prediction errors. | Immediate memory consolidation; value updating; solving the credit assignment problem in reinforcement learning. | Robust and universally accepted across multiple paradigms. |
| Forward Replay | Anterograde (e.g., A → B → C → D) | Pauses prior to movement initiation; hesitation at decision points (Vicarious Trial and Error). | Prospective planning; spatial working memory; predicting and selecting future trajectories. | Robust; strongly predictive of subsequent behavioral choices. |
| Preplay | Anterograde (Constructive) | Rest periods prior to novel exploration; abstract cognitive tasks requiring generalization. | Establishing latent cognitive maps; preparing neural assemblies for rapid encoding of novel, unexperienced environments. | Controversial; heavily debated regarding pre-existing network assemblies versus generalization of prior schemas. |
Methodological Innovations in Human Replay Detection
Historically, the study of hippocampal replay has relied heavily on invasive electrophysiological recordings in rodents, utilizing implanted tetrodes to record the extracellular spikes of large ensembles of individual place cells. Because placing dense intracortical electrode arrays in healthy human subjects is generally not feasible outside of rare presurgical epilepsy monitoring, translating the concept of replay to human cognition required methodological innovations capable of detecting fast, sparse sequence reactivations using non-invasive imaging techniques such as magnetoencephalography (MEG) and functional magnetic resonance imaging (fMRI).
Temporally Delayed Linear Modeling
A major breakthrough in human replay research has been the development of Temporally Delayed Linear Modeling (TDLM) 2329. TDLM is a domain-general analytical framework designed to extract fast, sequential neural reactivations from the macroscopic signals recorded by MEG or electroencephalography (EEG). Because MEG signals contain significant temporal autocorrelations, spatial leakage, and rhythmic background noise (such as the 10 Hz alpha rhythm) that can easily masquerade as sequential patterns, traditional rodent analysis methods - which rely on discrete spike times and template matching on linear tracks - are mathematically incompatible with human non-invasive data 23.
TDLM circumvents these confounds by operating entirely within a "decoded state space." Rather than looking at raw sensor data, researchers first train non-linear machine-learning classifiers on the MEG data while the human subject is actively engaged in a task (e.g., viewing a specific sequence of images or performing a motor sequence). These classifiers learn the unique, multivariate neural signature associated with each specific item or state, utilizing L1 regularization to ensure sparsity and out-of-sample generalization 2324. During subsequent resting periods, TDLM applies these classifiers to the spontaneous, continuous MEG data to generate a dynamic time series of state probabilities.
The core of TDLM is a two-level general linear modeling (GLM) approach 2329. The first GLM step uses multiple linear regression to quantify the empirical transition matrix - testing whether the decoded probability of state A at time $t$ systematically predicts the probability of state B at time $t + \Delta t$. Crucially, by including all states in a single model and adding phase-shifted regressors, TDLM explicitly controls for auto-correlations and background brain oscillations. The second GLM step projects this empirical transition matrix against the hypothesized sequence defined by the experimental task, resulting in a single statistical metric of "sequenceness" 2329. Because it relies on a graph-based GLM framework, TDLM allows researchers to cleanly address second-order statistical questions - such as whether forward replay differs from reverse replay, or how replay strength correlates with subsequent behavioral performance across different human subjects - without the need for continuous linear track environments.
Behavioral Evidence of Rapid Skill Consolidation
The application of TDLM to human MEG data has provided direct evidence that waking replay occurs in humans and is functionally relevant to both episodic and procedural learning. In a landmark study examining the acquisition of a sequential motor skill (rapidly typing a specific numeric sequence, such as "41324"), researchers observed distinct replay of the trained motor sequence during the brief 10-second rest intervals interleaved between periods of active practice 12.
The human waking replay events detected in this paradigm exhibited temporal characteristics strikingly similar to rodent replay. The sequential neural patterns were replayed in highly compressed bursts lasting approximately 50 milliseconds, representing an approximate 20-fold temporal compression relative to the speed of the actual physical behavior 1. Replay was detected in both forward and reverse temporal directions, recruiting a broad network encompassing the hippocampus, entorhinal cortex, and contralateral sensorimotor cortex 12.
Crucially, the frequency of these replay events was approximately three times higher during the short rest intervals interleaved within the training blocks compared to pre-training or post-training baseline rest periods 12. Furthermore, the subjects who exhibited the highest rates of waking replay during these brief pauses also demonstrated the most significant improvements in typing speed and accuracy. These findings suggest that waking replay is a primary neural mechanism underlying the "spacing effect" - the well-documented psychological phenomenon wherein learning is vastly improved when practice is divided by frequent, brief rest periods compared to continuous, massed practice 12. The data imply that the brain utilizes these micro-rests to rapidly bind and consolidate discrete action representations into fluid, automated skills via temporally compressed neocortical-hippocampal replay.
| Parameter | Invasive Rodent Electrophysiology | Non-Invasive Human MEG (via TDLM) |
|---|---|---|
| Signal Source | Extracellular spikes from single neurons (tetrodes/silicon probes). | Macroscopic magnetic fields generated by large cortical/hippocampal populations. |
| Data Representation | Discrete spike timing and burst rates. | Continuous decoded state probabilities. |
| Primary Confounds | Spike sorting errors; limited spatial coverage of the brain. | Temporal autocorrelation; spatial signal leakage; robust background oscillations (e.g., alpha). |
| Sequence Detection Method | Template matching; rank-order correlation; Bayesian decoding of position. | Two-stage General Linear Modeling (TDLM) testing empirical transitions against task graphs. |
Unconscious Neural Reactivation Versus Conscious Mind Wandering
The discovery that the human brain intensely processes recent memories during waking rest naturally intersects with psychological research on "mind wandering," daydreaming, and rumination. While an external observer might classify both states as quiet wakefulness or off-task behavior, the underlying neural dynamics and the level of conscious awareness associated with waking hippocampal replay versus prolonged mind-wandering appear to be distinct, albeit related, phenomena.
The Default Mode Network and Spontaneous Thought
Mind wandering is typically characterized phenomenologically as a spontaneous shift of conscious attention away from immediate external stimuli toward internally generated, task-unrelated thoughts, often involving autobiographical memories or anticipated future scenarios 2526. Neuroanatomically, this state is heavily correlated with the activation of the Default Mode Network (DMN), a distributed network encompassing the medial prefrontal cortex, posterior cingulate cortex, precuneus, and medial temporal lobe structures including the hippocampus 2627.
Recent research suggests that hippocampal SWRs may serve as the neurophysiological triggers that initiate episodes of conscious mind wandering. SWRs occur during the exact behavioral states permissive to mind wandering (disengagement from sensory input and stable immobility) and propagate intensely into the core cortical hubs of the DMN 27. In this framework, a spontaneous SWR in the hippocampus acts as an unconscious, associative search mechanism. By initiating a brief, highly compressed reactivation of a memory trace, the SWR injects a signal into the DMN. If this signal clears a certain threshold of cortical activation, it may evolve from a rapid, unconscious 50-to-100-millisecond electrophysiological ripple into a slower, sustained, and consciously experienced daydream 2728.
Phenomenological and Temporal Distinctions
Despite this potential causal link, waking replay and conscious mind wandering operate on fundamentally different timescales and levels of awareness. The highly compressed temporal nature of replay (20x compression, occurring within tens of milliseconds) precludes it from being the direct substrate of conscious experience, which unfolds in real-time. Dreams and daydreams are experienced subjectively at a timescale that mirrors waking life, whereas SWR-mediated replay is a transient, sub-second burst 35.
Furthermore, experimental manipulations of memory reactivation suggest a functional divergence based on the level of conscious awareness. When memories are cued and retrieved consciously during task engagement, the neural dynamics are distinct from those observed during spontaneous, unconscious replay during rest periods 29. The benefits of memory reactivation for long-term consolidation appear more robust when the reactivation occurs implicitly during a truly "offline" brain state - characterized by sensory decoupling and a lack of conscious deliberation - rather than during active, conscious rehearsal 29.
This distinction becomes particularly relevant in the context of psychopathology. Chronic, uncontrollable mind wandering is a hallmark of rumination and anxiety. Some researchers hypothesize that maladaptive forms of hippocampal replay may underlie these conditions. In this model, pathological rumination is driven by the excessive, generalized reverse replay of aversive past experiences, preventing proper contextual updating. Conversely, clinical worry may be associated with excessive forward replay of potential threat scenarios, continuously flooding the mPFC with anticipated negative outcomes 24. Thus, while the conscious rumination of mind wandering and the rapid electrophysiology of waking replay both involve the hippocampus retrieving past information, replay is best understood as a fast, unconscious computational process that strengthens synaptic pathways, whereas mind wandering represents the slower, conscious elaboration of the semantic and episodic narratives those pathways support.
| Feature | Waking Hippocampal Replay | Conscious Mind Wandering / Daydreaming |
|---|---|---|
| Timescale | Highly compressed (tens to hundreds of milliseconds; ~20x faster than reality). | Uncompressed; unfolds in real-time similar to physical experience. |
| Level of Awareness | Unconscious; implicit computational process. | Conscious; explicit narrative experience. |
| Primary Network Engagement | Hippocampus, Entorhinal Cortex, focused target ensembles in mPFC. | Default Mode Network (DMN), extensive cortical engagement. |
| Psychopathological Correlates | Potential mechanistic basis for persistent threat updating if dysregulated. | Correlated with rumination (past-focused) and worry (future-focused) in depressive and anxiety disorders. |
Implications for Systems Consolidation Theories
The robust evidence for high-fidelity waking replay across species necessitates a reevaluation of classical models of systems memory consolidation. The debate primarily centers on how episodic memories - richly detailed representations of specific events bound to spatial and temporal contexts - are maintained over the lifespan, and the specific role of the hippocampus in their long-term retrieval.
The Standard Consolidation Model
The Standard Consolidation Model (SCM) posits that the hippocampus acts strictly as a temporary memory structure. According to the SCM, an episodic memory is initially encoded via rapid synaptic changes in the hippocampus, which binds distributed cortical features of the experience into a coherent trace. Over time - and largely during sleep - repeated reactivation (replay) of this trace gradually trains the slower-learning neocortex. Eventually, the memory becomes fully consolidated within neocortical networks and is rendered completely independent of the hippocampus 373830.
The SCM elegantly explains the temporal gradient of retrograde amnesia observed in patients with hippocampal damage, where recent memories are lost but remote memories acquired decades prior are largely spared because they have fully transitioned to cortical storage 3730. Under this view, waking replay is simply an acceleration of the offline cortical training process typically associated with sleep.
Multiple Trace Theory and Trace Proliferation
In stark contrast, the Multiple Trace Theory (MTT) argues that the hippocampus is permanently required for the retrieval of truly episodic, context-rich memories, regardless of their age 373141. MTT concedes that the SCM correctly describes the consolidation of semantic memories (generalized facts extracted from experience), which do become hippocampus-independent. However, for episodic memories, MTT proposes that every time a memory is retrieved or spontaneously replayed, a new, parallel memory trace is formed within the hippocampus, linking the original event to the new context of retrieval 3031. Over time, frequently retrieved memories accumulate multiple hippocampal traces, making them more resilient to partial hippocampal damage.
The characteristics of waking replay provide compelling neurobiological support for elements of Multiple Trace Theory. The observation that waking replay is highly sensitive to current behavioral contexts, reward gradients, and ongoing task demands implies that replay is not merely a passive transfer mechanism slowly copying files to a neocortical hard drive. Instead, waking replay actively reconstructs and updates memory representations in real-time, integrating past outcomes with present goals to guide immediate decisions 1030.
Furthermore, the finding that both forward and reverse replay events drive highly specific, concurrent excitation and inhibition within the prefrontal cortex suggests an ongoing, dynamic dialogue between the hippocampus and the neocortex 12. This continuous bidirectional modulation is consistent with the MTT view that the hippocampus remains actively tethered to cortical ensembles, perpetually refreshing and multiplying traces as memories are utilized throughout an organism's life. The emergence of awake replay data effectively blurs the strict boundary between "consolidation" (traditionally an offline, stabilizing process) and "retrieval" (an active, online process), suggesting that both are continuously interwoven through rapid SWR events during waking idle states 6.
| Theoretical Feature | Standard Consolidation Model (SCM) | Multiple Trace Theory (MTT) |
|---|---|---|
| Role of Hippocampus | Temporary relay station and initial binder of memory elements. | Permanent obligate structure for the retrieval of detailed episodic memories. |
| Ultimate Memory Storage | Neocortex (fully independent of the hippocampus over time). | Distributed across Hippocampus and Neocortex (semantic elements separate, but episodic elements remain hippocampal). |
| Effect of Retrieval/Replay | Reinforces the transfer of the singular trace to the neocortex. | Creates a new, parallel hippocampal trace linked to the new retrieval context (trace proliferation). |
| Explanation of Retrograde Amnesia | Remote memories survive hippocampal damage because they are safely stored in the cortex. | Remote memories survive partial hippocampal damage because they possess a greater quantity of distributed traces due to repeated retrieval over time. |
Conclusion
Research into hippocampal replay during waking states has fundamentally broadened the scientific understanding of memory systems. Far from a dormant repository, the hippocampus functions as a dynamic simulation engine. During fleeting moments of waking rest, the hippocampus orchestrates highly compressed, sequential firing patterns locked to sharp-wave ripple oscillations. These replay events propagate via direct anatomical pathways to the prefrontal cortex, selectively exciting relevant cortical ensembles while inhibiting competing networks to facilitate rapid, precise synaptic plasticity.
The directionality of these sequential patterns underscores their diverse cognitive utility. Reverse replay acts retrospectively, assigning value to preceding actions and driving the rapid, offline consolidation of reward-based memories. Forward replay operates prospectively, sweeping through possible future trajectories to allow an organism to evaluate spatial decisions before physical execution. In humans, non-invasive imaging utilizing state-space decoding has confirmed that waking replay is a vital mechanism underlying rapid skill acquisition, operating implicitly during micro-pauses in behavior.
Ultimately, waking replay bridges the gap between the stabilization of the past and the prediction of the future. The fact that episodic sequences are continually replayed and updated during wakefulness supports theories of memory that view the hippocampus not merely as a temporary relay station, but as an indispensable, lifelong computational partner to the neocortex in the organization and retrieval of complex contextual experience.