Heterochronic parabiosis and systemic rejuvenation factors
Introduction to Heterochronic Parabiosis
Heterochronic parabiosis is a specialized surgical and experimental procedure in which the circulatory systems of two living organisms of different ages are connected, allowing them to share a single, continuous blood supply. The technique relies on the physiological exchange of whole blood - comprising cells, plasma, and an array of soluble factors - between the conjoined organisms 12. The concept of joining two living animals, known as parabiosis, dates back to 1864 when it was pioneered by the physiologist Paul Bert to investigate shared circulatory dynamics. Its application to the biology of aging, however, was primarily initiated by gerontologist Clive McCay in the mid-twentieth century 2. Following a period of declining interest in the 1970s due to complications such as "parabiotic disease" - a mysterious condition involving tissue rejection and high mortality - the technique was notably resurrected in 2005 by researchers at Stanford University, sparking a modern renaissance in geroscience 234.
By establishing a shared circulatory system between a young animal and an old animal, researchers are uniquely positioned to differentiate between cell-autonomous and non-autonomous mechanisms of aging 5. The heterochronic parabiosis model simultaneously interrogates two distinct, bidirectional biological processes. First, it measures the effect of an aged systemic milieu on a young organism, a process that reliably induces the acceleration of aging phenotypes. Second, it measures the effect of a young systemic milieu on an aged organism, which has been shown to induce significant systemic rejuvenation 67. When these heterochronic pairs are compared against isochronic control pairs - comprising two animals of the exact same age, either young-young or old-old - the experimental design isolates the specific impact of circulating systemic factors on tissue regeneration, stem cell exhaustion, and cellular senescence 26.
The foundational premise of heterochronic parabiosis rests on the observation that tissue regenerative capacity declines with age due to a diminished responsiveness of tissue-specific stem and progenitor cells, rather than their complete depletion or an irreversible intrinsic cellular failure 12. Cellular and organ function is heavily dictated by the systemic environment, which includes a complex network of endocrine signals, immune cells, inflammatory cytokines, and extracellular vesicles circulating in the blood 89. When surgically joined, the parabionts achieve approximately fifty percent blood chimerism, meaning the blood is thoroughly homogenized between the two partners over a short physiological period 9. The consequence of this homogenization is profound. The old parabiont benefits from continuous exposure to young circulating factors and the enhanced detoxification capacity of the young partner's organs, such as the liver and kidneys. Conversely, the young parabiont is subjected to the inflammatory and pro-geronic factors prevalent in the aged blood, resulting in a measurable decline in functional capacity 101112. This mutual exchange allows researchers to systematically map the biochemical pathways that regulate tissue regeneration and determine precisely how those pathways become altered by the aging process 13.
Physiological and Cellular Impacts of Blood Exchange
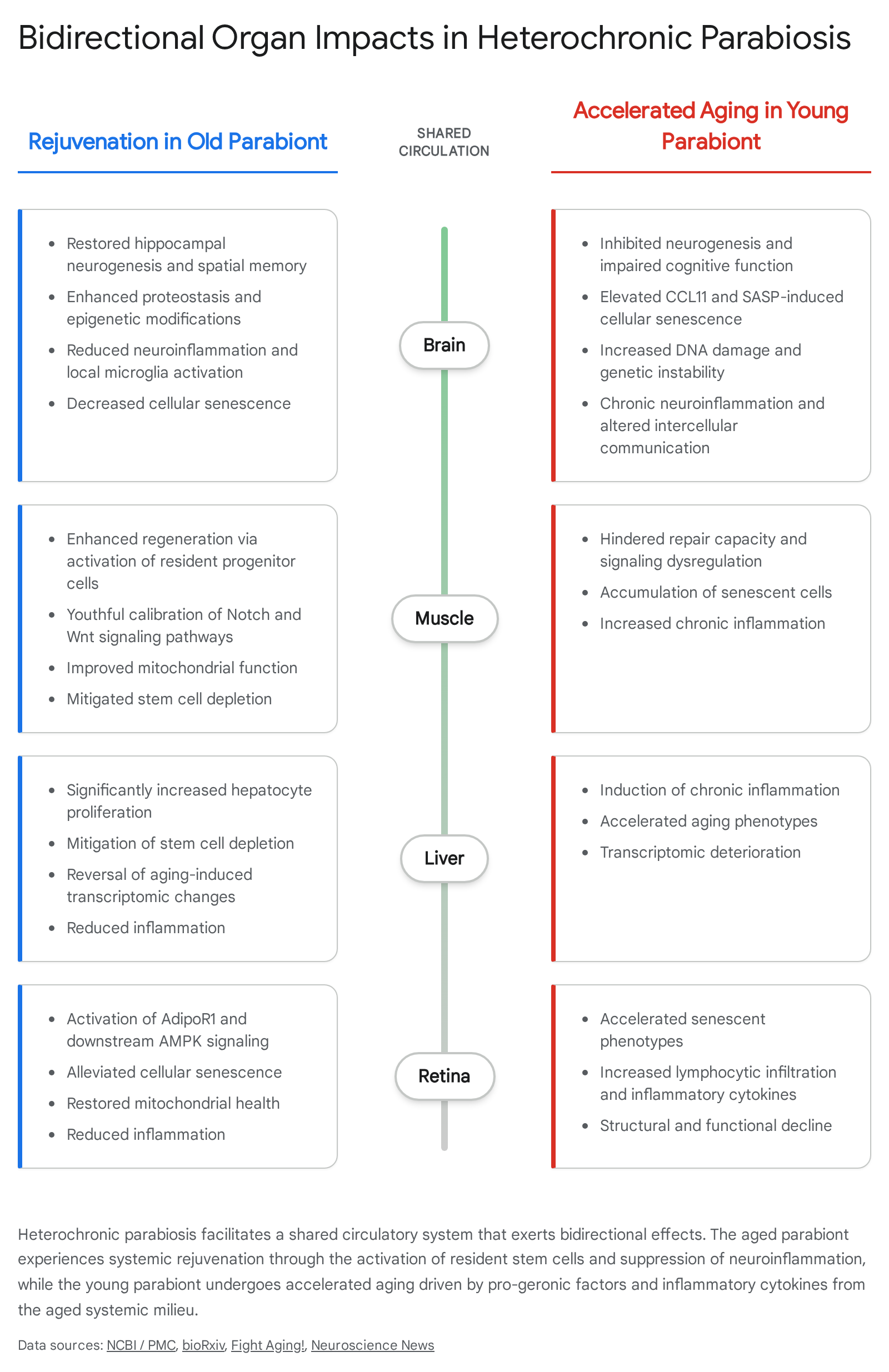
Extensive experimentation using the heterochronic parabiosis model has demonstrated that the biological age of cells and tissues is highly malleable. Exposure to a young systemic environment can reverse epigenetic markers, transcriptomic profiles, and metabolomic signatures traditionally associated with irreversible aging 414. Single-cell transcriptomic profiling of murine organs following parabiosis has revealed profound dynamic changes across multiple distinct tissues, identifying specific gene regulatory networks and ligand-receptor interactions that drive either the reversal of aging or the acceleration of aging 15.
Neurological and Cognitive Rejuvenation
The brain is exceptionally susceptible to the effects of systemic aging, and heterochronic parabiosis has yielded significant findings regarding cognitive and neurological rejuvenation. Transcriptomic analysis of brain cells during aging and heterochronic parabiosis reveals that endothelial cells are highly responsive to the shared circulatory environment, exhibiting strong dynamic changes in their gene expression that directly influence blood-brain barrier integrity and neurovascular coupling 15.
In aged mice paired with young partners, researchers observe a marked improvement in cerebral vascular remodeling, increased hippocampal neurogenesis, enhanced spatial memory, and better olfactory discrimination 81214. The young systemic milieu restores neurogenesis by encouraging the proliferation of oligodendrocyte precursor cells and reversing age-related hindrances in their differentiation into mature, functional oligodendrocytes. This process promotes robust remyelination at demyelinated sites within the aged central nervous system 14. Proteomic and transcriptomic data confirm that the rejuvenation process enhances proteostasis and positive epigenetic modifications in the brain, reducing the burden of senescent cells and dampening neuroinflammation 1216.
Muscle, Liver, and Retinal Regeneration
The rejuvenating effects of young blood extend to multiple peripheral organs, operating primarily through the reactivation of resident stem cell populations rather than the introduction of new stem cells from the young donor. In skeletal muscle, early studies observed that injured muscle in old isochronic parabionts regenerated poorly, reflecting the typical age-related decline in healing. However, when an old mouse was paired with a young mouse, muscle regeneration was robustly restored. This enhancement was driven by the activation of resident aged progenitor cells, known as satellite cells, rather than the engraftment of circulating stem cells from the young partner 12. The systemic environment calibrates biochemical signaling pathways - such as Notch, Wnt, and TGF-beta/pSmad signaling - to youthful levels, overcoming the inhibitory state imposed by the aged muscular niche 13.
Similarly, hepatocyte proliferation in the liver is significantly increased in aged heterochronic parabionts. The young systemic environment mitigates hepatic senescence and restores the regenerative capacity of liver progenitor cells, allowing the old liver to repair tissue damage at a rate comparable to a much younger organism 189.
Recent applications of single-cell RNA sequencing to retinal tissue have also uncovered the mechanisms driving visual decline and regeneration. Aging naturally compromises the retina through increased cellular senescence and widespread mitochondrial dysfunction. Heterochronic parabiosis experiments demonstrate that young systemic factors alleviate these senescent phenotypes in the eye. Specifically, the receptor AdipoR1 and its downstream AMPK signaling pathway have been identified as central mediators in retinal rejuvenation. Pharmacological activation of AdipoR1 with agonists such as AdipoRon has been shown to mimic the benefits of young blood, improving mitochondrial health, reducing neuroinflammation, and reversing senescence markers in the aged murine retina 17.
Accelerated Aging in Young Parabionts
While heterochronic parabiosis reliably rejuvenates the old organism, the procedure imposes a severe biological toll on the young partner. Exposure to old blood actively drives aging phenotypes in young tissues, inhibiting neurogenesis, impairing spatial learning and memory, and reducing overall synaptic plasticity 1418.
In the brain of the young parabiont, the influx of aged systemic factors leads to altered intercellular communication, increased DNA damage, and long-term genetic instability 16. Immune function in young heterochronic partners undergoes accelerated aging, and the regenerative capacity of their skeletal muscle is significantly hindered when exposed to the old systemic environment 28. In studies specifically examining the lacrimal glands, young mice exposed to an older systemic milieu exhibited increased lymphocytic infiltration, specifically involving marginal zone B cells and plasma cells. This infiltration was accompanied by elevated levels of inflammatory cytokines, including interferon-gamma and interleukin-1 beta 14. These findings underscore the extreme potency of the pro-geronic factors circulating in aged blood, demonstrating that the aging process is actively driven by systemic circulating signals rather than passive cellular decay.
Mechanisms of Action: The Addition Versus Dilution Debate
The dramatic phenotypic changes observed in heterochronic parabiosis have sparked an ongoing and highly active scientific debate regarding the primary mechanism of action driving these cellular transformations. Researchers historically questioned whether tissue rejuvenation stems primarily from the addition of youthful, anti-geronic factors to the old animal, or from the dilution of harmful, pro-geronic factors that naturally accumulate in the old animal's blood over time 10.
The Young Blood Addition Hypothesis
In the years immediately following the 2005 revival of parabiosis research, scientific consensus heavily favored the addition hypothesis. It was posited that young blood contains a rich abundance of signaling molecules, hormones, and growth factors that actively instruct aged, dormant cells to repair and regenerate 101116. Extensive proteomic screening studies identified specific blood-borne factors, such as Growth Differentiation Factor 11 and the hormone oxytocin, that appeared to act as potent age-modifying agents capable of rejuvenating stem cell niches when administered exogenously to older subjects 191619.
Under this prevailing paradigm, early clinical and translational efforts focused almost entirely on isolating these youthful factors or transfusing entire fractions of young plasma into older patients. Biopharmaceutical efforts operated on the assumption that supplementing a depleted biological reserve was the necessary and sufficient step required to reverse age-related decline and restore cellular homeostasis 10.
The Harmful Factor Dilution Paradigm
Over the subsequent decade, evidence mounted suggesting that the accumulation of harmful factors in aged blood plays a far more dominant role in organismal aging than the mere loss of youthful factors. In this framework, the young animal in a parabiotic pair serves effectively as a living biological filter, utilizing its robust liver and kidneys to clear metabolic waste, inflammatory markers, and senescent cell secretions from the shared circulation 11.
The "bad old blood" hypothesis asserts that physiological aging is characterized by the chronic secretion of pro-inflammatory cytokines, chemokines, and senescence-associated secretory phenotype factors 1014. When shared through parabiosis, the old systemic environment rapidly induces severe aging phenotypes in the young animal, a phenomenon that is not easily explained by a simple lack of youthful factors, but rather by the active presence of toxic biological inhibitors 1418.
Neutral Blood Exchange as an Experimental Intervention
To systematically test the addition versus dilution paradigms, researchers developed the Neutral Blood Exchange model. In a Neutral Blood Exchange procedure, researchers remove approximately fifty percent of the blood plasma from an old mouse and replace it with a neutral, synthetic physiological fluid consisting of standard saline supplemented with five percent albumin. This effectively dilutes the old plasma without introducing any young circulating factors whatsoever 1012.
The results of Neutral Blood Exchange experiments have provided robust support for the dilution hypothesis. A single Neutral Blood Exchange procedure triggers a massive resetting of the systemic signaling milieu to a youthful, pro-regenerative state 12. In older mice subjected to the exchange, researchers observed enhanced muscle and liver repair, promoted hippocampal neurogenesis, reduced neuroinflammation (evidenced by significantly fewer activated CD68-positive microglia), and improved behavioral performance in novel object and whisker discrimination tests 12.
Furthermore, comparative proteomics demonstrated that simply diluting old blood plasma yields a marked increase in the circulating determinants of brain maintenance and repair. This suggests that removing inhibitory senescence-associated factors relieves the suppression of endogenous regenerative pathways, allowing the aged body to heal itself 12. Neutral Blood Exchange was found to have a stronger and more robust rejuvenating effect on the aged brain than the systemic administration of the targeted senolytic drug ABT-263. This indicates that resetting the total signaling milieu via dilution is highly effective at reducing the propagation of peripheral senescence to the central nervous system, surpassing the efficacy of targeted cellular ablation 12.
Key Circulating Factors in Systemic Aging
Decades of exhaustive proteomic and transcriptomic analyses have identified specific circulating proteins, chemokines, and extracellular vesicles that act as the primary molecular mediators of systemic aging and rejuvenation 914.
Pro-geronic Factors
Pro-geronic factors accumulate steadily with age, driving cellular senescence and actively inhibiting tissue repair across multiple organ systems. The chemokine CCL11, also known as Eotaxin-1, is found to be highly elevated in the plasma and cerebrospinal fluid of aging humans and mice. CCL11 is heavily implicated in impaired spatial learning and reduced hippocampal neurogenesis, promoting cellular senescence by triggering pro-oxidant and pro-inflammatory signaling pathways 114.
Beta2-Microglobulin, a component of the major histocompatibility complex class I, acts as another potent circulating factor that negatively regulates cognitive and regenerative functions in the adult hippocampus. Increased systemic levels of Beta2-Microglobulin actively inhibit the proliferation and differentiation of neural progenitor cells, contributing directly to age-related cognitive decline 914. Additionally, classic markers of cellular senescence such as p16Ink4a and p21Cip1, alongside senescence-associated secretory phenotype factors like Interleukin-1 beta, Interleukin-6, Tumor Necrosis Factor-alpha, and Monocyte Chemoattractant Protein-1, are highly elevated in aged blood. These factors induce systemic chronic inflammation, a state commonly referred to in geroscience as "inflammaging" 520.
Anti-geronic Factors
Conversely, several biological factors that are abundant in young plasma exhibit potent regenerative properties. Growth Differentiation Factor 11, a member of the Transforming Growth Factor-beta superfamily, was initially identified for its remarkable ability to reverse age-related cardiac hypertrophy. Subsequent studies confirmed its role in enhancing neural and skeletal muscle regeneration 1916. The hormone oxytocin has also been documented as a critical age-modifying factor that promotes muscle regeneration and stimulates dormant cell division 119.
Most recently, small extracellular vesicles have emerged as highly potent mediators of intercellular communication, transferring youthful nucleic acids and proteins between cells. A landmark 2024 study published in Nature Aging by researchers at Nanjing University demonstrated that injecting small extracellular vesicles derived from young plasma into twenty-month-old male mice extended their median lifespan by 22.7 percent, allowing the mice to reach up to 1,266 days of age. The extracellular vesicle injections ameliorated age-related functional decline across the hippocampus, skeletal muscles, heart, testes, and bones by fundamentally improving mitochondrial energy metabolism 21222324.
Comparison of Circulating Mediators in Aging
| Factor or Biological Component | Classification | Primary Mechanism of Action | Tissue Target and Resulting Effects |
|---|---|---|---|
| CCL11 (Eotaxin-1) | Pro-geronic | Triggers pro-oxidant and pro-inflammatory pathways; induces senescence secretion. | Brain: Actively inhibits hippocampal neurogenesis and impairs spatial memory. 114 |
| Beta2-Microglobulin | Pro-geronic | MHC-I component; negatively regulates neural progenitor cell development. | Brain: Impairs broad cognitive and regenerative functions. 914 |
| MCP-1 / IL-6 / TNF-α | Pro-geronic | Mediates systemic chronic inflammation and deep cellular senescence. | Systemic: Drives "inflammaging" and tissue degradation across multiple organs. 520 |
| Growth Differentiation Factor 11 | Anti-geronic | TGF-beta superfamily signaling; heavily modulates cellular differentiation. | Heart, Muscle, Brain: Reduces cardiac hypertrophy; aids rapid tissue regeneration. 19 |
| Oxytocin | Anti-geronic | Hormone signaling promoting resident stem cell division. | Muscle: Restores skeletal muscle cell proliferation. 119 |
| Small Extracellular Vesicles | Anti-geronic | Transfers youthful nucleic acids and proteins to alter cellular metabolism. | Systemic: Improves mitochondrial energy metabolism; significantly extends lifespan. 212223 |
Clinical Translation and Biopharmaceutical Development
The profound biological effects observed in murine models of heterochronic parabiosis have spurred a robust, well-funded, and occasionally controversial effort to translate blood exchange principles into human therapeutics 10. The modern biopharmaceutical landscape ranges from rigorously controlled clinical trials utilizing specific, highly purified plasma fractions to commercial anti-aging clinics offering off-label plasma exchange therapies.
Plasma Fraction Trials in Neurodegeneration
In the pursuit of viable, scalable therapies for severe neurodegenerative diseases, biopharmaceutical companies like Alkahest, operating as a subsidiary of the global healthcare company Grifols, have developed proprietary plasma fractions derived from young human donors. These fractions, notably GRF6019 and GRF6021, are engineered to isolate beneficial proteins while removing deleterious or unnecessary plasma components 2425.
In a Phase II clinical trial assessing the candidate GRF6019 in patients with mild-to-moderate Alzheimer's disease, forty participants were randomized to receive intravenous infusions of either 100 milliliters or 250 milliliters of the plasma fraction. The dosing regimen consisted of infusions for five consecutive days during the first week, and again for five consecutive days during the thirteenth week 2526. The trial successfully met its primary safety and tolerability endpoints. Crucially, secondary endpoints revealed that participants maintained their cognitive and functional status over the entire six-month monitoring period. Patients demonstrated negligible cognitive decline as measured by standard industry batteries, including the 11-item Alzheimer's Disease Assessment Scale-cognitive subscale, the Mini-Mental State Examination, and the Alzheimer's Disease Cooperative Study Activities of Daily Living scale 2528.
Simultaneously, the fraction GRF6021 has been actively investigated in a Phase II trial for subjects suffering from Parkinson's disease with associated cognitive impairment. Completed in 2020, the study indicated that the infusions were well-tolerated and demonstrated positive preliminary effects on cognitive endpoints for the Parkinson's cohort 2427. Furthermore, independent academic trials conducted at Stanford University evaluated the safety of infusing one unit (approximately 250 milliliters) of young fresh frozen plasma from male donors aged 18 to 30 into patients with Alzheimer's disease. The trial, involving eighteen screened patients undergoing four weekly infusions, confirmed the fundamental feasibility and tolerability of the intervention. With no serious related adverse events reported, the trial laid essential groundwork for larger, double-blinded, placebo-controlled efficacy studies 30.
Therapeutic Plasma Exchange
Based on the compelling evidence supporting the plasma dilution paradigm, Therapeutic Plasma Exchange has emerged as a highly active area of clinical investigation for longevity and neuroprotection. Therapeutic Plasma Exchange involves mechanically filtering the patient's blood to remove old, toxin-laden plasma, replacing the volume with FDA-approved albumin or sterile saline solutions 2829.
The landmark Alzheimer Management by Albumin Replacement study utilized Therapeutic Plasma Exchange with albumin replacement to treat Alzheimer's disease. The results indicated that plasmapheresis combined with targeted albumin replacement could successfully slow cognitive decline in severe Alzheimer's patients by physically removing pro-inflammatory circulating factors from the central nervous system and systemic circulation 3031. Today, specialized longevity and regenerative medicine clinics leverage Therapeutic Plasma Exchange to actively reduce age-related plasma factors, remove circulating inflammatory mediators, and optimize systemic cellular function. This procedure is often incorporated into broader integrative medical protocols encompassing hormone optimization and peptide therapy 2832.
Extracellular Vesicles and Next-Generation Approaches
While therapeutic plasma exchange and whole-plasma transfusions present significant logistical and regulatory challenges, next-generation biotechnology startups in Asia and the United States are rapidly pivoting toward highly refined biological technologies. Companies are increasingly focused on epigenetic reprogramming, targeted senolytics, and advanced biomaterial administration to replicate the effects of parabiosis without the need for vast quantities of human donor blood 36.
In South Korea, where the biotechnology sector is rapidly expanding to compete with China's output, startups like MxT Biotech are utilizing advanced microfluidic platforms to engineer complex cell therapies. Concurrently, companies such as ROKIT Healthcare and Dx&Vx are advancing regenerative medicine via 3D bio-fabrication and mRNA vaccine technologies aimed at tissue regeneration and metabolic disease 33343536. Following the discovery of the remarkable lifespan-extending properties of small extracellular vesicles by Chinese researchers in 2024, the commercial extraction and administration of specific vesicular components from young blood has generated significant global interest. However, scientists caution that transitioning these findings from murine models to human clinical reality requires rigorous, multi-year primate trials and standard pharmacological development protocols, fundamentally advising against the direct, unregulated application of blood exchange techniques 222337.
Summary of Clinical Implementations
| Intervention Type | Target Disease or Condition | Mechanism and Clinical Delivery | Notable Trials or Clinical Entities |
|---|---|---|---|
| Plasma Fractions | Alzheimer's & Parkinson's Disease | Purified administration of young plasma proteins (GRF6019/6021) via intravenous infusion. | Alkahest Phase II trials; Stanford University academic trials. 24252630 |
| Therapeutic Plasma Exchange | Neurodegeneration & Systemic Aging | Mechanical filtration of old plasma; replacement with FDA-approved albumin. | AMBAR study; Holistique Clinic; Proactive Longevity. 28293031 |
| Extracellular Vesicles | Systemic Aging & Tissue Degeneration | Extraction and injection of sEVs containing nucleic acids to boost mitochondrial function. | Nanjing University murine trials; emerging Asian biotech startups. 21223635 |
Global Regulatory Frameworks for Blood and Plasma Therapies
The rapid commercialization of plasma-based rejuvenation therapies and advanced regenerative medicines has prompted regulatory agencies globally to establish strict, modernized guidelines. These frameworks govern the collection, processing, and clinical use of biological products, ensuring that clinical translation prioritizes patient safety and rigorous evidence generation.
United States Food and Drug Administration Oversight
In 2019, the United States Food and Drug Administration issued a stark, public warning against commercial establishments offering "young plasma" infusions for unproven health and wellness benefits. Commercial entities that had capitalized on the early parabiosis hype, such as the start-up Ambrosia - which charged consumers up to $8,000 per liter for young plasma - were effectively forced to cease operations following the regulatory intervention 338.
The regulatory agency reaffirmed in recent updates that there is currently no proven clinical benefit for the infusion of young donor plasma to prevent or treat conditions such as normal systemic aging, memory loss, Alzheimer's disease, or Parkinson's disease outside of strictly controlled clinical trials 3038. Plasma administration within the United States remains approved exclusively for indications outlined in the formally recognized Circular of Information for the Use of Human Blood and Blood Components, which primarily covers critical care applications rather than longevity interventions 38.
European Medicines Agency Regulations
The European Medicines Agency regulates plasma-derived therapies with extreme rigor, treating them either as standard medicinal products or as Advanced Therapy Medicinal Products, depending entirely on the extent of their processing and cellular manipulation 3940.
To streamline oversight and remain globally competitive, the European Medicines Agency fully implemented the Clinical Trials Regulation via the centralized Clinical Trials Information System. This mandate required all ongoing trials across the European Union to transition to this unified framework by January 2025 4146. The regulation aims to accelerate the approval of innovative therapies while ensuring high transparency and data integrity across all member states 42. Furthermore, the agency's Priority Medicines scheme facilitates accelerated clinical development for highly promising Advanced Therapy Medicinal Products. However, because physiological aging itself is not formally classified as a disease indication under current European frameworks, securing authorization pathways for purely "anti-aging" therapeutics remains a complex regulatory hurdle 340.
Regulatory Pathways in Japan and South Korea
Asian regulatory bodies have purposefully adopted more flexible, expedited frameworks to foster rapid innovation and attract global investment in regenerative medicine.
In Japan, the Pharmaceuticals and Medical Devices Agency operates under the Pharmaceuticals, Medical Devices, and Other Therapeutic Products Act and the Act on the Safety of Regenerative Medicine. A definitive hallmark of the Japanese regulatory system is its conditional and time-limited approval pathway. Regenerative medical products that successfully demonstrate safety and preliminary efficacy in early trials can receive conditional market approval for up to seven years. During this period, developers must collect post-market clinical data to confirm long-term efficacy for full authorization 43444546. This highly predictable, rapid regulatory environment - which consistently completes reviews in nine to twelve months - has established Japan as a premier destination for global biotechnology trials 4748.
South Korea follows a similar, highly agile dual-track model governed by the Ministry of Food and Drug Safety. The Act on Advanced Regenerative Medicine and Advanced Biopharmaceuticals, officially implemented in 2020, establishes a comprehensive framework for managing cellular therapies, gene therapies, and biologic products. South Korea allows expedited regulatory review for therapies addressing serious diseases with no viable alternative treatments. The nation has also actively explored conditional health insurance reimbursement to generate crucial real-world evidence for advanced regenerative products, positioning itself to aggressively compete with China's booming biopharmaceutical sector 44495051.
Ethical Considerations and Socioeconomic Implications
The ongoing pursuit of systemic rejuvenation through human blood and plasma products raises profound bioethical concerns. The field constantly straddles the difficult line between innovative, life-saving medicine and the potential for systemic socioeconomic exploitation 1952.
Donor Exploitation and Social Inequality
The prospect of treating age-related decline with the blood of young individuals has drawn frequent media comparisons to "vampirism" and dystopian science fiction 352. The primary ethical hazard centers entirely on the commodification of the human body. In the United States, which is one of the few global jurisdictions where donors can be legally financially compensated for plasma, the commercial plasma industry largely relies on younger, lower-income demographics to sustain its massive collection volumes 5359.
Bioethicists and critics argue that financial remuneration heavily incentivizes high-frequency donations among impoverished populations. This practice potentially leads to long-term, unmonitored health risks for vulnerable donors, including severe iron deficiency, depleted plasma proteins, and compromised immune function 5954. Consequently, the World Health Organization strongly advocates for Voluntary Non-Remunerated Donations to protect donor welfare and ensure the inherent safety of the global blood supply, explicitly warning against the socio-economic pressures introduced by paid donation models 5954. Should young plasma therapies or isolated plasma fractions achieve mainstream clinical efficacy for longevity, the resultant surge in commercial demand could exponentially exacerbate the exploitation of young, socioeconomically disadvantaged donors specifically to serve a wealthy, aging elite 5255.
Bioethics Nationalism and Supply Chain Vulnerabilities
The global reliance on plasma has birthed a complex geopolitical phenomenon recently termed "bioethics nationalism." Because many affluent nations strictly ban the financial compensation of plasma donors on moral and ethical grounds, they consistently fail to collect sufficient plasma to meet their own domestic healthcare needs. Consequently, the United States - which represents only five percent of the global population - currently supplies approximately sixty-eight percent of the world's raw plasma used to manufacture essential plasma-derived medicinal products 5355.
This dynamic creates a profound ethical paradox: countries enforcing strict bioethical bans on remuneration indirectly rely on the compensated, often low-income donor systems of the United States to supply their hospitals with critical therapies for immunodeficiencies, rare diseases, and severe trauma 5355. The European Medicines Agency has explicitly recognized this severe vulnerability in their medical supply chain. In response, the agency has formally advised the European Commission to leverage the proposed Critical Medicines Act to heavily support the development of alternative synthetic therapies, and to aggressively boost regional unremunerated plasma donation awareness in order to stabilize local supply chains 5657.
Future Directions in Geroscience
Research into heterochronic parabiosis has incontrovertibly demonstrated that biological aging is not an irreversible, strictly cell-autonomous decay. Rather, it is a highly flexible biological state dictated in large part by the systemic circulating environment 719. The scientific transition from the "addition" hypothesis - which focused on transfusing young blood - to the "dilution" and neutral blood exchange paradigms represents a critical maturation in the field of geroscience. This conceptual shift actively redirects clinical focus away from unproven, ethically dubious young plasma transfusions and toward targeted, synthetic interventions designed to clear senescence-associated secretory phenotype factors and resolve chronic inflammaging 121658.
As major biopharmaceutical entities refine their developmental approaches - moving toward highly scalable technologies like targeted specific plasma fractions, senolytics, therapeutic plasma exchange, and synthesized extracellular vesicles - the global reliance on human donor plasma for direct longevity interventions is expected to steadily decrease 222828. Future clinical frameworks in longevity medicine depend heavily on establishing universally validated biomarkers of aging, navigating incredibly complex international regulatory landscapes like the European Union's Clinical Trials Regulation and Japan's Pharmaceuticals and Medical Devices Act, and addressing the stringent bioethical mandates necessary to ensure safe, equitable access to age-modifying therapeutics.