Gut microbiome influence on human mood and behavior
The concept of a bidirectional communication network between the gastrointestinal tract and the central nervous system - formally designated as the microbiome-gut-brain axis - has evolved into a primary focal point in contemporary neuroscience, gastroenterology, and psychiatry. The human gastrointestinal tract harbors a complex ecological community comprising over 100 trillion microbial cells, including bacteria, viruses, fungi, and archaea, which collectively contain over 150 times the number of genes found in the human genome 12. Theoretical frameworks mapping the connection between these microbial populations and human psychological states have transitioned from early twentieth-century observations of "melancholic" intestinal flora to sophisticated multi-omics research that identifies precise biochemical, neural, and immunological signaling pathways 2344.
Within this paradigm, specific live microorganisms and microbiota-targeted interventions that yield measurable mental health benefits have been designated as "psychobiotics," a scientific classification introduced into the literature in 2013 3568710. While preclinical murine models consistently demonstrate profound behavioral and neurochemical alterations following microbiome modulation, translating these findings to human clinical populations remains a persistent scientific challenge. Parallel to this rigorous scientific investigation, a highly lucrative commercial dietary supplement market has rapidly co-opted the language of the gut-brain axis. This intersection of emerging science and commercial interest requires a critical delineation between established biological mechanisms, evolving clinical evidence, and unsupported marketing claims.
Biological Mechanisms of Gut-Brain Communication
Communication between the gastrointestinal microbial biomass and the brain is not mediated by a singular anatomical conduit, but rather through an overlapping, bidirectional network of neural, endocrine, immune, and metabolic pathways 48910.
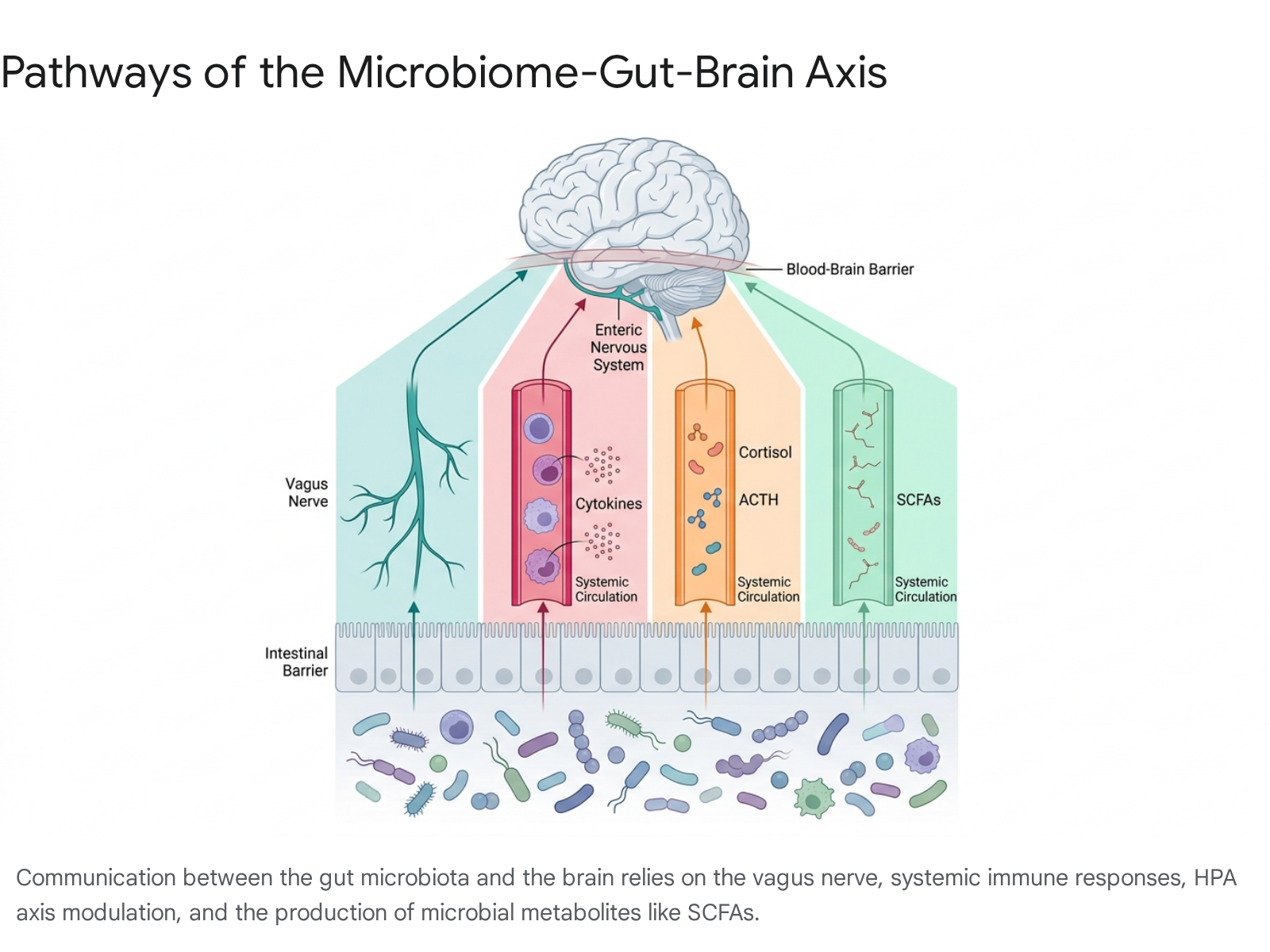
Neural Pathways and Enteric Nervous System Signaling
The most direct anatomical link between the gut and the brain is the vagus nerve, a primary component of the parasympathetic nervous system that extends from the brainstem to the enteric nervous system (ENS). The ENS, heavily embedded within the lining of the gastrointestinal system, acts semi-independently to control digestion while maintaining constant afferent (gut-to-brain) and efferent (brain-to-gut) communication with the central nervous system 4811.
Preclinical studies indicate that the behavioral effects of certain psychobiotic strains are abolished following subdiaphragmatic vagotomy, confirming that vagal afferent signaling is an essential anatomical requirement for specific microbiome-mediated mood alterations 248. Microbes within the intestinal lumen do not directly touch the vagus nerve; rather, they stimulate specialized epithelial cells known as enteroendocrine cells. These cells, sometimes referred to as neuropod cells, interface directly with vagal nerve endings, translating microbial chemical signals into electrical impulses that travel to the brainstem and subsequently to cortical areas involved in emotional regulation 81213.
Neurotransmitter Synthesis and Serotonergic Constraints
The gut microbiota significantly influences the synthesis and regulation of major neurotransmitters. A widely cited physiological metric indicates that approximately 90% to 95% of the body's total serotonin (5-hydroxytryptamine, or 5-HT) is produced in the gastrointestinal tract by enterochromaffin cells 411141516. However, serotonin generated in the gut is incapable of crossing the blood-brain barrier (BBB) 1617. Commercial marketing claims that suggest gut-produced serotonin directly enters the brain to elevate mood represent a fundamental mischaracterization of human neurobiology.
Instead, gut-derived serotonin acts locally to regulate intestinal motility, mucosal secretion, and the activation of vagal afferents that subsequently transmit signals to the brain 41517. Furthermore, the microbiota indirectly influences central serotonin levels by modulating the systemic availability of tryptophan, the circulating amino acid precursor to serotonin. Specific microbial populations can direct tryptophan away from the inflammatory kynurenine pathway and toward serotonin synthesis, increasing the pool of precursors available to cross the BBB 121521. In addition to serotonin, various microbial taxa independently synthesize other neuroactive compounds, including gamma-aminobutyric acid (GABA), dopamine, and noradrenaline, which exert localized effects on the ENS and systemic stress responses 4810111718.
Endocrine Pathways and HPA Axis Modulation
The gut microbiota interacts extensively with the host's endocrine system, primarily by modulating the hypothalamic-pituitary-adrenal (HPA) axis. The HPA axis governs the body's physiological response to psychological and physical stress, culminating in the release of cortisol from the adrenal glands 2410.
The foundational evidence for microbial influence on the HPA axis derives from experiments utilizing germ-free mice - rodents bred in sterile environments devoid of any microbiome. These germ-free subjects exhibit markedly exaggerated HPA axis responses to physical stressors, releasing excess corticosterone 44. Crucially, this hyper-reactive phenotype can be reversed by early-life intestinal colonization with specific Bifidobacterium species 44. In human clinical populations, gastrointestinal dysbiosis is frequently correlated with HPA axis hyperactivity and altered cortisol awakening responses, suggesting that a diverse and balanced microbial ecosystem serves a necessary calibrating function for baseline stress reactivity 151721.
Immune Modulation and Systemic Neuroinflammation
Systemic inflammation is increasingly recognized as a core pathophysiological driver of depressive and anxiety disorders 423. The gut microbiome serves as the primary regulator of the host immune system, with the intestinal mucosa housing the largest compartment of immune cells in the human body. Pathological dysbiosis often leads to a compromise of the intestinal epithelial barrier - a condition colloquially referred to as "leaky gut" - which permits the translocation of lipopolysaccharides (LPS) and other microbe-associated molecular patterns (MAMPs) from the intestinal lumen into systemic circulation 152319.
This endotoxemia triggers a systemic immune cascade, stimulating macrophages and dendritic cells to produce pro-inflammatory cytokines such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) 41219. Elevated circulating cytokines can compromise blood-brain barrier integrity, subsequently activating microglia (the brain's resident immune cells) and inducing states of neuroinflammation 1020. Neuroinflammation actively alters neurotransmitter metabolism, reducing the availability of monoamine precursors and further activating the HPA axis, a cycle strongly linked to the pathogenesis of clinical depression 415. Effective psychobiotic strains function mechanistically by fortifying epithelial tight junctions, decreasing LPS translocation, and upregulating anti-inflammatory cytokines like IL-10, thereby shielding the central nervous system from peripheral inflammatory signals 415231921.
Microbial Metabolites and Short-Chain Fatty Acids
Beyond structural immune interactions, gut microbiota ferment indigestible dietary fibers to produce bioactive metabolites, most notably short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate 810222324. SCFAs are potent systemic signaling molecules that cross the intestinal epithelium to interact with free fatty acid receptors (e.g., FFAR2 and FFAR3) located on enteroendocrine and immune cells 4810.
Butyrate is of particular neurological interest. It supports blood-brain barrier integrity, regulates microglial homeostasis, and acts biochemically as a histone deacetylase (HDAC) inhibitor 341025. By inhibiting HDACs, butyrate promotes epigenetic changes that enhance the expression of brain-derived neurotrophic factor (BDNF), a protein critical for neurogenesis and synaptic plasticity that is frequently depleted in depressed patients 41926. Lower levels of fecal SCFAs have been repeatedly observed in clinical populations diagnosed with major depressive disorder, and SCFA supplementation in preclinical models demonstrates reliable antidepressant effects 10.
| Signaling Pathway | Primary Mediators | Mechanism of Action | Strength of Current Evidence |
|---|---|---|---|
| Neural | Vagus nerve, Enteric Nervous System | Direct electrical and chemical signaling from enteroendocrine cells to the brainstem. | Strong (Preclinical); Emerging (Human) |
| Endocrine | HPA Axis, Cortisol | Microbial modulation of stress hormone cascades and baseline reactivity. | Moderate to Strong (Human) |
| Immune | Cytokines (IL-6, TNF-α), LPS | Maintenance of epithelial barrier; suppression of systemic and neuroinflammation. | Strong (Preclinical & Human) |
| Metabolic | SCFAs (Butyrate), Tryptophan | BBB integrity, microglial regulation, provision of neurotransmitter precursors. | Strong (Preclinical & Human) |
Evaluation of Clinical Efficacy in Psychiatric Disorders
Despite the extensive mechanistic validation established in animal models, the clinical efficacy of psychobiotics in human populations presents a complex and nuanced landscape. Syntheses of randomized controlled trials (RCTs) indicate that probiotic interventions exert highly strain-specific, heterogeneous effects across varying mental health outcomes, with notable differences between clinical and healthy sub-populations.
Meta-Analytical Findings in Depressive Disorders
Recent large-scale meta-analyses and umbrella reviews published between 2023 and 2025 demonstrate that psychobiotics exert a modest to moderate effect on reducing depressive symptoms. A comprehensive 2025 meta-analysis encompassing 72 RCTs and over 6,000 participants revealed a statistically significant reduction in depression scores (Standardized Mean Difference [SMD] = - 0.53; 95% CI: - 0.67 to - 0.39) when comparing probiotic, prebiotic, and synbiotic interventions against placebo 2728.
A narrower 2023 meta-analysis focusing exclusively on clinically diagnosed depressed populations analyzed 13 high-quality RCTs. This review observed an overall SMD of - 0.34 for depression severity, favoring the treatment group 29. Subsequent sensitivity analyses within the same dataset revealed effect sizes as large as - 0.96 (a highly significant clinical reduction) for specific single-strain and multi-strain probiotic interventions when isolated from prebiotics, which failed to show significant independent efficacy for depression 29.
These findings generally indicate that multi-species or targeted single-strain probiotics, administered over periods ranging from 4 to 12 weeks, operate effectively as adjunctive treatments for mild-to-moderate depression 2630. Researchers, however, uniformly caution that high statistical heterogeneity plagues these trials. Variables such as the baseline severity of depression, the specific bacterial formulation utilized, host dietary habits, and individual variations in baseline microbiome composition dramatically shift treatment outcomes, underscoring the necessity for precision medicine approaches moving forward 30313233.
Meta-Analytical Findings in Anxiety and Stress
The clinical data regarding anxiety disorders are markedly more conflicting. While the aforementioned 2025 meta-analysis reported statistically significant, albeit small, reductions in anxiety symptoms across 49 studies (SMD = - 0.44; 95% CI: - 0.59 to - 0.28) 2728, other rigorous umbrella reviews have deemed the evidence for generalized anxiety inconclusive 34.
Interventions frequently fail to separate from placebo in healthy individuals exhibiting low baseline stress. For example, a recent umbrella review highlighted that while probiotics showed an SMD of - 0.19 for anxiety in clinical populations, they showed no significant effect (SMD = - 0.10) in healthy participants 34. This discrepancy suggests that psychobiotics function primarily by repairing a pathologically dysregulated gut-brain axis rather than acting as a universal anxiolytic capable of dampening normative stress responses in otherwise healthy states 103435.
| Mental Health Outcome | Average Effect Size (SMD) | Intervention Type Yielding Best Results | Population Efficacy |
|---|---|---|---|
| Major Depressive Symptoms | - 0.34 to - 0.53 | Single-strain and multi-strain probiotics | Effective as adjunctive therapy in clinically diagnosed patients. |
| Anxiety Symptoms | - 0.19 to - 0.44 | Multi-strain probiotics | Inconsistent; most effective in patients with high baseline stress or clinical diagnosis. |
| Cognitive Performance | Negligible to - 0.10 | Synbiotics / Targeted strains | Limited efficacy; highly variable based on test metrics. |
| Sleep Quality | - 0.39 | Lactobacillus & Bifidobacterium | Moderate improvements noted in insomnia severity scales. |
The Translational Gap in Microbiome Research
The starkest challenge within the field of nutritional psychiatry is the profound discrepancy between preclinical animal data and human clinical trials - a systemic issue known as the translational gap 15363738.
Biological Limitations of Murine Models
The foundational evidence for the gut-brain axis was constructed almost entirely upon murine (mouse and rat) models, utilizing techniques such as fecal microbiota transplantation (FMT) and behavioral assays in germ-free environments. However, rodent models possess profound biological, immunological, and ecological differences from humans 137.
Rodents practice coprophagy (the consumption of feces), a behavior that rapidly homogenizes the microbiome across co-housed subjects within a colony, artificially reducing variance 44. Furthermore, laboratory mice are bred for generations to maintain genetic uniformity and are housed in highly controlled, sterile environments with standardized, unvarying diets 3744. Conversely, human populations present vast genetic diversity, consume highly variable and geographically distinct diets, and are exposed to a lifetime of compounding environmental pollutants, antibiotics, and psychosocial stressors 1244.
The Failure of Lactobacillus rhamnosus JB-1
A prominent historical example of this translational gap involves the probiotic strain Lactobacillus rhamnosus JB-1. In landmark preclinical studies, JB-1 administration significantly reduced anxiety- and depression-like behaviors in an anxious mouse strain, lowered stress-induced corticosterone release, and directly altered the central expression of GABA receptors via vagal pathways 213940.
Based on these exceptional murine results, researchers advanced the strain to a rigorous, randomized, double-blind, placebo-controlled crossover trial involving healthy human males. The participants were subjected to the Socially Evaluated Cold Pressor Test (SECPT) to induce acute stress, alongside comprehensive electroencephalography (EEG) and cognitive assessments. The results were entirely negative: JB-1 failed to demonstrate superiority over placebo across all measures, including subjective mood, HPA axis cortisol response, basal and stimulated cytokine inflammation panels, and cognitive performance 213940. This stark failure underscores a critical reality: exceptional efficacy in a murine behavioral model provides no guarantee of psychotropic activity in the human central nervous system.
Challenges in Gastric Survival and Viability
A secondary mechanical factor contributing to heterogeneous clinical outcomes is the biological challenge of probiotic delivery. The human stomach is an aggressive immunological barrier designed to destroy ingested pathogens via a highly acidic environment, typically operating at a pH between 1.5 and 3.5 4142. This extreme acidity inadvertently decimates many orally ingested commercial probiotic strains before they can transit to the colon.
In vitro research utilizing simulated gastric juice demonstrates that survival rates vary drastically depending on the specific bacterial strain, the protective format of the supplement, and the presence of concurrent buffering agents like food 424350. While specific strains, such as Lactobacillus rhamnosus GG and spore-forming Bacillus coagulans, possess innate natural resilience to gastric acid and bile salts 414445, other unprotected strains experience viability reductions exceeding 10^6 CFU (Colony Forming Units) within merely 5 minutes of acid exposure 53.
Biochemical analyses reveal that survival in acidic conditions is often dependent on cellular metabolism. For example, the presence of glucose allows certain Lactobacillus strains to generate ATP via glycolysis, which is then utilized by the F0F1-ATPase proton pump to actively expel protons from the cell, thereby maintaining internal pH homeostasis 45. Formats also dictate viability; un-encapsulated freeze-dried powders frequently demonstrate survival rates below 1%, whereas targeted delivery systems, enteric-coated capsules, or probiotics suspended in biological matrices (such as dairy or juice) exhibit substantially higher passage rates 42435053.
Colonization Resistance Versus Transient Persistence
Modern multi-omics research has further complicated the narrative of probiotic supplementation by defining the phenomenon of "colonization resistance." The prevailing consumer assumption is that ingested probiotics permanently engraft into the gut lining, fundamentally altering the host's long-term microbial composition. Rigorous invasive human trials have proven this assumption largely false.
When empiric probiotics are administered to humans, they encounter marked competitive resistance from the host's deeply entrenched indigenous microbiome 544647. Rather than establishing permanent mucosal colonies, most commercial probiotics exhibit transient, highly individualized shedding. They pass through the gastrointestinal tract, interacting temporarily with the immune system and producing local metabolites, but are systematically cleared from the body once supplementation ceases 42544647. While these transient microbes can still exert measurable, beneficial immunomodulatory and neuroactive effects during their transit, the lack of persistent engraftment severely limits the duration of their therapeutic efficacy 4246.
| Survival Factor | Variable Impact on Probiotic Efficacy |
|---|---|
| Stomach pH | Highly acidic environments (pH < 2.0) cause massive bacterial die-off for unprotected strains. |
| Delivery Matrix | Enteric capsules, microencapsulation, and whole-food matrices (dairy/juice) significantly increase gastric transit survival compared to free powders. |
| Metabolic Substrates | Presence of glucose/sugars allows bacteria to power proton pumps, resisting acid degradation. |
| Colonization Resistance | Native host microbiomes actively prevent permanent engraftment of foreign probiotic strains, resulting in transient efficacy. |
Strain-Specific Clinical Evidence in Human Trials
Due to the vast genetic diversity among bacterial species, the clinical efficacy of a psychobiotic is inextricably bound to its specific strain designation (e.g., Lactobacillus rhamnosus GG), rather than merely its genus and species (Lactobacillus rhamnosus) 145758. Blanket claims regarding the neurological benefits of "Lactobacillus" are scientifically imprecise. The following specific strains have demonstrated reproducible psychobiotic outcomes in human clinical trials.
Bifidobacterium longum 1714
Bifidobacterium longum 1714 represents one of the most thoroughly investigated psychobiotic strains targeting stress response. In randomized, double-blind, placebo-controlled trials, healthy human volunteers subjected to acute physical and social stress tests exhibited a significantly blunted cortisol response and reported lower perceived daily stress when supplemented with B. longum 1714 484950.
Furthermore, functional neuroimaging and electroencephalogram (EEG) studies revealed that the strain actively modulated central brain activity. Supplementation promoted an increase in calming theta-wave activity in the frontal and cingulate cortices, while simultaneously decreasing beta-3 wave activity in the hippocampus during resting states 484950. These neurophysiological shifts correlated directly with improvements in subjective vitality and mental fatigue scores on the Short Form Health Survey-36 (SF-36), suggesting a tangible physiological capability to regulate neural circuits involved in emotional response and cognitive resilience 485152.
Lactobacillus plantarum PS128
Lactobacillus plantarum PS128 has demonstrated significant anxiolytic and stress-reducing properties, particularly in occupational cohorts. In a rigorous 8-week clinical trial involving information technology professionals exhibiting high baseline psychological stress (Perceived Stress Scale scores ≥ 27), PS128 administration led to significant reductions in self-perceived job stress, state anxiety, and circulating cortisol levels 5354.
Additional placebo-controlled trials have indicated that PS128 can meaningfully improve subjective sleep parameters. In patients presenting with insomnia, PS128 supplementation not only improved subjective ratings of depression and fatigue but objective miniature polysomnography demonstrated a reduction in brainwave activity during deep sleep and a decreased frequency of nocturnal awakenings 55.
Dietary Interventions: Global Microbiomes and Fermented Foods
While isolated probiotic capsules dominate the commercial supplement market and standardized clinical trial designs, epidemiological and nutritional research increasingly emphasizes the role of complex dietary matrices and foundational eating habits in modulating the gut-brain axis.
Global Microbiome Diversity and Traditional Diets
The baseline composition of the human gut microbiome is highly dependent on long-term dietary patterns. Western populations, consuming diets characterized by high quantities of processed foods, refined carbohydrates, and animal proteins, consistently exhibit microbiomes dominated by the Bacteroides genus and demonstrate lower overall alpha diversity 246756.
Conversely, non-Western, traditional, and hunter-gatherer populations - such as the Hadza of Tanzania or rural populations in Papua New Guinea - who consume high-fiber, diverse, plant-based diets, exhibit highly resilient microbiomes dominated by Prevotella and robust fiber-degrading bacterial taxa 24675657. Dietary fiber serves as the primary and essential substrate for the microbial production of beneficial SCFAs, like butyrate, which are necessary for neurological health 2324.
Research demonstrates that increasing the consumption of diverse plant species (upwards of 30 different species per week) significantly enhances the abundance of beneficial, anti-inflammatory bacteria, such as Faecalibacterium prausnitzii, compared to consuming large quantities of only a few plant types 2324. This stark contrast has led researchers to caution that commercial probiotics, historically isolated from the gastrointestinal tracts of Western populations, may be limited in their efficacy. Correcting severe Western dysbiosis likely requires foundational dietary modifications to mimic traditional, high-fiber eating patterns to provide a hospitable environment for psychobiotic bacteria to thrive 246757.
Whole Food Matrices Versus Isolated Supplements
Fermented foods - such as kefir, kombucha, sauerkraut, traditional yogurt, and kimchi - deliver diverse live microbial communities alongside bioactive metabolites, peptides, and prebiotics generated naturally during the fermentation process 194270. Unlike isolated freeze-dried capsules, these complex whole-food matrices protect microbes during harsh gastric transit and provide the necessary fibrous substrates (prebiotics) required for microbial survival and proliferation upon reaching the colon 19224250.
Large observational cohort studies link higher regular consumption of fermented foods with reduced neuroticism and lower social anxiety, particularly in populations possessing high genetic or environmental risks for anxiety disorders 2270. However, strict interventional human trials utilizing whole fermented foods are sparse compared to highly funded supplement trials. Paradoxical findings have occasionally emerged in the literature, such as associations between high fermented food intake and slightly increased depressive symptoms in specific, highly stressed student populations, suggesting that the interaction between dietary fermentation and individual psychopathology remains highly complex and unresolved 2270.
Regulatory Frameworks and Marketing Claims
The translation of early microbiome science into consumer products has birthed a massive global commercial industry, with the broader probiotic market projected to reach values approaching $100 billion 7158. In this rapid expansion, the aggressive marketing language of dietary supplements routinely outpaces the verified clinical evidence, prompting strict responses and new compliance architectures from federal regulatory agencies.
United States: FTC and FDA Enforcement
In the United States, the Food and Drug Administration (FDA) and the Federal Trade Commission (FTC) share regulatory jurisdiction over health-related products; the FDA primarily regulates product labeling, while the FTC regulates advertising and marketing claims 7359. Psychobiotics sold as dietary supplements are explicitly not approved by the FDA to diagnose, cure, mitigate, or treat any disease. In 2021, the FDA issued severe warning letters to multiple companies for illegally marketing dietary supplements as treatments for clinical depression and anxiety, classifying such therapeutically marketed products as unapproved new drugs 6076.
Concurrently, the FTC's sweeping 2023 Health Products Compliance Guidance established rigorous new standards for advertisers 736162. The FTC strictly requires "competent and reliable scientific evidence" - generally defined as high-quality, randomized, placebo-controlled human clinical trials - to substantiate any advertised health benefit claim 736162. The FTC explicitly states that consumer testimonials, reliance on animal studies, or the inclusion of the standard Dietary Supplement Health and Education Act (DSHEA) disclaimer ("This statement has not been evaluated by the FDA...") will not shield a company from liability for deceptive marketing claims 73596263. Furthermore, marketing strategies that extrapolate generalized physiological facts - such as stating "the gut produces 90% of your serotonin" to imply that consuming a specific probiotic pill will elevate brain serotonin and cure depression - are heavily scrutinized as deceptive, as they ignore complex biological realities like the blood-brain barrier 141664.
Europe: EFSA Guidelines and Scientific Rigor
In Europe, the European Food Safety Authority (EFSA) maintains equally, if not more, stringent evaluation criteria for psychological and neurological health claims on food and supplements. EFSA guidance dictates that claims regarding "neurological function," "improved attention," or "improved mood" must be objectively measured by validated, standardized psychometric tests (such as the Profile of Mood States [POMS] or Multiple Affect Adjective Check List [MAACL]) in human populations 65.
Extrapolations from preclinical animal models or generic physiological processes are routinely and categorically rejected by the EFSA panel. Furthermore, EFSA requires companies to demonstrate a plausible, specific biological mechanism of action for the exact product formulation in humans. This rigorous standard has resulted in the repeated rejection of numerous commercial applications seeking to legally link botanical or microbial extracts to anxiety reduction or mood elevation, ensuring that only robustly proven interventions reach the consumer market 6582.
Conclusions
The microbiome-gut-brain axis is a validated, bidirectional physiological network that profoundly influences human behavior, immune function, and neurochemistry. The scientific consensus firmly establishes that systemic neuroinflammation, compromised intestinal epithelial barriers, and reduced microbial diversity contribute significantly to the pathophysiology of mood disorders. "Psychobiotics" represent a legitimate, highly promising frontier in precision psychiatric medicine, acting primarily through systemic immunomodulation, HPA axis calibration, and direct vagal nerve signaling.
However, translating this complex science into consumer therapies requires rigorous clinical skepticism. The clinical efficacy of psychobiotics is highly strain-specific, dose-dependent, and frequently transient, with a substantial failure rate when translating seemingly miraculous interventions from murine models to diverse human populations. While specific, highly researched strains such as B. longum 1714 and L. plantarum PS128 show genuine clinical promise for stress resilience and sleep architecture, they act as subtle modulators rather than standalone replacements for comprehensive psychiatric care. As regulatory bodies like the FTC and EFSA increasingly crack down on unsupported marketing, the future of microbiome-based mental health interventions lies not in hyperbolic "serotonin-boosting" claims, but in precision biomarker targeting and foundational dietary modifications that rebuild the structural integrity of the human gut ecosystem.