Glymphatic system and Alzheimer's protein clearance in sleep
The mammalian central nervous system is unique among the body's organ systems due to its lack of classically defined lymphatic vessels within the brain parenchyma. For decades, the mechanisms by which the brain cleared metabolic waste and maintained fluid homeostasis remained an open question. The characterization of the glymphatic system - a glia-dependent perivascular network responsible for the brain-wide clearance of soluble proteins and neurotoxic metabolites - has fundamentally redefined the understanding of neurological waste removal 123. Operating primarily during sleep, this macroscopic fluid transport system facilitates the continuous convective exchange of cerebrospinal fluid (CSF) and interstitial fluid (ISF) 456. Impairments in glymphatic transport are now recognized as a critical pathophysiological mechanism in the accumulation of amyloid-beta (Aβ) and tau proteins, the primary histological hallmarks of Alzheimer disease (AD) and related neurodegenerative proteinopathies 478.
Anatomical Pathways and Cerebrospinal Fluid Dynamics
The anatomical architecture of the glymphatic system relies on a sequence of perivascular spaces that parallel the cerebral vasculature.
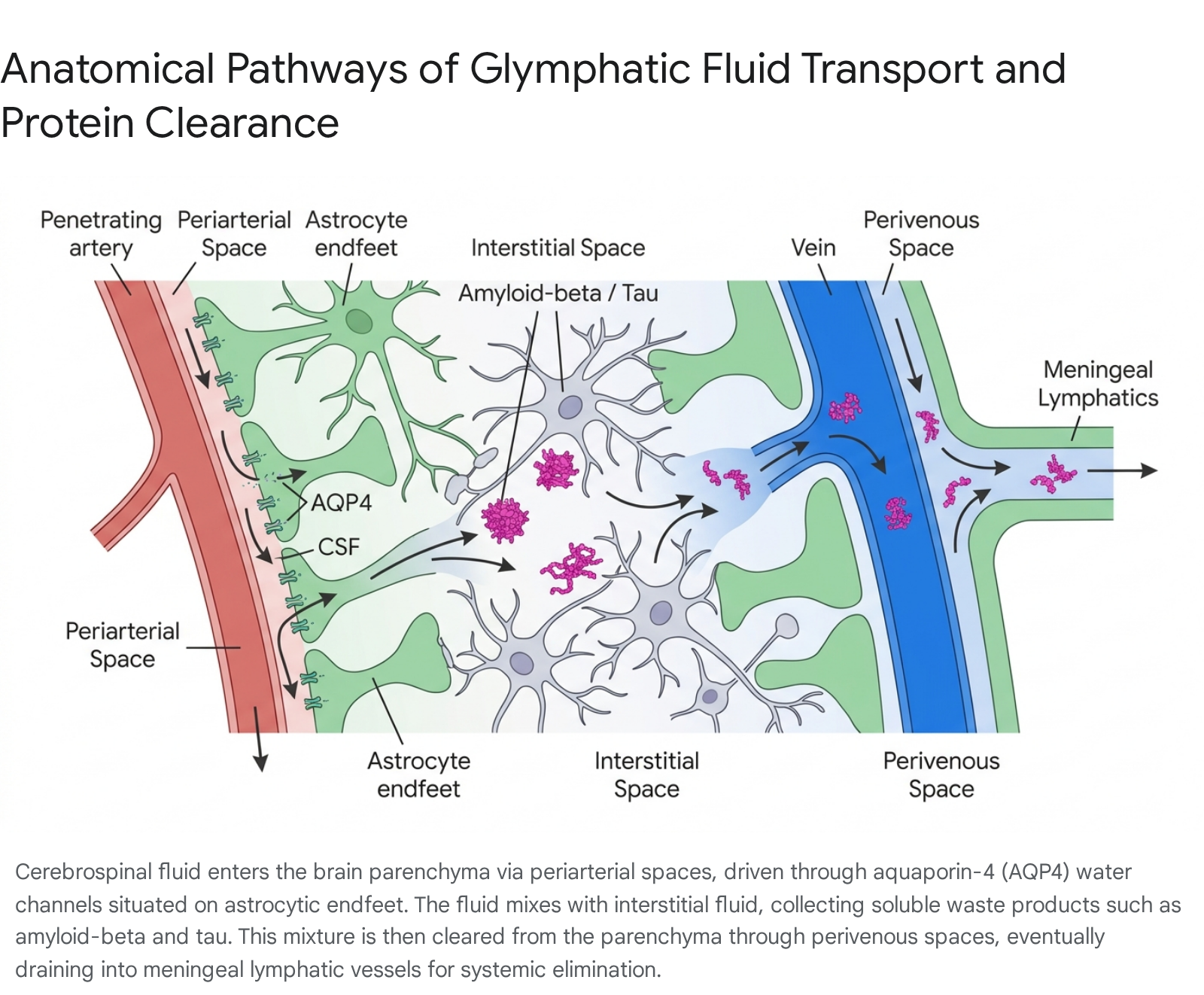
CSF, produced primarily by the choroid plexus within the brain's ventricles at a rate of approximately 500 milliliters per day in humans, circulates into the subarachnoid space 91011. From the subarachnoid space, CSF is driven into the brain parenchyma along the periarterial spaces (Virchow-Robin spaces) surrounding penetrating cerebral arteries 3512.
The transition of fluid from the periarterial space into the dense neuropil relies on a highly specialized cellular interface formed by astrocytic endfeet. These glial extensions completely ensheath the cerebral microvasculature 312. The astrocyte endfeet express exceptionally high densities of the aquaporin-4 (AQP4) water channel, which is anchored to the astrocytic membrane by the dystrophin-associated protein complex (DAPC), specifically through binding with α-syntrophin 313. The dense perivascular polarization of AQP4 channels creates a low-resistance pathway that allows CSF to enter the interstitium rapidly. Once inside the extracellular space, the CSF mixes with ISF, collecting soluble metabolic waste products, misfolded proteins, and excess neurotransmitters 141516.
The resulting solute-laden fluid is propelled by convective bulk flow toward the venous system, draining along the perivenous spaces 917. Experimental animal models demonstrate that arterial pulsations - specifically the rhythmic expansion of the arterial wall during the cardiac cycle - serve as the primary motive force driving fluid convection through these perivascular pathways 1819.
Genetic models underscore the critical role of AQP4 in this process. Mice bearing a genetic deletion of AQP4 (AQP4 KO) or those lacking the α-syntrophin anchoring protein (Snta1 KO) demonstrate a 25% to 60% reduction in glymphatic CSF tracer influx 3121520. In these AQP4 KO models, the loss of fluid convection leads to the stagnation of ISF movement. As a result, KO mice exhibit a 5% to 10% larger brain volume, increased overall water content, and enlarged interstitial spaces compared to wild-type mice, despite possessing similar vascular density and baseline CSF production rates 1215.
Sleep-State Dependency and Noradrenergic Modulation
Glymphatic function is highly synchronized with the sleep-wake cycle, transitioning from a state of relative dormancy during wakefulness to robust activation during sleep 152122. This phenomenon is driven by profound physical changes within the brain's microarchitecture. Experimental murine studies utilizing real-time iontophoresis and two-photon imaging reveal that the volume of the brain's interstitial space expands by 40% to 60% during natural sleep, primarily during the deep, non-rapid eye movement (NREM) stage known as slow-wave sleep (SWS) 152223.
The expansion of the extracellular compartment drastically reduces hydraulic resistance to fluid flow, permitting the rapid and efficient convective exchange of CSF and ISF 12224. NREM SWS is characterized by highly synchronized, high-amplitude delta waves (1-4 Hz) on an electroencephalogram (EEG) 1520. The presence of these prominent slow-wave rhythmic oscillations appears mechanically linked to fluid propulsion. Conversely, during wakefulness and rapid eye movement (REM) sleep, the extracellular space constricts, effectively halting convective glymphatic clearance and limiting solute movement to passive diffusion 121.
The gating mechanism for this state-dependent fluid dynamic relies primarily on noradrenergic signaling 5721. Norepinephrine, synthesized by the locus coeruleus, functions as the central nervous system's primary arousal neuromodulator. High noradrenergic tone during wakefulness limits cerebrovascular compliance and constricts the extracellular space. The onset of NREM sleep initiates an acute suppression of locus coeruleus firing, lowering central norepinephrine levels 57. This chemical shift triggers vascular relaxation, diminishes brain parenchymal resistance, and simultaneously expands the ISF volume 5720. Pharmacological studies mimicking this state by applying adrenergic antagonists directly into the CSF replicate the 60% expansion of the interstitial volume, even in awake animals, providing definitive evidence of the noradrenergic control of brain fluid transport 525.
Prolonged periods of wakefulness or sleep deprivation directly inhibit these processes. A single night of acute sleep deprivation significantly increases the production and interstitial accumulation of Aβ 52021. Chronic sleep fragmentation and a diminished proportion of SWS - common features of aging and preclinical neurodegenerative diseases - compound this failure in continuous waste removal, establishing a vicious biological cycle where toxic protein buildup further degrades sleep quality 2126.
Protein Efflux Mechanisms: Amyloid-Beta and Tau
The buildup of Aβ plaques and neurofibrillary tangles of hyperphosphorylated tau defines the primary pathology of AD 627. AD pathophysiology is driven by an ongoing imbalance between the synaptic-metabolic release of these proteins and their subsequent clearance 2728. The brain utilizes overlapping systems to manage this clearance, including local proteolytic degradation by microglial phagocytosis, transcellular transport across the blood-brain barrier (BBB), and glymphatic efflux into the CSF 62027.
Multicompartmental Models of Plasma Exchange
To systematically quantify how these proteins leave the brain, recent human clinical trials have applied multicompartment mass balance modeling 202930. A standard six-compartment pharmacokinetic model maps Aβ and tau production in neurons, release into the ISF, and subsequent clearance 2030. These proteins exist in varying forms: monomeric Aβ40 and unphosphorylated tau (np-tau) generally resist aggregation, whereas Aβ42 and phosphorylated tau (p-tau) are highly aggregation-prone 2030.
In a landmark multi-site randomized crossover clinical trial involving 39 healthy human participants (aged 49-66), researchers used a novel transcranial multifrequency impedance spectroscopy device to measure brain parenchymal resistance during natural sleep and sleep deprivation 202931. The study demonstrated that sleep-active glymphatic clearance significantly increased morning plasma levels of AD biomarkers 202931. During normal sleep, components of sleep-active physiology - specifically elevated EEG delta power, increased pulse transit time (indicating higher cerebrovascular compliance), and reduced brain parenchymal resistance - acted as the dominant mechanisms driving the overnight clearance of Aβ40, Aβ42, np-tau181, np-tau217, and p-tau181 from the brain to the peripheral blood 2029.
Conversely, during periods of enforced sleep deprivation, parenchymal resistance remained high, and glymphatic clearance stalled 20. In the waking state, the dominant factor governing morning plasma biomarker levels shifted from clearance to the ongoing synaptic-metabolic release of the proteins 20. Additionally, the trial's mass-balance data confirmed that enhanced glymphatic flow preferentially increased the clearance of the highly toxic, aggregation-prone species (Aβ42 and p-tau181) relative to their non-aggregating counterparts 20.
The Role of Glymphatic Flow in Tau Clearance
While the glymphatic clearance of Aβ has been recognized for over a decade, the system's role in clearing tau has only recently been fully established. Tau is an intracellular microtubule-associated protein; however, active synaptic release and cellular death deposit tau into the extracellular space 14. Once in the interstitium, extracellular tau seeds have the potential to spread pathology from neuron to neuron, following anatomical networks in a prion-like manner 814.
In vivo microinjection studies led by Takeshi Iwatsubo have proven that tau is fundamentally dependent on glymphatic convection for removal 1416. When fluorescently labeled human tau is injected into the striatum of wild-type mice, the protein rapidly diffuses, peaks in the CSF within six hours, and is largely cleared from the parenchyma within 48 hours 14. In contrast, in AQP4 knockout mice with impaired fluid convection, the clearance of tau is severely delayed 14. The delayed removal leads to profound pathological consequences: tauopathy mice lacking AQP4 accumulate twice the amount of insoluble p-tau, lose 20% more cortical neurons, and exhibit a 25% thinner cortex compared to mice with functional glymphatic pathways 14. These findings indicate that sluggish perivascular fluid dynamics act as a direct accelerant for tau aggregation and subsequent neurodegeneration.
Comparative Physiology: Human Versus Murine Clearance Kinetics
Experimental reliance on transgenic mouse models requires careful calibration when translating glymphatic mechanics to human physiology. Quantitative metrics comparing the rate and efficiency of Aβ clearance between species reveal substantial differences 3233.
The murine brain clears Aβ at an exceedingly high velocity. The half-life of human Aβ40 injected into a mouse brain ranges from 0.63 hours to 2.0 hours 32. In contrast, human mass balance indices indicate that the human brain clears only about 7% of its Aβ40 burden per hour, corresponding to a much longer half-life of approximately 10 hours in healthy, cognitively normal individuals 32. In human patients with AD, this clearance rate slows further to 5.2% per hour, extending the half-life to roughly 13 hours 32. Analysis of tau kinetics reveals an even slower process in humans, with average tau half-lives estimated at approximately 23 days 5.
The relative division of labor between BBB transport and glymphatic bulk flow also demonstrates species-specific tendencies. Using a modified brain efflux index, researchers estimate that in wild-type mice, roughly 62% of intracerebrally injected Aβ40 is cleared directly across the BBB via endothelial receptors such as low-density lipoprotein receptor-related protein 1 (LRP1) and P-glycoprotein (P-gp), while 38% is cleared via parenchymal degradation and glymphatic transport into the CSF 3233. Comparative in vitro models highlight that mouse endothelial cells (bEnd3) demonstrate a 30-fold higher rate of Aβ40 uptake and a 15-fold higher rate of degradation compared to human brain endothelial cells (hCMEC/D3) 3233. In humans, while BBB efflux remains important, the substantially lower efficiency of human endothelial transport suggests that humans may rely more heavily on glymphatic bulk flow for daily waste maintenance than mice do 632.
Summary of Aβ Clearance Kinetics by Species
| Kinetic Parameter | Human Brain | Murine Brain (Mouse Model) |
|---|---|---|
| Aβ40 Brain Half-Life | ~10 hours (Healthy) / ~13 hours (AD) 32 | 0.63 to 2.0 hours 32 |
| Aβ Clearance Rate | ~7.0% per hour 32 | At least 5x faster than human rate 32 |
| Tau Brain Half-Life | ~23 days 5 | Extracellular fraction cleared <48 hours 14 |
| Endothelial Uptake Efficiency | Low relative to mouse models 32 | 30-fold higher than human in vitro 32 |
| Primary Evaluated Transport | Glymphatic clearance heavily implicated 620 | ~62% BBB Efflux (LRP1/P-gp dependent) 3233 |
Terminal Efflux: Arachnoid Granulations and Meningeal Lymphatics
Once glymphatic fluid containing Aβ and tau has successfully traversed the brain parenchyma and entered the perivenous space, it empties into the subarachnoid CSF. The final physiological step requires the expulsion of this solute-heavy CSF from the cranial vault into the peripheral systemic circulation 910. The exact anatomical structures governing this terminal drainage have been a subject of intense historical and contemporary debate.
For the entirety of the 20th century, anatomical consensus held that CSF exited the subarachnoid space almost exclusively through arachnoid granulations (and smaller arachnoid villi) 101134. These are outcroppings of arachnoid membrane tissue that project directly into the dural venous sinuses (such as the superior sagittal sinus) 343536. Arachnoid granulations operate as unidirectional, pressure-dependent valves; when intracranial CSF pressure exceeds central venous pressure, the valves open, allowing CSF to pass into the venous bloodstream 36.
This monolithic view was challenged by the recent rediscovery and functional characterization of meningeal lymphatic vessels (mLVs) embedded within the dural leaflets lining the skull and spinal column 91011. The central nervous system utilizes these fully functional classic lymphatic vessels to drain fluid, immune cells, and macromolecules directly into the deep cervical lymph nodes 935. Experimental ablations or ligations of the dorsal dural lymphatics in mice result in the severe accumulation of Aβ and tau within the brain and meninges, accompanied by profound cognitive impairment 91037.
Current physiological models suggest a dual-outflow system 935. While arachnoid granulations may serve primarily as a pressure-release valve for water content to maintain intracranial fluid homeostasis, the meningeal lymphatic network acts as the dedicated immunological and waste-clearance pathway 3538. The proportion of fluid utilizing each pathway remains contested and highly species-dependent, with classic studies on large mammals (such as sheep) estimating that between 14% and 47% of injected tracer drains via the extracranial lymphatic system 939. Additional perineuronal drainage routes - particularly the flow of CSF along the olfactory nerves through the cribriform plate and into the nasal mucosa lymphatics - also contribute significantly to total volumetric efflux 91038.
Neuroimaging Markers and Clinical Assessment
Translating glymphatic physiology from laboratory animal models to human clinical practice requires reliable, non-invasive diagnostic methodologies. Direct visualization of glymphatic flow using intrathecal injections of gadolinium-based contrast agents is invasive, poses safety risks, and is largely unsuitable for large-scale epidemiological studies 1936. To overcome this, researchers have developed advanced magnetic resonance imaging (MRI) proxy markers.
DTI-ALPS: Quantifying Perivascular Diffusivity
The Diffusion Tensor Image Analysis Along the Perivascular Space (DTI-ALPS) index is currently the most widely utilized non-invasive imaging marker for glymphatic function 194041. The technique evaluates the diffusion of water molecules specifically within the perivascular spaces surrounding the deep medullary veins 194243.
At the level of the lateral ventricular body, the deep medullary veins and their associated perivascular spaces run perpendicular to the ventricular wall (the x-axis). In this same spatial region, the projection fibers of the corona radiata run craniocaudally (the z-axis), and the association fibers of the superior longitudinal fasciculus run anteroposteriorly (the y-axis) 4143. The DTI-ALPS index mathematically isolates water diffusivity along the x-axis relative to the orthogonal fiber tracts 4143. An ALPS index approaching 1.0 indicates severe glymphatic stagnation, whereas a higher index indicates robust, functional perivascular flow 1744.
Clinical applications of the DTI-ALPS index reveal strong correlations between structural perivascular integrity and neurological health 1941. In healthy aging populations, the ALPS index demonstrates a strict negative correlation with age, confirming that glymphatic efficiency naturally degrades over time 18. In individuals with AD, the index is significantly reduced and positively correlates with scores on the Montreal Cognitive Assessment (MoCA) and the Mini-Mental State Examination (MMSE), tracking alongside the progressive loss of memory, executive function, and visuospatial abilities 4045. The DTI-ALPS index is also acutely sensitive to localized vascular insults; following an ischemic stroke, the index drops precipitously in the infarcted hemisphere within 24 to 72 hours, returning toward baseline only after partial recovery by day seven 1944.
Transcranial Impedance Spectroscopy
Complementing MRI techniques, continuous, non-invasive assessment of the glymphatic system has recently been achieved through multifrequency electrical impedance spectroscopy 2946. Applied via wearable ear-mounted devices, this technology monitors brain parenchymal resistance dynamically throughout the night 4647. Because parenchymal resistance is inversely proportional to the volume of the extracellular space, impedance fluctuations accurately map the real-time expansion and contraction of the brain's interstitial fluid compartments during shifting sleep stages 202946. By measuring resistance at two-minute intervals, this technology bridges the gap between static daytime MRI scans and the highly dynamic, state-dependent nature of overnight brain clearance 4647.
Epidemiological Disparities and Cardiovascular Comorbidities
The integrity of the glymphatic system is deeply intertwined with broader cardiometabolic health and sleep architecture. Large-scale epidemiological meta-analyses reveal that clinical sleep disorders - including obstructive sleep apnea (OSA), insomnia, and REM sleep behavior disorder (RBD) - co-occur with structural markers of glymphatic dysfunction at a pooled prevalence of 44.9% 4. In particular, RBD is a severe prognostic indicator; a vast majority of individuals with RBD eventually progress to α-synucleinopathies like Parkinson's disease, and DTI-ALPS scores in these patient cohorts are profoundly depressed 2248.
Glymphatic impairment also offers a compelling mechanistic explanation for the pronounced racial and ethnic disparities observed in global AD incidence. In the United States, epidemiological data indicates that African Americans are roughly two times more likely, and Hispanics 1.5 times more likely, to develop AD and other dementias compared to White Americans 4950. Genetic profiling demonstrates that these disparities are not driven by higher frequencies of the APOE ε4 allele; rather, they are strongly correlated with a disproportionately high burden of cardiovascular risk factors 4950.
Hypertension and type 2 diabetes - conditions that exhibit highly skewed demographic prevalence - are actively destructive to perivascular clearance 424951. Arterial stiffening induced by chronic hypertension blunts the rhythmic vascular pulsations required to physically drive CSF along the periarterial spaces 1851. Correspondingly, imaging studies confirm that both hypertensive patients and those with uncontrolled type 2 diabetes display significantly lower DTI-ALPS indices compared to matched normotensive and normoglycemic controls 1851. Therefore, targeted management of systemic vascular health presents a direct pathway for preserving glymphatic function and mitigating dementia risk across diverse populations 4249.
Methodological Controversies in Glymphatic Research
The glymphatic model, while serving as a dominant framework in modern neuroscience, faces ongoing methodological scrutiny regarding the exact physics of brain fluid flow.
In 2024, a highly publicized study published in Nature Neuroscience by Nicholas Franks and colleagues challenged the core tenet of sleep-enhanced clearance 52. The research team injected inert fluorescent dye tracers directly into the caudate putamen (deep brain tissue) of mice 52. Utilizing an optical fiber implanted in the frontal cortex, they measured the reappearance of the dye to calculate a diffusion coefficient across varying states of wakefulness and sleep 52. The authors reported that the diffusion coefficient remained constant regardless of the animal's sleep state, leading them to conclude that interstitial fluid moves primarily through simple diffusion rather than bulk convective flow 52. Furthermore, they observed that dye was retained in the brain tissue longer during sleep and anesthesia, arguing that brain clearance is actually more efficient during wakefulness 52.
This conclusion drew immediate and severe rebuttals from the architects of the glymphatic hypothesis, including Maiken Nedergaard and Jonathan Kipnis 52. Critics highlighted substantial methodological artifacts in the Franks study. Primarily, the direct injection of fluid into the dense brain parenchyma inherently damages tissue and artificially spikes localized intracranial pressure 52. By disrupting the delicate physiological pressure gradients that drive CSF into the periarterial spaces, the injection method effectively collapses the very convective system it aims to measure 52. Furthermore, anatomists noted the lack of any known perivascular pathway connecting the deep caudate putamen to the frontal cortex measurement site, rendering the tracking of glymphatic flow across white-matter tracts conceptually flawed 52. Finally, critics argued that measuring localized dye diffusion within the parenchyma is fundamentally different from measuring the absolute clearance of waste out of the brain vault 52. Subsequent independent meta-analyses of diverse AQP4 knockout lines have consistently reaffirmed the existence of sleep-active convective bulk flow 3.
Pharmacological Interventions in Sleep Medicine
With the recognition that optimized sleep architecture directly mediates the removal of Aβ and tau, the pharmaceutical industry has aggressively pivoted toward neuropreventative sleep medicine. Traditional hypnotics (such as benzodiazepines and z-drugs) often induce sedation at the expense of natural slow-wave sleep 5253. As a result, newer pharmacological classes are being investigated for their ability to consolidate sleep and actively augment glymphatic clearance.
Dual Orexin Receptor Antagonists (DORAs)
Orexin (hypocretin) is an excitatory neuropeptide that sustains wakefulness 54. Dual orexin receptor antagonists (DORAs) block orexin signaling, promoting the natural onset of sleep without the severe disruptions to deep sleep architecture seen with older sedative-hypnotics 54. Early proof-of-concept studies using the DORA suvorexant demonstrated that a 36-hour administration protocol in healthy middle-aged adults resulted in a 10% to 15% reduction in phosphorylated tau and a 10% to 20% reduction in Aβ concentrations in the CSF 5455.
To evaluate the long-term disease-modifying potential of this class, the PAD-DORA (Prevention of Alzheimer Disease with Daridorexant) Phase 2 clinical trial was initiated 5256. Daridorexant (Quviviq) is a highly selective DORA noted for its favorable safety profile and minimal next-morning residual sleepiness 5355. The double-blind, placebo-controlled trial enrolled 240 cognitively unimpaired adults who were randomized to receive 50 mg of daridorexant or a placebo nightly for 12 months 525657. Participants were included regardless of baseline sleep complaints, provided they did not have active dementia (MoCA >21) or severe untreated OSA 5357.
The primary biological outcome of the PAD-DORA trial is the change from baseline in the plasma ratio of phosphorylated tau181 to unphosphorylated tau181 (p-tau181/np-tau181) 525657. Secondary endpoints include variations in the Aβ42/Aβ40 plasma ratio, astrocytosis markers (GFAP), and longitudinal cognitive preservation measured by the Preclinical Alzheimer Cognitive Composite (PACC) score 5256. If successful, daridorexant would establish the orexin pathway as a viable pharmacological lever for prophylactic brain health maintenance 53.
Noradrenergic Suppression via ACX-02
An alternative pharmacological approach seeks to bypass the induction of sleep entirely by directly replicating the neurochemical conditions of glymphatic activation. Because locus coeruleus-derived norepinephrine acts as the biological brake on perivascular flow, suppressing central noradrenergic tone artificially dilates the interstitial space and reduces parenchymal resistance 5725.
ACX-02 is a novel investigational combination therapy consisting of dexmedetomidine (an intravenous α2A-adrenergic agonist that strongly suppresses central norepinephrine) and midodrine (an oral peripheral α1-adrenergic agonist) 2528. While dexmedetomidine successfully enhances EEG delta power and mimics SWS, it typically induces systemic hypotension, which reduces the arterial pulsations necessary for glymphatic flow 725. The addition of midodrine stabilizes systemic arterial blood pressure, thereby maintaining optimal cerebrovascular compliance 2558.
In a set of crossover clinical trials involving healthy older adults, a continuous infusion of ACX-02 (0.7 μg/kg/h of dexmedetomidine plus 10 mg of midodrine) resulted in a 9% to 10% absolute increase in the brain-to-blood plasma clearance of both Aβ and tau over a single 4.25-hour intervention 2528. Bayesian mediation analysis confirmed that the primary drivers of this augmented clearance were the treatment's ability to induce dense EEG slow waves and actively lower resistance to perivascular fluid flow 2528.
Non-Pharmacological and Behavioral Interventions
Beyond pharmacotherapy, mechanical and acoustic modulation of sleep physiology is actively being translated into clinical tools designed to augment glymphatic clearance without systemic drug exposure 26.
Closed-Loop Acoustic Stimulation
Acoustic stimulation of slow-wave sleep utilizes automated, wearable EEG headbands (such as the DREEM 2 device) to track the brain's electrical rhythms in real time 5960. The underlying premise is that slow-wave delta oscillations can be synthetically deepened and extended through precise auditory pacing 6162.
The devices operate on a closed-loop algorithm: upon detecting the onset of endogenous NREM slow waves, the system delivers short (50-millisecond) auditory tones exactly phase-locked to the "up-phases" of the oscillations 6061. Triggering the auditory cortex precisely during the up-phase reliably induces further rhythmic slow oscillations, thereby increasing the total duration and amplitude of SWS without causing conscious arousal 6061. Conversely, poorly timed stimuli during the down-phase of the wave actively interfere with the rhythm 61. Pilot trials deploying these devices in the homes of older adults and patients with amnestic mild cognitive impairment have successfully demonstrated significant enhancements in both sleep depth and next-day declarative memory consolidation 265960. By maximizing the highly synchronized delta waves required for the expansion of the interstitial space, acoustic stimulation represents a highly scalable, non-invasive therapeutic vector for optimizing daily brain washing.
Airway Pressure and Respiratory Mechanics
For populations suffering from obstructive sleep apnea, the physical act of sleep acts as a barrier to, rather than an enabler of, glymphatic clearance. OSA causes severe sleep fragmentation, hypoxia, and dramatic intrathoracic pressure swings that impede venous return from the brain 4863. Observational trials (such as the GLIMPSE OSA study) are currently mapping how standard-of-care continuous positive airway pressure (CPAP) therapy restores glymphatic function 64. By stabilizing respiration, CPAP not only allows patients to successfully enter N3 slow-wave sleep but also normalizes the venous pressure gradients required for terminal CSF efflux into the dural lymphatics and systemic circulation 6364.
Conclusions and Future Directions
The characterization of the glymphatic system has provided a unifying mechanical link between the quality of human sleep and the pathogenesis of neurodegenerative proteinopathies. The brain's reliance on a highly orchestrated, state-dependent fluid transport network underscores the necessity of continuous, uninterrupted slow-wave sleep for the prevention of Alzheimer disease. During periods of low noradrenergic tone, the rapid influx of CSF through astrocytic AQP4 channels successfully flushes the parenchyma of neurotoxic amyloid-beta and tau, routing them toward the meningeal lymphatics for ultimate disposal.
While historical limitations in imaging constrained glymphatic research to rodent models, the advent of DTI-ALPS indexing and wearable transcranial impedance spectroscopy has successfully translated these biological principles to human subjects. The resulting data confirm that glymphatic stagnation is a primary driver of cognitive decline and a core mediator of the racial and ethnic disparities seen in global dementia prevalence. Consequently, targeting the glymphatic system through pharmacological interventions like dual orexin receptor antagonists and noradrenergic modulators, as well as via non-invasive acoustic stimulation, represents the most promising frontier in preventative sleep medicine. Future clinical development must focus on optimizing these interventions for chronic, long-term use to secure the structural integrity of the aging brain.