Glymphatic clearance in sleep loss and neurodegeneration in aging
Introduction to Brain Fluid Homeostasis
The central nervous system (CNS) presents a unique physiological paradigm due to its lack of a conventional lymphatic network for the clearance of metabolic waste. For decades, the precise mechanisms governing the removal of interstitial solutes from the brain parenchyma remained elusive. The characterization of the glymphatic system - a brain-wide, glial-dependent perivascular network - has redefined the understanding of cerebral fluid homeostasis. This macroscopic clearance system facilitates the exchange of cerebrospinal fluid (CSF) and interstitial fluid (ISF), propelling the efflux of soluble proteins and neurotoxic metabolites, including amyloid-beta (Aβ) and tau, out of the brain environment 12.
Research over the past decade has established that glymphatic function is highly state-dependent, exhibiting profound enhancement during sleep and significant suppression during wakefulness 34. However, this vital clearance mechanism is highly vulnerable to the aging process. The structural degradation of astrocytic networks, combined with age-related vascular stiffening and progressive alterations in sleep architecture, collectively precipitate a failure in brain waste clearance. This resulting glymphatic impairment establishes a pathological environment that fosters protein aggregation, acting as a critical, potentially causal link between chronic sleep loss and the onset of neurodegenerative disorders such as Alzheimer's disease (AD) 15.
This report provides a comprehensive analysis of the biophysical drivers of the glymphatic system, explores the ongoing methodological debates regarding cerebral fluid dynamics, and synthesizes global epidemiological data on sleep architecture and cognitive decline. Furthermore, it details the emerging pharmacological and non-pharmacological therapeutic strategies currently in clinical trials aimed at restoring glymphatic function in aging populations.
Anatomical and Biophysical Foundations of Cerebral Clearance
The Paravascular Pathway and Aquaporin-4
The architectural foundation of the glymphatic system relies on the intricate arrangement of the neurovascular unit. Cerebrospinal fluid, primarily synthesized by the choroid plexus, enters the subarachnoid space and is subsequently driven into the brain parenchyma along the periarterial spaces surrounding penetrating cerebral arteries 26. The outer boundary of these perivascular spaces (often referred to as Virchow-Robin spaces) is structurally defined by the overlapping endfeet of astrocytes, a specialized type of glial cell 27.
These astrocytic endfeet are uniquely characterized by a dense, highly polarized expression of the aquaporin-4 (AQP4) water channel 89. AQP4 channels serve as the primary bidirectional conduits for water movement between the perivascular space and the glial syncytium, thereby regulating the influx of CSF into the deeper interstitial space. Once in the parenchyma, the CSF mixes with ISF and accumulates metabolic byproducts generated by ongoing neuronal and glial activity. This fluid mixture is then directed toward perivenous spaces, where it exits the brain tissue 69.
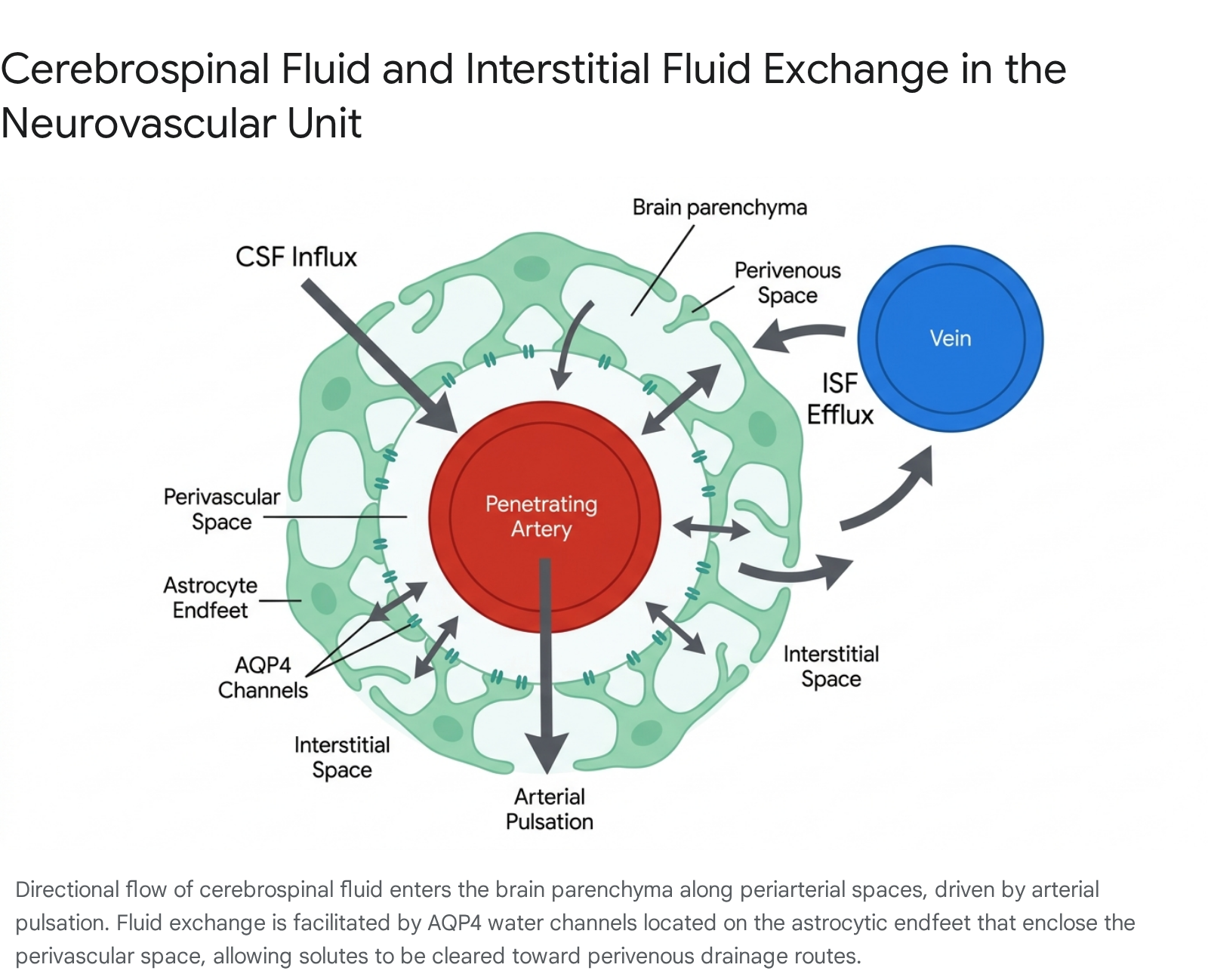
Downstream Lymphatic Integration and the Nasopharyngeal Plexus
While the glymphatic system dictates intra-parenchymal clearance, the ultimate removal of these waste products from the CNS requires integration with the peripheral lymphatic system. Recent anatomical research has expanded the understanding of these efflux routes. Traditionally, arachnoid granulations and dural meningeal lymphatic vessels were considered the primary drainage sites 710.
However, emerging evidence identifies the nasopharyngeal lymphatic plexus (NPLP) as a critical hub for CSF outflow. Connected to upstream lymphatic vessels near the cavernous sinus, pituitary gland, and through the cribriform plate, the NPLP actively drains CSF into the deep cervical lymph nodes 11. While there is no direct anatomical cellular linkage between the glymphatic perivenous spaces and the NPLP, these structures function synergistically. The glymphatic network delivers solute-laden ISF to the subarachnoid space and meningeal borders, where the NPLP and dural lymphatics capture the fluid to sustain the balance of cerebral fluids and finalize the systemic clearance of neurotoxic proteins 11.
Biophysical Drivers of Fluid Transport
The movement of fluid through the dense cerebral parenchyma necessitates substantial mechanical driving forces. The leading biophysical driver of glymphatic influx is arterial pulsation entrained to the cardiac cycle. High-speed particle tracking in vivo has demonstrated that CSF bulk flow in the perivascular space is pulsatile and synchronizes directly with the heartbeat, pushing fluid deep into the brain alongside penetrating arterioles 71213.
Beyond cardiac pulsatility, respiratory mechanics also play a critical role. While arterial pulsation provides positive pressure for influx, respiratory movements influence venous hemodynamics, exerting a negative-pressure "suction" effect that facilitates the efflux of ISF from the brain into the perivenous spaces 1314.
Furthermore, very low-frequency (VLF) vasomotor waves (less than 0.1 Hz) originating from arterial smooth muscle have recently been identified as potent drivers of solute convection. During sleep, these slow vasomotor waves increase in speed and amplitude, driving robust volumetric changes in the perivascular spaces that propel fluid through the tissue 1314.
Noradrenergic Regulation and the Sleep-Wake Cycle
The efficiency of glymphatic transport is not static; it fluctuates dramatically based on the organism's state of arousal. In awake states, glymphatic flow is highly restricted, whereas during sleep or general anesthesia, clearance rates increase significantly 12. The primary biological mechanism regulating this transition is central noradrenergic tone 47.
Norepinephrine, a neuromodulator predominantly secreted by the locus coeruleus in the brainstem, is a principal inducer of arousal. During wakefulness, high levels of extracellular norepinephrine restrict the brain's interstitial volume, thereby increasing parenchymal resistance to fluid flow. Furthermore, norepinephrine acts directly on choroid plexus epithelial cells to reduce overall CSF synthesis 47.
Upon transitioning to non-rapid eye movement (NREM) sleep, locus coeruleus firing declines, leading to a sharp drop in norepinephrine. This state shift results in a structural expansion of the extracellular space by up to 60%, drastically lowering resistance and permitting a surge of CSF-ISF exchange 24. During deep sleep, the brainstem releases residual, tiny waves of norepinephrine approximately once every 50 seconds. These infraslow oscillations trigger rhythmic pulsations in the brain's blood vessels, serving as a synchronized physiological pump that drives glymphatic clearance 14. Experimental pharmacological blockade of norepinephrine receptors in awake rodents successfully mimics this sleep state, inducing extracellular expansion and initiating glymphatic transport comparable to levels seen in naturally sleeping animals 47.
The Convection Versus Diffusion Debate
The Interstitial Bulk Flow Paradigm
Since its formal characterization in 2012, the glymphatic hypothesis has fundamentally reshaped the study of neurodegeneration. Central to this model is the premise that solute movement through the brain interstitium occurs via bulk convective flow - a pressure-driven movement of the solvent (water) that sweeps solutes along indiscriminately of their molecular size 1215.
Initial studies utilizing in vivo two-photon microscopy and dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) demonstrated that tracers injected into the subarachnoid CSF moved rapidly into the brain with size-independent kinetics, a hallmark of bulk flow 12. The dependency of this system on AQP4 was also established early on; AQP4 knockout mice exhibited a reported 55% to 70% reduction in interstitial solute clearance, cementing the channel's role in facilitating convective fluid exchange 289.
Methodological Critiques and Diffusion-Dominated Models
Despite widespread acceptance, the biophysical exactness of convective bulk flow within the dense neuropil has been the subject of persistent debate. A rigorous critique emerged in a 2024 study published in Nature Neuroscience by researchers at Imperial College London, who utilized advanced photobleaching and fluorescent dye diffusion techniques to challenge the core tenets of the glymphatic model 1716.
The Imperial College team reported that the diffusion coefficient of fluorescent dyes remained constant across states of wakefulness, sleep, and sedation, concluding that solutes move passively through the tissue via diffusion rather than being actively propelled by CSF bulk flow 1717. More controversially, their direct measurements indicated that the clearance of dyes from the frontal cortex actually slowed during sleep and anesthesia compared to the awake state 171617. Other critiques have highlighted inconsistencies in replicating the drastic clearance impairments in AQP4 knockout lines, suggesting that prior data on solute transport in the brain extracellular space may have been misinterpreted 812.
Reconciling Conflicting Transport Dynamics
The conflicting findings between convective and diffusive models often stem from divergent experimental methodologies. Proponents of the glymphatic system argue that the experimental techniques utilized by the dissenting studies introduce significant artifacts. Specifically, inserting needles to inject tracers directly into the brain parenchyma disrupts the delicate, natural pressure gradients that drive glymphatic transport, causes local tissue damage, and artificially elevates intracranial pressure (ICP), thereby suppressing physiological convective flow 151716.
When non-invasive methods are utilized, such as tracking tracers administered via the cisterna magna, evidence supports a dual-mechanism paradigm. Recent MRI studies mapping regional solute speeds document differences that cannot be explained by diffusion alone 15. The current consensus among a broad segment of the field is a hybrid model: thermal diffusion dictates the movement of molecules over short distances within the dense interstitial space, while convective bulk flow governs rapid, long-distance transport along the periarterial and perivenous pathways 1215. Ultimately, from a clinical perspective, the precise ratio of convection to diffusion is less critical than the measurable reality of net transport and macroscopic waste clearance, which are consistently shown to be sleep-dependent 15.
Sleep Architecture Variations and Cross-Species Translation
Neurophysiological Oscillations in Non-Rapid Eye Movement Sleep
Sleep is a heterogeneous state characterized by distinct neurophysiological stages. The homeostatic drive for sleep, which accumulates during wakefulness, is primarily dissipated during NREM sleep, particularly during the deepest phase known as slow-wave sleep (SWS) or stage N3 118.
During SWS, the electroencephalogram (EEG) is dominated by high-amplitude, low-frequency delta waves (0.5 - 4 Hz) 18. The efficiency of glymphatic fluid transport correlates directly with the magnitude of this slow-wave activity (SWA) 1. The synchronization of neuronal populations during delta oscillations facilitates the large-scale hemodynamic and vasomotor fluctuations required to drive CSF into the perivascular spaces 1019. Consequently, experimental or pathological fragmentation of SWS leads to a catastrophic decline in the clearance of brain waste 1.
Divergence in Rodent and Human Sleep Patterns
The vast majority of fundamental glymphatic research relies on rodent models. However, translating these findings to human pathology requires careful consideration of cross-species variations in sleep architecture. While the basic stages of NREM and REM sleep are conserved, the temporal dynamics and continuity of sleep differ significantly.
Humans naturally exhibit a consolidated, monophasic sleep pattern, typically experiencing 90-minute sleep cycles 20. In contrast, laboratory mice (e.g., C57BL/6 strains) possess a polyphasic sleep pattern consisting of highly fragmented, short sleep bouts characterized by frequent brief awakenings. The mean duration of a sleep cycle in rodents is approximately 10 minutes 1820. Furthermore, while human studies utilize scalp electrodes measuring broad cortical networks, rodent studies rely on invasive local field potential recordings, complicating direct electrophysiological comparisons 18.
| Sleep Architecture Parameter | Human (Homo sapiens) | Laboratory Mouse (Mus musculus) | Implications for Glymphatic Research |
|---|---|---|---|
| Typical Sleep Pattern | Consolidated, Monophasic | Fragmented, Polyphasic | Rodent glymphatic clearance must operate efficiently in brief windows; human clearance may rely on sustained deep sleep. |
| Average Cycle Length | ~90 minutes | ~10 minutes 20 | Translating the duration required for adequate Aβ clearance requires temporal scaling. |
| EEG Measurement | Scalp electrodes (macro-network) | Invasive deep electrodes (micro-network) 18 | Local field potentials in rodents may capture highly localized fluid dynamics not visible on human scalp EEGs. |
| Biological Lifespan | ~70-80+ years | ~24 months 1821 | Age-related glymphatic degradation in rodents is compressed, posing challenges for modeling chronic human neurodegeneration. |
Postural Influences on Glymphatic Efficiency
In addition to neurophysiological states, the physical orientation of the body during rest directly influences the fluid mechanics of the brain. Utilizing MRI and fluorescent tracers, studies in both rats and mice have assessed CSF-ISF exchange across varying body postures.
The data robustly demonstrate that glymphatic transport and the subsequent clearance of metabolic waste are most efficient when animals are in the lateral recumbent position, as compared to prone or supine positions 722. In the prone position (with the head upright), the overall CSF-ISF exchange is markedly reduced, accompanied by an increased, redirected CSF influx into the spinal cord and neck vasculature rather than the cerebral parenchyma 22. The lateral position represents the natural resting posture for rodents, dogs, cats, and humans, suggesting an evolutionary adaptation wherein sleep posture evolved to optimize the anatomical alignment of perivascular pathways and sympathetic tone for maximal waste clearance 22.
Epidemiological Evidence on Sleep Duration and Cognition
The Inverted U-Shaped Risk Profile
While molecular neuroscience defines the mechanics of the glymphatic system, global epidemiological studies provide macroscopic evidence linking sleep patterns to cognitive preservation. A consensus has emerged demonstrating an inverted U-shaped relationship between sleep duration and cognitive health; both short and long sleep durations are associated with measurable functional decline and increased dementia risk 2324.
Short sleep (typically defined as ≤ 6 hours per night) fundamentally restricts the total absolute time available for the glymphatic system to perform maintenance. Consequently, metabolic wastes, including Aβ and tau, accumulate at a rate that outpaces clearance 125. Alternatively, chronic long sleep (typically ≥ 9 or 10 hours) also correlates with cognitive impairment. However, researchers posit that long sleep is rarely the cause of glymphatic failure; rather, it serves as a biomarker for poor underlying sleep quality, high sleep fragmentation, or an ongoing compensatory response to early-stage neurodegeneration and neuroinflammation 232426.
Global Cohort Studies on Aging and Functional Disability
Analyses of large-scale, non-Western cohorts underscore the universality of the sleep-cognition link across diverse genetic and socioeconomic backgrounds. Data drawn from the World Health Organization's Study on Global AGEing and Adult Health (SAGE) evaluated older adults across China, Ghana, India, the Russian Federation, South Africa, and Mexico 25.
The SAGE study found that individuals reporting intermediate sleep durations (7 to 8 hours per night) exhibited significantly higher composite cognitive z-scores - measuring verbal recall, digit span, and verbal fluency - than those in the short or long sleep categories 2527. Furthermore, sleep quality scores were directly and positively correlated with cognitive performance. The China Health and Retirement Longitudinal Study (CHARLS), analyzing a cohort of 15,526 middle-aged and older Chinese adults, corroborated these findings. The CHARLS data highlighted that the negative impact of long sleep (≥ 10 hours) on overall cognitive function was highly pronounced, closely followed by extremely short sleep (≤ 4 hours) 2328.
| Epidemiological Cohort | Population & Scope | Key Findings Regarding Sleep and Cognition |
|---|---|---|
| SAGE (WHO) | Older adults in 6 middle-income nations (China, Ghana, India, Mexico, Russia, South Africa) 25. | Intermediate sleep (7-8 hours) yielded highest cognitive z-scores. Short (<6h) and long (>9h) sleepers exhibited significant cognitive deficits 25. |
| CHARLS | 15,526 middle-aged/older adults in China 23. | Revealed a pronounced inverted U-shaped relationship. Severe sleep extremes (≤4h or ≥10h) strongly predicted functional and cognitive disability 2328. |
| Seattle Longitudinal Study (SLS) | 1,104 adults in the USA tracked over 25 years 29. | High variability in individual sleep duration across decades was a strong prospective predictor of future cognitive decline 29. |
Cultural Variations in Sleep Architecture and Health Outcomes
While the physiological requirement for restorative sleep is biologically conserved, the average duration of sleep varies substantially across different global cultures. Epidemiological data highlight that countries in East Asia, notably Japan and South Korea, consistently report the shortest national average sleep durations, often nearly an hour less than populations in Western Europe or Oceania (e.g., France, New Zealand, Australia) 303132.
Interestingly, these macro-level differences in sleep duration do not universally correspond to variations in national health outcomes 3133. A study analyzing sleep across 71 countries determined that nations with lower average sleep durations did not exhibit lower life expectancies or higher rates of cardiometabolic disease. Instead, health outcomes appear heavily mediated by "cultural fit." Individuals whose personal sleep durations align closely with the established cultural norms of their specific country tend to enjoy better health outcomes, suggesting that humans exhibit a degree of phenotypic flexibility in their sleep needs that is strongly shaped by environmental and cultural contexts 3133.
Age-Related Glymphatic Degradation
Aquaporin-4 Depolarization and Astroglial Senescence
The most significant known risk factor for developing neurodegenerative dementias is age 1. The normal aging process is accompanied by profound structural changes within the neurovascular unit that physically dismantle the glymphatic network.
In a young, healthy brain, AQP4 channels are highly polarized, clustered almost exclusively at the astrocytic endfeet that sheath the penetrating blood vessels. This strategic localization is critical for maintaining the low-resistance pathway for CSF-ISF exchange 512. During aging, astrocytes undergo reactive changes and cellular senescence, resulting in a dramatic loss of this polarization. AQP4 channels migrate away from the perivascular endfeet and become diffusely distributed across the entire astrocytic soma 5. Quantitative studies in aging mice reveal that this widespread loss of perivascular AQP4 polarization is significantly associated with a 40% impairment in the clearance of parenchymal Aβ 5.
Vascular Remodeling and Loss of Arterial Compliance
Because arterial pulsation is the primary kinetic driver of glymphatic flow, the mechanical health of the cerebrovasculature is intimately tied to clearance efficiency. Normal aging induces widespread arteriosclerosis and vascular stiffening, which blunts the kinetic energy transferred from the cardiac cycle to the perivascular space 510.
In aging models, the vessel wall pulsatility of intracortical arterioles is reduced by approximately 27%. This reduction in pulsatility strongly correlates with a decline in macroscopic CSF-ISF exchange 5. Notably, studies indicate that the actual extracellular volume fraction and tissue tortuosity do not differ significantly between young and old brains, indicating that the failure of clearance is primarily a kinetic and mechanical transport issue rather than a structural collapse of the interstitial space itself 5.
Biomarker Kinetics and Amyloid-Beta Aggregation
The confluence of AQP4 depolarization, vascular stiffening, and age-related disruptions in NREM sleep architecture creates an environment where metabolic waste production outpaces clearance. The dynamics of how Aβ and tau are cleared have been quantified in recent human trials.
A highly cited 2026 study demonstrated that glymphatic clearance during normal sleep actively shifts the ratios of aggregation-prone biomarker species reaching the peripheral plasma. Efficient overnight clearance rapidly transports highly amyloidogenic species (such as Aβ42 and p-tau181) out of the brain parenchyma. By removing these prone species swiftly, the system denies them the necessary time to aggregate into larger, insoluble oligomers and plaques 3. Consequently, an efficient sleep cycle results in a higher proportion of monomeric forms reaching the plasma, increasing their respective plasma ratios in the morning 3. Conversely, when glymphatic transport is impaired by aging or sleep deprivation, clearance stalls, allowing these proteins to form large pathological aggregates that physically cannot pass through the perivascular spaces, accelerating the onset of Alzheimer's pathophysiology 23.
Pharmacological Interventions and Clinical Trials
The identification of glymphatic failure as an upstream mechanism in neurodegeneration has sparked a wave of pharmacological innovation. Therapies currently in clinical and preclinical development target either the structural components of the transport pathway (AQP4) or the neurophysiological state that drives it (noradrenergic signaling).
Modulation of Aquaporin-4 Dynamics
Directly modulating the AQP4 channel offers a localized approach to managing fluid flow across the blood-brain barrier and within the perivascular space. Depending on the clinical indication, both inhibitors and facilitators of this channel are under investigation.
| Investigational Drug | Mechanism of Action | Target Indication / Clinical Status | Impact on Fluid Dynamics |
|---|---|---|---|
| AER-271 (SIM0307) | Intravenous AQP4 Inhibitor (prodrug delivering active AER-270) | Severe ischemic stroke / Phase I trials completed 343536. | Designed to block pathological water influx during ischemia, arresting cerebral edema and reducing intracranial pressure 63437. |
| TGN-020 | AQP4 Inhibitor | Brain ischemia and edema / Preclinical 383940. | Pre-treatment significantly reduces the percentage of brain swelling volume and cortical infarction size following ischemic injury 3839. |
| TGN-073 | Orally available AQP4 Facilitator | Alzheimer's disease, dementia / Preclinical 404142. | Increases AQP4-mediated water flux, enhancing interstitial fluid turnover and significantly reducing insoluble Aβ accumulation in the parenchyma 36404142. |
For neurodegenerative diseases like Alzheimer's, facilitators such as TGN-073 hold significant promise. Preclinical MRI studies indicate that administering TGN-073 to normal brains increases apparent diffusion coefficients and parenchymal tracer uptake, confirming that facilitating AQP4 lowers resistance to water transport and enhances the convective flushing required to clear metabolic waste 4041.
Noradrenergic Suppression and ACX-02 Trials
An alternative to targeting the structural channels is to manipulate the autonomic state of the brain to induce a "sleep-like" environment for clearance. Because elevated norepinephrine suppresses glymphatic flow, pharmacological dampening of central noradrenergic tone can theoretically enhance transport. Dexmedetomidine, an alpha-2 adrenergic agonist commonly used as a sedative, has shown efficacy in preclinical models by increasing CSF influx, repolarizing AQP4, and promoting waste clearance 43.
However, dexmedetomidine induces systemic hypotension, which inadvertently reduces the arterial pulsatility necessary to drive the fluid. To solve this, a novel fixed-dose combination therapy termed ACX-02 was developed, combining intravenous dexmedetomidine with oral midodrine (a peripherally acting alpha-1 adrenergic agonist that maintains systemic blood pressure) 4447.
Two notable cross-over clinical trials (including NCT07432997) assessed ACX-02 in healthy older adults 444748. The administration of ACX-02 successfully suppressed central noradrenergic tone without dropping blood pressure, resulting in increased EEG slow waves, enhanced cerebrovascular pulsatility, and reduced parenchymal resistance 4447.
Crucially, dynamic shifts in plasma mass balance indices confirmed that this pharmacological enhancement increased the brain-to-blood clearance of Aβ and tau by approximately 9% to 10% during a single 4.25-hour sleep opportunity 4447.
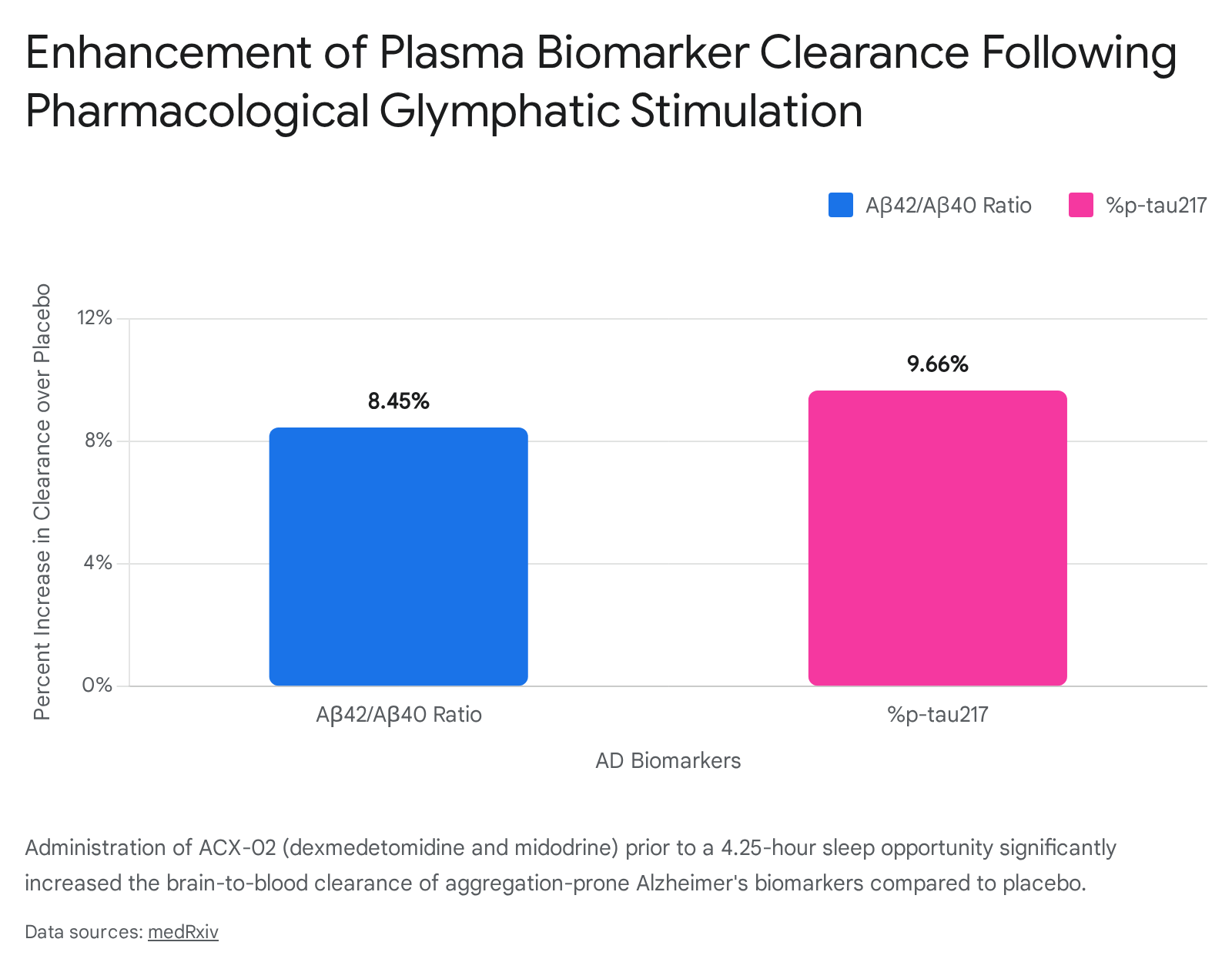
Bayesian mediation analysis confirmed that the induction of slow waves and the reduction of resistance were the key mediators of this enhanced clearance, proving that glymphatic transport can be safely and effectively augmented in humans to combat Alzheimer's-related proteinopathies 444749. Care must be taken with generic sleep aids, however; studies indicate that standard hypnotics like zolpidem can actually reduce restorative norepinephrine waves by 50% and drop fluid transport by 30%, highlighting that chemically forced unconsciousness does not equate to functional glymphatic sleep 1445.
Non-Pharmacological Neuromodulation and Device Interventions
Transcranial Magnetic and Pulse Stimulation
Given the challenges of long-term pharmacological interventions, non-invasive device stimulation is emerging as a potential avenue to boost glymphatic function. Repetitive Transcranial Magnetic Stimulation (rTMS), specifically theta-burst stimulation (TBS), has shown promise in older adults with mild cognitive impairment.
A recent sham-controlled trial evaluated changes in the DTI-ALPS index following ten consecutive days of TBS. The study revealed that TBS interventions significantly modulated glymphatic function, resulting in a quantifiable increase in the ALPS index 46. This effect was particularly pronounced in individuals carrying the APOE $\epsilon$4 allele (a major genetic risk factor for AD), who characteristically exhibit lower baseline ALPS indices. Within this subgroup, the TBS-induced improvements in glymphatic function correlated directly with improvements in memory, demonstrating the clinical relevance of targeted electromagnetic neuromodulation 46.
Similarly, Transcranial Pulse Stimulation (TPS) utilizes acoustic pulses to enhance local cerebral blood flow and neurovascular coupling. Because glymphatic influx depends heavily on arterial pulsatility, restoring local perfusion via TPS may indirectly re-establish the mechanical drive necessary for solute transport, offering a complementary strategy for mild to moderate Alzheimer's patients 47.
Behavioral Factors and Exercise Limitations
Behavioral modifications remain a foundational approach to protecting brain clearance. Maintaining cardiovascular health directly preserves the arterial compliance needed to drive CSF. However, the timing of physical exercise is highly relevant. While regular voluntary exercise improves baseline systemic vascular compliance and ultimately boosts waste clearance during subsequent rest periods, active exercise itself does not increase glymphatic flow. During acute physical exertion, the central release of norepinephrine spikes, actively suppressing the glymphatic network to prioritize cerebral energy metabolism and alertness 7. Therefore, the cognitive benefits of exercise are realized asynchronously, heavily dependent on the quality of the sleep cycle that follows.
Conclusion
The discovery and subsequent mapping of the glymphatic system have provided a unifying mechanistic framework for understanding the profound clinical link between sleep loss, aging, and neurodegeneration. Brain waste clearance is a dynamic, mechanically demanding process reliant on precise neurovascular coupling, polarized aquaporin-4 channels, and the infraslow physiological oscillations uniquely generated during deep non-rapid eye movement sleep.
Normal aging systematically disrupts these components, precipitating a state of chronic clearance failure that allows highly amyloidogenic proteins to aggregate into pathological lesions. While academic debates persist regarding the exact biophysical balance of convection and diffusion within the deeper cortical tissue, the overarching clinical imperative remains clear: protecting and enhancing macroscopic fluid transport is essential for cognitive longevity. With the advent of novel aquaporin facilitators, targeted noradrenergic inhibitors like ACX-02, and non-invasive neuromodulation techniques, the preservation of the glymphatic system represents a highly viable and rapidly advancing frontier in the prevention and treatment of Alzheimer's disease and related dementias.