Glycine and N-acetylcysteine for glutathione and mitochondrial aging
Biological Aging and Oxidative Stress Mechanisms
Biological aging is defined by a progressive deterioration of cellular and physiological function, driven by a complex interplay of molecular deficits. In the field of geroscience, nine primary hallmarks of aging have been identified, encompassing genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, cellular senescence, stem cell exhaustion, altered intercellular communication, and mitochondrial dysfunction 12. A prevailing biological mechanism linking many of these diverse hallmarks is the steady accumulation of oxidative stress (OxS) 34. Oxidative stress arises when the production of reactive oxygen species (ROS) - toxic but naturally occurring byproducts of cellular metabolism - exceeds the body's endogenous antioxidant defense capacity 356.
At the center of the cellular defense apparatus against reactive oxygen species is glutathione (GSH), a tripeptide molecule composed of glutamate, cysteine, and glycine 78. Glutathione operates as the master intracellular antioxidant in human physiology, actively neutralizing free radicals, supporting hepatic detoxification pathways, and facilitating vital cellular repair mechanisms 797. Within the mitochondria, glutathione plays an especially critical role in neutralizing the superoxide radicals generated during the production of adenosine triphosphate (ATP) via the electron transport chain 589.
However, intracellular glutathione concentrations decline significantly with advancing age 1310. This age-associated depletion compromises mitochondrial efficiency, leading to an environment of chronic oxidative stress 345. The resulting mitochondrial dysfunction triggers a cascade of systemic metabolic and physical declines, manifesting as systemic insulin resistance, elevated chronic inflammation (inflammaging), endothelial dysfunction, and the degradation of skeletal muscle strength and cognitive capacity 1611. Restoring intracellular glutathione levels has therefore emerged as a primary interventional target in translational geroscience and longevity research.
Endogenous Glutathione Synthesis Pathway
The biological effort to restore glutathione levels relies heavily on understanding its natural synthesis and degradation pathways. Glutathione synthesis is an intracellular process facilitated by the gamma-glutamyl cycle, requiring two distinct enzymatic steps 1216. In the first step, the amino acids glutamate and cysteine are combined to form gamma-glutamylcysteine, a reaction catalyzed by the enzyme glutamate-cysteine ligase (GCL) 12. GCL is a heterodimer consisting of a catalytic subunit (GCLC) and a modifier subunit (GCLM) 13. In the second step, glycine is added to the gamma-glutamylcysteine intermediate to form the final glutathione tripeptide, a reaction catalyzed by the ATP- and magnesium-dependent enzyme glutathione synthetase (GS) 12.
While the human body maintains abundant supplies of glutamate, the intracellular availability of cysteine and glycine acts as the rate-limiting bottleneck in the synthesis of glutathione 614. As individuals age, systemic concentrations of these two non-essential amino acids frequently decline, leading to a suppression of GCLC, GCLM, and GS expression 131516.
Extracellular Degradation by Gamma-Glutamyl Transpeptidase
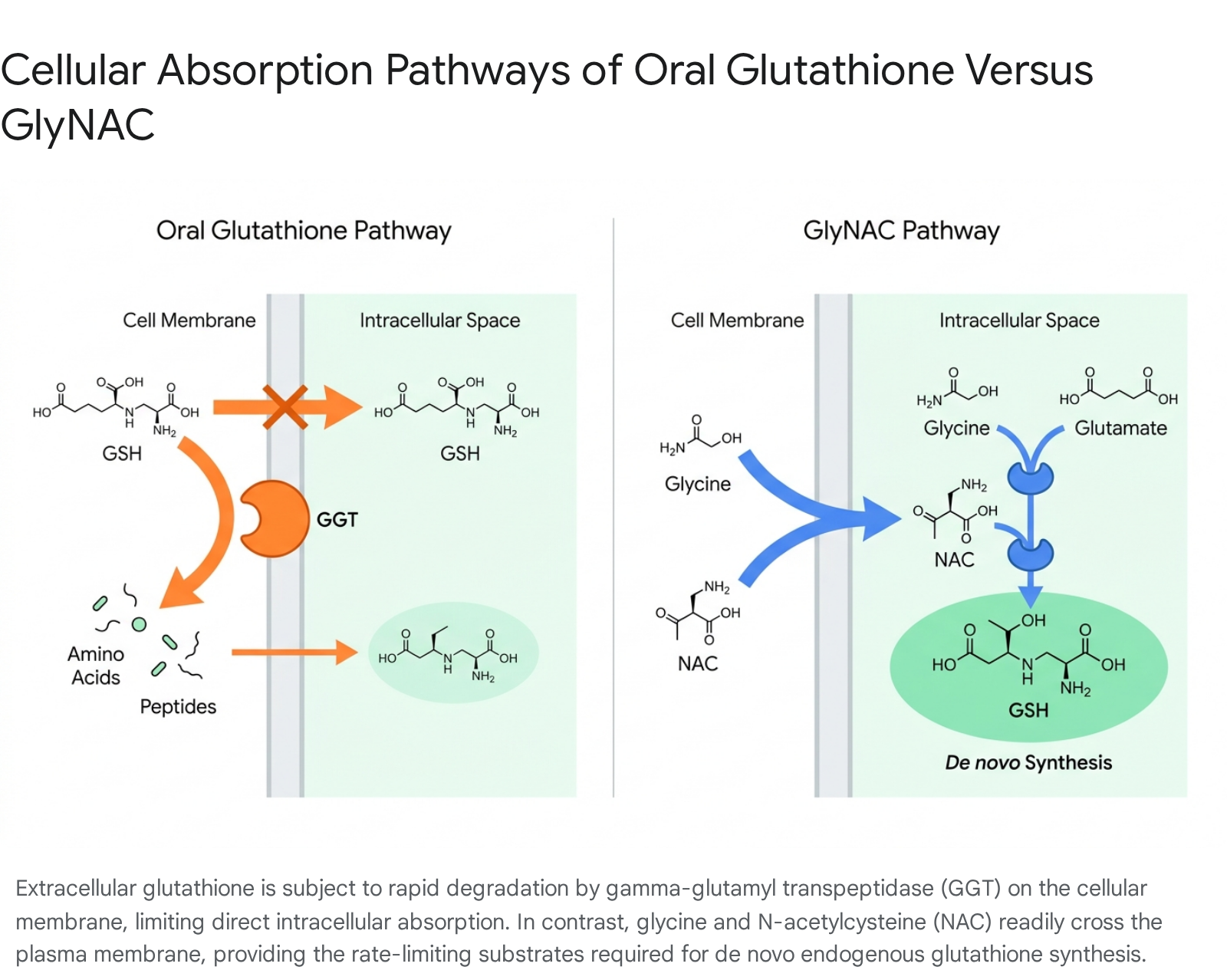
Direct oral supplementation of the intact glutathione molecule is highly inefficient due to its rapid enzymatic degradation in the gastrointestinal tract and systemic circulation 1012. Glutathione contains an unusual peptide linkage between the amino group of cysteine and the carboxyl group of the glutamate side chain 16. Because of this unique gamma-glutamyl bond, standard intracellular peptidases cannot degrade it 16.
However, when ingested orally, exogenous glutathione encounters gamma-glutamyl transpeptidase (GGT), a ubiquitous enzyme located on the outer leaflet of the plasma membrane of cells in various tissues, including the small intestine, liver, kidneys, and bile ducts 121617. GGT acts as the universal facilitator of glutathione breakdown 12. It aggressively hydrolyzes extracellular glutathione, cleaving the gamma-glutamyl bond to release free glutamate and the dipeptide cysteinylglycine (Cys-Gly) 1218. The Cys-Gly dipeptide is subsequently broken down by dipeptidases into free cysteine and glycine 1819.
Because of this rapid and comprehensive extracellular degradation, standard oral glutathione supplements demonstrate systemic bioavailability rates of less than 1%, rendering them largely ineffective for elevating intact intracellular glutathione concentrations 920.
Bioavailability Profiles of Glutathione Supplementation Modalities
Given the physiological challenges of direct glutathione administration, multiple delivery technologies and alternative precursor strategies have been developed to bypass the GGT barrier.
The efficacy of these formulations is measured by their ability to increase systemic plasma concentrations and, more importantly, intracellular red blood cell (RBC) concentrations of reduced glutathione.
| Delivery Modality | Bioavailability Characteristics | Mechanism of Action and Clinical Efficacy | Limitations |
|---|---|---|---|
| Standard Oral Glutathione | Poor (< 1%). Highly variable absorption 7920. | Degraded by gastrointestinal peptidases and membrane-bound GGT. Minimal intact absorption 101216. | Generally ineffective for systemic elevation of GSH. High degradation rate before reaching target cells 8920. |
| Liposomal Glutathione | Moderate to High. 1.9-fold higher cellular uptake than standard oral forms 21. | A phospholipid bilayer (liposome) encapsulates the intact GSH molecule, protecting it from digestive breakdown and facilitating intestinal transport 722. | High per-dose cost. Effects are rapidly reversible, returning to baseline within 1 month of discontinuation 727. |
| Micellar Glutathione (LMG) | High. Peak response (Cmax) up to 2.43-fold higher than standard forms 23. | Utilizes micelle structures to enhance solubility and gastrointestinal absorption 23. | Clinical data relies on short-term (30-day) pharmacokinetic studies rather than long-term aging markers 23. |
| Sublingual Glutathione | Moderate. Bypasses the gastrointestinal tract 810. | Rapidly absorbed via capillaries under the tongue, directly entering the bloodstream to avoid first-pass metabolism 810. | Variable absorption rates depending on individual mucosal permeability 810. |
| Intravenous (IV) Glutathione | Maximum (100%). Complete systemic delivery 789. | Direct delivery into the bloodstream for acute medical use, achieving immediate systemic elevation 79. | Requires strict medical supervision. Carries risks of anaphylaxis and hepatotoxicity. Used primarily for acute toxicosis, not daily supplementation 7927. |
| N-Acetylcysteine (NAC) Monotherapy | High (Approx 70%+ oral bioavailability) 2425. | Provides the rate-limiting amino acid cysteine for endogenous GSH synthesis 72425. | Fails to address concomitant age-related glycine deficiencies. Can occasionally worsen oxidative stress in specific animal models 7. |
| GlyNAC (Glycine + NAC) | High. Rapid absorption of both precursors within 60 minutes of ingestion 2126. | Provides combined substrates to bypass cellular synthesis bottlenecks, forcing robust endogenous intracellular GSH production 5732. | Requires large daily clinical doses (~4.8g to 7.2g). Long-term longitudinal human safety data remains under study 93334. |
Rationale for Precursor Supplementation
Rather than attempting to deliver the intact tripeptide, the alternative pharmacological strategy relies on providing the cellular precursors required for de novo endogenous synthesis. The administration of N-acetylcysteine (NAC) is utilized instead of standard L-cysteine because the added acetyl group stabilizes the molecule during digestion and systemic transport 78. This stabilization allows NAC to reliably reach the intracellular space, where it is subsequently deacetylated and converted into usable L-cysteine 734.
Glycine, historically viewed primarily as a basic structural amino acid with low metabolic priority, is now recognized as an essential component for maintaining optimal glutathione redox status 1534. Clinical data indicates that older adults exhibit significantly lower baseline levels of erythrocyte glycine compared to younger adults 16. When researchers supplement aging cohorts with NAC alone or glutathione alone, the metabolic improvements are often marginal or absent 7.
The combination of these two amino acids provides the exact stoichiometric substrates necessary to overcome the synthesis bottleneck. Researchers tracking clinical outcomes refer to this resultant biological synergy as the "Power of 3" - indicating that the combined presence of glycine, cysteine, and the resulting intracellular glutathione is fundamentally required to effectively correct cellular deficiencies and modulate aging hallmarks 5735.
Reversal of Mitochondrial Dysfunction and Metabolic Flexibility
Mitochondria are the primary source of cellular energy, utilizing two main metabolic pathways for ATP generation: mitochondrial fatty acid oxidation (MFO) and mitochondrial glucose oxidation (MGO) 137. In healthy, metabolically flexible young adults, mitochondria preferentially utilize fatty acids for energy during fasting states 727.
Aging, coupled with the decline in intracellular glutathione, severely disrupts this metabolic balance. In older adults, mitochondrial metabolism shifts inefficiently, resulting in suppressed fatty acid oxidation and an excessive, inefficient reliance on glucose oxidation 3727. High-resolution respirometry analyses from clinical trials confirm this phenotype. For example, when comparing older adults to young adults, older populations consistently demonstrate a significant reduction in fat-burning capacity coupled with elevated glucose reliance, culminating in a higher respiratory quotient 37828.
By restoring intracellular glutathione via GlyNAC supplementation, cells regain the necessary antioxidant capacity required to protect the mitochondrial electron transport chain from continuous superoxide damage 78. Normalizing the redox balance triggers a rapid molecular recalibration of energy metabolism. Clinical measurements demonstrate that providing GlyNAC to older adults results in an upregulation of mitochondrial fatty acid oxidation by up to 78%, and a corresponding 65% decrease in the reliance on mitochondrial glucose oxidation 3727. This correction allows cells to produce energy efficiently, simultaneously reducing insulin resistance by 64% and lowering fasting insulin levels by 65% 3727.
Central Nervous System Energy Demands and Brain Glucose Steal
The restoration of mitochondrial fuel pathways carries profound implications for human neurology and cognitive aging. The brain is an energy-intensive organ that relies almost exclusively on glucose for metabolic fuel 1129. Because of the impaired fatty acid oxidation observed in aging, peripheral non-brain organs incorrectly utilize systemic glucose as their primary energy source 1129.
This creates a systemic metabolic deficit. Utilizing advanced tracer methodologies, researchers have documented a "brain glucose steal" phenomenon 1129. The excessive glucose utilization by impaired peripheral tissues deprives the central nervous system of its required fuel 1129. By restoring mitochondrial fatty acid oxidation in peripheral tissues, GlyNAC supplementation forces non-brain organs back into physiological fatty-acid utilization. This systemic correction halts the stealing of glucose, maximizing glucose availability for the brain. This metabolic realignment is theorized to be a primary driver behind the significant improvements observed in age-related cognitive decline following GlyNAC supplementation 1129.
Preclinical Evidence in Murine Models
Translational research in geroscience relies heavily on rodent models to map biological pathways prior to human application. Extensive studies on naturally aged C57BL/6J mice have established the foundational evidence for the biological efficacy of the glycine and N-acetylcysteine combination.
When aged mice (90 weeks old) were supplemented with a GlyNAC-enriched diet, researchers observed a robust correction of systemic age-associated defects. Most notably, the continuous dietary intervention increased the absolute lifespan of the aged mice by 24% compared to age-matched controls 5. The supplemented mice also exhibited corrected glutathione synthesis pathways, improved mitochondrial fatty acid oxidation, and lower systemic body fat and liver fat content 13.
Subsequent murine investigations focused specifically on the aging central nervous system to understand the physiological basis of age-associated cognitive decline 283031. Because mice do not naturally develop Alzheimer's Disease, studying old, un-engineered mice provides a highly accurate representation of natural, biological brain aging 28. Analyses of the old mouse brain tissue revealed multiple molecular deficits, including severe glutathione deficiency, elevated markers of oxidative stress (measured via thiobarbituric acid reactive substances, or TBARS), genomic damage, and high levels of neuroinflammation 112830. Furthermore, the aged brains demonstrated suppressed expressions of vital neurotrophic factors, including brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), and nerve growth factor (NGF) 112830.
Eight weeks of targeted GlyNAC supplementation successfully corrected these brain defects. The intervention elevated neurotrophic factor levels, restored glucose transporter expressions (GLUT1 and GLUT3), and significantly improved behavioral cognitive performance, as evidenced by aged mice navigating complex mazes faster and with fewer errors 283032.
Human Clinical Trials in Older Adults
The translation of these preclinical murine findings into human interventions has yielded significant clinical data through a series of pilot studies and randomized controlled trials, predominantly focused on older adults.
The 24-Week Pilot Study
The initial human clinical proof-of-concept was established in a 36-week open-label trial involving eight older adults (aged 71 to 80 years) 4915. Participants were supplemented with weight-based dosing of GlyNAC for 24 weeks, followed by a mandatory 12-week washout period.
The 24-week intervention produced marked reversals across multiple physiological systems. Participants demonstrated complete correction of red blood cell glutathione deficiency, decreased oxidative stress, and the reversal of mitochondrial dysfunction 4835. Clinically, these cellular corrections mapped directly to improved physical markers: grip strength increased significantly, gait speed accelerated, and cognitive test performance improved 435.
However, following the 12-week cessation of the supplement, researchers noted that the majority of the biochemical and clinical benefits rapidly declined 43533. This data indicates that the intervention must be maintained continuously to preserve the anti-aging benefits, as the fundamental biological drivers of aging reassert themselves once the precursor supply is withdrawn 833.
The 16-Week Placebo-Controlled Randomized Clinical Trial
To validate the pilot findings with higher statistical rigor, researchers conducted a 16-week double-blind, randomized, placebo-controlled trial involving 24 older adults (aged 61 - 80 years, with a BMI > 27) and a reference cohort of 12 healthy young adults (aged 21 - 40 years) 13. Older adults were randomized to receive either a daily combination of 100 mg/kg of glycine and 100 mg/kg of NAC, or an isonitrogenous alanine placebo administered at 200 mg/kg/day 3.
The results of the 16-week intervention confirmed the prior pilot observations while drastically expanding the documented scope of the physiological improvements.
| Biomarker / Physiological Metric | Older Adults (Baseline) vs Young Adults | Older Adults (Post-16 Week GlyNAC) | Older Adults (Placebo Group) |
|---|---|---|---|
| Total RBC Glutathione (tGSH) | 66-70% lower than young adults 13. | Increased by 225%, reaching young adult levels 1344. | No significant change 713. |
| Oxidative Stress (TBARS / F2-isoprostanes) | Significantly elevated (up to 5x higher) 4434. | Decreased by 72%, neutralizing baseline elevation 44. | No significant change 37. |
| Inflammatory Marker: Interleukin-6 (IL-6) | Markedly elevated (inflammaging) 744. | Decreased by 78% 744. | No significant change 7. |
| Inflammatory Marker: TNF-alpha (TNF-α) | Markedly elevated 44. | Decreased by 54% 744. | No significant change 7. |
| Mitochondrial Fatty Acid Oxidation (MFO) | Suppressed, reliant on glucose 37. | Increased by 78%, restoring metabolic flexibility 37. | No significant change 3. |
| Insulin Resistance (HOMA-IR) | Over 4x higher than young adults 7. | Decreased by 64% 734. | No significant change 7. |
| Physical Function (Gait Speed / Strength) | Lower baseline speed and grip strength 3. | Significant increases in gait speed and 6-minute walk distance 3. | Continual gradual decline 3. |
Molecular analyses from this trial indicated improvements in seven of the nine recognized aging hallmarks 123. Notably, GlyNAC supplementation resulted in the significant upregulation of mitophagy and Pax7 expression, alongside the downregulation of 8-OHdG, p16INK4α, and muscle POT1 expression 3. These specific protein expression shifts indicate direct mechanistic impacts on the loss of proteostasis, stem-cell exhaustion, genomic damage, cellular senescence, and telomere attrition 3.
Postprandial Oxidative Stress Protection
Further sub-analyses of the randomized cohort, published in Innovation in Aging, explored the metabolic resilience of older adults during acute dietary stress. When subjected to a carbohydrate-rich glucose meal, older adults normally experience a severe, prolonged spike in oxidative stress and inflammatory markers compared to young adults 3536. Specifically, two hours after a glucose challenge, older adults exhibited drastically higher percentage increases in both TBARS and IL-6 36.
The 16-week continuous GlyNAC supplementation effectively protected the older adults against these acute glucose-driven inflammatory and oxidative spikes. Following the intervention, the postprandial inflammatory response in the older cohort was maintained at levels comparable to the younger, highly resilient cohort, suggesting that GlyNAC confers robust protection against the daily cellular damage induced by standard carbohydrate ingestion 3536.
Independent Verification and Demand-Dependent Efficacy
While the trials utilizing 16-to-24-week interventions on older populations yielded robust results, an independent randomized, controlled clinical trial conducted by Lizzo et al. (2022) provided crucial nuance to the efficacy profile and pharmacokinetics of GlyNAC.
Lizzo et al. evaluated 114 generally healthy older adults (mean age 65 years) utilizing three different fixed daily dose regimens (2.4 g, 4.8 g, and 7.2 g daily, divided in a 1:1 ratio) over a shorter 14-day observation period 263448. The primary predefined endpoint - the broad systemic increase of total whole-blood glutathione (GSH-T) across the entire cohort - was not statistically met 2634.
However, rigorous post-hoc analysis revealed a distinct biological divergence within the subject pool. A specific subset of the older adults characterized by high baseline oxidative stress (measured as being above the median for malondialdehyde) and low baseline glutathione levels (below the median) did demonstrate a highly significant increase in glutathione generation when exposed to the medium (4.8 g) and high (7.2 g) daily doses 263334. Conversely, the low dose (2.4 g) failed to elicit a consistent benefit even in the deficient subjects 33. Pharmacokinetic tracking showed that oral intake of GlyNAC at the active doses led to efficient, dose-dependent absorption of glycine and oxidized cysteine into the blood plasma within 60 minutes of ingestion 26.
This independent data establishes a vital biological reality regarding precursor supplementation: GlyNAC does not indiscriminately force the hyper-accumulation of glutathione in healthy populations. Rather, the intervention operates on a physiological demand basis 333437. It supplies rate-limiting substrates that the body only actively utilizes to synthesize glutathione when systemic demand is elevated - namely, when oxidative stress is high and endogenous antioxidant reserves are depleted 3337. Consequently, the clinical benefits of GlyNAC are most pronounced in older adults or populations burdened with chronic metabolic distress, and may provide limited physiological change in highly active, healthy adults with intact endogenous antioxidant capabilities 3337.
Investigations in Cognitive Decline and Alzheimer Disease
Given the established mechanisms of restoring the brain glucose steal and improving spatial memory in murine models, clinical attention has pivoted toward neurodegenerative disorders. Alzheimer's disease (AD) and mild cognitive impairment (MCI) are characterized by profound energy failure in the brain, neuroinflammation, and extreme oxidative stress - all targets successfully modulated by GlyNAC in preclinical settings 3550.
Current clinical efforts include early-phase trials specifically designed to evaluate GlyNAC's therapeutic potential in neurodegenerative populations. An active Phase 1 trial at Baylor College of Medicine is testing the intervention in individuals aged 55 to 85 with confirmed brain changes related to Alzheimer's disease 50. The goal is to determine if mitigating oxidative damage and enhancing cellular energy production can effectively slow or reverse the clinical progression of memory degradation 3550.
Safety Profile and Theoretical Long-Term Risks
In human clinical trials published to date, the combination of glycine and N-acetylcysteine has exhibited a strong safety and tolerability profile, though long-term longitudinal data remains a notable gap in the literature.
The robust clinical benefits noted in reversing aging hallmarks have been achieved using significant, weight-based dosing equivalent to 100 mg/kg of glycine and 100 mg/kg of NAC per day 3. For an average 70 kg adult, this equates to approximately 14 grams of total daily amino acid supplementation. Fixed-dose trials similarly indicate that higher doses (4.8 to 7.2 grams total per day) are required to move the biological needle in deficient individuals 263334.
Short-term clinical trials lasting up to 24 weeks have reported no serious adverse events linked to the supplementation 12633. The most frequently noted side effects are mild gastrointestinal disturbances, including nausea, bloating, and diarrhea, which are common pharmacological responses to high-dose oral amino acid intake 3338. Standard clinical guidelines suggest titrating the dose gradually over several weeks to mitigate these digestive issues 33. Crucially, markers of liver and kidney function have remained stable across all studied human cohorts 7.
Despite this promising short-to-medium-term safety data, critical evidence gaps and theoretical risks remain regarding prolonged, multi-year use. * Metabolic Animal Models: Extrapolations from select animal models warrant cautious monitoring. While healthy animals exhibit longevity benefits, specific metabolic animal models (e.g., obese or severely insulin-resistant mice) subjected to chronic, massive doses of isolated glycine have demonstrated worsening glucose intolerance driven by enhanced hepatic gluconeogenesis 38. * Neurological Morphology: Some animal studies tracking chronic high-dose glycine administration over 5 months have observed transient brain morphological changes, such as astrocyte hypertrophy, though overt neurotoxicity was not recorded 38. * Antioxidant Over-Suppression: While NAC possesses a well-established acute safety profile, theoretical concerns exist regarding the long-term, high-dose use of isolated antioxidants 733. Reactive oxygen species act as essential physiological signaling molecules in specific acute scenarios (such as exercise adaptation and immune response). Excessive, continuous suppression of ROS via heavy antioxidant loads may theoretically blunt beneficial physiological adaptations 4852. * Off-Target Proliferation: Furthermore, some murine studies have raised questions regarding off-target cellular proliferation and cancer promotion under constant, massive antioxidant supplementation, prompting calls for long-term safety validation 4852.
Until longitudinal human safety data exceeding 24 weeks is established, GlyNAC supplementation remains an experimental, yet highly evidence-backed nutritional approach to expanding human healthspan. By addressing the molecular root causes of biological aging - specifically glutathione depletion and mitochondrial dysfunction - the precise combination of glycine and N-acetylcysteine offers a potent, mechanistic intervention against the biological deterioration of age.