Gene Therapy for Biological Aging
Biological Foundations of Aging and Geroscience
Biological aging is defined as a complex, progressive deterioration of cellular function and physiological integrity, representing the primary risk factor for the majority of chronic non-communicable diseases, including neurodegeneration, cardiovascular disease, and metabolic syndromes 12. The scientific framework for understanding this physiological decline relies on a categorized set of biological mechanisms known as the hallmarks of aging. Originally formalized in 2013 as nine distinct biological hallmarks, the consensus paradigm was expanded and reorganized in 2023 to encompass twelve interconnected mechanisms that drive the aging process 34.
The contemporary model of biological aging categorizes these twelve hallmarks into three functional tiers: primary, antagonistic, and integrative 15. Primary hallmarks reflect the root causes of cellular damage and include genomic instability, telomere attrition, epigenetic alterations, and a loss of proteostasis 15. As cellular damage accumulates, antagonistic hallmarks emerge as compensatory responses that initially serve protective roles but eventually become deleterious when chronically activated 1. These include deregulated nutrient sensing, mitochondrial dysfunction, and cellular senescence 15. Finally, the integrative hallmarks manifest the systemic physiological consequences of the prior tiers, encompassing stem cell exhaustion, altered intercellular communication, disabled macroautophagy, chronic inflammation, and dysbiosis 45. Disabled macroautophagy was distinctly separated from the loss of proteostasis in 2023, acknowledging that autophagic processes target not only proteins but entire organelles and non-proteinaceous macromolecules 5.
| Tier Classification | Hallmark of Aging | Biological Mechanism | Clinical Implication |
|---|---|---|---|
| Primary | Genomic Instability | Accumulation of DNA damage over time. | Increased mutation rates and cancer vulnerability. |
| Primary | Telomere Attrition | Shortening of chromosomal protective caps. | Replicative arrest and tissue degradation. |
| Primary | Epigenetic Alterations | Dysregulation of gene expression and transcription. | Loss of specific cellular identity and function. |
| Primary | Loss of Proteostasis | Failure of protein folding and clearance mechanisms. | Toxic protein aggregation (e.g., Alzheimer's). |
| Antagonistic | Deregulated Nutrient Sensing | Overactivation of mTOR and insulin/IGF-1 signaling. | Metabolic diseases, obesity, and type 2 diabetes. |
| Antagonistic | Mitochondrial Dysfunction | Decline in cellular bioenergetics and ATP production. | Systemic energy deficits and neurodegeneration. |
| Antagonistic | Cellular Senescence | Permanent cell cycle arrest and SASP secretion. | Chronic tissue inflammation and functional decay. |
| Integrative | Stem Cell Exhaustion | Depletion of regenerative progenitor cell pools. | Impaired tissue repair and regenerative capacity. |
| Integrative | Altered Intercellular Communication | Disrupted endocrine and paracrine signaling networks. | Systemic physiological discoordination. |
| Integrative | Disabled Macroautophagy | Failure to recycle damaged organelles and cellular waste. | Intracellular toxicity and cellular death. |
| Integrative | Chronic Inflammation | Persistent, low-grade systemic immune activation. | Exacerbation of virtually all age-related morbidities. |
| Integrative | Dysbiosis | Disruption of the microbiome composition and function. | Impaired nutrient absorption and immune dysregulation. |
The interdependence of these mechanisms implies that targeted attenuation of a single hallmark frequently induces secondary modifications across the systemic network 5. Historically, research in the biology of aging relied heavily on reductionist methodologies, focusing on single-gene mutations that significantly altered the lifespan in simple model organisms 66. However, extensive genomic sequencing and comparative biological analyses have demonstrated that the genetic determination of human aging is highly polygenic, driven by a vast array of genetic variants with small individual effect sizes localized within key interrelated metabolic networks 7.
Explaining functional changes associated with aging requires a multidisciplinary systems biology approach, utilizing high-throughput single-cell sequencing, epigenomics, and multi-omic network mapping to analyze the complex, stochastic nature of biological aging 66. Computational models have identified conserved signaling pathways across species - such as the FoxO and mTOR signaling cascades - demonstrating that longevity is regulated by highly interconnected molecular hubs 7. Furthermore, recent transcriptomic analyses suggest that the vulnerability of specific genes to age-related decline is heavily influenced by physical gene length; long genes accumulate more stochastic DNA damage, leading to systemic decreases in long-gene expression across various tissues and contributing significantly to the aging phenotype 8. This systemic perspective dictates that isolated gene therapies must be evaluated not only for their direct target efficacy but also for their broad pleiotropic effects on cellular metabolism, tissue architecture, and overarching longevity networks.
Telomerase Modification and Cellular Senescence
The development of gene therapies targeting primary and antagonistic hallmarks has advanced through both formal clinical pathways and experimental regulatory environments. One of the most widely debated therapeutic targets in preliminary human and animal longevity trials is telomerase reverse transcriptase (TERT).
Mechanisms of Telomere Attrition and Repair
Telomeres are repetitive, non-coding DNA sequences located at the termini of chromosomes, forming a structural cap that protects the cell from sensing natural chromosome ends as severe DNA damage 2. Due to the end-replication problem of DNA polymerase, telomeres progressively shorten with each cellular division. Once telomeres reach a critically shortened length, the cell either enters apoptosis or transitions into a state of replicative arrest known as cellular senescence 2. Senescent cells accumulate in aging tissues, secreting a localized mixture of pro-inflammatory cytokines, proteases, and growth factors known as the senescence-associated secretory phenotype (SASP), which actively drives surrounding healthy cells toward dysfunction 9. Interventions aiming to express human telomerase reverse transcriptase (hTERT) seek to synthesize and elongate these chromosomal caps, thereby extending the cellular replicative lifespan and preventing senescence 211.
Clinical and Preclinical Controversies
The biotechnology company BioViva has historically championed hTERT gene therapies. In 2015, the company's CEO, Elizabeth Parrish, underwent offshore self-experimentation, receiving gene therapies designed to lengthen telomeres and increase muscle mass, positioning the company at the forefront of unregulated anti-aging interventions 101314. In 2021, BioViva announced preliminary results from a non-FDA-sanctioned trial involving five patients diagnosed with mild to moderate cognitive impairment 11. The study utilized viral vectors to deliver both hTERT and the Klotho gene - a gene encoding a protein associated with neuroprotection and the reduction of beta-amyloid plaques 211. The company reported that the median telomere length increased in four of the patients, and cognitive testing demonstrated significant improvements across the cohort 1113.
In 2022, BioViva, in collaboration with academic researchers from Rutgers University and Harvard Medical School geneticist George Church, published an animal study in the Proceedings of the National Academy of Sciences (PNAS) evaluating cytomegalovirus (CMV) vectors delivering TERT and follistatin genes 101112. The publication claimed profound results: an intranasal and injectable CMV vector delivering TERT extended the median lifespan of 18-month-old mice by 41.4%, while the follistatin vector yielded a 32.5% lifespan extension 12. The authors asserted that the treated cohorts exhibited improved blood glucose tolerance, mitigated hair loss, reduced sarcopenia, and reversed signs of mitochondrial dysfunction without an increased risk of cancer 12.
However, the validity of these claims became heavily contested shortly after publication. Independent scientific sleuths on the post-publication peer-review platform PubPeer, notably Dr. Elisabeth Bik and science writer Frank Swain, identified significant irregularities in the published data, including oversaturated western blots, differing black values, and the direct splicing and duplication of images across multiple figures 1011. Following these public allegations, the Rutgers University Office of Research Regulatory Affairs initiated an internal review 11. Despite the authors issuing two corrections in 2022 and 2023 attempting to provide new data that purportedly reaffirmed their experimental conclusions, Rutgers officially requested a retraction in August 2024 due to persistent data discrepancies in Figures 1 and 3 101113. PNAS formally retracted the paper, marking the first retraction in George Church's career; Church concurred with the retraction, citing inadequate backups of raw datasets, although BioViva's leadership contested the decision 1011. This incident highlights the critical necessity for rigorous, transparent validation when evaluating the efficacy of viral gene therapies aiming to modulate the fundamental hallmarks of aging.
Myostatin Inhibition and Sarcopenia
Separate from telomerase interventions, the targeted inhibition of specific muscle-degrading proteins has emerged as a distinct branch of longevity therapeutics. Follistatin (FST) acts as a natural inhibitor of myostatin and activin, proteins responsible for restricting muscle development and promoting tissue inflammation 181415. Animals naturally deficient in myostatin exhibit dramatic muscle hypertrophy, leading researchers to target this pathway to combat sarcopenia 16. Age-related muscle loss presents a significant morbidity risk in older populations, leading to frailty, metabolic dysfunction, increased incidence of falls, and loss of independence 2.
Follistatin Plasmid Gene Therapy
Plasmid-based follistatin gene therapy has been developed as an alternative to viral vectors, utilizing circular DNA to induce temporary, non-integrating gene expression 17. Minicircle, a biotechnology startup, initiated human trials using a follistatin plasmid (FST-344) complexed with polyethyleneimine (PEI) 17. Administered via a single subcutaneous injection into adipose tissue, the non-heritable therapy purportedly yields systemic follistatin expression lasting approximately one year 15.
Minicircle's preliminary data release on 43 adult human subjects reported a mean fat-free mass gain of 1.96 pounds and a body fat reduction of 0.87%, changes that were directionally conserved across varied age groups and sexes 1617. The intervention was generally well-tolerated; however, minor adverse events were observed, most notably a transient increase in low-density lipoprotein (LDL) cholesterol by approximately 8 mg/dL in roughly one-third of the cohort 151617. The researchers hypothesized that the anabolic requirement for lean mass gain necessitated localized cholesterol export from the liver, contributing to the mild lipid elevation 17. It is critical to note that these trials lacked double-blind, placebo-controlled protocols, making the results difficult to distinguish from standard lifestyle modifications or the placebo effect 16.
Regulatory Arbitrage in Special Economic Zones
To circumvent the protracted timelines and stringent requirements of the United States Food and Drug Administration (FDA), companies developing experimental longevity therapies frequently utilize regulatory arbitrage, operating within offshore Special Economic Zones (SEZs) 1819. Minicircle, along with other longevity-focused biotechs like Unlimited Bio, conducts its clinical operations primarily in Próspera, a Zone for Employment and Economic Development (ZEDE) situated on the island of Roatán in Honduras 1818.
Próspera operates under a customized regulatory framework designed to attract biomedical innovation. The jurisdiction enacted "Próspera Health Services Regulation A," a legal structure that explicitly permits the provision of experimental gene and plasmid therapies, regenerative medicine, and institutional review board-overseen medical research that has yet to receive FDA approval 202122. This regulatory flexibility allowed Unlimited Bio to rapidly incorporate and prepare a Phase I/II trial intended to study a combinatorial regimen of plasmid gene therapies targeting both VEGF (vascular endothelial growth factor, to stimulate capillary formation) and follistatin 1819. Unlimited Bio expects to dose participants in 2026, measuring efficacy through age-related biomarkers such as VO2 max and cognitive function tests, with the explicit goal of marketing the therapy directly to consumers immediately following preliminary trials 181819.
While these SEZs facilitate exceptionally rapid clinical iteration - allowing companies to move from incorporation to human dosing in a matter of months - the global regulatory consensus remains highly skeptical. Industry analysts and regulatory experts note that data gathered under these permissive frameworks is unlikely to be accepted by major agencies like the FDA or the European Medicines Agency (EMA) for formal drug approval 1818. The FDA typically demands rigorous, large-scale randomized controlled trials for broad, non-rare indications like aging, making the small-scale, healthy-volunteer data generated in Próspera insufficient for global market entry 1818. Consequently, these offshore ventures function primarily within the domain of medical tourism, offering unapproved biohacking interventions to self-funded patients 1828.
In Vivo Base Editing for Metabolic Aging Targets
While systemic anti-aging gene therapies target the overarching physiological clock, highly precise gene-editing technologies are being deployed against specific metabolic nodes that drive accelerated systemic aging, most notably cardiovascular disease.
Targeting PCSK9 in Cardiovascular Disease
Atherosclerotic cardiovascular disease (ASCVD) is the leading cause of premature mortality worldwide, heavily mediated by lifelong elevated levels of low-density lipoprotein cholesterol (LDL-C) 2324. The proprotein convertase subtilisin/kexin type 9 (PCSK9) gene acts as a critical negative regulator of hepatic LDL receptors; it degrades the receptors responsible for clearing LDL-C from the bloodstream 23. Gain-of-function mutations in PCSK9 cause familial hypercholesterolemia (FH), a genetic disorder characterized by dangerously high cholesterol levels from birth, while loss-of-function variants confer robust, lifelong protection against hypercholesterolemia and subsequent ASCVD events 2325.
To therapeutically mimic these protective loss-of-function variants, Verve Therapeutics developed VERVE-101, an investigational in vivo CRISPR base-editing therapy designed to permanently inactivate the PCSK9 gene in the liver 2627. Unlike traditional CRISPR-Cas9 systems that induce double-strand DNA breaks - which carry significant risks of unpredictable insertions, deletions, and chromosomal translocations - VERVE-101 utilizes an adenine base editor 27. Delivered via lipid nanoparticles (LNPs), the therapy guides the base editor to the specific PCSK9 sequence, where it catalyzes a highly precise single adenine-thymine (A-T) to guanine-cytosine (G-C) substitution 27. This specific edit introduces a nonsense mutation that halts PCSK9 protein production, permanently downregulating the protein after a single intravenous infusion 27.
Pharmacodynamics and Clinical Safety Halts
The Phase 1b Heart-1 trial for VERVE-101 enrolled adult patients living with heterozygous familial hypercholesterolemia (HeFH), severe, advanced ASCVD, and uncontrolled hypercholesterolemia despite maximal oral lipid-lowering therapy 2528. The open-label, single-ascending dose study evaluated four escalating dose cohorts: 0.1, 0.3, 0.45, and 0.6 mg/kg 2829.
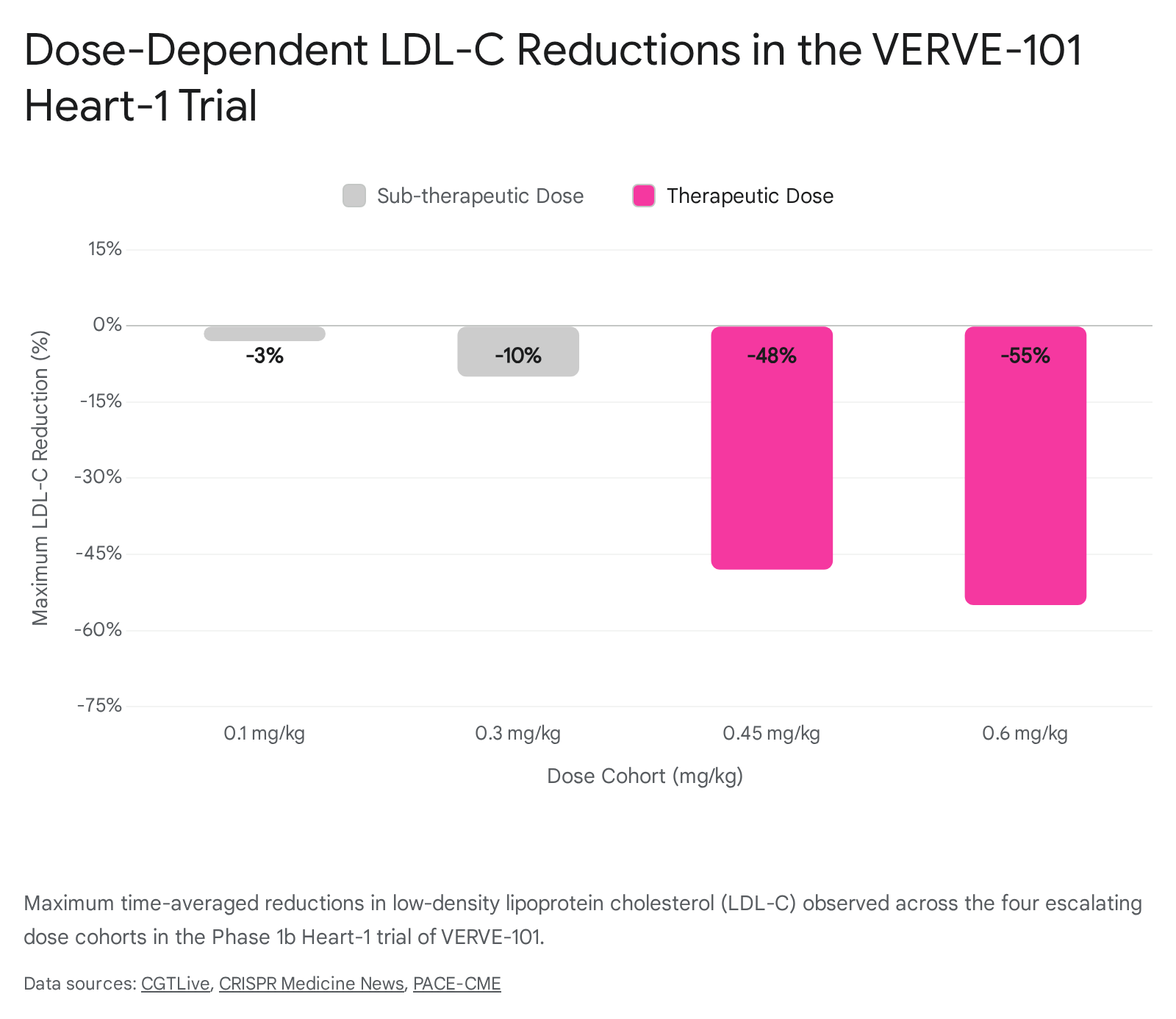
At sub-therapeutic doses (0.1 and 0.3 mg/kg), the therapy exhibited minimal clinical effect. However, at therapeutic doses, the pharmacodynamic response was unprecedented for an in vivo editing medicine. Patients in the 0.45 mg/kg cohort achieved time-averaged blood PCSK9 protein reductions of up to 84% and corresponding LDL-C reductions of up to 48% from baseline 2528. The single patient in the highest dose cohort (0.6 mg/kg) achieved a 55% reduction in LDL-C that proved durable for over 180 days post-infusion 252628.
Despite these groundbreaking efficacy markers, the trial encountered severe safety hurdles. In April 2024, Verve Therapeutics voluntarily paused enrollment following an adverse event in the sixth patient of the 0.45 mg/kg cohort 29. Within four days of dosing, the patient experienced a Grade 3 drug-induced transient elevation in alanine transaminase (ALT) alongside Grade 3 drug-induced thrombocytopenia 29. Although the patient remained asymptomatic and the laboratory abnormalities resolved within days, the independent data safety and monitoring board recommended a comprehensive investigation 29.
Next-Generation Iterations and Alternative Targets
Subsequent preclinical investigations utilizing a non-targeting guide RNA - designed to preclude any actual base editing - replicated the laboratory abnormalities in animal models, suggesting that the adverse events were driven primarily by the first-generation lipid nanoparticle delivery system rather than the CRISPR base editor mechanism 30.
Consequently, Verve reprioritized its clinical pipeline, shifting focus to the Heart-2 clinical trial for VERVE-102 252930. VERVE-102 utilizes the identical base editor and guide RNA for PCSK9 but is housed within a proprietary N-acetylgalactosamine (GalNAc)-LNP delivery system designed to allow for low-density lipoprotein receptor-independent delivery, effectively bypassing the inflammatory triggers of the original LNP 2529. Furthermore, the company initiated the Pulse-1 Phase 1b trial for VERVE-201, a distinct base-editing candidate targeting the ANGPTL3 gene to treat refractory hypercholesterolemia and homozygous familial hypercholesterolemia (HoFH) 30.
Epigenetic Reprogramming and Cellular Rejuvenation
The most profound recent paradigm shift in longevity gene therapy involves partial epigenetic reprogramming. This technique targets the epigenome - the biochemical markers (such as DNA methylation patterns) dictating gene expression - aiming to revert aged or damaged cells to a youthful transcriptional state without fundamentally altering the underlying DNA sequence 3132.
Partial Reprogramming via OSK Transcription Factors
Cellular reprogramming relies on the transient expression of specific transcription factors. The original Yamanaka factors - Oct4, Sox2, Klf4, and c-Myc (collectively OSKM) - are capable of driving mature somatic cells into a state of full pluripotency, generating induced pluripotent stem cells (iPSCs) 3340. However, continuous systemic expression of OSKM in living organisms is highly toxic, causing cellular dedifferentiation and resulting in the rapid formation of lethal teratomas.
To harness the rejuvenating potential of reprogramming while mitigating oncogenic risk, researchers developed partial reprogramming protocols. By transiently expressing only a subset of the factors - specifically Oct4, Sox2, and Klf4 (OSK), omitting the oncogene c-Myc - cells can shed age-associated epigenetic marks and restore youthful function without losing their established cellular identity 313334.
Preclinical studies validate the profound systemic benefits of this approach. In a landmark 2024 study, researchers demonstrated that systemically delivered adeno-associated viruses (AAVs) encoding an inducible OSK system in 124-week-old wild-type mice (an extremely advanced age) extended the median remaining lifespan by 109% compared to wild-type controls 3335. The partial reprogramming rejuvenated hepatic gene expression, reversed age-related metabolic decline, and improved cumulative physical frailty scores without inducing widespread tumorigenesis 3335. Similar interventions leveraging OSK mRNA delivered specifically to fibrotic livers have demonstrated the capacity to shift the hepatic microenvironment from a fibrotic to a regenerative state by downregulating fibrogenic mediators and halting extracellular matrix deposition 4034.
Clinical Translation in Optic Neuropathies
The first direct translation of partial epigenetic reprogramming into human clinical trials is being spearheaded by the biotechnology firm Life Biosciences with their lead candidate, ER-100 (AAV2-OSK) 3637. In January 2026, the FDA cleared the Investigational New Drug (IND) application for ER-100, marking a historic milestone as the first-ever cellular rejuvenation therapy utilizing epigenetic reprogramming to reach human trials 3145.
ER-100 aims to treat severe age-related optic neuropathies, specifically non-arteritic anterior ischemic optic neuropathy (NAION) and open-angle glaucoma (OAG) 3137. Both conditions precipitate severe, irreversible vision loss due to damage to the retinal ganglion cells (RGCs), the primary neurons responsible for transmitting visual signals from the eye to the brain 37. Preclinical data gathered from nonhuman primates demonstrated that intravitreal injections of ER-100 allowed for the safe, controlled expression of OSK, successfully restoring DNA methylation patterns to a youthful state and driving measurable improvements in visual function, verified at the cellular level by pattern electroretinograms (pERG) 3137.
The Phase I, first-in-human clinical trial (NCT07290244) utilizes a sequential cohort design. It begins with a dose-escalation phase in OAG patients, where independent Data Safety Monitoring Boards evaluate the 28-day safety profiles of sentinel participants before expanding to a selected dose cohort of NAION patients 3132. Given that retinal neurons are post-mitotic and the eye is highly compartmentalized, the ocular microenvironment provides an ideal, immunoprivileged testing ground for cellular rejuvenation prior to targeting highly proliferative, systemic organs 45.
| Clinical Trial Candidate | Developer | Primary Indication | Biological Target | Phase / Status |
|---|---|---|---|---|
| VERVE-101 | Verve Therapeutics | HeFH / ASCVD | PCSK9 Base Editing | Phase 1b (Heart-1); Enrollment paused due to LNP safety. |
| VERVE-102 | Verve Therapeutics | HeFH / ASCVD | PCSK9 Base Editing | Phase 1b (Heart-2); Enrolling, utilizes advanced GalNAc-LNP. |
| ER-100 | Life Biosciences | OAG / NAION | OSK Epigenetic Reprogramming | Phase I; FDA IND cleared January 2026. |
| FST-344 | Minicircle | Age-related Frailty | Follistatin Plasmid | Phase I (Offshore/Honduras); Completed initial cohorts. |
Synthetic Delivery Platforms for Genetic Medicines
The successful translation of genetic interventions into systemic anti-aging therapies is strictly contingent on the efficiency, safety, and organ-specificity of the delivery vehicle. The industry is currently executing a broad shift from immunogenic viral vectors toward chemically defined synthetic lipid platforms.
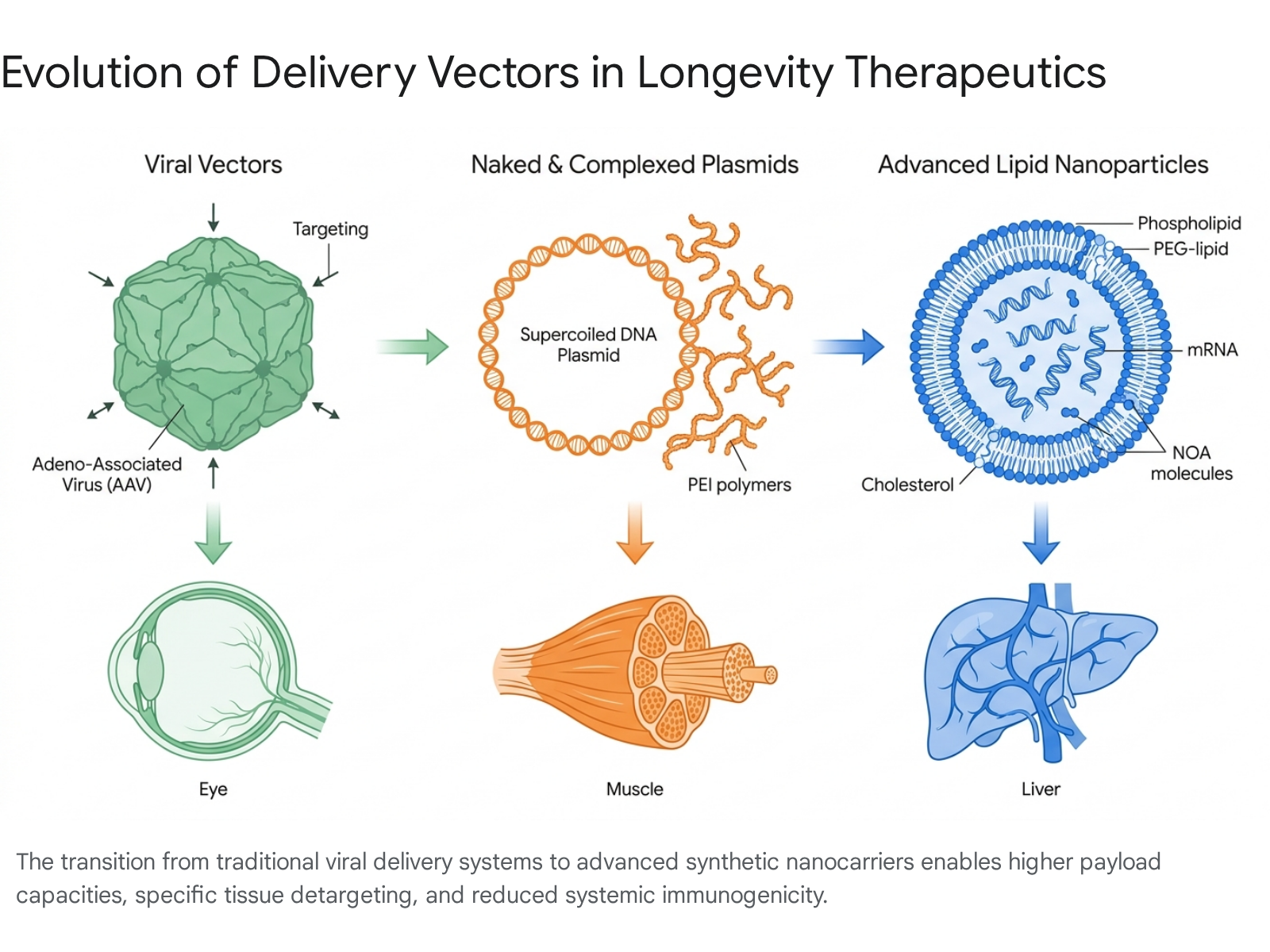
Limitations of Viral Vectors and Plasmids
Adeno-associated viruses (AAVs) remain the gold standard for localized gene therapy due to their low risk of insertional mutagenesis and high tropism for post-mitotic tissues, such as the eye and central nervous system 3738. However, AAVs possess strict payload limitations (roughly 4.7 kilobases), restricting the delivery of large multi-gene cassettes 39. Furthermore, systemic AAV administration faces severe hurdles regarding pre-existing patient immunity (neutralizing antibodies) and dose-dependent hepatotoxicity 39. While cytomegalovirus (CMV) vectors offer massive carrying capacities and historically low immunogenicity, their stability, manufacturing scale-up, and data reproducibility remain significant challenges within the field 1112.
Unpackaged plasmid DNA bypasses the immunogenic limitations of viral shells and allows for the transient expression of non-integrating episomal DNA. Formulations utilizing polyethyleneimine (PEI) to complex plasmids have demonstrated clinical tolerability 17. Nonetheless, bare plasmids and rudimentary polymer complexes often struggle with low in vivo transfection efficiencies, rapid degradation by nucleases, and an inability to penetrate specific deep-tissue targets.
Innovations in Lipid Nanoparticle Architecture
Lipid nanoparticles (LNPs), catapulted to global prominence via mRNA vaccines, offer unparalleled manufacturing scalability and modularity. However, their broad application in geroscience faces significant barriers: high acute toxicity upon systemic dosing, a strong default accumulation in the liver, and exceptionally low endosomal escape rates leading to payload degradation within the cell 40.
Recent biochemical engineering advancements address these specific bottlenecks: * Rapidly Hydrolyzing LNPs (RD-LNPs): Traditional LNPs persist in tissues for extended durations, contributing to sustained cellular toxicity. Researchers have developed acid-degradable lipids utilizing an 'azido-acetal' linker that rapidly hydrolyzes upon encountering the acidic environment of the endosome 40. RD-LNPs achieve rapid endosomal disruption within minutes, drastically improving the delivery of mRNA to the liver, lung, spleen, and brain while minimizing long-term tissue persistence 40. * Phospholipid-Free Target Specificity: To achieve selective hepatocyte targeting for partial cellular reprogramming in fibrotic livers, the H4T3_F6 LNP formulation was synthesized utilizing natural unsaturated fatty alcohol-based ionizable lipids 34. This simplified three-component, phospholipid-free LNP achieved mRNA delivery efficacy matching standard benchmarks while exhibiting enhanced selectivity for hepatocytes over immune cells. This targeted delivery allows OSK mRNA to reverse extracellular matrix deposition and fibrogenic signaling without triggering off-target systemic effects 4034. * Anti-Inflammatory pDNA-LNPs: Encapsulating plasmid DNA within LNPs historically triggers intense acute inflammation via the cGAS-STING innate immune pathway 41. By incorporating nitro-oleic acid (NOA) - an endogenous lipid that naturally inhibits STING - into the LNP architecture, NOA-pDNA-LNPs effectively subvert this inflammatory cascade 41. This specific chemical engineering ameliorates serious inflammatory responses in vivo, enabling safe, prolonged transgene expression at levels up to 11.5 times greater than standard mRNA-LNPs at day 32, offering a highly viable platform for durable longevity gene therapies 41. * GlycoLNPs for Liver Detargeting: Because LNPs naturally accumulate in the liver, treating systemic aging requires vectors that can specifically avoid hepatic clearance. Researchers have engineered GlycoLNPs by substituting neutral helper lipids with neutral glycolipids, achieving charge-independent liver detargeting and shifting delivery significantly toward the spleen and non-hepatic tissues 42.
| Delivery Vector System | Primary Payload Category | Innate Immunogenicity | Advancements & Tissue Tropism |
|---|---|---|---|
| Adeno-Associated Virus (AAV) | DNA (Small Cassettes) | High (Neutralizing antibodies) | High affinity for post-mitotic tissues (CNS, Retina). Used in ER-100. |
| Cytomegalovirus (CMV) | DNA (Large/Multi-Gene) | Moderate | Broad tropism, historically unstable in long-term data replication. |
| PEI-Complexed Plasmids | Episomal Plasmid DNA | Low | Localized intramuscular or subcutaneous delivery. Utilized in FST-344. |
| Lipid Nanoparticles (LNP) | mRNA / Base Editors | Moderate | Predominantly hepatic. Advanced GalNAc-LNPs bypass LDL receptors. |
| Rapidly Hydrolyzing LNPs | mRNA | Low (Due to rapid clearance) | Azido-acetal linkers enable systemic delivery across liver, lung, spleen, and brain. |
| NOA-pDNA-LNPs | Plasmid DNA | Low (Suppresses cGAS-STING) | Sustained expression of DNA via subversion of innate inflammatory cascades. |
Regulatory Frameworks and Clinical Trial Design
The path toward commercialization for therapies targeting the root biology of aging introduces unprecedented challenges for traditional regulatory bodies. The existing frameworks of the FDA and the EMA were fundamentally designed to evaluate interventions treating specific, universally recognized pathologies rather than underlying systemic biological decay 3951.
Adaptive Trial Protocols for Rare Indications
Current regulatory paradigms do not classify biological aging as an actionable clinical indication. Consequently, biotechnology firms developing longevity therapies must strategically position their clinical trials to target distinct age-related diseases with strictly defined endpoints 183951. Life Biosciences successfully bridged their epigenetic reprogramming technology into the clinic by targeting specific optic neuropathies (NAION and OAG) rather than applying for a generalized cellular rejuvenation trial 3137.
Furthermore, clinical trials for rare genetic indications often face insurmountable logistical barriers regarding small patient populations 52. To adapt, the FDA has issued guidance promoting innovative trial designs in the cell and gene therapy sector. Traditional randomized controlled trials are increasingly substituted with single-arm studies - where patients serve as their own controls - externally controlled patient data models, and adaptive Bayesian designs 52. The agency also encourages the use of natural history protocols to establish historical baselines for rare diseases, allowing for accelerated approval using novel surrogate endpoints (such as specific biomarker fluctuations or protein expression levels) with the caveat of extensive post-market confirmatory studies 3952.
Manufacturing and Post-Market Complexities
The FDA actively issues guidance regarding the specific Chemistry, Manufacturing, and Controls (CMC) considerations for therapies incorporating human genome editing. For highly precise interventions like CRISPR base editing, the agency demands exhaustive assessments of off-target editing outcomes and potential unintended consequences of on-target editing 43. Furthermore, regulatory bodies require voluntary consent to extensive long-term follow-up programs before trial enrollment, acknowledging that gene therapies may yield delayed phenotypic effects or late-onset immunogenicity long after the primary trial endpoints have been evaluated 5243. These stringent safety guardrails, while necessary for patient protection, substantially inflate the capital requirements and timelines necessary for clinical development 44.
Financial Landscapes and Institutional Investment
The capital requirements for gene therapy development are immense, driven by the complexities of longitudinal clinical trial execution, intensive toxicological screening, and Good Manufacturing Practice (GMP) requirements for nanoparticle and viral vector scale-up 44. Traditional venture capital often hesitates at the lengthy horizons inherent to aging biology. However, substantial philanthropic commitments and specialized funds are actively restructuring the financial landscape to support the transition of geroscience from academia to the clinic.
Philanthropic Capital and Global Funding Initiatives
The Hevolution Foundation, a massive nonprofit entity headquartered in Saudi Arabia, acts as a primary catalyst for global geroscience research 45. Armed with a reported mandate of deploying up to $1 billion annually into the healthspan sector, Hevolution operates largely beyond the immediate return-on-investment timelines of traditional pharmaceutical capital 45. The foundation heavily funds academic research and early-stage biotechnology firms exploring epigenetic reprogramming, cellular senescence, and novel delivery mechanics 4546.
In early 2024, Hevolution led a $50 million Series A financing extension - anchoring the round with a $20 million direct impact investment - into Aeovian Pharmaceuticals to advance their platform of selective mTORC1 inhibitors, targeting diseases of aging and severe neurological conditions 4547. In addition to direct equity investments in companies like Tune Therapeutics, which is developing epigenetic silencing drugs delivered via LNPs, Hevolution aggressively seeds the academic pipeline 4548. Through its Geroscience Research Opportunities (HF-GRO) and New Investigator awards, the foundation funds post-doctoral fellowships across North America, Europe, and Latin America, aiming to foster international scientific convergence on the biology of aging 49.
Late-Stage Venture Capital in Rejuvenation Biotechnology
Concurrently, late-stage venture capital is successfully identifying and scaling mature gene therapy targets. In early 2026, Life Biosciences closed a fully-subscribed $80 million Series D financing round to fuel the Phase I clinical trials of their ER-100 epigenetic reprogramming candidate in optic neuropathies 60. This funding is also earmarked to advance their broader Partial Epigenetic Reprogramming (PER) platform across additional systemic organ targets, aiming for comprehensive cellular rejuvenation 60. Such substantial institutional support indicates a rapidly maturing market where validated, targeted applications of longevity gene therapies are achieving the requisite clinical data and financial milestones necessary for broad integration into modern healthcare.