Future developments and speculation in longevity science
Introduction and Baseline Assessment
The trajectory of geroscience has historically been characterized by an acute asymmetry between preclinical success and human clinical validation. Over the past several decades, interventions that dramatically extended healthspan and absolute lifespan in yeast, nematodes, and murine models have frequently failed to replicate those systemic effects in human cohorts. However, the scientific and regulatory landscape of longevity medicine in 2026 represents a structural departure from previous eras. Driven by the advent of next-generation epigenetic biomarkers, the repurposing of highly efficacious metabolic drugs, and unprecedented capital deployment, the field is transitioning from exploratory biological research into a clinically actionable medical discipline 112.
Over the next decade (2026 - 2036), the longevity sector will undergo a distinct bifurcation regarding clinical adoption. Near-term clinical deliverables will rely heavily on the off-label and repurposed use of existing, patent-expired, or recently approved pharmacological agents - most notably glucagon-like peptide-1 (GLP-1) receptor agonists and mechanistic target of rapamycin (mTOR) inhibitors - guided by increasingly precise multi-omic aging clocks 3456. Conversely, the more radical interventions aimed at reversing the fundamental hallmarks of aging, specifically partial cellular reprogramming and targeted senolytics, remain highly speculative for systemic human use and are likely to be confined to localized, disease-specific applications within this ten-year horizon 87. This report provides an exhaustive analysis of the current state of longevity science, evaluating regulatory frameworks, diagnostic biomarker maturity, systemic pharmacological interventions, senotherapeutics, cellular reprogramming, and the overarching socio-economic forces shaping the field.
The Evolution of Regulatory Frameworks for Aging
A persistent bottleneck in the translation of longevity science into clinical practice is the regulatory framework governing drug approval and clinical trial design. The United States Food and Drug Administration (FDA) and the European Medicines Agency (EMA) traditionally operate on a "one disease, one drug" model 89. Because biological aging is not currently classified as a distinct pathological indication, pharmaceutical developers must adapt gerotherapeutics to fit within standard disease criteria 810.
The Disease Classification Debate
There is a growing divergence between global health organizations and regional regulatory bodies regarding the fundamental classification of aging. The World Health Organization (WHO), in its International Classification of Diseases (ICD-11), deliberately avoids labeling aging as a disease. The medical community opposed previous proposals to classify "senility" or "old age" as diagnostic categories, citing concerns that such labels would pathologize older adults, exacerbate ageism, and lead to the dismissal of severe underlying symptoms as natural consequences of aging 9. Consequently, the WHO utilizes the specific term "ageing-associated decline in intrinsic capacity," prioritizing the measurement of functional deterioration over a disease classification 9.
The FDA maintains the stance that aging is a natural biological process, requiring developers to target specific age-related diseases (e.g., osteoarthritis, macular degeneration, or cardiovascular disease) as proxies for the aging process 89. However, the landscape is experiencing a gradual shift toward composite clinical endpoints. The design of the Targeting Aging with Metformin (TAME) trial, approved by the FDA, serves as a vital proof-of-concept for the field 813. Rather than targeting a single pathology, TAME is designed to measure a composite outcome that includes the delay of cardiovascular events, cancer, dementia, and overarching mortality 811. If successful, TAME could compel regulatory agencies to define new marketing approvals and indications for gerotherapeutics, incentivizing further pharmaceutical investment 8.
Frailty Guidelines and the European Approach
The EMA has taken a pragmatic approach to geriatric medicine, prioritizing the baseline characterization of frailty in clinical trial populations. Recognizing that chronological age is a suboptimal predictor of drug susceptibility and adverse outcomes, the EMA's Committee for Medicinal Products for Human Use (CHMP) requested its Geriatric Expert Group to draft specific guidance on frailty 12. This supplements the International Council for Harmonisation (ICH) E7 guidelines on geriatric populations 1213.
The EMA now requires the use of validated instruments, such as the Short Physical Performance Battery (SPPB), to assess physical frailty at baseline in trial participants 1214. This cultural shift within European clinical trials, strongly supported by the European Organisation for Research and Treatment of Cancer (EORTC), aims to ensure that trial populations accurately reflect the physiological reality of older, multi-morbid patients 15. EMA Scientific Advisers have emphasized that precise descriptions of study populations are critical, as the benefit-risk balance of a gerotherapeutic or oncological drug shifts significantly depending on the patient's baseline frailty and intrinsic capacity 1215.
Qualification of Biomarkers and Surrogate Endpoints
For longevity drugs to reach the market efficiently, the field requires validated surrogate endpoints - biomarkers that reliably predict long-term clinical benefit so that trials do not have to wait decades to observe mortality data 1617. Regulators currently demand robust, longitudinal evidence that a surrogate endpoint truly predicts clinical benefit, generating an arduous barrier to entry for novel longevity compounds 1617.
The Biomarker Qualification Program
The FDA's Biomarker Qualification Program (BQP), formalized under the 21st Century Cures Act, was intended to streamline the use of novel biomarkers in drug development 18. However, a 2025 analysis revealed systemic challenges in the program's efficacy. While 61 biomarker projects were accepted into the BQP over its first eight years, only eight achieved full qualification, and critically, none of the qualified biomarkers were surrogate endpoints 18. Surrogate endpoint projects face significantly longer qualification timelines due to the complex validation required to prove that a biological signal reliably translates to meaningful patient health improvements 1618.
The EMA provides a similar pathway through its Qualification of Novel Methodologies for Medicine Development 1920. While the EMA has successfully qualified tools like the GFR slope as a validated surrogate endpoint for chronic kidney disease, broad systemic aging biomarkers have yet to receive formal surrogate endpoint qualification 1920. Consequently, developers of longevity therapeutics must still rely on traditional, lengthy clinical outcomes or attempt to use diagnostic biomarkers for trial enrichment rather than primary efficacy endpoints 1719.
Modalities of Biological Age Measurement
The ability to quantify biological aging independent of chronological time is the foundational technology enabling modern geroscience. Over the past decade, epigenetic clocks - algorithms analyzing genome-wide DNA methylation (DNAm) patterns - have evolved rapidly, separating into distinct generations based on their training parameters and predictive utility 212522.
The Progression of Epigenetic Clocks
First-generation epigenetic clocks, such as the Horvath and Hannum clocks, were trained exclusively to predict chronological age 2122. While mathematically impressive, their clinical utility in longevity medicine was limited. An optimal biomarker for geroscience must predict an individual's health trajectory, disease vulnerability, and mortality risk rather than simply calculating the number of years since birth 2123.
Next-generation (second and third-generation) models were engineered specifically to associate with health and lifestyle variables 21. Second-generation clocks, such as PhenoAge and GrimAge, incorporated health-related phenotypic data (e.g., white blood cell counts, smoking history) and mortality risk directly into their underlying algorithms 222425. Third-generation models, most notably DunedinPACE, represent a paradigm shift by measuring the pace or velocity of biological aging 222425. Trained entirely on longitudinal phenotypic changes across multiple organ systems over 20 years within the Dunedin birth cohort, DunedinPACE acts as a "speedometer" for aging rather than a static odometer 222526.
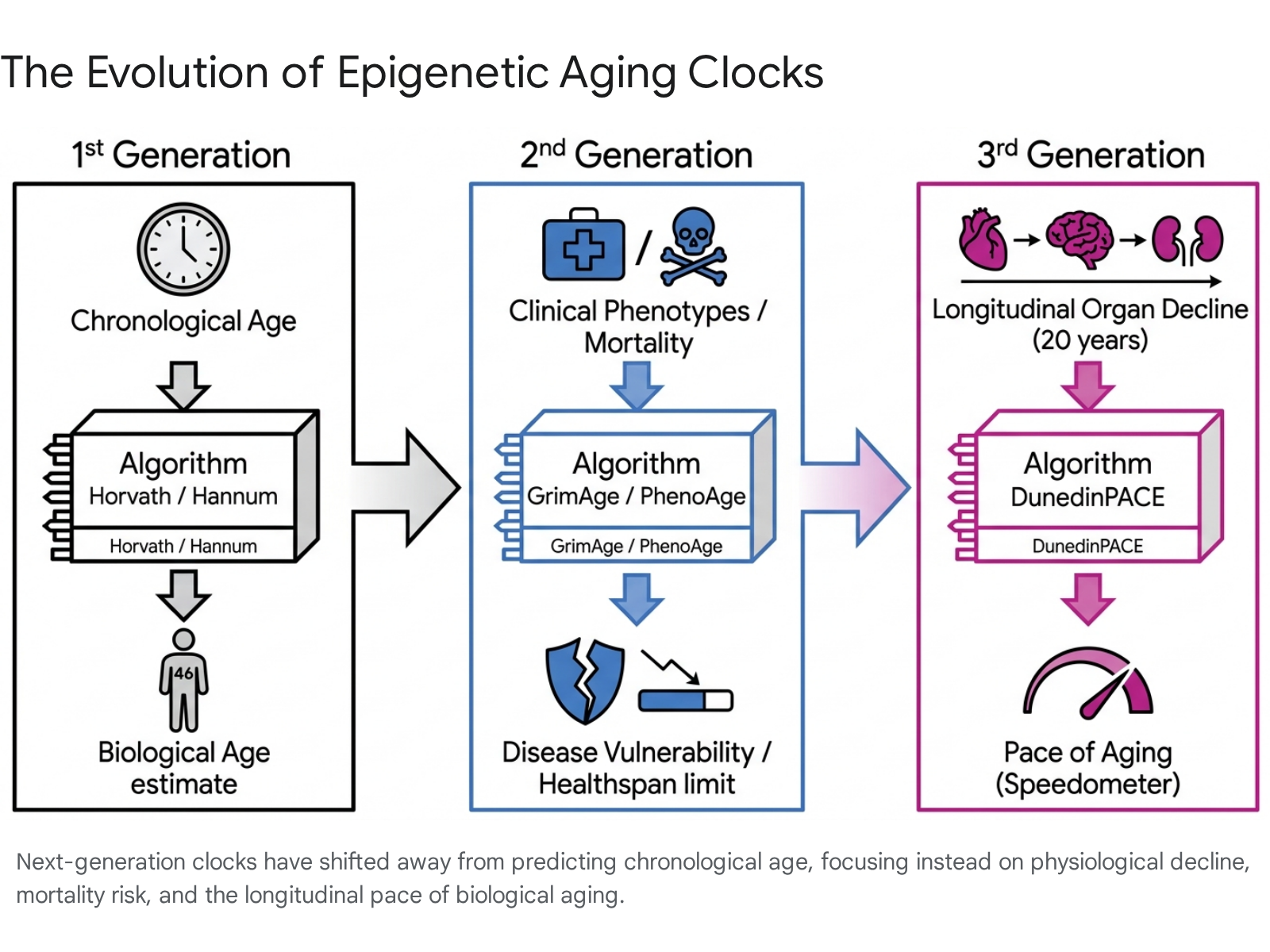
In clinical validation debates throughout 2025 and 2026, GrimAge and DunedinPACE established themselves as the industry standards 23252728. Large-scale statistical analyses conducted by the National Institute on Aging revealed that GrimAge consistently outperforms other clocks, as well as telomere length measurements, in predicting overall mortality 2627. Conversely, DunedinPACE has demonstrated exceptional sensitivity to short-term longitudinal interventions 2325. In the Berlin Aging Study II (BASE-II), which tracked over 1,000 individuals across 16-year intervals, DunedinPACE showed the strongest and most consistent associations with cognitive performance declines and increasing frailty indices, whereas first-generation clocks showed no clinically relevant signals 2528. For example, cognitive decliners in the cohort aged at a biological rate of 1.075 years per chronological year, indicating a distinctly accelerated pace of aging compared to healthy maintainers 28.
| Epigenetic Clock Generation | Prominent Algorithm Models | Primary Training Modality | Clinical Utility and Predictive Focus |
|---|---|---|---|
| First-Generation | Horvath (2013), Hannum (2013) | Chronological Age vs. DNA methylation | Low clinical utility; predicts chronological time rather than physiological health trajectory 2122. |
| Second-Generation | PhenoAge, GrimAge | Clinical Phenotypes, Blood Biomarkers, Mortality Risk | High; effectively predicts disease vulnerability, multi-morbidity risk, and lifespan limits 222526. |
| Third-Generation | DunedinPACE | Longitudinal multi-organ deterioration over 20 years | Very High; measures the dynamic rate (speed) of biological decline and is highly responsive to short-term lifestyle or pharmacological interventions 2325. |
Proteomic and Multi-Omic Integration
While epigenetic clocks dominate the commercial landscape, singular modalities are increasingly viewed as insufficient for a holistic assessment of organismal aging. By 2025, researchers introduced advanced proteomic aging clocks capable of analyzing the vast array of circulating proteins in blood plasma 2529. A landmark Nature Medicine study identified 204 key proteins that predict biological age and align closely with the incidence of eighteen major chronic diseases, including cardiovascular, renal, lung diseases, and neurodegeneration 29.
The advantage of proteomic clocks lies in their dynamic, highly responsive nature. Because proteins reflect the immediate, real-time physiological state of the body, these clocks react to therapeutic interventions over months, offering faster feedback loops than epigenetic alterations 229. As the field matures, the scientific consensus is shifting toward comprehensive multi-omic profiling, wherein epigenetic, proteomic, metabolomic, and microbiome data are aggregated to provide an organ-specific assessment of biological resilience 252224.
Evaluation of Systemic Pharmacological Interventions
The most immediate clinical deliverables in longevity science rely on pharmaceutical agents already approved for discrete pathological indications. The period between 2023 and 2026 yielded substantial human data on these repurposed drugs, clarifying their true geroprotective efficacy beyond foundational murine models.
The Paradigm Shift of GLP-1 Receptor Agonists
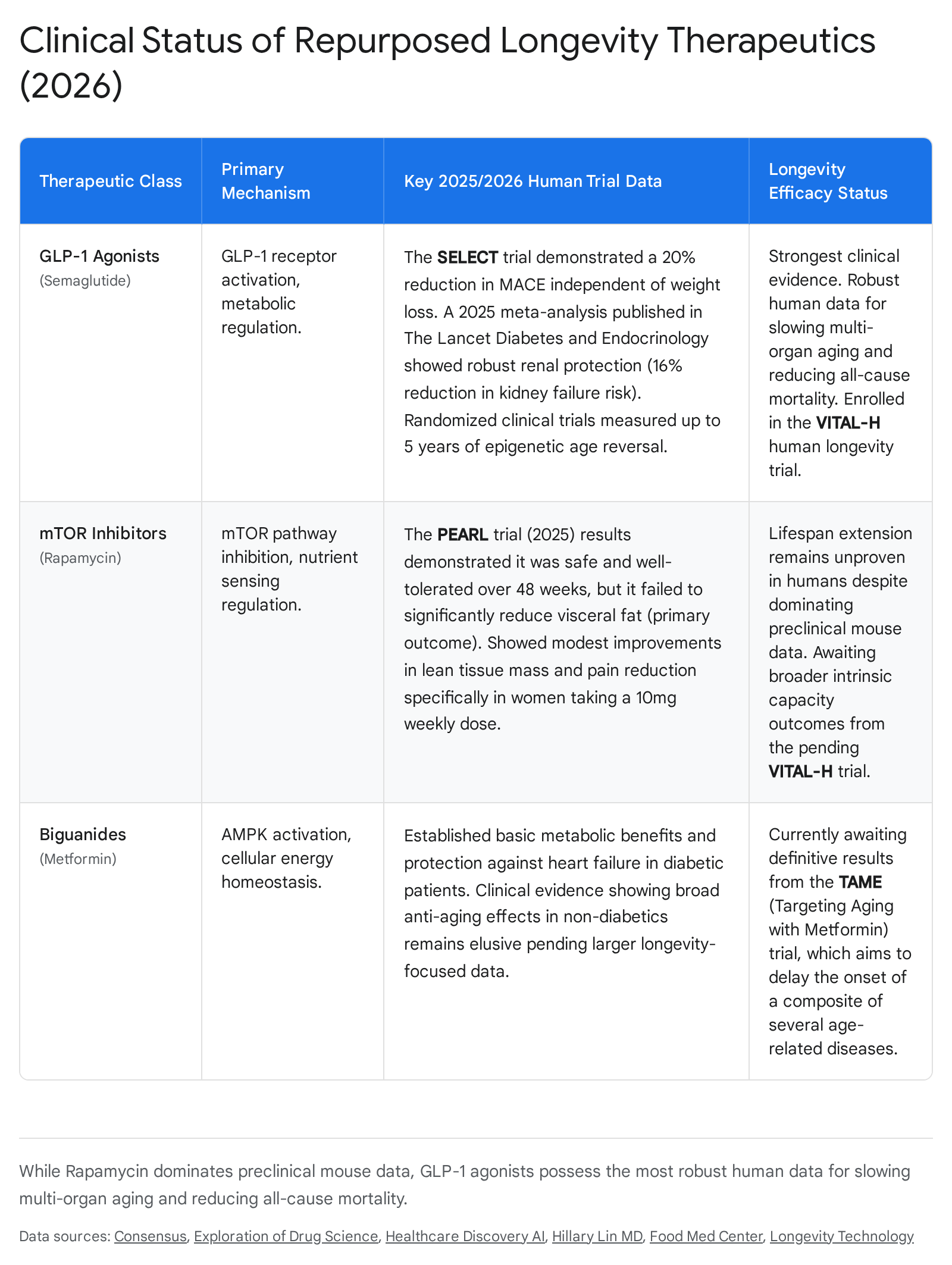
The most significant disruption to the longevity pipeline has been the integration of Glucagon-Like Peptide-1 (GLP-1) receptor agonists (e.g., semaglutide, tirzepatide, orforglipron) into geroscience frameworks. Initially commercialized exclusively for type 2 diabetes and obesity, a rapidly expanding body of clinical research indicates that these compounds exert systemic, multi-organ geroprotective effects that operate upstream of caloric balance 330.
A critical 2025 sub-analysis of the SELECT trial published in The Lancet demonstrated that semaglutide reduced major adverse cardiovascular events (MACE) by 20% and all-cause mortality by 19% (4.3% incidence versus 5.2% in the placebo group) 3. Crucially, this cardiovascular protection occurred irrespective of the degree of fat mass reduction 3. Furthermore, a 2025 meta-analysis in Lancet Diabetes and Endocrinology synthesizing renal evidence across GLP-1 trials revealed that these drugs reduced the risk of kidney failure by 16% and slowed the decline in glomerular filtration rate by 22%, representing a meaningful deceleration of renal aging in large randomized cohorts 3. GLP-1s are also proving effective against metabolic dysfunction-associated steatohepatitis (MASH), with Phase 3 trials underway and semaglutide securing FDA approval for the condition 331.
The molecular basis for these observations was corroborated by a landmark 32-week, double-blind, randomized, placebo-controlled phase 2b trial in 2025. This trial provided the first clinical evidence that semaglutide directly modulates epigenetic aging 3. Utilizing multiple DNA methylation clocks on patients with HIV-associated lipohypertrophy, the trial recorded extraordinary biological age reversals: a 3.1-year reduction on PCGrimAge, a 4.9-year reduction on PhenoAge, and a 9% slowing in the pace of aging via DunedinPACE 3. Consequently, leading institutions are now structuring trials to position GLP-1 agonists directly as longevity therapeutics rather than merely weight management tools 3.
Mechanistic Target of Rapamycin Inhibitors
Rapamycin (sirolimus), an mTOR inhibitor and immune-modulator, has long been considered the most consistently replicated lifespan-extending drug in mammalian laboratory history, reliably yielding 10% to 25% lifespan extensions across diverse murine strains 113233. Translating this mechanism to humans, however, requires navigating the delicate balance between nutrient sensing optimization and systemic immunosuppression.
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial, published in 2025, represents the first long-term (48-week), randomized, placebo-controlled trial evaluating low-dose, intermittent rapamycin (5 mg or 10 mg weekly) specifically for longevity in healthy adults 4323839. The clinical outcomes were highly nuanced. Rapamycin failed to achieve its primary endpoint of visceral fat reduction, showing an effect size of essentially zero 432. However, secondary outcomes revealed modest, sex-specific signals: women taking the 10 mg weekly dose experienced statistically significant improvements in lean tissue mass and pain reduction, alongside improved bone mineral density 43238.
Crucially, the PEARL trial confirmed that low-dose, intermittent rapamycin is safe and well-tolerated over a year in healthy populations, distinct from the severe side effects associated with daily, high-dose organ transplant protocols 43233. Nonetheless, as of 2026, there is no definitive human trial proving that rapamycin extends absolute human lifespan or broadly reverses biological aging 3233.
The VITAL-H Trial: Harmonizing Interventions and Endpoints
Recognizing the need for head-to-head comparisons, researchers launched the VITAL-H trial, backed by $38 million in federal funding 634. Enrolling up to 1,000 healthy adults aged 60 to 65 in South Texas, the study aims to test rapamycin, semaglutide, and dapagliflozin (an SGLT2 inhibitor) against a placebo 634. Guided by principal investigator Dr. Elena Volpi, the trial's primary endpoint is "intrinsic capacity" - a comprehensive metric encompassing physical resilience and mental functionality 634. The explicit goal of VITAL-H is to force the FDA to recognize intrinsic capacity as a meaningful, regulatory-grade endpoint, marking a watershed moment in testing longevity drugs under rigorous paradigms 6.
In parallel, combination therapies are being explored to address drug cessation challenges. The TrialX NCT07092618 trial is actively recruiting patients discontinuing GLP-1 therapies, evaluating whether the administration of metformin alone, or combined with rapamycin and low-dose naltrexone (LDN), can sustainably maintain metabolic health and prevent weight regain 3035.
The Status of Nutritional Modulators and NAD Precursors
The narrative surrounding over-the-counter dietary supplements in longevity science is increasingly characterized by stringent scientific scrutiny and tempered expectations, shifting focus away from early speculative compounds.
The Decline of Resveratrol
Resveratrol, a polyphenol found in red grapes, was once hailed as a revolutionary molecule following early 2000s studies suggesting it extended lifespan by activating SIRT1 pathways 363744. By 2026, it has been largely abandoned by leading geroscience clinicians 36373846. The Interventions Testing Program (ITP) - the gold standard for rigorous preclinical lifespan testing - demonstrated unequivocally that resveratrol has no effect on the lifespan of male or female mice when administered at any stage of life 3637.
Retrospective analyses revealed that early positive murine studies were an artifact; mice were fed an extremely high-fat, life-threatening diet consisting of 60% fat, primarily coconut oil 36. Resveratrol mitigated these severe metabolic conditions but failed to extend the maximum lifespan of healthy animals 36. Furthermore, over 150 human clinical trials have yielded neutral or conflicting results, plagued by the compound's exceedingly low systemic bioavailability and failure to directly stimulate sirtuin activity in robust human models 3738.
Nicotinamide Mononucleotide and the Regulatory Environment
Nicotinamide Adenine Dinucleotide (NAD+) precursors, specifically Nicotinamide Mononucleotide (NMN) and Nicotinamide Riboside (NR), remain heavily researched due to the established age-related decline of NAD+ metabolism 39404142. Clinical trials consistently confirm that oral NR and NMN supplementation can safely and reliably elevate circulating whole-blood NAD+ levels by approximately 130% to 150% in healthy middle-aged and older adults over short durations 394051.
Despite this proof-of-biology, translating elevated NAD+ levels into tangible physiological improvements has proven difficult. Recent meta-analyses of human trials reveal minimal to no significant improvements in lipid profiles, glucose control, cognitive function, or muscle performance 394041. Evidence suggests that rather than direct cellular absorption, gut bacteria may convert NMN and NR into nicotinic acid (NA) prior to raising circulating NAD+ levels, complicating the pharmacodynamics of these supplements 4151. Next-generation precursors, such as trigonelline, are currently under investigation for superior serum stability and muscle targeting capabilities 42.
The regulatory status of NMN has also experienced significant volatility. In late 2022, the FDA issued determinations to specific suppliers, such as SyncoZymes and Inner Mongolia Kingdomway, that NMN was excluded from the definition of a dietary supplement due to its prior investigation as a pharmaceutical drug by MetroBiotech 435344. This triggered widespread market removals. However, following extensive litigation and a citizen petition led by the Natural Products Association (NPA), the FDA formally reversed its stance in late 2025 435345. Under the Dietary Supplement Health and Education Act (DSHEA) "race to market" provision, the FDA acknowledged sufficient evidence that NMN was marketed as a supplement in the U.S. prior to its authorization for drug investigation, rendering it lawful for continued sale 444546.
The Narrowing Translation of Senolytic Therapies
Senescent cells are damaged cells that cease dividing but resist apoptosis, accumulating in tissues over time. They secrete a highly active cocktail of inflammatory cytokines, chemokines, and proteases known as the senescence-associated secretory phenotype (SASP), which actively drives tissue dysfunction 5747. The premise of senolytics - drugs that selectively induce apoptosis in these accumulating cells - is biologically elegant, but clinical translation has encountered substantial friction 747.
First-Generation Senolytics in Clinical Settings
The academic foundation of the senolytics pipeline relies heavily on first-generation, repurposed compounds, primarily the combination of dasatinib (a tyrosine kinase inhibitor used in leukemia) and quercetin (a plant flavonoid), collectively referred to as D+Q 71148.
In early-phase trials, D+Q demonstrated vital proof-of-biology. A 2023 phase 1 feasibility trial in patients with early symptomatic Alzheimer's disease showed that intermittent D+Q administration achieved successful central nervous system penetration and favorable tolerability 7. However, larger trials targeting systemic aging have yielded mixed results. A 2024 phase 2 randomized trial of D+Q in postmenopausal women failed to reduce the primary bone-resorption marker overall, though positive signals in bone-formation markers were observed specifically in patients with the highest baseline senescence burden 7. Similarly, a randomized pilot trial in idiopathic pulmonary fibrosis (IPF) found D+Q to be safe but failed to produce meaningful improvements in pulmonary or physical-function measures compared to a placebo 7.
Ongoing studies, such as the NCI-funded SEN-SURVIVORS Phase II trial (NCI-2021-13203), are testing D+Q and fisetin to reduce frailty in adult survivors of childhood cancer, a population highly susceptible to accelerated senescence 49. Furthermore, Mayo Clinic researchers are investigating a sequential protocol of dasatinib, quercetin, fisetin, and temozolomide targeting residual disease in glioma patients (NCT07025226), attempting to leverage senolytics against the inflammatory microenvironment of tumors 5051.
The Pivot Toward Localized Administration
Despite ongoing research, the consensus in 2026 is that sweeping systemic application of senolytics carries an unfavorable potency-safety tradeoff. Broad systemic clearance of senescent cells can impair wound healing and trigger unanticipated toxicities, while resistance mechanisms developed by senescent tissues complicate monotherapy approaches 747.
Consequently, the senolytics pipeline is narrowing into a highly selective, localized, and biomarker-driven discipline 7. The strongest clinical prospects currently involve targeted delivery to specific tissues where the senescence burden is clear and systemic exposure is minimized. UNITY Biotechnology's UBX1325, a senolytic BCL-xL inhibitor delivered via localized injection directly into the eye for diabetic macular edema, exemplifies this approach 7. Similarly, the dermatology sector is advancing topically applied senolytics, such as Rubedo Life Sciences' RLS-1496, which entered phase 1 trials in late 2025 for inflammatory skin conditions and actinic keratosis 7. Immorta Bio's SenoVax, an investigational senolytic immunotherapy, further highlights the pivot toward precise, immunology-driven clearance mechanisms in oncology 52.
Partial Cellular Reprogramming and Epigenetic Rejuvenation
While pharmacological agents and senolytics operate downstream to mitigate damage, partial cellular reprogramming represents a direct intervention to reverse the core epigenetic architecture of aging. Derived from Shinya Yamanaka's 2006 discovery that four transcription factors (Oct4, Sox2, Klf4, and c-Myc, or OSKM) can convert adult somatic cells into induced pluripotent stem cells (iPSCs), geroscience researchers are attempting to apply these factors transiently in vivo 646553. The objective is to reset the epigenome to a youthful transcriptional state without causing the cells to completely de-differentiate, lose their functional identity, or form teratomas 646554.
Mesenchymal Drift and Alternative Payloads
The fundamental biological risk of cellular reprogramming is identity loss. A comprehensive 2025 study led by researchers at Altos Labs mapped how cellular aging drives "mesenchymal drift," a process where differentiated cells gradually lose their strict functional identity and drift toward chaotic, scar-forming phenotypes 54. While transient OSKM expression can reverse this drift, uncalibrated activation erases cell memory entirely, causing catastrophic tissue disintegration and oncogenesis 5354.
To circumvent these risks, the industry is aggressively moving away from the standard Yamanaka cocktail. By 2026, entities such as NewLimit are utilizing advanced AI architectures to screen thousands of non-Yamanaka transcription factor payloads 8. In early 2026, NewLimit announced the discovery of payloads capable of rescuing regenerative function and reversing multiple youthful features in old human hepatocytes (liver cells) while strictly preserving cell type 855.
Translational Timelines and Corporate Pipelines
The commercial landscape of cellular reprogramming is highly capitalized, with over $4 billion invested across top companies, yet it remains clinically nascent 870. Each major entity is pursuing distinct delivery and payload strategies to navigate regulatory constraints 8707172.
| Biotechnology Company | Primary Technology Platform | Target Delivery Vector and Indication | 2026 Clinical Development Status |
|---|---|---|---|
| Altos Labs | Comprehensive cellular rejuvenation programming (Epigenetic resetting) | Systemic and highly varied pre-clinical models | Preclinical. Achieved robust in vivo partial reprogramming reversal without oncogenesis in animal models (2025), but no disclosed human trial registry 8547071. |
| Life Biosciences | OSK factors (excluding oncogene MYC) | AAV gene therapy delivered via intravitreal injection to the eye | Phase 1 Human Dosing (Q1 2026). Target indications include NAION and Glaucoma 87172. |
| NewLimit | AI-discovered novel transcription factors | Lipid Nanoparticle (LNP)-mRNA targeting liver tissues | Preclinical. Scaling manufacturing for candidate payloads; targeting alcohol-related liver disease 8557256. |
| Retro Biosciences | OSKM and alternative payloads | Ex-vivo T-cell reprogramming | Preclinical (Reprogramming). While operating an active autophagy trial (RTR242), their reprogramming track targets human trials by late 2026 8717256. |
| Turn Biotechnologies | ERATM Platform (mRNA delivery) | mRNA delivery targeting Dermatology and Osteoarthritis | IND preparation underway. Focus on transient, non-permanent dosing similar to therapeutic vaccines 72. |
The timeline for widespread, systemic cellular reprogramming in humans extends significantly beyond the next decade. Current clinical strategies strictly limit exposure to localized, highly controlled environments - such as the immune-privileged eye, ex-vivo blood cells, or highly targeted liver delivery - to mitigate oncogenic risk and appease regulatory safety standards 872.
The Socio-Economic Architecture of the Longevity Industry
The transition of geroscience from theoretical biology to clinical reality carries profound global socio-economic implications. The rapidly aging global population, combined with skyrocketing chronic disease management costs, renders the traditional reactive healthcare model financially unsustainable 55758.
Capital Allocation and Transnational Policy
The longevity sector is experiencing a structural shift in capital allocation, moving beyond consumer wellness and speculative venture capital into institutionally funded healthcare solutions 171. While total biotech venture capital faced broader market contractions, funding for dedicated healthspan science has surged, reaching an estimated $75 billion globally by 2024 - 2025 59.
The Hevolution Foundation has emerged as a dominant transnational force in this ecosystem. Headquartered in Riyadh, the non-profit foundation had deployed over $400 million in grant funding and direct investments to more than 250 scientists globally by early 2025 5960. Hevolution's explicit mandate is to expand "healthspan" - the period of life spent in functional independence - rather than merely extending absolute chronological lifespan 59. Through initiatives like the Global Healthspan Summit (GHS) and the Impetus Grants program (which features rapid three-week funding decisions for bold aging biology projects), philanthropic capital is systematically derisking early-stage basic science that traditional pharmaceutical companies avoid 59616263.
At the 2025 Global Healthspan Summit, regulatory alignment was a central theme. The CEO of the Saudi Food and Drug Authority (SFDA), Dr. Hisham S. Aljadhey, emphasized that regulatory involvement must evolve beyond mere product approval to actively influence the entire healthspan lifecycle, encompassing disease prevention, targeted nutrition, and early biotechnology adoption 6364.
Healthspan Extension as a Macroeconomic Imperative
A common critique of longevity medicine is the fear of "longevity inequality," wherein bespoke, highly expensive rejuvenation therapies are accessible only to the ultra-wealthy, exacerbating global health disparities 82. However, health economists suggest a more democratized trajectory 582.
The near-term pharmacological pipeline is heavily reliant on small molecules (e.g., metformin, generic rapamycin, and eventually off-patent GLP-1 agonists) that are extremely inexpensive to manufacture at scale 82. Furthermore, macroeconomic models indicate that delaying the biological onset of aging by just 2.2 years could generate global savings of $7 trillion over fifty years by compressing the morbidity window and reducing the enormous costs associated with frailty and dementia 582. The economic framework of Quality-Adjusted Life Years (QALYs) estimates that effective longevity interventions provide between 1.07 and 5.32 additional QALYs per individual 5. Because the market for aging interventions encompasses the entire human population, therapies will benefit from unprecedented economies of scale 82. This dynamic directly incentivizes governments, public health authorities, and insurance providers to subsidize gerotherapeutics, integrating biological age testing into routine clinical practice as a financially imperative preventative measure 11582.