Functions and Significance of the Default Mode Network
Paradigm Shift in Systems Neuroscience
For several decades, cognitive neuroscience operated under the assumption that the human brain was primarily a reactive organ, remaining at a relatively idle baseline until stimulated by external tasks, cognitive demands, or sensory input. This prevailing paradigm shifted fundamentally with the formal identification of the default mode network (DMN), a large-scale, distributed neural system that demonstrates highly synchronous, robust baseline activity during periods of wakeful rest 123. Initially identified through positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) as a consistent set of brain regions that systematically "deactivated" during externally oriented, attention-demanding cognitive tasks, the DMN is now understood to be anything but a passive physiological baseline 14.
The intrinsic activity of the DMN represents active, self-generated internal mentation. This internal cognitive processing encompasses a vast array of high-level psychological operations, including self-referential thought, autobiographical memory retrieval, mental time travel (encompassing both retrospective episodic memory and prospective episodic future thinking), and social cognition or "theory of mind" 134. The continuous, dynamic integration of these complex internal states allows individuals to maintain a cohesive autobiographical narrative and simulate potential future scenarios based on past experiences 13. Consequently, the DMN has emerged as a central focus in systems neuroscience, functioning as a critical neural hub that orchestrates the boundary between internal representational models and external reality.
Recent clinical interventions have also highlighted the plasticity of the DMN. Modulating DMN activity is increasingly viewed as a therapeutic target for improving cognitive clarity and emotional regulation. For example, mindfulness-based neurofeedback (mbNF) has been shown to reduce DMN overactivity by 30% to 40% and decrease specific hyperconnectivity - such as between the subgenual anterior cingulate cortex (sgACC) and the medial prefrontal cortex - by up to 35% 1. Such adaptations not only facilitate emotional regulation but also enhance the functional integration between large-scale networks, optimizing the brain's ability to transition between introspection and task-oriented cognition 1.
Anatomical Structure and Functional Subsystems
The DMN is not a single, monolithic anatomical structure. It is a heterogeneous and highly interconnected network comprising distinct cortical nodes distributed across the brain. The canonical core of the DMN includes the posterior cingulate cortex (PCC), the adjacent precuneus, the medial prefrontal cortex (mPFC), and the bilateral angular gyri (AG) located in the inferior parietal lobule 134.
Advances in resting-state functional connectivity, independent component analysis (ICA), and data-driven clustering algorithms have demonstrated that the overall DMN can be parsed into functionally specialized subnetworks. These subsystems operate in tandem to support distinct but overlapping cognitive domains 25. The macroscale architecture of the DMN is generally organized into three primary subsystems, each demonstrating distinct connectivity profiles and specialized functional output 356.
| Subsystem | Primary Anatomical Nodes | Cognitive and Functional Specialization |
|---|---|---|
| Core Subnetwork | Anterior medial prefrontal cortex (aMPFC), posterior cingulate cortex (PCC), bilateral angular gyri (AG). | Supports generalized self-referential processing, perceptually decoupled cognition, and acts as the integrative coordinating hub linking the other DMN subsystems 136. |
| Medial Temporal Subnetwork | Hippocampal formation, parahippocampal cortex, retrosplenial cortex (RSC), ventral posterior inferior parietal lobule. | Heavily implicated in memory-based constructive processes, including episodic memory retrieval, contextual scene construction, and spatial navigation 356. |
| Frontotemporal Subnetwork | Dorsomedial prefrontal cortex (dmPFC), lateral temporal cortex, temporal pole, temporoparietal junction (TPJ). | Primarily supports abstract, semantic, and social cognition, including mentalizing (theory of mind), language comprehension, and understanding the emotional states of others 16. |
The precise coordination among these subsystems enables fluid transitions between recalling a specific past event (mediated by the Medial Temporal subsystem), extracting its semantic or social meaning (mediated by the Frontotemporal subsystem), and integrating it into one's continuous sense of identity (mediated by the Core subsystem) 356.
Sender and Receiver Microarchitecture
While canonical resting-state parcellations define the DMN through broad functional co-activation, high-resolution microstructural and effective connectivity mapping has revealed a secondary, orthogonal organizational axis. This axis defines the directional flow of information into and out of the network. High-resolution directional functional connectivity analysis indicates that the DMN contains distinct "receiver" (afferent-biased) and "sender" (efferent-biased) zones interleaved within the same macro-regions, such as the PCC and mPFC 789.
Receiver-like subregions exhibit intrinsic connectivity biases toward heteromodal association cortices. These zones are preferentially engaged when individuals must integrate incoming external perceptual inputs with internal knowledge structures, such as making a socially grounded evaluation of a perceived face 7810. In contrast, sender-like subregions exhibit stronger efferent projections to distributed sensorimotor and attentional systems 7910. These efferent zones are preferentially recruited during memory-guided decisions that rely entirely on decoupled, internally generated representations without concurrent perceptual input 710. This microarchitectural double dissociation provides a mechanistic explanation for how the DMN manages its immense versatility, allowing it to support both internally generated memory retrieval and externally oriented social and semantic comprehension without cross-modal interference 7911.
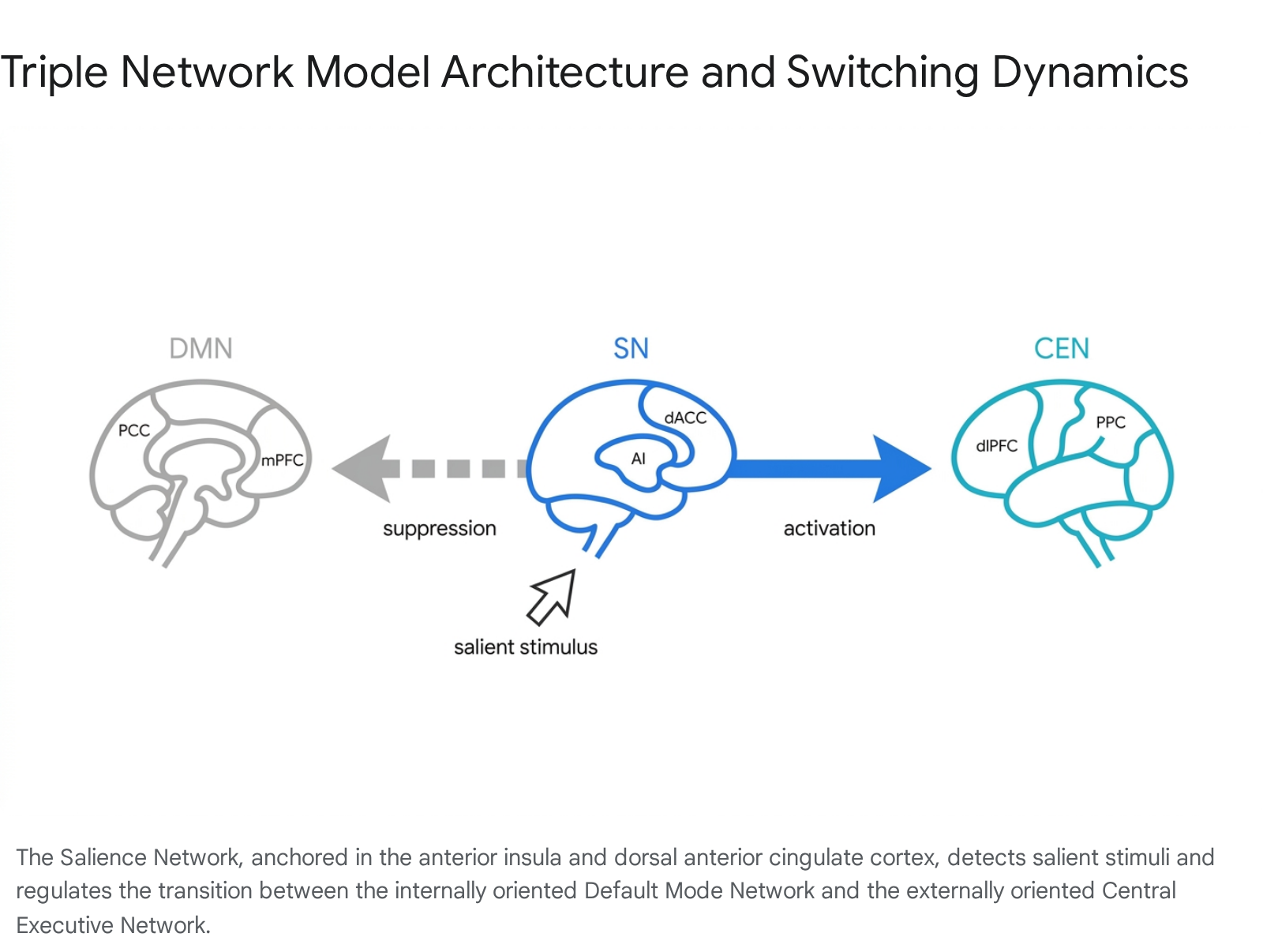
The Triple Network Model
To fully contextualize the neurobiological role of the DMN, it must be examined not in isolation, but through its dynamic interactions with other large-scale brain networks. The most prominent paradigm for understanding this macro-level brain organization is the "Triple Network Model," which posits that cognitive function and dysfunction are largely governed by the interactions among the Default Mode Network, the Central Executive Network (CEN), and the Salience Network (SN) 121314.
The Central Executive Network, sometimes referred to as the frontoparietal network, is anchored in the dorsolateral prefrontal cortex (dlPFC) and the posterior parietal cortex (PPC) 141314. It is specialized for externally directed, cognitively demanding tasks, such as working memory manipulation, complex problem-solving, and rule-based decision-making 1413. In a healthy baseline state, the DMN and CEN exhibit a fundamentally anti-correlated relationship. When attention is directed outward toward a demanding task, CEN activity increases while DMN activity is actively suppressed to prevent internal thoughts from distracting the individual 131417.
Salience Network Mediation
The moderation of this push-pull dynamic is governed by the Salience Network, which consists primarily of the anterior insula (AI) and the dorsal anterior cingulate cortex (dACC) 141415. The SN acts as the brain's internal moderator, continuously monitoring the external environment and internal homeostatic states to identify highly relevant or salient emotional, sensory, and cognitive stimuli 1419.
Upon detecting a salient stimulus that requires focused attention, the SN - particularly the right anterior insula - initiates a rapid signaling cascade. It causally engages the CEN to deploy working memory and task-execution resources, while simultaneously inhibiting the DMN to suppress ongoing mind-wandering and self-reflection 141920. Evidence for this causal switching mechanism is robust. Intracranial electroencephalography (EEG) recordings from human participants engaged in memory tasks have demonstrated a consistent pattern of directed information flow originating in the anterior insula and traveling to both the DMN and the frontoparietal networks 16. Furthermore, experimental interventions using transcranial magnetic stimulation (TMS) targeted at SN sites have been shown to directly enhance connectivity within the CEN while altering DMN engagement, definitively confirming the SN's role as a network switch 20. Conversely, when external cognitive demands subside, the SN facilitates the disengagement of the CEN and the reactivation of the DMN, returning the brain to an introspective state 1419.
Disruptions to this switching mechanism are profound. When the SN fails to adequately suppress the DMN during cognitive tasks, the brain experiences "default mode interference," a phenomenon linked to attention lapses, error proneness, and reduced cognitive flexibility 1717.
DMN Engagement in Goal-Directed Behavior
Historically, the DMN was strictly labeled as a "task-negative" network due to its widespread deactivation during focused external tasks. However, recent fMRI and cognitive literature has conclusively demonstrated that this binary classification is a vast oversimplification 41819. The DMN plays an active, vital role in several forms of externally driven, goal-directed behavior, challenging the assumption that it is exclusively dedicated to internal mentation.
Increased activity within DMN hubs - particularly the core and medial temporal subnetworks - has been consistently observed during major cognitive transitions, such as switching between distinct external task domains or restarting an active task following a brief rest period 1819. For example, studies utilizing multi-voxel pattern analysis (MVPA) during demanding task-switch experiments reveal distinct activity patterns within the DMN for different stimulus domains 19. This suggests that the DMN is heavily involved in context updating and the retrieval of new cognitive paradigms or "rules" when environmental demands shift abruptly, requiring the individual to integrate spatial, self-referential, and temporal information 1819.
Furthermore, the DMN is recruited during automated behavioral tasks where deep external focus is no longer required, allowing the brain to allocate resources elsewhere 418. It is also highly active during external tasks that explicitly demand internal constructive processes, such as evaluating social scenarios, making complex moral and ethical judgments, or generating creative semantic associations 4620. When individuals attempt to understand the beliefs and intentions of others - an active, externally prompted task - the frontotemporal subnetwork of the DMN is robustly engaged 620. Therefore, the DMN is best characterized not as task-negative, but as a system that operates at the intersection of the external environment and the internal constructive self 3.
Lifespan Trajectories of Functional Connectivity
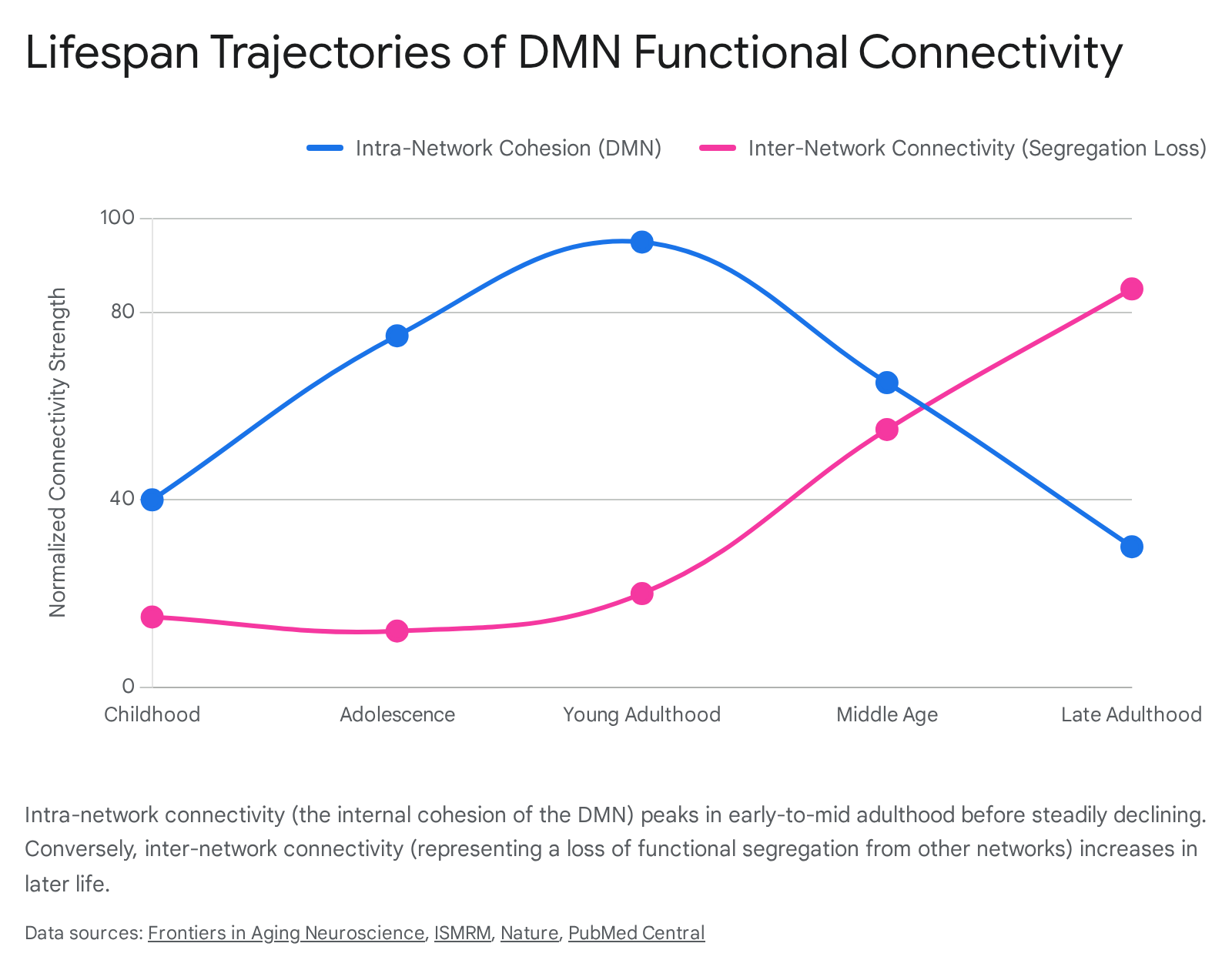
The intrinsic functional architecture of the DMN is not static; it undergoes profound structural and functional transformations from fetal development through late adulthood. The trajectory of DMN functional connectivity broadly resembles an inverted U-shape, characterized by early-life integration, mid-life optimization, and late-life functional decline and dedifferentiation 212223.
Fetal Development and Maturation
In the fetal and infant brain, resting-state networks are highly segregated and primarily isolated to local regions 23. A "proto-DMN" exists, but it lacks the long-range antero-posterior cohesion seen in mature adult brains, indicating that early development is primarily focused on independent functional systems rather than broad integration 23. Throughout childhood and adolescence, the DMN undergoes a period of robust strengthening. This is characterized by progressively increased functional coupling between anterior hubs (such as the mPFC) and posterior hubs (such as the PCC and precuneus) 2124.
This critical period of increased integration corresponds directly with the cognitive maturation of complex social reasoning, abstract problem-solving, and the consolidation of personal identity 2425. Extensive neuroimaging data, drawing from tens of thousands of scans globally, indicate that the global mean network variance and regional connectivity organization of the DMN peak in the late third and early fourth decades of life. This peak finalizes the functional architecture of the higher-order association cortices 22.
Aging and Network Segregation
During healthy aging, the highly optimized functional organization of the brain begins to naturally dedifferentiate. The most reliable functional hallmark of the aging DMN is a significant and progressive decrease in intra-network functional connectivity 52627.
| Lifespan Stage | Intra-Network Connectivity (DMN Cohesion) | Inter-Network Connectivity (Segregation) | Cognitive Correlates |
|---|---|---|---|
| Childhood / Adolescence | Rapidly increasing; strengthening of the antero-posterior axis 2124. | Highly segregated; networks operate independently 2123. | Development of theory of mind, self-identity, and future planning 24. |
| Early to Mid-Adulthood | Peak integration and structural cohesion; optimized local and global variance 22. | Stable segregation; efficient switching mediated by the Salience Network 1419. | Peak executive function, complex semantic retrieval, and emotional regulation. |
| Late Adulthood / Aging | Progressive decline; significant weakening of long-range coupling (e.g., mPFC to PCC) 527. | Loss of segregation; DMN increasingly couples with task-positive networks 2128. | Generalized cognitive slowing, shifts in working memory, and attentional allocation 2729. |
Advanced subject-specific fMRI analyses analyzed in native space rather than normalized space demonstrate severe localized degradations; for instance, the mean correlation between the supramarginal gyrus and the superior-frontal cortex in the right hemisphere drops from roughly 0.5 in young participants to 0.04 in elderly subjects 29. Furthermore, older adults consistently show a flattened and slowed time course of blood oxygen level-dependent (BOLD) responses, exhibiting smaller and slower deactivations of the DMN during externally focused tasks 30.
Concurrently, while intra-network cohesion declines, inter-network connectivity - the functional coupling between the DMN and previously independent networks like the CEN or the Dorsal Attention Network - tends to increase 2128. This loss of network segregation suggests that older adults may recruit broader, less specialized cortical regions to maintain cognitive performance, compensating for reduced local efficiency 2129. Alternatively, this may reflect a degradation of the Salience Network's ability to cleanly toggle the anti-correlated networks 521. Interestingly, aging effects are not entirely uniform across the network; older adults occasionally demonstrate stronger connectivity in specific subsystems, such as the dorsal PCC, which heavily interfaces with cognitive control regions, indicating dynamic compensatory resource reallocation 5.
Neurodevelopmental Pathologies
Because the DMN is a vast integrative hub with exceptionally high metabolic demands, it is exquisitely sensitive to both structural and functional disruption. Aberrant DMN connectivity is a widely recognized biomarker across a broad spectrum of neurodevelopmental disorders, psychiatric conditions, and neurodegenerative diseases.
Attention-Deficit/Hyperactivity Disorder (ADHD)
In ADHD, dysfunction heavily revolves around the "default mode interference" hypothesis. In typical neurobiology, the Salience Network successfully suppresses the DMN when an external task requires focused attention 1717. In individuals with ADHD, there is an attenuated antagonism between the DMN and the CEN. Due to weakened Salience Network coupling, the DMN fails to fully deactivate during goal-oriented tasks, leading to the intrusion of introspective, task-irrelevant thoughts 1736. This hyperconnectivity between the DMN and task-relevant networks correlates strongly with poor response control, higher commission error rates, and clinical impulsivity 31.
Furthermore, specific presentations of ADHD display unique connectomic signatures. Network-based statistical (NBS) and graph-theoretical analyses reveal that the Combined presentation type (ADHD-C) is differentiated from the Inattentive presentation type (ADHD-I) by significantly reduced intra-network connectivity within the DMN 32. Genetic studies indicate that this altered functional connectivity may be linked to variants in monoaminergic pathways, specifically monoamine oxidase A and B (MAOA and MAOB), highlighting the genetic underpinning of DMN dysregulation 17.
Autism Spectrum Disorder (ASD)
Autism Spectrum Disorder shares a high rate of comorbidity with ADHD, and while both disorders exhibit Triple Network dysregulation, their DMN profiles diverge based on the nature of their clinical phenotypes 253334.
| Pathological Feature | Autism Spectrum Disorder (ASD) | Attention-Deficit/Hyperactivity Disorder (ADHD) |
|---|---|---|
| DMN Intra-Network Status | Mixed trajectory: Often overconnected in childhood, but predominantly underconnected in adolescence and adulthood 2425. | Altered structure; failure of the DMN to deactivate efficiently during active tasks 3632. |
| Inter-Network Interactions | Reduced connectivity between the DMN and the Salience Network (SN) 33. | Reduced anti-correlation between the DMN and Central Executive Network (CEN); increased cross-network integration 1731. |
| Primary Clinical Correlate | Deficits in social cognition, "theory of mind," and processing of complex interpersonal cues 2425. | Attentional lapses, persistent mind-wandering, hyperactive behaviors, and poor inhibitory control 173631. |
In adolescents and adults with ASD, the DMN is predominantly characterized by functional underconnectivity, particularly between anterior and posterior hubs 2425. Because the DMN overlaps significantly with the "social brain," this generalized underconnectivity - and specific decoupling from temporoparietal regions - correlates directly with the severity of social impairments, altered perceptual feature integration, and difficulties in moral decision-making 2425.
Psychiatric and Neurodegenerative Pathologies
Major Depressive Disorder and Schizophrenia
The Triple Network Model provides a profound explanatory framework for psychiatric illnesses. In Major Depressive Disorder (MDD), the DMN often exhibits localized hyperconnectivity, specifically within the subgenual anterior cingulate cortex (sgACC). This hyperconnectivity creates a negative associative-processing bias, neurologically locking the patient into cycles of depressive rumination and negative self-referential thought 11420. Conversely, in schizophrenia, structural and functional abnormalities within the anterior insula (the core of the Salience Network) result in deviant salience mapping. The brain incorrectly assigns high salience to internally generated thoughts or speech within the DMN, directly contributing to the manifestation of auditory hallucinations and clinical psychosis 1414.
Alzheimer's Disease and Dementias
In neurodegenerative medicine, the DMN is uniquely and profoundly implicated in the pathophysiology of Alzheimer's disease (AD). The spatial distribution of early amyloid-beta ($\beta$-amyloid) plaques maps almost perfectly onto the core structural nodes of the DMN, particularly the posterior cingulate cortex, precuneus, and medial prefrontal cortex 264135.
Resting-state fMRI studies indicate that the functional connectivity abnormalities seen in AD represent an aggressive, accelerated pattern of normal aging. AD patients display severe, progressive breakdowns in intra-DMN connectivity, beginning in the medial temporal subsystems (resulting in the classic episodic memory deficits seen in early AD) and eventually spreading to the lateral parietal hubs 2636. Longitudinal connectivity trajectories are highly predictive of cognitive decline; for instance, the rate of annual decline in DMN connections is exponentially faster (up to 13.6 times) in individuals presenting with $\beta$-amyloid plaques compared to those with isolated cerebrovascular dementia 41. Furthermore, patients with progressive mild cognitive impairment (MCI) who transition to full dementia exhibit specific aberrant synchronizations within the DMN that are absent in patients with stable MCI 37.
Current hypotheses surrounding this pathogenesis suggest a metabolic vulnerability model. The chronic high baseline metabolic activity and continuous neuronal firing within the DMN make it uniquely susceptible to $\beta$-amyloid toxicity. This creates a destructive positive feedback loop: the high neural activity drives local metabolic stress, accelerating localized $\beta$-amyloid deposition, which in turn further disrupts DMN connectivity and accelerates widespread neuronal death 35.
Interestingly, network disruption profiles are specific to distinct neurodegenerative conditions. While AD is characterized by DMN fragmentation, behavioral variant frontotemporal dementia (bvFTD) presents with an inverse pattern: significantly reduced Salience Network connectivity alongside abnormally increased intrinsic connectivity within the DMN 36. In Parkinson's Disease (PD) with cognitive impairment, the DMN shows abnormal hyperconnectivity between the PCC and areas such as the angular gyrus and precuneus, indicating that diverse pathologies disrupt the large-scale networks in highly specific, condition-dependent ways 38.
Global Demographic Diversity in Network Neuroscience
A critical and persistent limitation in contemporary connectome research is the overwhelming reliance on functional and genetic data derived from Western, Educated, Industrialized, Rich, and Democratic (WEIRD) populations. Datasets representing populations in Africa, South Asia, and diverse racial cohorts worldwide remain severely sparse, which systematically restricts the generalizability of established network biomarkers and targeted therapeutics 394048.
Findings in African and African-American Populations
Recent analyses incorporating diverse populations have revealed significant deviations from established Western neurobiological benchmarks. For instance, advanced neuroimaging studies assessing octogenarians in the United States demonstrated that older Black adults exhibited significantly higher gray matter microstructural integrity - measured via mean diffusivity - compared to their White counterparts 41. This higher structural integrity persisted even when adjusting for higher systemic vascular risks, indicating distinct neuroprotective or compensatory network configurations that are not captured in predominantly White cohorts 41.
Furthermore, large-scale genomic initiatives, such as the African Ancestry Neuroscience Research Initiative led by the Lieber Institute, have illuminated the profound impact of genetic diversity on neurological risk factors. In post-mortem brain samples from individuals of African descent, researchers identified over 2,500 genes whose expression varied significantly based on the proportion of African ancestry 42. Crucially, ancestry-informed genetic variations primarily impacted non-neuronal cells, such as the immune and vascular cells that support networks like the DMN, potentially explaining the differing prevalence rates of Alzheimer's and stroke across demographics 42. Conversely, variations in psychiatric traits (which implicate the DMN and SN) were largely driven by environmental exposures rather than genetic ancestry 42.
In Africa itself - the continent with the highest global genetic diversity - the lack of Findable, Accessible, Interoperable, and Reusable (FAIR) brain datasets severely limits the exploration of these mechanisms 394048. New initiatives, such as the African Somatic and Stem Cell Bank, are actively generating deeply phenotyped biological lines to capture this unique genetic variation, aiming to provide cellular models for studying DMN-related neurodegeneration 48.
Findings in South Asian Populations
Genetic and functional differences are equally critical in South Asian populations, where mixed dementia cases account for nearly half of all diagnoses 4143. In cohorts analyzed through the Longitudinal Aging Study of India (LASI-DAD), whole-genome sequencing identified distinct genetic risk architectures 44. While some variants recognized in European populations (like APOE $\epsilon$4) remain predictive of DMN decline, researchers identified rare missense mutations (e.g., in the PICALM and TSPOAP1 genes) that are vastly more prevalent in South Asian individuals and correlate strongly with executive function and working memory decline 44. Additionally, the elevated prevalence of Type 2 diabetes within South Asian communities introduces unique, compounding vascular risk factors that uniquely affect executive control networks in tandem with the DMN 4143.
Advancing a truly global framework for neuroscience that adequately powers diverse, longitudinal functional imaging studies is paramount. Without integrating these populations, the scientific community cannot definitively establish which dynamics of the default mode network represent universal human biology versus those shaped by population-specific genetic, environmental, and epigenetic variables 404546.
Conclusion
The default mode network operates as the highly sophisticated neurological substrate for the continuous internal narrative of human consciousness. Far from a passive resting state, it is an anatomically complex, highly integrated system of functional subsystems and directional micro-zones that seamlessly bridge past autobiographical memories, future projections, and complex social simulations. Governed by the rapid switching mechanics of the salience network, the DMN exists in a delicate, continuous equilibrium with task-positive regions like the central executive network.
When this dynamic equilibrium degrades - whether through the developmental miswirings seen in ADHD and ASD, the psychiatric signaling failures of depression and schizophrenia, or the toxic metabolic cascades of Alzheimer's disease - core aspects of personal identity, memory, and cognitive control are profoundly compromised. As neuroimaging technology and computational modeling continue to evolve, future research must prioritize the integration of underrepresented global demographics into longitudinal cohorts. Only through a diverse, genetically and environmentally comprehensive approach can science fully resolve the protective mechanisms and vulnerabilities that govern this vital network across the entirety of the human lifespan.