Extracellular vesicle and exosome therapy for aging
The pursuit of interventions capable of delaying, halting, or reversing the biological aging process has driven significant innovation in regenerative medicine. In recent years, therapeutic focus has shifted from cell-based interventions - such as mesenchymal stem cell transplantation - to cell-free paradigms. At the center of this shift are extracellular vesicles, colloquially and commercially referred to as exosomes. Marketed heavily for applications ranging from aesthetic skin rejuvenation to systemic lifespan extension, extracellular vesicle therapy occupies a highly volatile intersection between rigorous scientific discovery and premature commercial exploitation.
While early preclinical evidence demonstrates profound regenerative potential, including the reversal of age-related functional declines in mammalian models, the clinical reality is considerably more complex. The administration of extracellular vesicles in humans is currently constrained by massive biological hurdles, a lack of standardized manufacturing protocols, and an almost universal absence of regulatory approval for therapeutic use in major jurisdictions. This report systematically examines the biological mechanisms of extracellular vesicles in the context of aging, evaluates the early preclinical and clinical evidence, compares the modality to existing regenerative treatments, and analyzes the profound disconnect between scientific reality and the booming direct-to-consumer medical tourism market.
Terminology and the MISEV2023 Guidelines
The scientific terminology surrounding cell-derived vesicles has historically been fraught with ambiguity, leading to the frequent misuse of the term "exosome" in both academic literature and commercial marketing. The regulatory and scientific consensus on this nomenclature is detailed in the Minimal Information for Studies of Extracellular Vesicles (MISEV2023) guidelines, published by the International Society for Extracellular Vesicles (ISEV) 123.
Evolution of Nomenclature
According to the MISEV2023 guidelines, the term "exosome" strictly refers to a specific biogenesis pathway. True exosomes are vesicles measuring 30 to 150 nanometers in diameter that originate from the inward budding of endosomes to form multivesicular bodies. These bodies subsequently fuse with the plasma membrane to release their contents into the extracellular space 1245. Because establishing this specific intracellular origin is technically arduous, the MISEV2023 guidelines strongly recommend the use of operational terms based on physical characteristics, such as "small extracellular vesicles" (sEVs) for particles under 200 nanometers, rather than assuming an exosomal origin 12. Despite these rigorous academic standards, the commercial sector has almost universally adopted "exosome" as a ubiquitous marketing buzzword, applying it to heterogeneous mixtures of vesicles, cellular debris, and uncharacterized extracellular particles 2.
Technical Challenges in Isolation and Characterization
The standardization of extracellular vesicle research and therapeutic development is hindered by specific technical challenges outlined in the MISEV2023 framework. Biological matrices naturally contain non-vesicular extracellular particles (NVEPs), lipoproteins, and protein aggregates that co-isolate with vesicles and may greatly outnumber them 24. In blood plasma, major confounding factors include platelets and lipoproteins (such as HDL and LDL) which overlap in size and density with extracellular vesicles 4. Furthermore, a "biomolecular corona" on the vesicle surface makes it difficult to distinguish true vesicle components from associated free proteins 4.
Other biofluids present unique isolation hurdles: urine contains the Tamm-Horsfall protein (THP) which traps vesicles in lattice-like networks; milk contains casein micelles that overlap in size with vesicles; and standard cell culture supplements, such as fetal bovine serum, are rich in their own vesicles, complicating the purification of the target therapeutic 4. Compounding these issues are the limits of detection (LOD) in characterization techniques like Nanoparticle Tracking Analysis (NTA) and Dynamic Light Scattering (DLS), which often overestimate size and assume spherical uniformity 4. Measuring total protein or lipid content often results in overestimations of vesicle concentration because these molecules are present in co-isolated non-vesicular components 46.
Biological Mechanisms of Rejuvenation
To understand the therapeutic potential of extracellular vesicles, it is necessary to examine their precise biological function and the signaling pathways they modulate to combat cellular senescence.
Intercellular Communication and Pathway Modulation
Extracellular vesicles function as a sophisticated intercellular communication system. Unlike stem cells, which exert effects through both cellular engraftment and paracrine signaling, small extracellular vesicles rely entirely on the targeted transfer of highly specific biological cargo 56. Encased in a lipid bilayer that protects them from enzymatic degradation in the extracellular environment, these vesicles transport a payload of proteins, lipids, messenger RNA (mRNA), and microRNA (miRNA) 7108.
As the human body ages, intrinsic cellular turnover declines, collagen synthesis decreases, and antioxidant defenses weaken. Concurrently, extrinsic factors induce oxidative stress and chronic, low-grade inflammation - a phenomenon frequently termed "inflammaging" 7109. When extracellular vesicles derived from young or optimal cellular sources, such as perinatal mesenchymal stem cells (MSCs) or young donor plasma, are introduced to this environment, they deliver regulatory instructions that alter the behavior of aged recipient cells 10910.
These vesicles counteract structural degradation by upregulating the production of proteins like collagen and elastin, primarily through the activation of the transforming growth factor-beta (TGF-β)/Smad signaling pathway 911. Simultaneously, they deliver cytokines and growth factors that inhibit matrix metalloproteinases (MMPs), which are the enzymes responsible for extracellular matrix breakdown 7. By reducing oxidative stress and modulating key inflammatory cascades, such as the NF-κB and MAPK pathways, extracellular vesicles suppress the senescence-associated secretory phenotype (SASP), thereby mitigating the toxic inflammatory environment characteristic of aged tissues 109.
MicroRNA Transfer and Transcriptional Reprogramming
The most profound mechanisms of extracellular vesicle-mediated rejuvenation involve the transfer of epigenetic regulators, specifically microRNAs. During the natural aging process, the circulating profile of miRNAs undergoes a dynamic and detrimental shift. Pro-aging miRNAs accumulate in the bloodstream, while rejuvenating miRNAs are systematically lost, which disrupts tissue homeostasis and exacerbates cellular energy deficits 12.
The cargo within therapeutic extracellular vesicles actively rewrites this imbalance. For example, specific exosomal miRNAs such as miR-1246 have been identified as activators of the TGF-β/Smad signaling pathway, enhancing collagen production and preventing the apoptosis of skin cells 11. Similarly, miR-767 plays a critical role in delaying the aging process by regulating fibroblast function, while miR-125b enhances angiogenesis, a process vital for tissue oxygenation and wound healing 11.
Reversal of Mitochondrial Dysfunction
The capability of young extracellular vesicles to correct systemic metabolic decline was elucidated in a landmark 2024 study published in Nature Aging by Chen et al. 13. Drawing on the historical principles of heterochronic parabiosis - where the circulatory systems of young and old animals are joined to produce rejuvenating effects - researchers isolated the specific blood components responsible for this phenomenon 1317.
The researchers demonstrated that small extracellular vesicles from the plasma of young mice contain abundant therapeutic miRNAs, specifically miR-664a-3p 121314. When delivered to aged tissues, these miRNA cargoes enter the recipient cells and stimulate the expression of Peroxisome proliferator-activated receptor-gamma coactivator 1-alpha (PGC-1α) 121314. PGC-1α is a master transcriptional regulator of mitochondrial biogenesis. By stimulating its expression both in vitro and in vivo, the young vesicles restored mitochondrial energy metabolism, compensated for existing mitochondrial deficits, and fundamentally enhanced cellular ATP supply 1213.
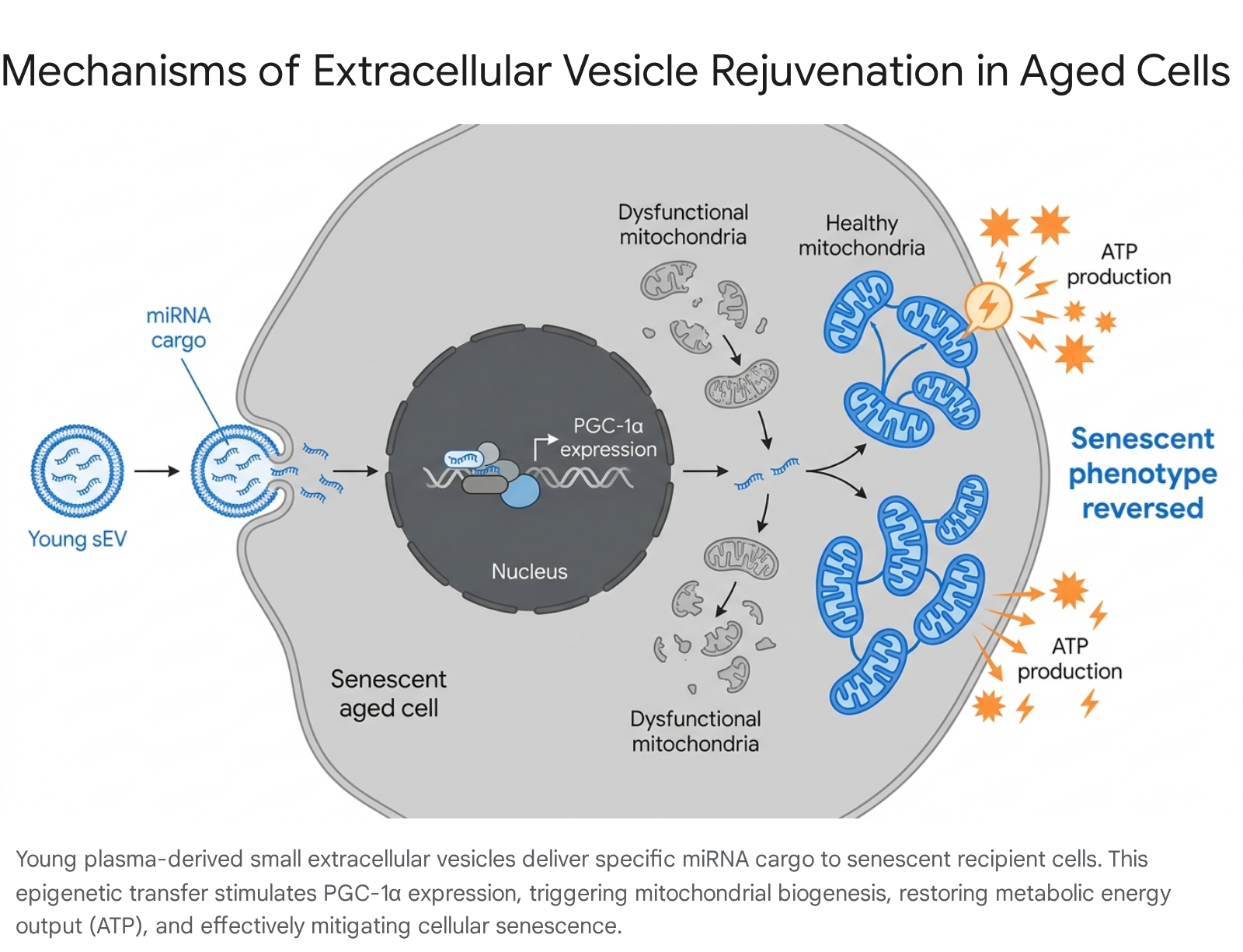
Quantitative proteomic analyses confirmed that the extensive alterations in the proteomes of aged tissues following young vesicle treatment were inextricably linked to these optimized metabolic processes 131719.
Regulation of Cellular Senescence and Apoptosis
The therapeutic efficacy of extracellular vesicles is deeply dependent on the biological age of the source material. Research analyzing the impact of extracellular vesicles on hematopoietic stem cells (HSCs) reveals a stark dichotomy between young and aged vesicles. Exosomes derived from young individuals maintain the viability and proliferation potential of HSCs without inducing senescence 15. Conversely, extracellular vesicles isolated from elderly donors actively stimulate senescence-related differentiation. Aged vesicles demonstrate an upregulation of BAX (a pro-apoptotic gene) and a significant downregulation of BCL-2 (an anti-apoptotic gene), actively promoting cellular death pathways in recipient cells 15.
Similar divergent effects are observed in macrophage modulation. In models of acute lung injury, young MSC-derived vesicles successfully alter macrophage phenotypes, reduce the recruitment of inflammatory macrophages, and alleviate pulmonary inflammation 16. Aging MSC-derived vesicles fail entirely in this regard; they show a significantly lower efficacy in internalization by target macrophages and possess an altered miRNA profile that renders them incapable of exerting protective, anti-inflammatory effects 16.
Evaluation of Preclinical and Clinical Evidence
While the theoretical mechanisms are well-established, the empirical evidence supporting extracellular vesicle therapy remains in a transitional phase. Current literature represents a robust foundation of preclinical animal models accompanied by early-stage, often small-cohort human clinical trials.
Cutaneous Regeneration and Dermatological Applications
The most visible and heavily commercialized application of extracellular vesicles is in aesthetic dermatology. Extracellular vesicles derived from human mesenchymal stem cells have demonstrated measurable efficacy in reversing both intrinsic chronological aging and extrinsic photoaging 7911.
A comprehensive 2025 analysis of twelve clinical studies examining the use of extracellular vesicles in treating various skin conditions indicated measurable improvements across diverse patient populations 1117. In clinical trials for acne scar treatment, improvements in scar depth and texture were observed in 76% of participants following combination therapy with microneedling 17. In wound healing paradigms, exosome-treated wounds exhibited closure rates 30% to 40% faster than control groups, characterized by superior collagen organization and reduced inflammatory markers 17.
A 2025 investigator-blinded, split-face trial compared the efficacy of adipose mesenchymal stem cell-derived (ASC) exosomes to platelet-rich plasma (PRP) for photoaged facial skin 10. Participants underwent radiofrequency microneedling treatments, with exosomes applied to one half of the face and PRP to the other. Histological analysis confirmed that the exosome application increased Type I collagen and glycosaminoglycans, successfully ameliorating wrinkling, dyschromia, erythema, and uneven texture at rates equal to or exceeding the PRP intervention 10. However, researchers note that because pure exosomes are large, complex biological molecules, their topical efficacy is heavily dependent on the delivery method. Cosmetic formulations applied to intact skin cannot penetrate the stratum corneum to reach the dermis where cellular regeneration occurs, limiting their utility to superficial epidermal hydration unless physically driven into the tissue via microneedling or ablative lasers 18.
Alopecia and Hair Restoration
Exosomes derived from mesenchymal stem cells have shown substantial promise in the treatment of androgenetic alopecia. Clinical research indicates that exosome treatments can stimulate hair follicle growth and extend the anagen (growth) phase of the hair cycle 11. In clinical trials, targeted exosome interventions increased hair density by an average of 28% and measurably improved hair thickness 17. For needle-averse patients, exosome therapy offers a minimally invasive alternative to traditional surgical hair transplantation, and early comparative studies suggest that a single treatment of highly concentrated exosomes can occasionally yield results superior to multiple sessions of traditional autologous therapies 1011.
Musculoskeletal Repair and Osteoarthritis
Beyond aesthetic applications, extracellular vesicles show considerable utility in treating degenerative musculoskeletal conditions, which represent a primary hallmark of functional aging. Mesenchymal stem cell-derived exosomes promote cartilage repair by modulating immune responses within the joint capsule, reducing localized inflammation, and maintaining synovial homeostasis 1019.
In clinical observations of knee osteoarthritis, patients receiving intra-articular exosome injections demonstrated accelerated healing and improved joint mobility 19. The recorded outcomes mirrored, and in certain cohorts slightly exceeded, those achieved with live stem cell therapies, but notably lacked the associated cellular risks 19. Similarly, in addressing age-related muscle loss (sarcopenia), preclinical data suggest that MSC exosomes stimulate the activation, proliferation, and differentiation of satellite cells, thereby promoting muscle regeneration and preserving long-term motor function 10. While these applications imply vast potential for extending functional healthspan, large-scale Phase II and Phase III human trials are still required to establish standardized dosing regimens and verify long-term efficacy 1718.
Systemic Lifespan Extension in Mammalian Models
The most striking, yet strictly preclinical, evidence for extracellular vesicles relates to systemic lifespan extension. The aforementioned 2024 Nature Aging study evaluated the impact of weekly intravenous injections of small extracellular vesicles isolated from the plasma of young mice. Administered to 20-month-old male mice, the intervention resulted in a median lifespan increase to 1,031 days - a 22.7% extension compared to the typical 840-day lifespan of the C57BL/6J male control cohort 12.
The longest-living subject in the treatment group survived for 1,266 days, the biological equivalent of a human living between 120 and 130 years 12. To contextualize this magnitude of effect, standard caloric restriction therapies in similar murine models typically extend median lifespan by roughly 16.4%, while pharmacological interventions like metformin and nicotinamide yield lifespan extensions of 5.8% and 4.2%, respectively 12. Beyond mere chronological extension, the intravenous vesicle treatment mitigated senescent phenotypes and reversed functional declines across multiple organ systems, including the hippocampus, heart, muscle, testis, and bone 1213. While this represents a monumental proof-of-concept for systemic anti-aging biology, extrapolating these murine results directly to human biology remains highly speculative. Human trials for systemic intravenous exosome lifespan extension do not currently exist outside of highly regulated, early-phase investigational studies.
Comparative Analysis of Regenerative Modalities
To accurately assess the current and future utility of extracellular vesicle therapy, the modality must be contextualized against the two dominant regenerative treatments currently utilized in medical and aesthetic practice: Mesenchymal Stem Cell (MSC) therapy and Platelet-Rich Plasma (PRP) therapy.
Extracellular Vesicles Versus Mesenchymal Stem Cells
Historically, live stem cell therapy has served as the gold standard in regenerative medicine. However, the transplantation of live, nucleated cells carries inherent biological and logistical risks. Because live stem cells possess the capability to self-replicate and differentiate, their administration raises valid clinical concerns regarding uncontrolled cellular proliferation, the potential for tumorigenesis, and immune rejection, particularly in allogeneic (donor-derived) applications 692021.
Extracellular vesicles are fundamentally acellular. Lacking a functional nucleus, they cannot self-replicate or differentiate, eliminating the primary risks of tumorigenesis and malignant transformation 1202228. This cell-free nature vastly improves their safety profile and reduces overall immunogenicity, making vesicles a superior candidate for "off-the-shelf" allogeneic therapies 69. Furthermore, vesicles are physically smaller (30 - 150 nm) than whole cells, permitting them to cross biological barriers - such as the blood-brain barrier - that typically restrict the migration of large stem cells 5. Logistically, extracellular vesicles are significantly more robust; they can be sterilized via standard non-destructive filtration, lyophilized (freeze-dried), and stored at room temperature or deep-frozen without the complex, costly cryopreservation protocols required to maintain the viability of whole stem cells 5192021.
Extracellular Vesicles Versus Platelet-Rich Plasma
Platelet-Rich Plasma (PRP) is an autologous therapy, meaning it is derived entirely from the patient's own biological material. Blood is drawn and centrifuged to concentrate platelets, which are then reinjected to release growth factors that stimulate localized tissue repair 2930. The primary advantage of PRP is its extreme safety profile; because the material is strictly autologous, there is virtually zero risk of allergic reaction, immune rejection, or communicable disease transmission 2923.
However, the efficacy of PRP is intrinsically limited by the biological age and baseline health status of the individual patient. A 50-year-old patient yields 50-year-old growth factors, which inherently possess diminished regenerative capacity compared to younger biology 24. In contrast, exosome therapies utilized in clinical settings are typically derived from highly screened perinatal tissues, such as umbilical cord Wharton's jelly, or young donor plasma. These sources represent "biological age zero," carrying a substantially higher concentration of potent growth factors, uncorrupted genetic instructions, and rejuvenating miRNAs that far exceed the signaling capability of aged autologous plasma 2324. Consequently, while PRP frequently requires multiple successive sessions to achieve subtle improvements, highly concentrated exosome therapies have demonstrated faster, more pronounced, and more targeted regenerative outcomes in early comparative trials 302325.
| Feature / Modality | Extracellular Vesicles (Exosomes) | Mesenchymal Stem Cells (MSCs) | Platelet-Rich Plasma (PRP) |
|---|---|---|---|
| Biological Origin | Acellular lipid vesicles derived from stem cells or plasma 729 | Live, nucleated multipotent cells 20 | Autologous blood concentrate 29 |
| Primary Mechanism | Epigenetic transfer (miRNA) and targeted signaling 109 | Paracrine signaling and cellular engraftment 28 | Release of growth factors from localized platelets 25 |
| Regenerative Potency | High ("biological age zero" if donor-derived) 24 | High (dependent on source tissue and culture) 20 | Variable (strictly dependent on the patient's biological age) 24 |
| Tumorigenic Risk | Negligible (cannot self-replicate without a nucleus) 2022 | Potential risk (live cellular proliferation in vivo) 621 | None (contains no proliferating or multipotent cells) 25 |
| Immunogenicity | Very Low (naturally immune-tolerant) 6 | Moderate (requires matching or specific umbilical sources) 22 | None (100% autologous material) 30 |
| Storage & Handling | Highly stable, can be lyophilized and easily filtered 520 | Complex cryopreservation required to maintain cell viability 19 | Prepared on-site, immediately prior to therapeutic use 2923 |
Technical Barriers to Clinical Translation
Despite their theoretical superiority and promising preclinical results, the translation of extracellular vesicles from the laboratory to systemic human clinical practice is severely bottlenecked by unresolved technical and biological limitations.
Manufacturing, Yield, and Standardization
The foremost hurdle in exosome therapeutics is the profound difficulty of isolating pure, functional vesicles at a commercial scale. Current isolation methodologies present a stark trade-off between absolute yield and product specificity. Techniques such as differential ultracentrifugation are prone to aggregating the vesicles or physically damaging their fragile lipid bilayers, while size exclusion chromatography is highly precise but suffers from exceptionally low volume yields 426. Advanced microfluidic isolation techniques remain in their nascent phases and similarly struggle with scalability 26.
Furthermore, extracellular vesicles are inherently heterogeneous. Because the molecular cargo of a vesicle is determined by the specific metabolic state of the parent cell at the exact moment of secretion, establishing batch-to-batch consistency for pharmaceutical grading is incredibly difficult 5620. Currently, there are no universally agreed-upon release criteria or definitive quality control assays capable of rapidly and comprehensively profiling the thousands of individual proteins and nucleic acids within a single batch of manufactured vesicles 518.
Structural Integrity and Storage Limitations
While exosomes are generally more stable than live stem cells, preserving their structural integrity during commercial distribution remains problematic. Some independent studies suggest that exosomes are optimally stable at - 80 °C, a temperature that is logistically impractical for widespread distribution in commercial aesthetic or outpatient settings 26.
Efforts to bypass deep-freezing through lyophilization (freeze-drying) have yielded poor results. A recent independent analysis conducted by the EXACT Oversight Bureau evaluated a dozen commercial aesthetic products labeled as containing "exosomes." The analysis found that 95% of these commercial products did not meet the basic structural criteria of a functional vesicle 36. The vast majority exhibited broken or non-intact bilipid membranes, directly attributed to destructive processing methods like lyophilization. Without an intact lipid membrane to protect and deliver the internal cargo, the vesicle is non-functional, rendering the commercial product biologically useless 36.
Pharmacokinetics, Clearance, and Surface Engineering
When evaluating systemic anti-aging therapies - such as intravenous infusions of exosomes - pharmacokinetics present a formidable barrier. Naturally occurring extracellular vesicles have a very short half-life in systemic circulation. When injected intravenously, they are rapidly recognized as foreign debris and cleared by the reticuloendothelial system, particularly by macrophages residing in the liver, spleen, and lungs 5626.
This rapid macrophage-dependent clearance drastically limits the concentration of therapeutic cargo that actually reaches intended target tissues, severely diminishing systemic efficacy 527. To overcome this biological limitation, biomedical engineers are exploring active surface modifications. One promising strategy involves sheathing the vesicles with CD47 - a naturally occurring "don't eat me" signaling protein that protects the vesicles from immune phagocytosis, thereby extending their circulation time and improving targeted organ accumulation 826. However, these modified semi-synthetic vesicles are complex to scale, introduce new variables of heterogeneity, and have yet to undergo the rigorous human safety testing required for clinical use 826. Without precise targeting mechanisms, systemic administration also raises concerns regarding off-target effects, where potent growth factors might inadvertently accumulate in unintended organs or hypothetically accelerate the growth of dormant, pre-existing dysplasias 83638.
The Commercial Landscape and Medical Tourism
The profound gap between early-stage preclinical promise and the lack of approved clinical protocols has given rise to a volatile, highly lucrative "gray market." Driven by massive consumer demand for longevity and anti-aging therapies, an international commercial landscape has emerged that frequently operates well ahead of the established clinical evidence.
Direct-to-Consumer Marketing and Economic Disparities
A systematic review of direct-to-consumer online advertising identified hundreds of businesses heavily promoting purported exosome products 2829. These clinics routinely market extracellular vesicles as panaceas for a litany of complex conditions. For instance, a review identified 38 businesses operating 60 clinics specifically marketing unproven exosome and stem cell products for COVID-19 and Long COVID, alongside claims for systemic aging, hair loss, autism, and neurodegeneration 2830.
Marketing strategies deliberately obscure the investigational nature of these products. Promotional claims often highlight the advanced molecular biology of exosomes - accurately citing genuine academic research regarding mRNA transfer and PGC-1α upregulation - but improperly apply these preclinical murine findings as guarantees of human clinical efficacy 3042. Furthermore, many businesses utilize deceptive regulatory framing, stating that their products are "FDA-registered." In regulatory terminology, registration merely indicates that a manufacturing facility has filed its existence with the government; it is entirely distinct from being "FDA-approved," which requires rigorous, multi-phase clinical trials demonstrating both safety and efficacy 54231.
The economics of exosome therapy reveal staggering markups indicative of high-demand, unregulated consumer markets. A comprehensive analysis of clinic pricing indicates that the cost of exosome therapy averages $11,322 per treatment, with some clinics charging up to $25,000 for systemic interventions, and aesthetic applications averaging around $5,000 382830. In stark contrast, stem cell biologists note that the actual manufacturing cost to produce a single dose of generalized exosome product in a laboratory setting can be as low as $50 to $100 38. This immense profit margin heavily incentivizes the rapid proliferation of clinics and encourages the premature commercialization of unproven biological materials 3638.
The Regulatory Arbitrage in Mexico
Because major Western regulatory bodies strictly limit the administration of unapproved biological products, a robust medical tourism industry has emerged, primarily concentrated in jurisdictions with more permissive or rapidly evolving regulatory frameworks.
Mexico has positioned itself as a premier destination for regenerative wellness, capitalizing heavily on its geographic proximity to patients in the United States and Canada 4432. The Mexican regulatory authority, COFEPRIS, issues specific licenses for clinics to infiltrate and bank stem cells, creating a formal legal framework that permits treatments that remain unavailable in the US 32. Furthermore, in 2025, Mexico instituted sweeping pharmaceutical regulatory reforms. These included updated Good Manufacturing Practice (GMP) standards (NOM-241-SSA1-2025) and the introduction of reliance pathways that allow COFEPRIS to accept certifications from the World Health Organization (WHO) and reference regulatory bodies, thereby accelerating the approval of advanced therapies and medical devices 4647. However, the quality of care remains heterogeneous. While top-tier luxury clinics in medical tourism hubs like Cancun and Puerto Vallarta operate state-of-the-art laboratories, lesser-regulated entities pose significant risks regarding product sterility and biological origin 44.
Permissive Frameworks in Panama, Thailand, and the UAE
Panama operates under an advanced regulatory environment specifically tailored to support specialized regenerative procedures. Unlike many Western nations with restrictive policies on donor tissues, Panama legally permits the use of allogeneic, donor-derived umbilical cord mesenchymal stem cells and their isolated exosomes 4448. These tissues are highly potent and immune-privileged, allowing patients to receive high-dose "Golden Cell" systemic rejuvenation without the necessity of undergoing autologous surgical extraction 44. Backed by the Ministry of Health (MINSA), Panama has established itself not merely as a clinical destination but as an active hub for legitimate pilot studies in regenerative protocols 48.
Thailand and the United Arab Emirates (UAE) manage massive volumes of international patients seeking anti-aging exosome protocols. These nations successfully combine JCI-accredited hospital infrastructures with luxury medical hospitality 443334. Clinics in these regions offer advanced exosome applications for aesthetics, orthopedics, and highly specific use-cases such as in vitro fertilization (IVF) support, often at costs up to 92% lower than equivalent treatments in Western markets 3334. However, the regulatory oversight in these high-volume regions can be fragmented, placing the burden of extensive due diligence entirely on the traveling patient to verify laboratory certifications and physician credentials 4435.
Global Regulatory Frameworks and Compliance
The classification of extracellular vesicles is a matter of intense global debate. Because they fall directly into the regulatory chasm between live cellular therapies and traditional molecular pharmaceuticals, authorities worldwide have adopted highly divergent frameworks to govern their development, manufacturing, and clinical use.
United States Food and Drug Administration Oversight
The position of the United States Food and Drug Administration (FDA) is unequivocal: as of 2026, there are absolutely zero FDA-approved exosome products for therapeutic use in humans 1742.
The FDA classifies exosomes intended for disease treatment, systemic administration, or anti-aging as unapproved new drugs and unlicensed biological products. They are strictly regulated under Section 351 of the Public Health Service (PHS) Act and the Federal Food, Drug, and Cosmetic Act 54252. To be legally administered in the US, an exosome product must undergo the full, decades-long pharmaceutical pipeline, beginning with an Investigational New Drug (IND) application subject to Institutional Review Board (IRB) oversight, advancing through Phase I-III clinical trials, and culminating in a Biologics License Application (BLA) 51742.
Because thousands of clinics have bypassed this process to capitalize on consumer demand, the FDA has dramatically escalated its enforcement actions. Between 2023 and 2026, the agency issued a continuous barrage of warning letters to manufacturers and distributors, including Kimera Labs, Chara Biologics, Evolutionary Biologics, New Life Medical Services, Platinum Biologics, Dynamic Stem Cell Therapy, and the Innate Healthcare Institute 54252. These letters cited severe federal violations, including marketing unapproved biologicals, distributing unlicensed products, and failing to validate fundamental sterility and manufacturing procedures 4252. Consequently, any clinic in the US offering systemic exosome injections outside of an authorized clinical trial is operating illegally within a gray market 42.
To circumvent these strict biological regulations, some cosmetic manufacturers have turned to Plant-Derived Exosomes (P-Exos) or plant-derived exosome-like nanoparticles (PELNs) 3636. Extracted from botanicals, these vesicles fall entirely outside the FDA's Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/P) framework 36. While they allow cosmetic companies to legally market "exosome" topical serums, plant exosomes intrinsically lack the human-specific biological payload (such as human mRNA and miRNA). They offer antioxidant and anti-inflammatory benefits, but cannot facilitate true human cellular epigenetic reprogramming 3636.
European and Asian Regulatory Paradigms
The European Medicines Agency (EMA) and the UK's Medicines and Healthcare products Regulatory Agency (MHRA) apply similarly stringent oversight to that of the FDA. In these jurisdictions, extracellular vesicles are classified as Advanced Therapy Medicinal Products (ATMPs) or biological medicinal products, subjecting them to the highest tiers of clinical evaluation and strict Good Manufacturing Practice (GMP) requirements prior to any market authorization 423755.
In contrast, Asian jurisdictions exhibit more dynamic and segmented frameworks. Japan operates under a unique dual-track system guided by the Pharmaceuticals, Medical Devices, and Other Therapeutic Products Act (PMD Act) and the Act on the Safety of Regenerative Medicine (ASRM) 5538. This framework permits conditional, time-limited approvals for regenerative products, allowing promising therapies to reach patients faster while mandatory post-market efficacy data is actively gathered 55.
South Korea, despite being a global leader in aesthetic medicine, tightly restricts human-derived materials. The Ministry of Food and Drug Safety (MFDS) explicitly bans human cells, tissues, and their culture media from being used as general cosmetic ingredients, reserving exosome applications strictly for clinical and pharmaceutical pathways 5539. Meanwhile, China's National Medical Products Administration (NMPA) recently integrated extracellular vesicles into its own ATMP framework, explicitly prohibiting the use of human-derived exosomes in commercial cosmetics, while classifying exosome-infused medical dressings as strictly regulated Class III medical devices 5539. In Taiwan, the Ministry of Health and Welfare (TFDA) has similarly banned human-derived exosomes in general cosmetics, allowing exemptions only for specifically approved case reviews 39. India governs extracellular vesicles under the Drugs and Cosmetics Act of 1940, treating them as biological medicinal products requiring stringent oversight from the Central Drugs Standard Control Organisation (CDSCO) 37.
| Jurisdiction / Agency | Primary Regulatory Classification for Exosomes | Commercial Cosmetic Status |
|---|---|---|
| United States (FDA) | Biological Product / Unapproved New Drug 42 | Strictly prohibited if human-derived; Plant-derived allowed 36 |
| European Union (EMA) | Advanced Therapy Medicinal Product (ATMP) 3755 | Prohibited if human/animal-derived 39 |
| United Kingdom (MHRA) | Biological Medicinal Product / ATMP 3755 | Regulated under general EU/UK cosmetic bans 37 |
| Japan (PMDA) | Regenerative Medicinal Product (Conditional Approval) 55 | Restricted under specific safety frameworks 38 |
| South Korea (MFDS) | Biological Product / Regenerative Medicine 55 | Explicitly banned as a cosmetic ingredient 39 |
| China (NMPA) | Advanced Therapy Medicinal Product (ATMP) 39 | Prohibited in cosmetics; Dressings are Class III devices 39 |
Documented Safety Risks and Adverse Events
The central justification for stringent global regulation is the preservation of patient safety. While the theoretical biological profile of exosomes is highly biocompatible and immune-tolerant, the practical administration of unregulated, mass-produced biological fluids carries severe, documented risks.
Contamination and Manufacturing Failures
Because human exosomes are derived from living human tissues or continuous cell cultures, the manufacturing process is highly vulnerable to contamination by bacteria, viruses, or fungi 3840. Traditional sterilization methods utilized for standard pharmaceuticals, such as intense heat or irradiation, destroy the fragile lipid membranes of the vesicles, rendering them biologically inert 38. Therefore, exosome production requires pristine, highly advanced aseptic processing.
When clinics purchase unregulated products from non-compliant laboratories, the results can be catastrophic. The FDA and the Centers for Disease Control and Prevention (CDC) have issued severe public safety alerts following clusters of adverse events. Notably, a cluster of incidents in Nebraska linked unapproved exosome products to severe bacterial infections, resulting in the hospitalization of 19 patients across eight states 54041. In severe cases documented by the FDA, the systemic administration of unapproved regenerative products has resulted in blindness, extreme immune reactions, and the formation of tumors 54241. Additionally, if the source cells are not meticulously screened, there is a theoretical risk that exosomes derived from mutated or pre-cancerous cells could transfer oncogenic signaling molecules, actively promoting tumor growth in the recipient 363840.
Cutaneous Complications from Off-Label Administration
Even in localized, aesthetic applications, profound risks persist. While exosomes are generally recognized as safe for superficial topical application immediately following laser or microneedling treatments (where they remain on the surface or enter shallow micro-channels), the direct intradermal injection of unapproved cosmetic exosome formulations is highly dangerous.
A 2025 clinical case series from South Korea documented four adult patients who developed severe, persistent cutaneous adverse reactions after receiving intradermal injections of cosmetic exosome products in non-clinical settings (such as beauty salons) 4243. These patients suffered from persistent erythema, deep nodules, granulomatous inflammation, and permanent scarring 4243. Treatments to resolve these severe complications required systemic and intralesional corticosteroids, ablative laser therapy, and surgical excision 4243. Even with intensive dermatological intervention, all patients experienced incomplete resolution and residual scarring, underscoring the absolute necessity for medical supervision and strict adherence to approved, topical routes of administration for cosmetic products 4243.
Conclusion
Extracellular vesicle therapy represents one of the most compelling and rapidly evolving frontiers in biogerontology and regenerative medicine. By harnessing the precise intercellular communication networks that natively govern tissue repair, and by delivering specific epigenetic payloads like microRNAs to stimulate mitochondrial biogenesis, exosomes offer a biologically plausible mechanism for the systemic reversal of age-related functional decline. Early preclinical evidence, particularly regarding massive lifespan extension in mammalian models, is profound. Furthermore, early clinical data in dermatology and orthopedics indicates that acellular vesicle therapy can match or exceed the regenerative benefits of live stem cell transplantation, while successfully circumventing the risks of cellular replication and immune rejection.
However, the current commercial landscape is dominated by extreme marketing hype that vastly outpaces clinical reality. The overwhelming majority of exosome products currently sold direct-to-consumer lack standardized quality control, possess unverified or compromised biological activity, and operate entirely outside the legal boundaries of FDA authorization. The immense technical challenges of isolating pure, functional vesicles at scale, combined with the rapid systemic clearance of these particles by the immune system in vivo, dictate that true systemic anti-aging therapies for humans remain years away from validated, approved clinical use.
For the field to transition successfully from an unregulated medical tourism gray market to mainstream, evidence-based medical science, industry stakeholders must prioritize the rigorous MISEV2023 characterization standards, pursue legitimate Investigational New Drug clinical pathways, and openly acknowledge the current biological and regulatory limitations of the modality. Until such comprehensive evidence is finalized and regulatory approvals are granted, extracellular vesicle therapy for aging remains a highly promising, yet fundamentally investigational, scientific frontier.