Evolutionary trade-off between longevity and reproduction
Introduction to Evolutionary Gerontology
Biological aging, defined scientifically as senescence, is characterized by a progressive, age-dependent decline in physiological integrity and cellular function, leading to an impaired response to environmental stress and an exponentially increasing vulnerability to death. From a strict evolutionary perspective, the ubiquitous presence of aging across most multicellular organisms presents a fundamental theoretical paradox. Natural selection consistently favors morphological and physiological traits that optimize an organism's survival and reproductive success. Given this optimization process, the universal, systematic deterioration of the adult organism appears inherently maladaptive 123.
Early attempts to resolve this evolutionary paradox, most notably championed by August Weismann in the late nineteenth century, relied heavily on group selection frameworks. Weismann initially posited that aging evolved as a programmed, adaptive mechanism intended to remove older, post-reproductive individuals from a population. This controlled demographic turnover, he argued, was necessary to free up finite environmental resources for younger, potentially more adaptable members of the species, thereby accelerating the evolutionary adaptability of the group as a whole 145.
However, the advent of the modern evolutionary synthesis in the mid-twentieth century largely dismantled these programmed, group-selection explanations for senescence. Evolutionary biologists, including George C. Williams and John Maynard Smith, demonstrated through rigorous population genetics that selection operates overwhelmingly at the level of the individual and their constituent genes, not at the level of the group or species 1677. Because the vast majority of organisms in wild, unmanaged populations succumb to extrinsic mortality factors - such as predation, infectious disease, exposure to harsh climates, and starvation - long before they ever reach an advanced chronological age, natural selection has virtually no opportunity to select directly for or against phenotypic traits that only manifest late in life 8910. The statistical probability of surviving to experience the deleterious effects of aging in the wild is minimal. Consequently, any genetic mutation that provides an immediate, early-life advantage to individual reproduction will rapidly spread through a population, even if that same gene carries severe survival penalties in later life, simply because the long-term penalties are rarely realized 237.
This paradigm shift firmly established that aging is not an active, adaptive genetic program designed to cull the elderly, but rather a non-adaptive by-product resulting from the inexorable decline in the force of natural selection with advancing chronological adult age. This consensus rests upon three classical, though interconnected, evolutionary frameworks: the Mutation Accumulation (MA) theory proposed by Peter Medawar, the Antagonistic Pleiotropy (AP) theory formalized by George C. Williams, and the Disposable Soma Theory (DST) developed by Thomas B. L. Kirkwood 12311.
While the Mutation Accumulation and Antagonistic Pleiotropy theories provide necessary population-genetic explanations for why senescence evolves, the Disposable Soma Theory provides the physiological and biochemical mechanisms. First articulated in 1977, the DST posits that aging is the inevitable, mechanical result of an evolutionary trade-off in the allocation of finite metabolic resources between reproduction and the maintenance of somatic tissues 12131415.
Origins and Core Principles of the Disposable Soma Theory
The foundational logic of the Disposable Soma Theory emerged not from classical ecology, but from mechanistic studies of cellular aging. In 1963, biochemist Leslie Orgel proposed the Error Catastrophe Theory of Aging, hypothesizing that senescence was triggered by a self-amplifying cascade of errors in the molecular machinery responsible for cellular transcription and translation 1215. While evaluating Orgel's theory, Thomas Kirkwood sought to reconcile these cellular mechanics with the overarching principles of evolutionary genetics. Kirkwood recognized that cells possess highly effective mechanisms for molecular proofreading and the excision of damaged DNA and misfolded proteins. However, the operation of these high-fidelity repair systems demands an immense expenditure of metabolic energy 1316.
The central tenet of the Disposable Soma Theory is that all organisms exist in environments characterized by finite resources - specifically energy and nutrients acquired through foraging. An organism must continuously partition these limited metabolic resources among competing, essential physiological demands: somatic growth, fat storage, reproductive effort, and continuous somatic maintenance 121317. Somatic maintenance encompasses a vast array of life-preserving biochemical functions, including DNA excision repair, the synthesis of antioxidant enzymes to neutralize reactive oxygen species (ROS), the maintenance of stem cell pools, immune system vigilance, and general protein homeostasis (proteostasis) 121519.
To preserve the continuity of a species across infinite generations, the organism is evolutionarily mandated to invest massive resources into maintaining the absolute genetic stability of the germline (the cellular lineage that produces eggs and sperm). However, the somatic cells - comprising the physical body that houses and protects the germline - only need to survive long enough to ensure the successful transmission of genetic material to the next generation 121415. Under the DST framework, the soma is evolutionarily "disposable" once its primary objective of maximizing reproductive fitness has been achieved 121415.
The Ecological Determinants of Energy Allocation
Evolutionary logic, driven by the realities of extrinsic environmental mortality, dictates that investing infinite metabolic energy into perpetual somatic repair is a mathematically unsound strategy. In wild ecosystems, animals face constant, stochastic threats from predators, pathogens, and environmental hazards. These extrinsic mortality factors establish a strict statistical maximum on an organism's life expectancy, entirely independent of any internal biological aging process 8141519.
If an organism faces high extrinsic mortality - for example, a small rodent that is highly likely to fall victim to a predator within its first year of life - any metabolic energy invested in building a durable, long-lasting body capable of surviving for twenty years is functionally wasted. The genetic lineage of a mouse that invests heavily in DNA repair at the expense of early reproduction will be rapidly outcompeted by a mouse that invests everything into maturing quickly and producing large litters before a predator inevitably kills it 8121419.
Natural selection ultimately optimizes the allocation of energy to maximize total lifetime reproductive output. The mathematically optimal strategy is to partition just enough resources to somatic maintenance to keep the organism in adequate physiological condition through its expected natural lifespan, while aggressively diverting the remainder of the energy budget to accelerate growth and maximize reproductive capacity 131415.
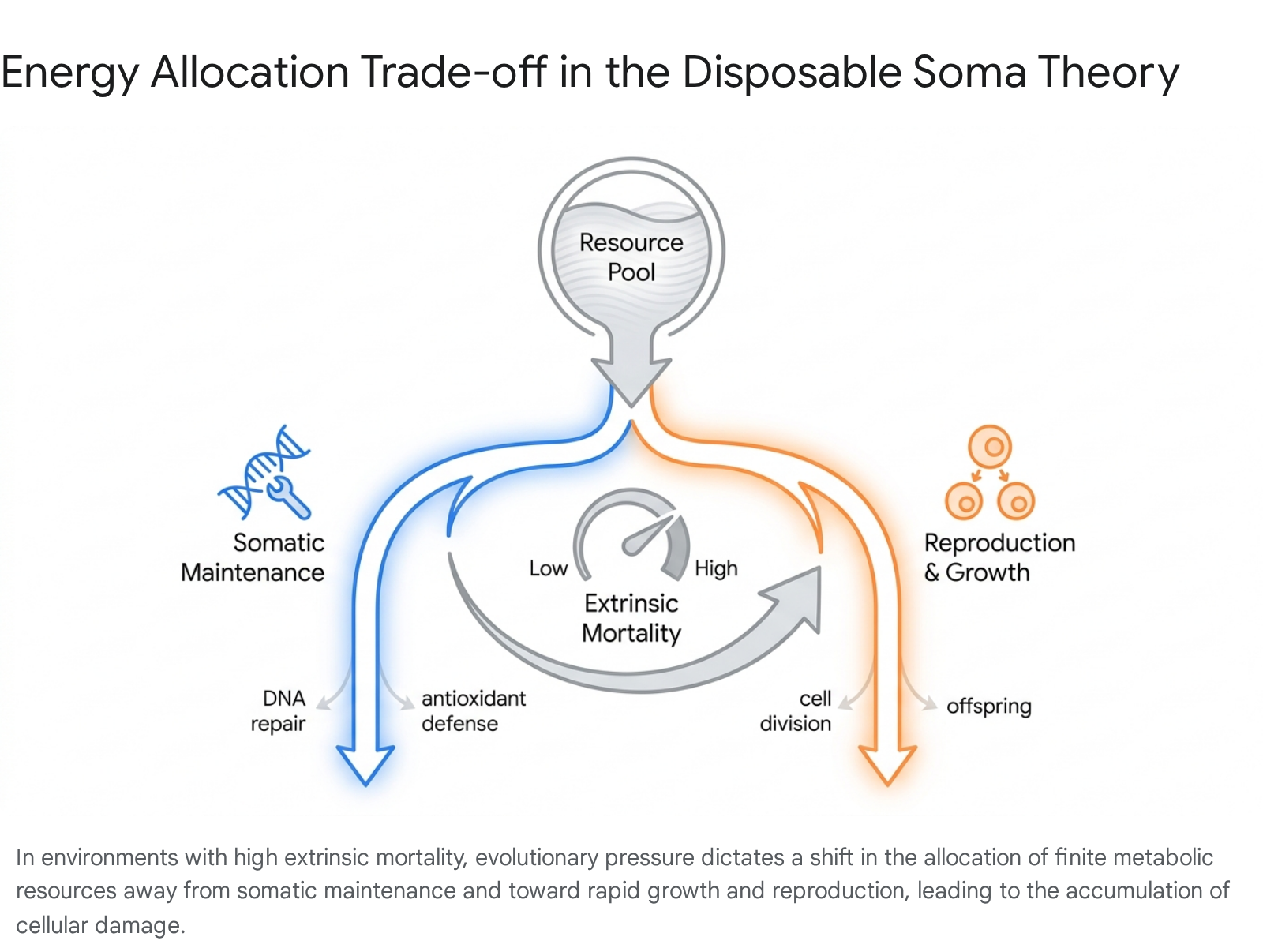
As a direct and unavoidable consequence of this sub-maximal, compromised investment in cellular repair, subtle molecular damage - such as oxidative damage to proteins, accumulation of genetic mutations, telomere attrition, and the depletion of functional stem cell niches - gradually accrues over time. This progressive, uncorrected accumulation of systemic damage is what manifests phenotypically as aging 1216.
Conversely, populations that evolve in highly protected environmental niches subject to low extrinsic mortality face different evolutionary pressures. Species that are insulated from predation - such as birds capable of flight, bats, or genetically isolated species residing on predator-free islands - experience a higher probability of long-term survival. Without the intense pressure of imminent extrinsic death, it becomes evolutionarily advantageous for these organisms to allocate a significantly larger proportion of their metabolic resources toward rigorous somatic maintenance 1218. This heightened investment in cellular repair limits early offspring mortality and allows the organism to engage in multiple reproductive bouts over an extended timeframe. As predicted by the Disposable Soma Theory, such isolated, low-mortality populations predictably evolve delayed primiparity (later age of first reproduction), lower rates of free radical generation, superior DNA repair mechanisms, and a vastly increased maximum lifespan 1221.
Mathematical Formalization: Dynamic Energy Budget Theory
To transition the conceptual framework of the Disposable Soma Theory into a rigorous, testable mathematical model, researchers frequently employ the Dynamic Energy Budget (DEB) theory. Developed as a formal metabolic theory, DEB provides a universal quantitative framework based on thermodynamic principles and mass-balance equations to describe how individual organisms assimilate and utilize resources across their entire life cycle 192021.
Within the standard DEB modeling architecture, energy acquired from food does not immediately translate into growth. Instead, assimilated energy enters a centralized physiological compartment termed the "reserve" 172022. The mobilization and subsequent utilization of this energy reserve are strictly governed by the "kappa-rule" ($\kappa$-rule). The parameter $\kappa$ dictates a fixed fraction of the mobilized energy flux that is relentlessly prioritized and allocated to somatic maintenance and structural growth. Under DEB assumptions, somatic maintenance represents an obligatory, demand-driven process; it must be paid first 171920. If the $\kappa$ fraction of mobilized energy becomes insufficient to cover basic somatic maintenance costs, structural growth ceases entirely, and the organism eventually faces starvation and death 1723.
The remaining complementary fraction of mobilized energy, defined mathematically as $1 - \kappa$, is routed toward a parallel metabolic pathway governing maturity maintenance and reproduction. During the embryonic and juvenile stages - when the organism feeds but cannot yet reproduce - this $1 - \kappa$ fraction drives developmental maturation, fueling the increase in physiological complexity and the installation of endocrine and reproductive regulation systems 202327. When the accumulated energy invested into this maturation pathway reaches a specific biological threshold (defined as puberty), the organism transitions to adulthood. At this point, the $1 - \kappa$ energy flux is permanently redirected from maturation into the active production of gametes and reproductive tissues 17202724.
The DEB model effectively translates the evolutionary trade-offs of the Disposable Soma Theory into specific, measurable physiological parameters. Species subjected to severe ecological pressures and high extrinsic mortality are modeled with life history strategies utilizing lower functional $\kappa$ values over time, forcing a massive diversion of metabolic reserves into the $1 - \kappa$ reproductive channel at the direct expense of long-term somatic structural durability 12152227. Some extensions of DEB theory adapt the fixed $\kappa$-rule to accommodate specific biological phenomena, such as hyperallometric scaling of reproductive energy in mature fish, or the cyclical, stage-specific energy allocations required for crustacean molting 2227. Regardless of the specific mathematical adjustments, the underlying logic remains consistent with Kirkwood's framework: energy utilized for creating the next generation is energy definitively stolen from the preservation of the current organism 19222725.
Molecular Mechanisms Mediating the Allocation Trade-off
When the Disposable Soma Theory was initially proposed, the precise intracellular mechanics responsible for measuring energy and shifting metabolic priorities were unknown. However, recent advances in molecular biology have identified a network of highly conserved, nutrient-sensing cellular signaling pathways that act as the definitive biological levers of this evolutionary trade-off. Chief among these regulatory systems are the mechanistic Target of Rapamycin (mTOR) pathway, the AMP-activated protein kinase (AMPK) pathway, and the Insulin/IGF-1 signaling (IIS) cascade 263127.
The mTOR and AMPK Regulatory Axis
The mechanistic Target of Rapamycin (mTOR) functions as the master anabolic switch within eukaryotic cells. Operating primarily within two distinct protein complexes (mTORC1 and mTORC2), mTOR intricately coordinates cellular growth, metabolism, and proliferation by integrating upstream signals regarding environmental inputs, including the presence of growth factors, systemic energy status (ATP levels), oxygen availability, and the abundance of specific nutrients, particularly essential amino acids like leucine 31272829.
In environments where nutrients are abundant, mTORC1 is heavily activated. This activation drives high-energy cellular processes required for rapid growth, muscle hypertrophy, and peak reproductive function, including the translation of mRNA and the synthesis of proteins, lipids, and nucleotides 2729. However, the activation of mTORC1 simultaneously triggers the potent suppression of essential catabolic and somatic maintenance processes. Most critically, active mTORC1 phosphorylates and inhibits ULK1 (unc-51 like autophagy activating kinase 1), effectively shutting down autophagy 312729. Autophagy is the vital intracellular housekeeping mechanism responsible for identifying, degrading, and recycling damaged organelles, misfolded proteins, and toxic metabolic by-products. By suppressing autophagy, chronic mTOR activation allows cellular damage to accumulate unchecked 3127.
Conversely, under conditions of nutrient scarcity, caloric restriction, or elevated physical exertion, the body's primary energy sensor, AMPK (AMP-activated protein kinase), is triggered by rising levels of cellular AMP. AMPK actively antagonizes the growth pathway by directly inhibiting mTORC1 and simultaneously phosphorylating activating sites on ULK1, thereby derepressing autophagy and shifting the cell from a state of rapid growth into a highly conservative state of intense somatic repair and cellular recycling 312728.
This biochemical antagonism perfectly mirrors the macro-evolutionary predictions of the Disposable Soma Theory. At a fundamental cellular level, an organism cannot simultaneously execute the metabolic programs for rapid, competitive growth (mTOR-driven anabolism) and rigorous somatic maintenance (AMPK-driven autophagy) 3135. In the context of modern human environments - characterized by sedentary lifestyles and the constant availability of high-calorie nutrition - the mTOR pathway remains chronically activated. This continuous, unyielding state of nutrient abundance traps the cells in a perpetual "growth" phase, suppressing vital autophagic repair and accelerating the systemic deterioration associated with aging, insulin resistance, and metabolic disease 3135.
Insulin/IGF-1 Signaling and Forkhead Box O3 (FOXO3)
The Insulin/IGF-1 signaling (IIS) pathway represents another deeply conserved neuroendocrine axis that intimately links nutrient status, growth, and maximum lifespan across taxa ranging from invertebrates to mammals 263031. Mutations or interventions that gently reduce the functionality of the IIS pathway consistently result in the most robust and highly reproducible extensions of healthspan and maximum lifespan observed in biological research 2630.
A critical downstream target mediating the longevity effects of the IIS pathway is a family of transcription factors known as Forkhead box O (FoxO). In mammals, FOXO3 acts as a central genetic executor of somatic maintenance. Under conditions of low insulin signaling or environmental stress, FOXO3 proteins avoid phosphorylation and freely translocate into the cell nucleus. Once there, FOXO3 binds to specific DNA enhancer sequences, orchestrating a massive, protective genetic program 313233.
FOXO3 directly upregulates a vast suite of target genes dedicated to extreme cellular resilience. It promotes the transcription of ROS scavengers (such as superoxide dismutase and catalase) to manage oxidative stress, initiates cell cycle arrest to allow time for DNA repair mechanisms to function, activates critical autophagy effectors (e.g., Pink1, Bnip3), and regulates the delicate homeostasis and self-renewal capacity of adult stem cell pools 31323340. Furthermore, during viral challenges or severe, irreparable cellular damage, FOXO3 triggers controlled cellular apoptosis (via genes like PUMA and Bim) to prevent the spread of dysfunction and reduce systemic tissue inflammation 3233.
The immense importance of this maintenance pathway is underscored by human genomic data. While the exact contribution of genetics to human longevity varies, specific single-nucleotide polymorphisms (SNPs) within the FOXO3 gene (such as rs2802292 and rs10457180) are among the extremely few genetic variants consistently and strongly associated with individuals living past 95 or 100 years of age across highly diverse global populations (e.g., Japanese, European, and American cohorts) 32334034. By sitting at the interface of stress detection and broad-spectrum genetic repair, FOXO3 acts as the ultimate molecular switch routing finite energy into the somatic maintenance branch predicted by the Disposable Soma Theory 3340.
Comparative Analysis of Classical Evolutionary Theories of Aging
To properly contextualize the impact and limitations of the Disposable Soma Theory, it must be carefully delineated from the other classical pillars of evolutionary gerontology: the Mutation Accumulation (MA) theory and the Antagonistic Pleiotropy (AP) theory. While all three models fundamentally agree that senescence results from a drastic decline in the force of natural selection at older ages, their proposed mechanistic drivers are entirely distinct.
Table 1: Comparison of Classical Evolutionary Theories of Aging
| Characteristic | Mutation Accumulation (MA) | Antagonistic Pleiotropy (AP) | Disposable Soma Theory (DST) |
|---|---|---|---|
| Primary Proponent | Peter Medawar (1952) | George C. Williams (1957) | Thomas B. L. Kirkwood (1977) |
| Core Mechanism | Passive accumulation of deleterious genetic mutations that only exert their effects in late, post-reproductive life. | Active evolutionary selection for pleiotropic alleles that confer early-life reproductive benefits but carry late-life costs. | Physiological, energetic trade-off between the allocation of finite resources to reproduction versus somatic tissue repair. |
| Nature of Genetic Action | Mutations are functionally neutral during early development; they escape selective pressure due to the "selection shadow." | A single gene possesses multiple, opposing phenotypic effects across the organism's life course. | Operates via broad, complex regulatory networks (e.g., mTOR, IIS) governing systemic energy metabolism and budgeting. |
| Evolutionary Driver | Genetic drift in wild populations where very few individuals survive to old age. | Direct selective advantage of robust early-life vitality, rapid maturation, and maximum early fecundity. | High extrinsic environmental mortality rendering long-term, indefinite somatic investment biologically futile. |
| Testable Predictions | Additive genetic variance and inbreeding depression should increase dramatically with advancing chronological age. | Negative genetic correlations should exist between early-life fecundity and late-life survival metrics. | Direct, positive correlations should exist between species lifespan, cellular DNA repair capacity, and antioxidant defenses. |
| Analytical Level | Strict Population Genetics. | Strict Population Genetics. | Evolutionary Physiology and Bioenergetics. |
23113536373839
In the broader literature, the Disposable Soma Theory is frequently described merely as a specialized physiological subset of Antagonistic Pleiotropy. Technically, the complex genetic architecture that commands the shift of metabolic resources toward aggressive early reproduction at the definitive expense of somatic maintenance does exhibit antagonistic pleiotropy: the genes boost early-life reproductive fitness while strictly guaranteeing late-life physical deterioration 33740.
However, leading theorists point out that DST diverges fundamentally from AP in its approach. The Disposable Soma Theory represents an optimality approach grounded in macroscopic metabolic economics, addressing the energetic costs of broad functions like DNA proofreading and immune surveillance. In contrast, Antagonistic Pleiotropy is traditionally a quantitative genetics approach, relying on the identification of specific, discrete alleles that harbor hardwired, dual-action phenotypic effects regardless of available metabolic energy 3637. Both theories are currently considered valid, complementary mechanisms that likely operate simultaneously within populations 43537.
Empirical Challenges and the Caloric Restriction Paradox
Comparative physiological biology provides a wealth of correlative evidence supporting the DST. Across highly divergent taxa, animal species with longer maximum lifespans generally exhibit demonstrably superior somatic maintenance mechanisms. This includes highly efficient DNA excision repair, substantially lower rates of free radical and ROS generation within mitochondria, and robust cellular antioxidant defenses 1519. Furthermore, targeted artificial selection experiments using the fruit fly Drosophila melanogaster have shown that selectively breeding lines for delayed reproduction directly leads to the evolution of significantly longer lifespans, while selecting for rapid early reproduction correspondingly shortens lifespan, exactly matching the reproduction-maintenance trade-off predicted by the DST 121536.
Despite this foundational support, modern empirical data sets have heavily challenged the universal applicability of the DST's core predictions, sparking intense academic debate.
The 2024 PNAS Reproduction Trade-off Study
A non-negotiable, central prediction of the strict Disposable Soma Theory is that energetic investments made into reproductive events forcefully deplete the resources available for somatic maintenance. This physiological diversion is expected to accentuate systemic cellular damage, permanently increase the rate of senescence, and ultimately shorten the lifespan of the breeding individual 25414243.
In a highly comprehensive 2024 study published in the Proceedings of the National Academy of Sciences (PNAS), researchers Speakman, Mitchell, and colleagues tested this hypothesis directly. The team conducted a lifetime analysis of over 120 breeding and non-breeding female C57BL/6J mice, rigorously quantifying reproductive output, dynamic body composition, total daily energy expenditure, multiple biomarkers of oxidative stress, and lifetime survival trajectories 41424344.
The results starkly contradicted DST predictions. The data confirmed that reproduction indeed carried an immediate, acute risk of mortality - specifically, sudden death due to mechanical complications encountered during parturition (the birthing process). Consequently, the survival curve for the breeding cohort dropped significantly during the active reproductive phase 4143. However, for those females that successfully survived the breeding period, there was absolutely no statistically significant residual effect on their subsequent lifespan. Once reproduction ceased, the breeding mice lived just as long, and aged at the same rate, as the virgin control group 414344.
Furthermore, extensive biochemical testing - conducted precisely at the peak of lactation, the period of absolute maximum energetic strain - revealed an overwhelming lack of evidence for elevated oxidative stress across eleven different tissue types. There were no significant correlations between a female's lifetime reproductive output and the accumulation of damage to proteins or DNA, nor were there compensatory changes in antioxidant enzyme levels (catalase, SOD, GPx) in vital organs like the liver 4143. The authors concluded that the higher mortality seen in reproducing females is purely a direct mechanical consequence of birthing, and does not carry the long-term physiological deterioration or trade-off in somatic maintenance predicted by the DST, thereby questioning the fundamental physiological basis of the theory 414344.
Defenders of the DST counter these findings by highlighting the artificiality of laboratory conditions. Laboratory mice are housed in perfectly insulated, predator-free environments with unrestricted, ad libitum access to highly nutritious food. Under conditions of limitless energetic abundance, the mice are never forced to choose between allocating energy to a litter or repairing their DNA; they have ample calories to do both. Therefore, the evolutionary trade-off forged over millions of years of resource scarcity may be completely masked in a laboratory setting 2541.
The Hyperfunction Theory Critique
Another significant area of debate arises from the physiological phenomenon of Caloric Restriction (CR). Across diverse species, reducing caloric intake by roughly 30% to 40% (without causing malnutrition) consistently and robustly extends both healthspan and maximum lifespan 254546.
According to a strict, literal interpretation of the Disposable Soma Theory's energetic logic, an organism subjected to a severe reduction in its overall energy supply should have fewer metabolic resources available to fund expensive somatic repair mechanisms. This enforced energy deficit should logically accelerate aging. Yet, empirically, CR delays aging 254546. To reconcile this, DST proponents argue that during severe food scarcity, the organism senses that bringing offspring into a harsh environment is futile. It therefore completely shuts down the highly expensive reproductive channel, diverting every remaining fraction of its limited energy budget directly into extreme somatic maintenance, hoping to outlive the famine 25.
However, critics like Mikhail Blagosklonny argue that this explanation is convoluted and paradoxical. Blagosklonny's alternative "Hyperfunction Theory" posits that aging is not a passive process of damage accumulation caused by a lack of repair resources. Instead, he views aging as a destructive, quasi-programmed process driven by the blind, aimless continuation of developmental growth pathways - specifically the nutrient-sensing mTOR pathway - long after the organism has reached adulthood 4547. Under the hyperfunction model, CR extends lifespan not by ingeniously reallocating a scarce energy budget toward DNA repair, but simply by starving and deactivating the hyper-functional, gerogenic mTOR pathway that actively drives cellular senescence 4547.
Boundary Conditions: Negligible Senescence in Nature
A definitive test of the validity of any gerontological theory is its ability to explain outlier species that defy the biological norm. Certain organisms exhibit a phenomenon known as "negligible senescence." These animals display absolutely no observable decline in physiological capacity, no reduction in reproductive output, and no statistical increase in mortality rate as they age 8484957. Notable examples include the Hydra (a freshwater polyp), the naked mole-rat (Heterocephalus glaber), and the Greenland shark (Somniosus microcephalus).
According to the DST, a complete absence of aging suggests an organism has evolved a life-history strategy that allocates sufficient metabolic resources to achieve 100% perfect somatic maintenance, repairing all systemic damage as rapidly as it occurs 1850. In 2024, researchers developed highly sophisticated mathematical models of the DST to identify the precise theoretical conditions under which natural selection would favor such total agelessness. The models revealed that evolution can select for the complete removal of a specific type of somatic damage only if two strict criteria are met: the mortality risk posed by the unchecked damage must be incredibly severe, and the metabolic energy required to execute the repair cannot impose an excessively large penalty on the organism's reproductive rate 1850.
The models further indicated that while agelessness is mathematically possible in very narrow biological contexts - such as in simple organisms like Hydra or Planaria that utilize highly pleiotropic repair processes (e.g., constant asexual fission and stem cell regeneration) - it is almost impossible to achieve in highly complex mammals. In complex organisms characterized by dozens of independent, differentiated organ systems, the sheer variety of damage types requires multiple, independent repair processes. The cumulative energetic cost of funding perfect repair across all these systems would incur an evolutionary penalty to reproduction that is simply too massive to survive natural selection 1850.
However, the genomic reality of exceptionally long-lived vertebrates illuminates the immense evolutionary mechanisms required to approach this threshold of negligible senescence.
The Greenland Shark
The Greenland shark is currently recognized as the longest-lived vertebrate on Earth, with a conservatively estimated lifespan exceeding 270 years and potentially reaching up to 500 years 5759. Its extreme longevity is coupled with physical traits indicating a profoundly low-energy life history: it inhabits the freezing depths of the North Atlantic and Arctic Oceans, exhibits an extremely slow mass-specific metabolic rate, and has a cruising speed of less than one meter per second 5759.
Remarkably, physiological assays of Greenland shark muscle tissue across various age cohorts show no significant variation or decline in metabolic enzyme activity with age, indicating that their baseline cellular metabolism does not deteriorate over centuries 59. Furthermore, despite living with heavy burdens of ocular parasites, molecular and histological examinations of sharks aged over 130 years reveal perfectly preserved, functional retinal structures adapted to 458-nanometer blue wavelength light, with no signs of age-related neural degeneration 51.
A 2024 chromosome-level assembly of the massive 6.45 Gb Greenland shark genome provided the genetic rationale for this extreme somatic durability. The genome contains a vast expansion of transposable elements (such as LTRs and LINEs) 57. Because high transposable element activity continuously threatens to cause catastrophic double-strand DNA breaks, the shark was forced to evolve compensatory mechanisms. Researchers identified 81 genes specifically duplicated in the Greenland shark that form a highly connected network entirely dedicated to DNA double-strand break repair. This includes multiple functional copies of the critical RAD51 gene, created via retrotransposition 57. Furthermore, key tumor suppressor and longevity genes show distinct adaptations; the TP53 gene (p53) features a unique three-lysine amino acid insertion that likely stabilizes its alpha helix, while genes like PARP14 (DNA repair) and MRPS35 (mitonuclear protein balance) show signatures of strong positive selection 57.
The Naked Mole-Rat
The naked mole-rat is a unique, subterranean murine species that exhibits extraordinary longevity, frequently exceeding 38 years in captivity - an age roughly ten times greater than similarly sized rodents like mice. Throughout this extended lifespan, naked mole-rats show almost no signs of reproductive decline, neurodegeneration, or susceptibility to cancer 484952.
The mole-rat's resistance to senescence is linked to extreme somatic investments in molecular fidelity. They exhibit drastically improved protein homeostasis (proteostasis) and enhanced translational fidelity compared to shorter-lived mammals, possessing a unique 28S ribosomal RNA cleavage that minimizes protein misfolding 485253. Additionally, aging is often driven by the progressive dysregulation of alternative splicing (AS). Analyses of naked mole-rat brain and spleen tissues reveal that their expression of splicing regulatory factors is maintained at levels nearly double that of mice, and these expression patterns remain remarkably stable and unchanged for at least two decades of adult life 49.
Both the Greenland shark and the naked mole-rat inhabit highly specialized ecological niches - the extreme deep ocean and heavily fortified, cooperative eusocial subterranean colonies, respectively. These environments are characterized by exceedingly low rates of extrinsic mortality from predation 59515253. This perfectly aligns with the macroscopic prediction of the Disposable Soma Theory: deep evolutionary safety from external hazards provides the necessary conditions for natural selection to heavily favor extreme energetic allocations toward specialized somatic maintenance programs.
Human Evolutionary Energetics and Post-Reproductive Survival
Human life history presents a series of unique evolutionary paradoxes that challenge classical life history models. Humans grow slower than expected for hominoids of equivalent size, yet reproduce faster. Most perplexingly, human females exhibit a uniquely extended post-reproductive lifespan, routinely surviving for decades in vigorous physiological condition after experiencing menopause and total reproductive cessation 5455.
According to a strict, individual-level interpretation of the Disposable Soma Theory and Antagonistic Pleiotropy, once an individual definitively ceases to reproduce, their direct Darwinian fitness drops to zero. Therefore, any metabolic energy expended on somatic maintenance beyond the age of last reproduction represents a catastrophic evolutionary waste 565758. Some early theories suggested that human menopause was simply an artifact of modern medicine artificially extending lifespan beyond the physiological viability limit of mammalian oocytes 5558. However, data from wild chimpanzee populations, such as those in the Ngogo community, confirm hormonal signatures of menopause at ages nearly identical to humans, indicating a shared evolutionary basis rather than a modern medical artifact 55.
To explain the selective advantage of extended human post-reproductive survival, evolutionary anthropologists rely on the Grandmother Hypothesis, heavily elaborated by Kristen Hawkes based on foundational concepts from George C. Williams 57585969. The hypothesis posits that during the ecological shifts of the Plio-Pleistocene era, the human diet became increasingly reliant on complex, deeply buried, or difficult-to-process foods (like deep tubers). This dietary shift extended the period of juvenile nutritional dependence 5558.
In this demanding ecological context, older females faced increasing risks with continuous childbirth. It became evolutionarily advantageous for them to cease their own direct, risky reproductive efforts and instead redirect their remaining energetic capacity toward provisioning, foraging for, and protecting their weaned grandchildren 555869. Observational data from modern hunter-gatherer societies, such as the Hadza of Tanzania, confirm that grandmothers are critical providers of energy-dense tubers, directly improving the health and survival rates of their grandchildren. This intense alloparenting allows their daughters to prematurely cease lactation and resume reproduction sooner, drastically increasing the family lineage's total lifetime reproductive output 575969.
The Grandmother Hypothesis forces a critical expansion of the Disposable Soma Theory's energetic calculus. Rather than viewing the energy budget solely at the isolated level of a single reproducing individual, human life history necessitates the concept of a "pooled energy budget" across an extended kin network or breeding community 54. The somatic maintenance of a post-menopausal grandmother is heavily favored by natural selection because her durable, healthy "soma" acts as a highly efficient engine for intergenerational energy transfer 405460.
Extensive demographic analyses, including massive datasets of 17th and 18th-century French-Canadian settlers, provide robust quantitative support for this mechanism. Living, co-resident maternal grandmothers allowed their daughters to increase their total number of children by an average of two, and increased the number of children surviving to age 15 by one. However, the evolutionary benefits of this "grandmother effect" have limits; the demographic data show that the survival benefits provided to grandchildren waned significantly when grandmothers passed the age of 75, or when geographic distance prevented direct interaction 61.
The genetic architecture supporting this uniquely human extension of robust somatic maintenance involves the powerful selection of specific longevity-associated alleles, such as the previously discussed variants in the FOXO3 and APOE genes. These genes actively buffer the older individual against metabolic, inflammatory, and cardiovascular decline, ensuring they remain physically and cognitively capable of executing complex foraging and social behaviors long after direct reproduction has ceased 323458.
Towards a Pluralistic Interpretation of Aging
The Disposable Soma Theory remains one of the most elegant, unifying, and influential frameworks in the history of evolutionary gerontology. By explicitly grounding the abstract, statistical forces of natural selection in the tangible, physical realities of thermodynamics, cellular metabolism, and physiological constraints, the DST successfully answers why the vast majority of living species inevitably deteriorate with age: achieving biological perfection is simply too metabolically expensive in a dangerous world 121347.
However, the rapid expansion of modern molecular biology, genomics, and advanced experimental gerontology necessitates a transition toward a "pluralistic interpretation" of the evolution of aging 1. The pluralistic framework acknowledges that no single, rigid theory can account for the staggering diversity of aging patterns observed across all taxa. The three classical theories - Mutation Accumulation, Antagonistic Pleiotropy, and the Disposable Soma Theory - are not mutually exclusive competitors. Rather, they operate simultaneously, synergistically, and with varying degrees of intensity depending entirely on the specific organism, its unique ecological niche, and its evolutionary life history 135.
As evidenced by the 2024 empirical challenges in mammalian models, the precise mechanical nature of the trade-off between reproduction and survival is far more complex than originally envisioned, and may be masked, bypassed, or heavily modified by varying states of nutrient availability and stress 14143. Furthermore, the discovery of highly conserved, nutrient-sensing regulatory networks like mTOR and the FOXO family of transcription factors has successfully bridged the theoretical gap between macro-evolutionary ecology and microscopic cellular biochemistry, proving conclusively that aging is driven by active, malleable signaling networks that mediate the delicate daily balance between rapid anabolism and protective somatic repair 3131.
Ultimately, while the rigid boundaries and absolute predictions of the classic Disposable Soma Theory are being continuously tested, refined, and occasionally contradicted by modern genomics and experimental biology, its core foundational premise - that biological longevity is fundamentally dictated by the unforgiving evolutionary economics of resource allocation - remains an indispensable tool for understanding the biology of aging.