Evolutionary trade-off between reproduction and longevity
Evolutionary Context and Theoretical Origins
The biological phenomenon of aging presents a fundamental paradox within the framework of evolutionary biology. If natural selection rigorously optimizes biological systems for survival and reproductive success, the ubiquitous existence of senescence - a generalized, progressive deterioration of physiological function and increased mortality risk over time - appears counterintuitive 123. The resolution to this paradox lies in understanding that evolution selects for the maximization of lifetime reproductive fitness, not for the maximization of chronological longevity 134.
Prior to the 1970s, the evolutionary basis of aging was dominated by two interconnected genetic frameworks. The first was the mutation accumulation theory, formulated by Peter Medawar in 1952 256. Medawar observed that in natural ecosystems, wild organisms face high rates of extrinsic mortality due to predation, disease, starvation, and environmental hazards 57. Because few individuals survive to an advanced age, the force of natural selection declines precipitously following reproductive maturity 35. Medawar theorized the existence of a "selection shadow" - a period in late life during which selective pressure is too weak to purge late-acting deleterious mutations from the gene pool, allowing them to accumulate via genetic drift and cause senescence 35.
Building upon this, George Williams introduced the antagonistic pleiotropy hypothesis in 1957 248. Williams proposed that certain pleiotropic alleles confer significant reproductive or developmental advantages early in life but exhibit detrimental, senescence-inducing effects later in life 28. Because the early-life benefits manifest when the organism is most likely to be alive and reproducing, natural selection strongly favors these alleles, embedding the mechanisms of eventual physiological decline into the genome as an unavoidable biological cost 248.
While mutation accumulation and antagonistic pleiotropy provided robust genetic explanations for aging, they lacked a comprehensive physiological mechanism. In 1977, British biologist Thomas Kirkwood introduced the disposable soma theory, bridging the gap between evolutionary population genetics and molecular cellular biology 19. Inspired by Leslie Orgel's 1963 error catastrophe theory - which modeled how random errors in macromolecular synthesis could exponentially compound - Kirkwood recognized that cellular maintenance is not a passive state but a highly energy-intensive active process 14.
The disposable soma theory posits that an organism operates with a finite budget of metabolic energy, which must be partitioned among competing physiological processes: basal metabolism, somatic growth, physical activity, reproduction, and somatic maintenance (the repair of molecular and cellular damage) 110. In an environment where extrinsic mortality guarantees a relatively short lifespan, an organism that diverts excessive metabolic resources to achieve perfect, indefinite somatic maintenance will be outcompeted by organisms that allocate those resources toward rapid growth and early reproduction 111. Consequently, the "soma" (the physical body) is merely a disposable vehicle tasked solely with ensuring the survival and propagation of the immortal "germ line" 412. Natural selection calibrates the investment in somatic repair to be strictly sufficient for the organism to reach reproductive maturity and fulfill its expected ecological reproductive potential, after which un-repaired molecular damage inevitably accumulates, resulting in senescence 1913.
Core Principles of Resource Allocation
The disposable soma theory fundamentally recharacterizes aging as an energetic and physiological optimality problem. The maintenance of cellular fidelity - spanning DNA repair, protein turnover, and oxidative stress mitigation - requires continuous metabolic expenditure 1414. Perfect maintenance is theoretically possible, as evidenced by the immortality of the germ line and organisms like Hydra vulgaris, which possess simple body plans with no strict division between soma and germ line cells and continuously renew their tissues without undergoing senescence 1112. However, for complex multicellular organisms, perfect maintenance represents an evolutionary misallocation of resources 911.
If an organism invests heavily in somatic protection, its probability of surviving to an advanced biological age increases, but this investment creates an energetic deficit that suppresses the generation of precursor reproductive cells and limits offspring output 15. Conversely, too low an investment in self-repair is equally disadvantageous, as the organism may succumb to internal molecular decay before reaching reproductive age 1. The evolutionary stable strategy mathematically dictates a sub-optimal level of investment in maintenance mechanisms 916. The organism accepts a gradual accumulation of faults - such as shortened telomeres, cross-linked proteins, and accumulating DNA mutations - as the necessary thermodynamic cost for maximizing early-life fecundity 113.
Cellular Mechanisms of Somatic Maintenance
While the disposable soma theory is a high-level evolutionary framework, its execution relies on distinct molecular and cellular processes. Organisms deploy complex networks to maintain genomic integrity, ensure protein homeostasis, and regulate epigenetic stability 141718. The specific degradation of these systems over time reflects the evolutionary cap on energy allocation for maintenance 1920.
Genomic Instability and DNA Repair Protocols
The cellular genome is under continuous assault from endogenous factors, predominantly reactive oxygen species (ROS) produced as metabolic byproducts during mitochondrial oxidative phosphorylation, as well as exogenous environmental mutagens 171821. These agents cause diverse forms of molecular damage, ranging from base modifications and bulky adducts to cross-links and highly lethal double-strand breaks (DSBs) 18. A primary biomarker for oxidative genomic damage is the oxidized base 8-oxo-2′-deoxyguanosine (8-oxodG), which promotes mutagenesis and cellular dysfunction if left unrepaired 18.
Cells utilize energy-intensive DNA repair machineries to preserve genomic fidelity. The base excision repair (BER) pathway is tasked with identifying and excising oxidized bases resulting from ROS exposure 1718. The nucleotide excision repair (NER) pathway addresses bulky DNA adducts and UV-induced photoproducts 18. When double-strand breaks occur, the repair protocol depends strictly on the cell's stage in the cell cycle. Actively proliferating cells predominantly utilize homologous recombination (HR), an accurate repair mechanism that relies on an undamaged sister chromatid as a template 17. However, somatic stem cells, which maintain tissue homeostasis, often reside in a metabolically quiescent state. Due to their lack of an actively replicating sister chromatid, quiescent stem cells must rely on non-homologous end joining (NHEJ) to resolve DSBs 17. NHEJ is a fast but highly error-prone mechanism that directly ligates broken DNA ends, frequently introducing insertions or deletions (indels) that cause permanent somatic mutations 1718.
As these imperfect repairs compound, DNA damage inevitably accumulates. This accumulation triggers cellular checkpoint responses characterized by the upregulation of cell cycle inhibitors, including p16Ink4a, p19Arf, and p53 17. The activation of these inhibitors forces the cell into irreversible cell cycle arrest (senescence), apoptosis, or differentiation, ultimately depleting the somatic stem cell pools required for tissue regeneration 1720. Recent cross-species comparative analyses of intestinal crypts across diverse mammals confirm that the somatic mutation rate is a dominant driver of aging, validating the disposable soma theory's premise that variations in somatic DNA repair investments correlate directly with species-specific lifespans 2022.
| DNA Repair Pathway | Primary Molecular Target | Cellular Context | Energetic/Fidelity Characteristics |
|---|---|---|---|
| Base Excision Repair (BER) | Oxidized bases (e.g., 8-oxodG), alkylated bases. | Ubiquitous across cell types. | Highly specific; clears endogenous ROS damage but capacity declines with age. |
| Nucleotide Excision Repair (NER) | Bulky DNA adducts, UV-induced photoproducts. | Ubiquitous across cell types. | Energy-intensive; mutations in NER genes lead to rapid progeroid syndromes. |
| Homologous Recombination (HR) | Double-strand breaks (DSBs). | Proliferating cells (S/G2 phase). | High fidelity; requires a sister chromatid template, making it unavailable to quiescent cells. |
| Non-Homologous End Joining (NHEJ) | Double-strand breaks (DSBs). | Quiescent cells, somatic stem cells. | Fast but low fidelity; error-prone ligation leads to accumulating indels and somatic mutations over time. |
Proteostasis and Cellular Waste Clearance
Cellular functionality is equally dependent on the continuous maintenance and turnover of the proteome. Protein homeostasis (proteostasis) requires regulating the synthesis, folding, trafficking, and degradation of all cellular proteins 1420. Two major degradation pathways are responsible for clearing damaged or misfolded proteins: the ubiquitin-proteasome system (UPS) and the lysosomal/macroautophagy pathway 1420.
The UPS handles the proteolytic degradation of 80 - 90% of all cellular proteins, precisely recognizing and degrading oxidized, misfolded, or otherwise dysfunctional monomeric proteins before they can aggregate 14. Conversely, macroautophagy acts as a broader stress-response mechanism, encapsulating large protein aggregates, damaged macromolecules, and entire dysfunctional organelles (like aged mitochondria) for lysosomal degradation 1720.
Maintaining these pathways is extremely energetically demanding. As the organism ages and metabolic resources become constrained according to evolutionary allocation parameters, the efficiency of both the UPS and autophagic clearance declines 1423. The loss of autophagy - demonstrated experimentally through the deletion of genes such as Atg7 or Fip200 - precipitates a lethal intracellular accumulation of ROS and toxic protein aggregates, accelerating hematopoietic stem cell depletion and driving systemic aging 1720. Under the disposable soma framework, the toxic aggregation of waste proteins that characterizes neurodegenerative diseases is the direct downstream consequence of an evolutionary strategy that limits the energy available for perpetual proteome turnover 1421.
Epigenetic Deterioration and Biological Clocks
Beyond physical damage to the DNA sequence, the disposable soma theory encompasses the progressive failure of the epigenome. Aging is universally characterized by predictable epigenetic alterations, including global DNA hypomethylation, targeted hypermethylation at specific promoter regions, altered histone modifications (such as changes in H3K27ac), and the widespread destabilization of heterochromatin 202324.
These epigenetic modifications disrupt cellular identity by aberrantly downregulating essential maintenance genes and upregulating inflammatory pathways, a process deeply intertwined with cellular senescence 2324. The predictability of these methylation patterns has enabled the development of highly accurate "epigenetic clocks" (such as the DunedinPACE clock), which track biological aging and predict mortality risk more effectively than chronological age or standard physiological biomarkers 242526.
A landmark 2025 study analyzing extensive human cancer genomic databases established a critical causal link between the somatic mutation theory and epigenetic aging 27. The research demonstrated that random somatic DNA mutations predictably trigger cascading epigenetic modifications across the genome, fundamentally altering gene expression networks 27. This finding aligns perfectly with the disposable soma framework: evolutionary resource limits permit stochastic somatic mutations, which subsequently serve as the mechanistic catalyst for widespread, predictable epigenetic decay and systemic aging 2027.
Mathematical Formalization of Resource Allocation
To evaluate the disposable soma theory beyond abstract evolutionary principles, researchers have formalized the concept into quantitative physiological and demographic models, primarily utilizing Dynamic Energy Budget (DEB) theory and Euler-Lotka frameworks 152829.
Dynamic Energy Budget (DEB) Theory
The standard DEB model provides a comprehensive thermodynamic framework detailing how individual organisms assimilate energy from food, store it as biological reserves, and mobilize those reserves to fuel competing metabolic demands 28. Within the DEB model, a critical allocation parameter is denoted as $\kappa$ (kappa). The model postulates that a fixed fraction, $\kappa$, of an organism's mobilized energy reserve is allocated strictly to somatic maintenance and structural growth 282930. The remaining fraction, $1 - \kappa$, is allocated to maturity maintenance, maturation (during juvenile stages), and reproduction (in fully grown adults) 282930.
To test the evolutionary optimality of this allocation, researchers estimated the DEB parameters for 276 diverse animal species across major phyla and chordate classes 282930. They calculated the hypothetical optimal value of $\kappa$ that would absolutely maximize reproductive output for fully grown adults under abundant food conditions. The analysis revealed that natural selection rarely optimizes purely for maximal immediate reproduction. Only 13% of the analyzed wild species exhibited a reproduction rate close to the theoretical maximum, while 83% of species possessed a $\kappa$ value significantly higher than the reproductive optimum 282930.
This demonstrates a strong empirical evolutionary pressure to over-allocate resources toward somatic maintenance relative to strict reproductive maximization 2830. While maintaining the soma indefinitely is energetically prohibited, maintaining it at a level sufficient to endure multiple reproductive cycles across an expected ecological lifespan requires a $\kappa$ value higher than what would yield maximum instant fecundity 2830. Notably, when the metabolic parameters of wild jungle fowl were compared to domestic agricultural chickens selectively bred for extreme meat and egg production, the domestic races exhibited $\kappa$ values perfectly aligned with absolute reproductive maximization, achieved through the systematic down-regulation of maintenance allocations 282930.
The Euler-Lotka Population Dynamics Framework
At the population level, the consequences of cellular resource allocation are evaluated using life-history models based on the Euler-Lotka equation 15. This mathematical framework integrates age-specific survival schedules ($l(x)$) and fecundity schedules ($m(x)$) to calculate overall lifetime evolutionary fitness ($r$) 15.
In these models, an organism's relative investment in cellular protection ($q$) exerts complex, interacting effects on both survival and reproduction 1531. Survival presents a hump-shaped relationship regarding resource allocation: as resources are directed toward somatic maintenance, survival initially increases due to reduced molecular damage 15. However, this comes at a steep reproductive cost. If an organism dedicates too many resources to protecting somatic cells, the energetic deprivation suppresses the generation and maturation of precursor reproductive cells, collapsing lifetime fecundity 15. The mathematical optimization of the Euler-Lotka equation definitively proves that unless environmental survival probability approaches absolute certainty, the maximum overall fitness is achieved by enforcing a sub-optimal maintenance setting that permits eventual physiological breakdown 151631.
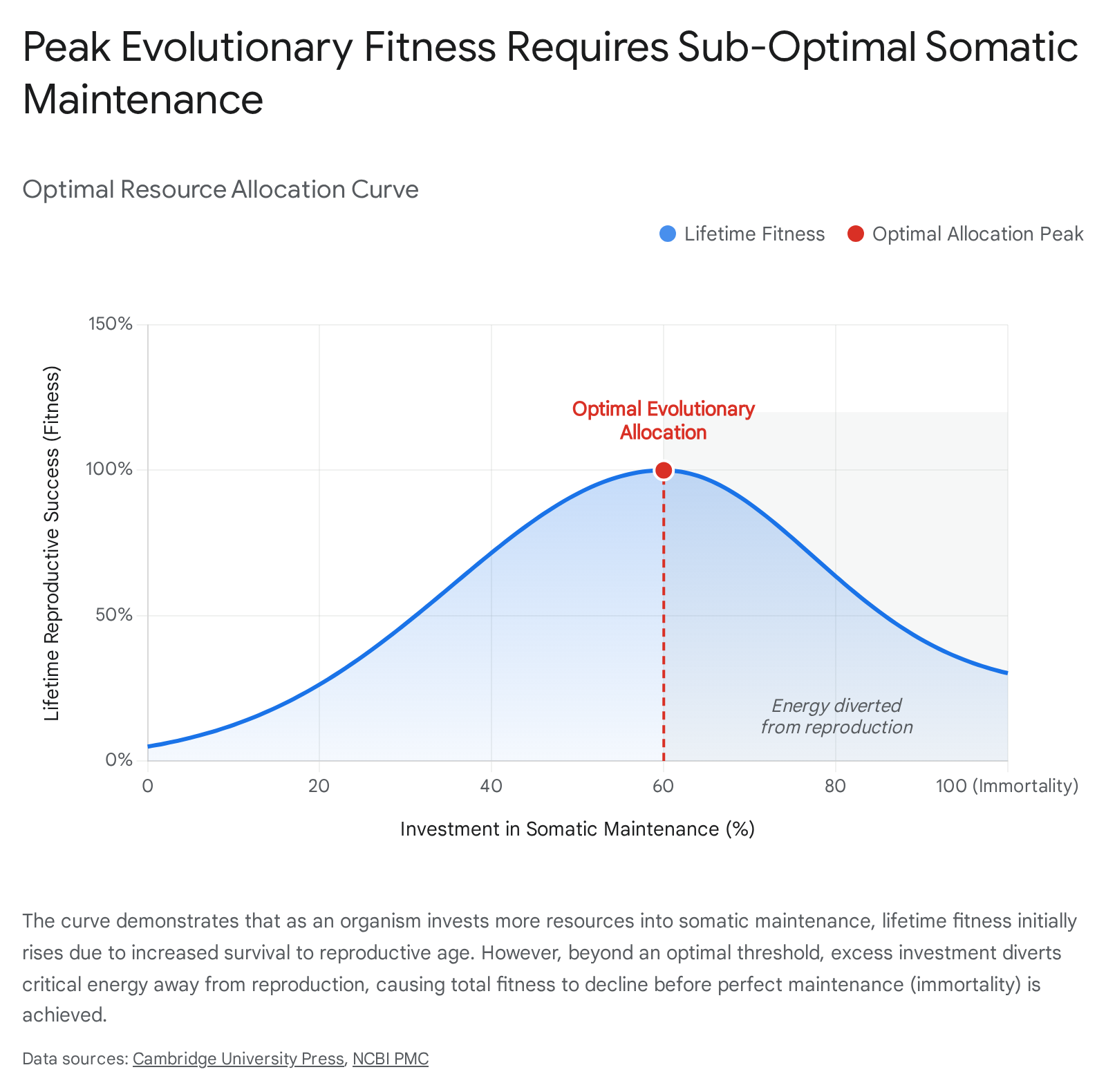
The Caloric Restriction Paradigm
The disposable soma framework offers the most widely cited evolutionary explanation for the physiological effects of caloric restriction (CR) 101632. Extensive experimental data across taxa, from yeast to non-human primates, demonstrate that restricting caloric intake by 20% to 50% without inducing malnutrition significantly extends both healthspan and maximum lifespan 103233.
Under normal, ad libitum feeding conditions, an organism allocates energy to both active reproductive efforts and baseline somatic maintenance 1032. However, when environmental food availability drops, the organism faces an acute survival threat. Darryl P. Shanley and Thomas Kirkwood developed a life-history model (the S-K model) demonstrating that attempting to reproduce during a famine is evolutionarily disastrous; the offspring are highly unlikely to survive, and the massive energetic cost of reproduction risks killing the parent 91016.
According to the disposable soma theory, CR triggers a highly conserved adaptive response. The organism deliberately suppresses reproductive pathways and redirects its scarce remaining energy directly into upregulating somatic maintenance functions, such as enhanced autophagy, improved error-checking, and augmented DNA repair protocols 11032. This physiological shift preserves the organism's reproductive value, reducing the intrinsic rate of senescence so the individual may outlive the famine and resume reproduction when resources return 1016. In a laboratory setting, because the "famine" never ends, this adaptive maintenance state is maintained indefinitely, resulting in dramatic lifespan extensions 32.
While the S-K model elegantly aligns with observations in female rodents, it faces challenges 1032. For example, male animals often do not suppress reproductive efforts under CR to the same extent as females, yet still experience lifespan extension 10. Furthermore, critics propose alternative models, such as the "clean cupboards" hypothesis, which argues that CR lifespan extensions are not due to increased energy allocation to repair (which is mathematically difficult to justify when total caloric intake is severely reduced), but rather due to reduced cellular waste production and metabolic turnover 32.
Empirical Exceptions and Biological Anomalies
If senescence is a universal thermodynamic inevitability driven by resource allocation constraints, the phenomenon should manifest universally across multicellular life 1422. However, advancing biodemographic research has identified profound exceptions that challenge the generalizability of the disposable soma theory 43435.
The Reproduction-Longevity Paradox in Eusocial Insects
The disposable soma theory explicitly posits a severe trade-off between reproduction and longevity 136. Yet, eusocial insects - including ants, bees, wasps, and termites - represent a stark contradiction to this fundamental rule 363738. Eusocial colonies feature extreme reproductive division of labor, where one or a few individuals (queens) completely monopolize reproduction while a vast caste of sterile or non-reproductive workers maintain the colony 3738.
Despite allocating almost entirely toward massive reproductive output (often laying thousands of eggs daily), eusocial queens exhibit lifespans orders of magnitude longer than their workers 373839. Termite queens can live over 15 to 30 years, ant queens generally live over a decade, while their respective worker castes perish within weeks or months 373839. Crucially, this immense lifespan divergence occurs without genomic variation; queens and workers share identical genetic blueprints, meaning the longevity divergence is entirely phenotypic and regulatory 3739.
Laboratory isolation studies on the ant Cardiocondyla obscurior demonstrate that when queens are reproductively activated, their survival rate actually increases relative to non-reproductive queens and workers, completely reversing the classical reproduction-longevity trade-off 3640. Evolutionary biologists hypothesize that this occurs because queens reside deep within protected nests, completely shielded from extrinsic mortality (predation, foraging hazards), which maintains the force of natural selection strong late into life 373839. Furthermore, mathematical modeling indicates a "queen effect": the demographic dynamics of eusocial reproduction inherently select for maternal longevity, overriding standard somatic maintenance trade-offs and allowing the activation of latent, highly efficient intrinsic survival mechanisms 363839.
Negligible Senescence in Reptiles
The disposable soma framework assumes that age-related deterioration is unavoidable. However, specific taxa exhibit "negligible senescence," characterized by no measurable decline in physiological function, maintained or increasing fertility, and mortality rates that remain stable or even decrease as the organism ages 344142.
Testudines (tortoises and turtles) have long been recognized for their extreme longevity, but recent rigorous demographic analyses have redefined their aging parameters 434344. A comprehensive 2022 study published in Science analyzed 52 species of turtles and tortoises residing in controlled zoological environments using the Species360 Zoological Information Management System (ZIMS) 434345. The researchers discovered that 75% of the studied species exhibited extremely slow or entirely negligible senescence, and 80% aged significantly slower than modern humans 4345.
This capability is largely attributed to indeterminate growth. Unlike mammals, which cease growing upon reaching sexual maturity, many reptiles continue to grow throughout their lives 4344. This life-history strategy requires continuous investment in cellular repair and tissue regeneration, bypassing the developmental endpoint that triggers senescence in mammals 354344. Genomic sequencing of giant tortoises (such as the Galapagos and Aldabra species) reveals distinct evolutionary strategies linked to this longevity, including lineage-specific expansions in DNA repair genes (XRCC6), enhanced immune mediators, and specific mutations in telomere-maintenance genes like DCLRE1B, which alter its binding interface with telomeric repeat binding factors 4346.
The Naked Mole-Rat Anomaly
Mammalian negligible senescence is incredibly rare, but the naked mole-rat (Heterocephalus glaber) presents a profound anomaly to physiological aging models 343847. Native to harsh, subterranean environments in the Horn of Africa, these rodents routinely live over 30 years in captivity - roughly nine times longer than similarly sized mice 3447.
Naked mole-rats exhibit a flat Gompertz mortality curve, meaning their risk of death does not increase as they age chronologically 3438. They maintain youthful body composition, show no decline in cardiac or neurological function, and breeding females experience no menopause or fertility decline even into their third decade 3447. Furthermore, despite residing in low-oxygen environments that generate high levels of oxidative stress, naked mole-rats show high retention of protein activity and unparalleled resistance to spontaneous neoplasm development 2134. Their ability to seamlessly repair oxidized protein moieties and effectively clear senescent cells prevents the accumulation of proteomic damage and the toxic senescence-associated secretory phenotype (SASP), challenging the assertion that high reproductive output inevitably compromises somatic maintenance in mammals 2134.
Botanical Exceptions and Modular Body Plans
The plant kingdom provides further evidence that biological immortality is mechanistically possible, provided the organism's structural plan accommodates it. The Great Basin bristlecone pine (Pinus longaeva) holds the record as the oldest individual, non-clonal organism on Earth, with verified chronological ages exceeding 5,000 years 424849.
Extensive physiological and genetic analyses of bristlecone pines ranging from 23 to over 4,700 years old reveal absolutely no evidence of mutational aging or senescence 50. There are no age-related changes in tracheid diameter, vascular cambial function, shoot growth increments, pollen viability, or seed germinability 50. Genomic sequencing of the 24-billion-base-pair bristlecone pine genome demonstrates that these trees possess significantly larger-than-average telomere lengths compared to other conifers, indicating profound cellular stability 4849. The survival of such ancient trees is facilitated by a modular body plan; sections of the organism can undergo localized necrosis due to external damage, while the surviving independent meristematic tissues continue generating fresh, non-senescent clones, rendering the concept of systemic organismal aging irrelevant 44850.
| Taxonomic Group | Representative Species | Evidence of Negligible Senescence | Distinctive Biological Mechanisms |
|---|---|---|---|
| Reptiles (Testudines) | Aldabra Giant Tortoise (Aldabrachelys gigantea) | 75% of analyzed species show negligible or extremely slow senescence; flat mortality curves. | Indeterminate growth; expansions in DNA repair (XRCC6) and telomere maintenance (DCLRE1B) genes. |
| Mammals (Rodentia) | Naked Mole-Rat (Heterocephalus glaber) | 30+ year lifespan; no age-related mortality acceleration; no reproductive decline; cancer immunity. | Extreme proteostasis efficiency; robust clearance of senescent cells; eusocial reproductive structure. |
| Plants (Conifers) | Great Basin Bristlecone Pine (Pinus longaeva) | 5,000+ year lifespan; no decline in cambial function, pollen viability, or seed germinability. | Modular growth plan allows compartmentalized tissue death; unusually long, stable telomeres. |
| Invertebrates (Cnidaria) | Freshwater Polyp (Hydra vulgaris) | Continuous asexual reproduction with no observable increase in mortality or decline in function over time. | Simple body plan with no strict division between somatic and germline cells; continuous stem cell renewal. |
Theoretical Critiques and Alternative Models
While the disposable soma theory has organized biogerontology for four decades, recent experimental data and competing theoretical models severely challenge its core premises 125152. The primary critique focuses on the assumption that reproduction generates a direct, long-term metabolic penalty that physically compromises somatic repair.
Empirical Failures of the Maintenance Trade-off
If the disposable soma theory is correct, allocating metabolic resources to reproduction must result in an accelerated decline in physiological homeostasis, culminating in increased late-life mortality 5354. A rigorous 2024 study tested this exact prediction using over 120 breeding and non-breeding female C57BL/6J mice 5355. Researchers conducted comprehensive biological inventories, measuring reproductive outputs, daily energy expenditure, body composition, systemic oxidative stress, and overall lifespan 5355.
The findings contradicted the foundational predictions of the disposable soma framework 5355. While reproduction was not biologically inconsequential - carrying an immediate, acute mortality risk due to the physical stresses of parturition - there was absolutely no evidence of a residual survival penalty once the females ceased breeding 5355. The breeding mice did not display elevated levels of oxidative stress, nor did they show accelerated long-term physiological deterioration compared to their non-breeding counterparts 5355. Higher mortality was exclusively a direct, immediate consequence of the act of reproduction itself, rather than the manifestation of a deferred "somatic maintenance debt" 5355.
The Hyperfunction Theory of Aging
The most formidable theoretical challenge to the disposable soma framework is the hyperfunction theory, proposed and championed by Mikhail V. Blagosklonny 51525456. Blagosklonny argues that the disposable soma theory rests on a subtle anthropomorphism: the concept of an "intelligent designer" or biological accountant rationally allocating scarce energy coins between a reproduction budget and a repair budget 52. In reality, Blagosklonny asserts, evolution behaves strictly as a "blind watchmaker" that selects purely for robust, rapid developmental growth and early-life survival, lacking any mechanism or foresight to plan for post-reproductive life 1252.
According to the hyperfunction model, aging is not the passive accumulation of random molecular damage caused by energy scarcity 1252. Instead, aging is an active, "quasi-programmed" process - the aimless, wasteful continuation of the exact same genetic programs that successfully drove early development 125254. The central driver of this process is the nutrient-sensing, growth-promoting mechanistic target of rapamycin (mTOR) signaling pathway 125256.
During youth, mTOR activation is essential; it drives cellular mass accumulation, metabolic growth, and sexual maturation, granting young organisms the physical robustness required to survive environmental hazards 1252. However, evolution provides no regulatory mechanism to "switch off" this growth pathway after developmental maturity is achieved 125256. Left unconstrained, this chronic post-developmental hyper-activation forces cells into an aberrant state of senescence 1252. Rather than merely arresting the cell cycle, hyper-activated mTOR drives cellular hypertrophy and the secretion of pro-inflammatory cytokines 1252. Ultimately, it is this active hyperfunction - not a passive failure of DNA repair mechanisms - that generates the specific, non-random organ pathologies (such as atherosclerosis, hypertension, and organ hypertrophy) that result in organismal death 1252.
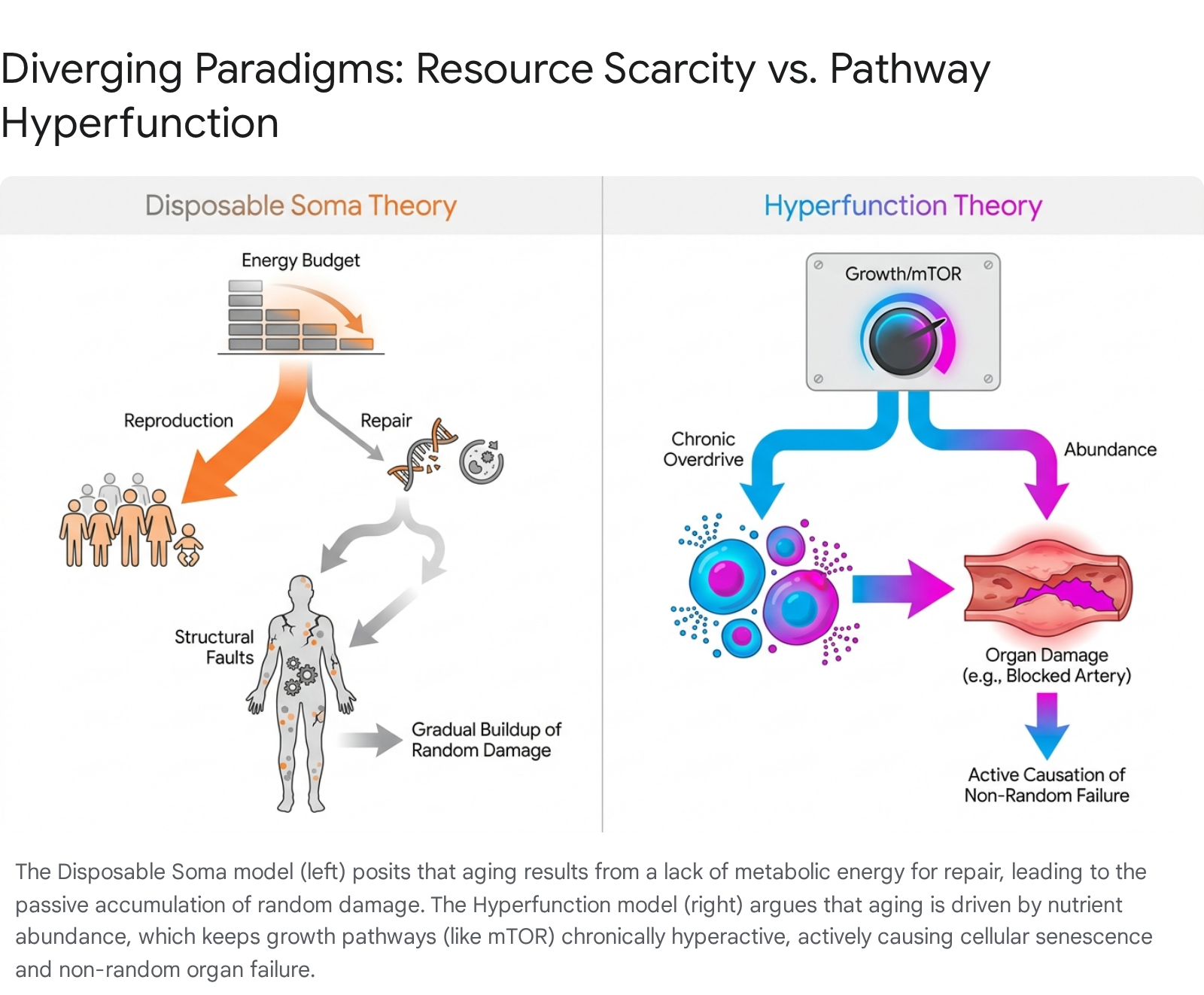
The hyperfunction theory resolves several physiological paradoxes that the disposable soma framework struggles to explain 1251. For instance, under the disposable soma model, increased food intake provides more metabolic resources, which logically should allow for greater somatic repair and extended lifespan. Yet, increased food intake invariably accelerates aging and shortens lifespan across species 12. The disposable soma theory attempts to reconcile this by arguing that caloric restriction (starvation) somehow forces the organism to reallocate energy to repair, a mathematically questionable proposition when total energy is severely depleted 1232.
Conversely, the hyperfunction theory provides a direct, mechanistic explanation: food intake activates the nutrient-sensing mTOR pathway 1254. Therefore, eating less (caloric restriction) dampens mTOR activation, effectively suppressing the quasi-programmed acceleration of aging 1254. This also explains the profound efficacy of rapamycin - a direct pharmacological inhibitor of mTOR - in extending healthspan and lifespan across diverse animal models, a phenomenon that cannot be easily reconciled with a model based strictly on passive energy scarcity 125152.
| Feature | Disposable Soma Theory (Kirkwood) | Hyperfunction Theory (Blagosklonny) |
|---|---|---|
| Primary Cause of Aging | Passive accumulation of stochastic molecular damage. | Active, aimless continuation of developmental growth programs. |
| Role of Natural Selection | Optimizes energy allocation between reproduction and somatic repair. | Blindly selects for robust early-life growth, ignoring post-reproductive consequences. |
| Mechanism of Caloric Restriction | Starvation triggers an adaptive reallocation of scarce resources to DNA repair. | Reduced food intake directly suppresses the nutrient-sensing mTOR pathway. |
| Cellular Senescence | Cells arrest to prevent damaged DNA from causing cancer. | Hyper-activated growth pathways in arrested cells drive hypertrophy and toxic secretions. |
| Disease Pathophysiology | Random cellular deterioration leads to general frailty. | Specific pathway hyperfunction drives non-random, targeted organ damage (e.g., atherosclerosis). |
Extension to the Microbial World
Historically, the disposable soma theory was deemed applicable only to multicellular organisms that exhibit a strict biological division between mortal somatic cells and immortal germline cells 41157. Single-celled microbes (prokaryotes and protists) were widely considered to be potentially immortal, as they reproduce via binary fission, ostensibly passing a pristine, rejuvenated copy of the genome to identical daughter cells 4257.
However, recent evolutionary arguments and highly sensitive observational techniques have expanded the explanatory scope of the disposable soma theory to encompass the entire microbial world 57. Research demonstrates that bacterial cell division is frequently morphologically and molecularly asymmetrical 425758. During division, bacteria such as Escherichia coli partition damaged macromolecules, oxidized proteins, and old cell poles unevenly between the two progeny 42.
One daughter cell inherits the newly synthesized cellular components (acting as the rejuvenated "germline"), while the other inherits the older cellular poles and the accumulated metabolic damage (acting as the aging "soma") 4257. Over successive divisions, the cell lineages that consistently inherit the older poles exhibit classic hallmarks of senescence: cumulatively slowed growth, decreased biomass production, and a statistically increased probability of death 42. This phenomenon suggests that the evolutionary trade-off between the energetic cost of absolute molecular fidelity and the competitive necessity of rapid reproduction is universally conserved, driving replicative and physiological aging even at the unicellular level 57.
Conclusion
The disposable soma theory represents a monumental paradigm shift in biogerontology, transitioning the scientific understanding of aging from an inevitable genetic destiny to a dynamic physiological trade-off governed by ecological constraints 1913. By recognizing that organisms operate within a finite metabolic budget, Thomas Kirkwood provided a comprehensive optimality framework that elegantly bridges evolutionary population genetics with the molecular realities of DNA repair, proteostasis, and epigenetic maintenance 11420. The theory successfully explains the broad evolutionary patterns of longevity, demonstrating why high-predation species invest in rapid, massive fecundity at the expense of somatic repair, while apex species invest in highly sophisticated, durable maintenance machineries 11359.
However, as the precision of modern experimental gerontology and longitudinal biodemography has advanced, the limits of the theory's universality have become apparent. The existence of modular organisms like the bristlecone pine and indeterminate growers like tortoises that exhibit negligible senescence, combined with the profound decoupling of reproduction and mortality in eusocial insects, indicates that aging is not an inescapable thermodynamic necessity for all biological architecture 35384350. Furthermore, rigorous empirical studies failing to detect residual survival penalties following intense reproduction challenge the fundamental premise of a zero-sum energetic ledger 5355.
Concurrently, alternative frameworks - most notably the hyperfunction theory - provide compelling evidence that biological aging may be less about the passive accumulation of un-repaired damage due to scarcity, and more about the active, toxic hyper-activation of growth pathways fueled by nutrient abundance 1254. Moving forward, a complete understanding of biological aging will require synthesizing the undisputed realities of metabolic resource constraints highlighted by the disposable soma theory with the complex, quasi-programmed regulatory mechanisms that drive species-specific life histories.