Evolutionary Mismatch and Modern Human Health
Evolutionary mismatch is a state of biological and ecological disequilibrium that occurs when an organism, having evolved specific adaptations in one environment, develops phenotypes that are harmful to its fitness or well-being when exposed to a novel environment 12. For the vast majority of the evolutionary history of Homo sapiens, a specific range of natural environments defined the parameters within which natural selection shaped human physiological and metabolic networks 2. However, rapid human-induced environmental change - driven by the advent of agriculture, widespread industrialization, and modern urbanization - has fundamentally outpaced the rate of human biological adaptation 3.
This temporal lag between rapid environmental transition and slow genetic evolution creates a fundamental conflict between ancient biological mechanisms and modern living conditions 134. The physiological systems that evolved to efficiently store energy during periods of scarcity, mount rapid inflammatory responses to acute pathogen exposure, and synchronize circadian rhythms to the natural solar cycle now generate pathological outcomes when exposed to continuous caloric abundance, hyper-sanitized environments, and artificial light at night 567.
Theoretical Foundations of Mismatch
To comprehensively evaluate evolutionary mismatch, researchers analyze the divergence between the ancestral environment (E1) - the ecological, dietary, and social context in which specific traits evolved - and the novel environment (E2) in which the organism currently resides 1. The ancestral environment for anatomically modern humans, spanning the Late Pleistocene (approximately 125,000 to 12,000 years ago), was characterized by frequent physical movement, acute stress responses to predators, and foraging diets 38.
By contrast, modern industrial environments (E2) are characterized by sedentary behaviors, persistent psychological stress, artificial lighting, noise pollution, and processed, calorie-dense foods 34. Evolutionary mismatch theory requires isolating the precise population, the traits in question, the inheritance mechanisms, and the differing selection pressures across E1 and E2 to fully understand the physiological costs of this divergence 1.
Categories of Environmental Disequilibrium
Mismatch theory delineates distinct mechanisms through which environmental disequilibrium manifests. These categories clarify whether a mismatch is driven by static genetic inheritance, lifelong developmental trajectories, or rapid behavioral shifts.
| Mismatch Category | Definition | Mechanism | Clinical Example |
|---|---|---|---|
| Genetic Mismatch | A fixed trait that was adaptive or neutral in E1 becomes deleterious in E2 due to slow genetic adaptation. | Altered selection pressures render historically beneficial or neutral inherited alleles maladaptive in novel contexts 12. | Sickle cell trait provides essential malaria protection but causes severe anemia in modern, malaria-free environments 9. |
| Developmental Mismatch | Phenotypes fixed during early development based on environmental cues become maladaptive if the adult environment differs. | Predictive adaptive responses (e.g., maternal constraints) misalign with later-life environmental realities 2. | Fetal undernutrition triggers energy-saving metabolism, predisposing the individual to obesity and diabetes if the adult environment is calorie-rich 1011. |
| Cultural Mismatch | Culturally transmitted behaviors, technologies, or dietary practices create new environmental parameters that conflict with baseline biology. | Rapid cultural niche construction significantly outpaces the rate of biological evolution across populations 1212. | The widespread adoption of highly processed foods and sedentary routines disrupts ancient metabolic homeostasis 34. |
Phenotypic Plasticity Constraints
Phenotypic plasticity - the capacity of a single genotype to produce distinct morphological, physiological, or behavioral phenotypes in response to environmental variation - serves as a primary biological mechanism for coping with environmental fluctuation 131415. By allowing organisms to alter trait expression during their lifetime, plasticity can mitigate the immediate consequences of evolutionary mismatch and facilitate colonization of new environments 14.
However, phenotypic plasticity is not limitless. Theoretical and empirical studies suggest that the evolution of perfect plasticity is constrained by the costs of maintaining sensory machinery, the reliability of environmental cues, and a lack of standing genetic variation 13. When modern environmental cues mimic ancestral ones but deliver different fitness outcomes, it creates an "ecological trap." For instance, an organism may prefer a novel environmental cue that mimics historical properties without receiving the expected fitness benefits 2. Furthermore, relaxed selection and variable selection intensities in modern human environments act as constraints on the evolution of optimal plasticity, leaving populations vulnerable when the novel environment falls entirely outside the historical range of reaction norms 1316.
Urbanization and Cardiometabolic Transitions
The global epidemic of non-communicable diseases (NCDs), including type 2 diabetes, hypertension, and cardiovascular disease, has often been characterized as an unavoidable consequence of human aging and extended lifespans. However, research in evolutionary medicine indicates that these "diseases of aging" are fundamentally driven by lifestyle transitions from non-industrial to urban environments 1718.
Built Environment and Physical Infrastructure
To isolate the specific environmental drivers of cardiometabolic decline, researchers have conducted large-scale within-population analyses of indigenous societies currently undergoing rapid market integration. Recent cross-sectional and biomarker studies of the Turkana pastoralists in Kenya (n = 3,692) and the Orang Asli mixed-subsistence groups in Peninsular Malaysia (n = 1,119) provide critical insights into this epidemiological transition 172019. Both populations are experiencing a rapid shift from subsistence-level, nomadic or semi-nomadic lifestyles to urban, market-integrated environments 1720.
Extensive factor analyses of these transitioning cohorts revealed that broad measures of urban infrastructure and market-derived material wealth are stronger predictors of declining cardiometabolic health than more proximate individual factors, such as specific dietary macro-components 2019. The built environment acts as an overarching constraint on behavior, enforcing sedentary lifestyles, altering daily physical mobility patterns, and dictating the accessibility of ultra-processed market goods. Turkana individuals who transition to urban environments exhibit significantly poorer health markers - including elevated body fat, cholesterol, and blood pressure - compared to their rural counterparts 20. Notably, being born in an urban area independently predicts adult cardiometabolic health, meaning life-long city dwellers experience the highest cumulative cardiovascular disease risk 20.
Epigenetic Aging Versus Functional Capacity
In rural, non-industrial settings, the Turkana and Orang Asli exhibit minimal to modest age-dependent increases in body size, lipid concentrations, and blood pressure, retaining robust cardiometabolic health into late adulthood 1718. Urban environments significantly amplify these age effects. In the Orang Asli population, urban individuals exhibited an 18.5% higher average body mass index (BMI) than rural individuals, a variation that mediated 26.0% of the relationship between urbanicity and epigenetic age acceleration 17.
Interestingly, while urbanization reliably accelerates cardiometabolic aging and alters epigenetic clocks, it does not universally accelerate all biomarkers of biological aging. Measures of generalized functional capacity, such as grip strength and walking speed, showed no consistent lifestyle-dependent age effects across the rural-urban gradient in these transitional populations 1718. This indicates that evolutionary mismatch is not a generalized accelerant of all somatic aging pathways, but rather uniquely detrimental to physiological systems governing energy metabolism, fat storage, and vascular health.
Microbiome Extinction and Dietary Transitions
The human microbiome functions as an accessory genetic reservoir that is highly responsive to environmental shifts, acting as a critical interface between ancestral host biology and modern dietary inputs 21. The rapid transition from complex, plant-heavy foraging diets to industrialized food systems has profoundly restructured the human gut ecosystem, resulting in widespread dysbiosis.
The Missing Microbes Hypothesis
The "Missing Microbes" hypothesis posits that the cumulative features of modern life - widespread antibiotic use, urban sanitization, cesarean deliveries, and highly processed diets - have precipitated the progressive extinction of ancient, co-evolved members of the human microbiota 5622. These indigenous microbes evolved symbiotically with human hosts over millions of years, providing essential immunoregulatory and metabolic functions in exchange for resources 5.
Genomic analyses of desiccated human paleofeces dating back over 1,000 years, recovered from dry caves in Utah and northern Mexico, demonstrate the stark reality of this microbial biodiversity loss. Researchers found that ancient microbiomes contained nearly 40% more unique microbial species than modern industrial populations 2324. Furthermore, ancient microbial populations incorporated significantly fewer genes related to antibiotic resistance and exhibited higher numbers of transposases, suggesting a greater capacity to adapt to highly varied, seasonal foraging diets 24.
Taxonomic Shifts and VANISH Bacteria
There is a consistent loss of specific bacterial species in modern populations, termed VANISH taxa (Volatile and/or Associated Negatively with Industrialized Societies of Humans) 56. Genera such as Treponema, Prevotella, and Succinivibrio, which dominate the microbiomes of hunter-gatherer and rural populations, are virtually absent in urban centers 2125. For example, the bacterium Treponema succinifaciens is consistently identified in ancient paleofeces and contemporary non-industrialized populations but is entirely missing from the microbiomes of industrialized individuals 2324.
In the absence of these ancient fiber-degrading networks, the modern gut is increasingly dominated by the Bacteroidaceae family 25.
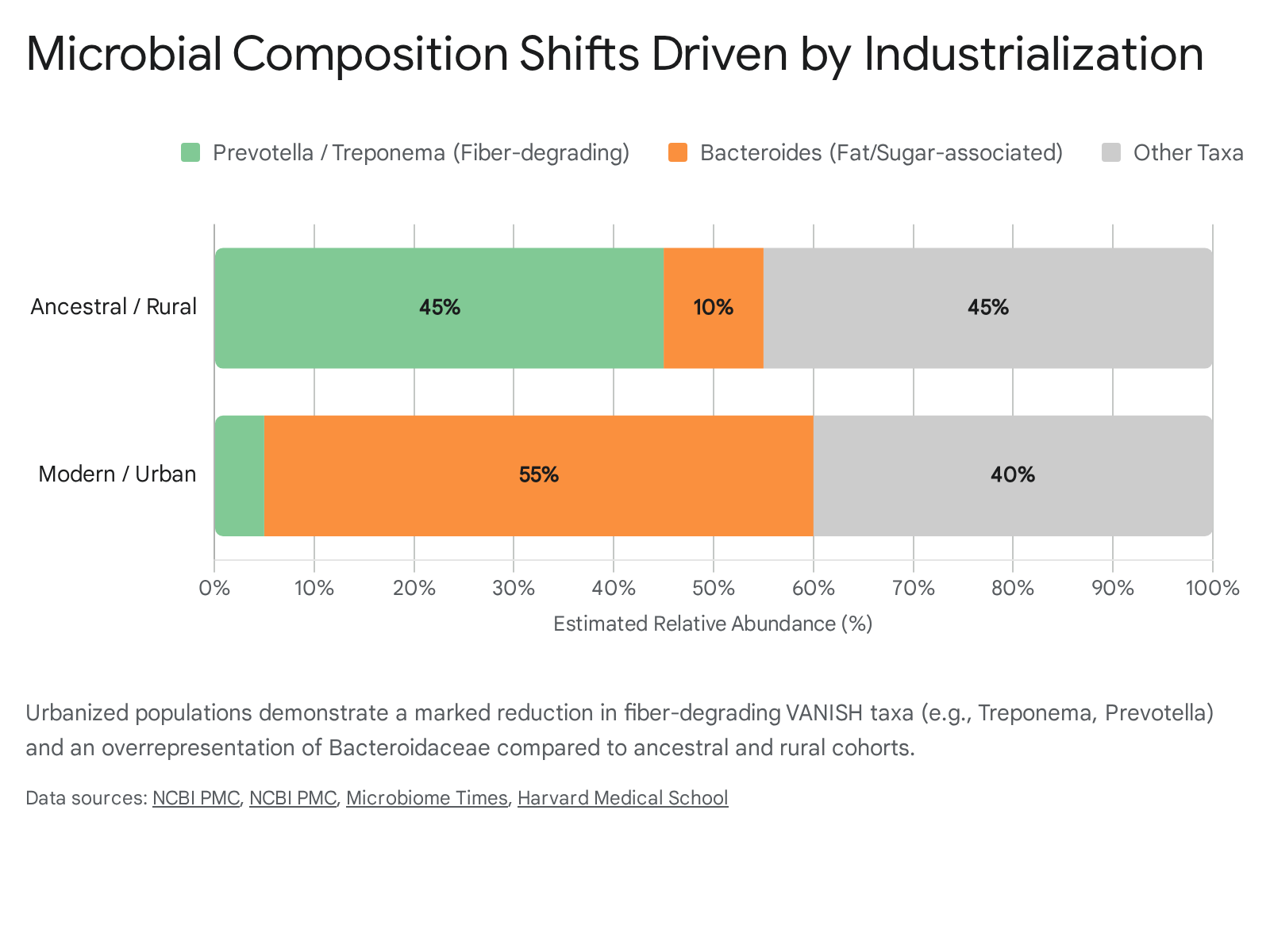
While multifunctional, the rise of mucin-degrading Bacteroides and the loss of Treponema are statistically linked to compromised insulin signaling and elevated fasting blood glucose. Observational studies confirm that individuals lacking these VANISH microbes have a significantly higher prevalence of diabetes, higher HbA1c levels, and higher BMI 626.
Substrate Fermentation and Short-Chain Fatty Acids
Ancestral diets provided a vast array of structurally distinct dietary fibers, including resistant starches, non-starch polysaccharides, beta-glucans, plant gums, and polyphenols 2728. These complex substrates dictate specific cross-feeding interactions among microbes. When gut microbes ferment diverse plant fibers, they generate short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate 2127. Butyrate, in particular, is the primary nutrient source for colonocytes and plays a critical role in maintaining the mucosal barrier and modulating local immune responses 2126.
The contemporary transition to diets deficient in complex plant fibers and saturated with refined carbohydrates directly starves these keystone microbial species. Industrialized microbiomes exhibit substantially reduced overall alpha diversity (the number of co-existing taxa) 52529. The reduction of SCFA-producing bacterial networks compromises mucosal immunity and promotes metabolic endotoxemia, contributing to the pathogenesis of autoimmune disorders, chronic systemic inflammation, and obesity 626.
Microbial Genetic Adaptation to Refined Carbohydrates
The physiological mismatch regarding modern diets extends beyond caloric density to alter the fundamental host-microbe metabolic axis. In environments of historical scarcity, seeking out sweet foods offered a survival advantage by providing immediate, safe energy 30. In the modern context of ubiquitous refined sucrose and high-fructose corn syrup, this drive is maladaptive.
Microbes themselves undergo rapid genetic adaptation to these novel nutrient pools. High-sugar diets select for specific microbial gene profiles adapted to rapid simple-sugar transport and metabolism, subsequently altering the production of secondary microbial metabolites 31. In several human populations, increases in microbial gene content dedicated to sugar metabolism and transport are robustly associated with the acceleration of type 2 diabetes and obesity 31. Secondary metabolites and toxins produced by these altered microbial communities can lead to the destruction of the intestinal epithelium, further exacerbating systemic metabolic dysfunction 31.
Circadian Rhythm Disruption and Hepatic Pathology
The evolutionary parameters of human sleep and metabolism were strictly shaped by the natural solar cycle and diurnal patterns of feeding and fasting. The widespread deployment of artificial light and the demands of modern socioeconomic schedules have profoundly disrupted the deeply conserved biological pathways that govern human circadian rhythms 3233.
The Light-Gut-Liver Axis
For millions of years, the onset of environmental darkness reliably initiated the secretion of melatonin from the pineal gland, signaling physiological rest and down-regulating daytime metabolic processes 7. Modern chronic exposure to artificial light at night (ALAN) - specifically blue-enriched light in the 460 - 480 nm spectrum emitted by digital screens and urban lighting - suppresses endogenous melatonin production and desynchronizes both the central master clock in the suprachiasmatic nucleus and peripheral clocks residing in organs such as the liver 734.
This circadian misalignment has revealed a complex pathological mechanism recognized as the "light-gut-liver axis."
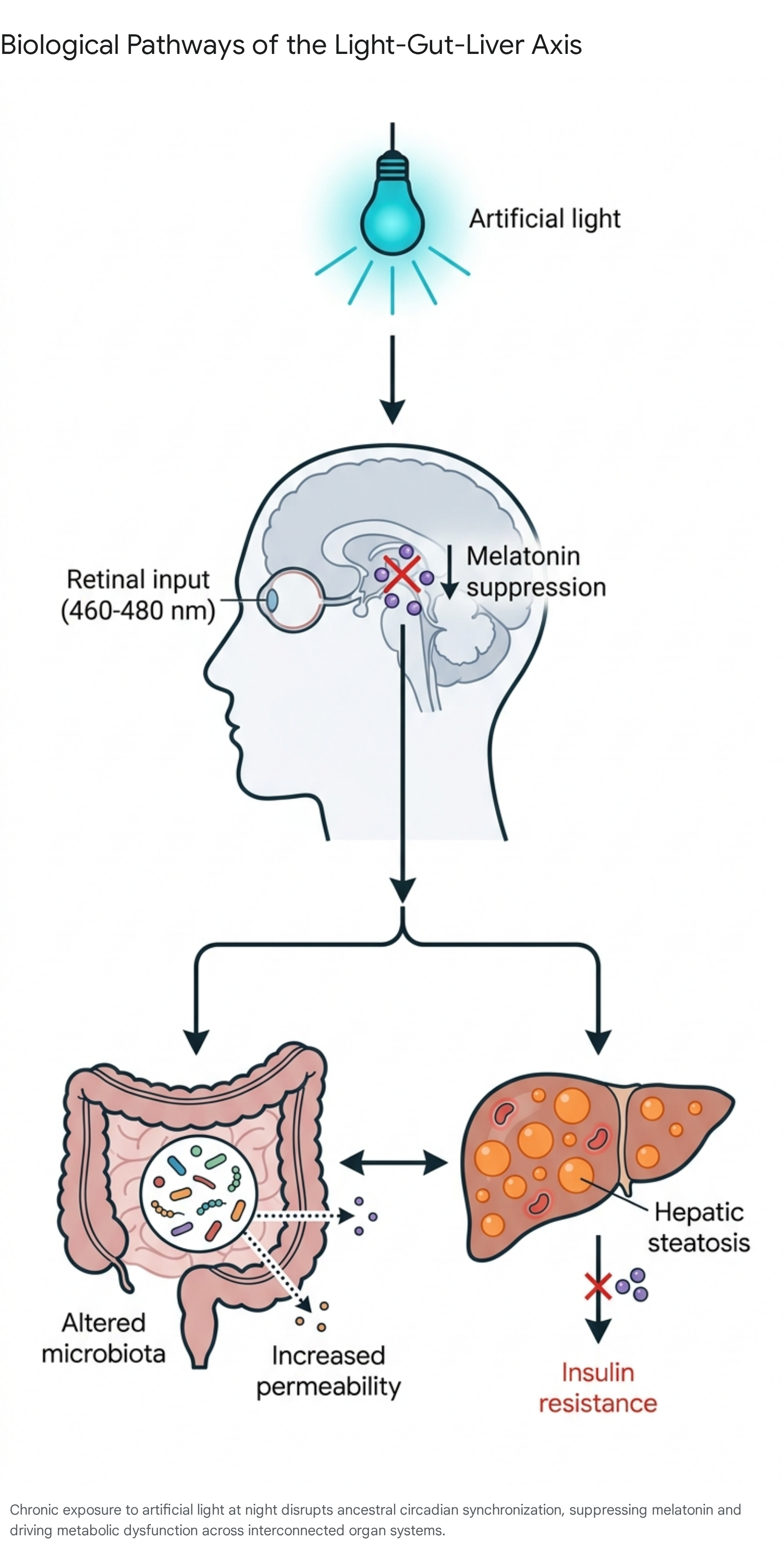
Disrupted circadian transcription factors (such as BMAL1 and CLOCK) trigger metabolic dyssynchrony, characterized by an abnormal overlap of anabolic and catabolic phases 3335.
This circadian overlap drives unabated hepatic de novo lipogenesis, leading to the accumulation of triacylglycerols (TAGs), lipid droplets within hepatocytes, and systemic insulin resistance 736. Concurrently, continuous dim light exposure induces gut dysbiosis and increased intestinal permeability. Experimental models subjected to three-month dark deprivation demonstrate a profound (more than five-fold) suppression of plasma melatonin, the development of hepatic steatosis, lipofuscin accumulation, damage to mitochondria, and the activation of cellular senescence markers (p16, p21, p53) 33. Together, these effects establish ALAN as a primary environmental driver of metabolic dysfunction-associated steatotic liver disease (MASLD) 73337. In human populations, particularly among older adults, increased outdoor nighttime light intensity correlates robustly with short sleep duration, compounding the overall metabolic risk 37.
Nutritional Influences on Sleep Architecture
The transition from ancestral dietary patterns to modern nutritional paradigms extends its deleterious effects directly into objective sleep physiology. Extensive target-trial emulations utilizing 4.8 thousand person-nights of high-resolution actigraphy and real-time dietary logging demonstrate that routine, day-to-day nutritional choices exert immediate and measurable effects on sleep architecture 3841.
Dietary Fiber and Restorative Sleep Metrics
Ancestral-style dietary inputs, specifically characterized by high fiber density and whole-plant diversity, are robustly associated with highly restorative sleep architectures. In these robust trial emulations, higher fiber intake was associated with a structured redistribution of sleep stages, including a +0.59 percentage point increase in deep (slow-wave) sleep and a +0.76 percentage point increase in REM sleep 3841. Simultaneously, high fiber consumption was linked to a -1.35 percentage point decrease in lighter, less restorative sleep and a reduction in the mean nocturnal heart rate by -1.14 bpm, indicating improved autonomic down-regulation 3841.
Conversely, modern dietary patterns characterized by higher percentages of saturated fat and refined sugars predict disrupted sleep profiles. Higher sugar intake correlates with increased arousals and sleep fragmentation, while a higher percentage of energy derived from saturated fats predicts less slow-wave sleep 39. Furthermore, modern meal-timing behaviors - specifically the consumption of heavy, calorie-dense evening meals - elevate nocturnal autonomic tone and increase sleep-onset latency 3841.
| Sleep Metric | Ancestral Diet Profile (High Fiber/Plant Diversity) | Modern Diet Profile (High Saturated Fat/Sugar/Late Timing) |
|---|---|---|
| Deep (Slow-Wave) Sleep | Increased proportion (+0.59 pp) 384139 | Decreased overall duration 39 |
| REM Sleep | Increased proportion (+0.76 pp) 3841 | Disrupted / Reduced duration 4139 |
| Arousals / Fragmentation | Reduced wake-after-sleep onset (WASO) 4041 | Increased frequency of nocturnal arousals 3941 |
| Autonomic Tone | Down-regulated (lower mean nocturnal heart rate) 3841 | Up-regulated (higher mean nocturnal heart rate) 3841 |
Microbiome Diversity and Cognitive Health
The connection between sleep and diet is highly mediated by the gut microbiome. Studies utilizing actigraphy coupled with microbiome sampling demonstrate that total microbiome diversity (measured via Shannon and Chao1 indices) is positively correlated with increased sleep efficiency and total sleep time, and negatively correlated with wake-after-sleep onset (WASO) 404142. Within specific phyla, the richness of Bacteroidetes and Firmicutes shows positive associations with sleep efficiency 42.
Moreover, this microbial-sleep axis influences cognitive and immunological markers. Microbiome diversity exhibits significant positive correlations with performance on abstract cognitive matching assessments, while showing complex regulatory relationships with inflammatory cytokines such as interleukin-6 (IL-6), a biomarker known to influence sleep architecture 4042. The modern loss of microbial diversity therefore cascades into degraded sleep quality and subsequent cognitive and immunological deficits.
Immune System Dysregulation
The human immune system evolved over millennia in an environment replete with varied microbial, parasitic, and environmental antigens. In the absence of these continuous exposures, the regulatory mechanisms of the immune system can become hyper-reactive, resulting in an inward attack against host tissues.
The Old Friends Hypothesis
Closely aligned with the Missing Microbes theory, the "Old Friends" hypothesis posits that routine early-life exposure to specific, harmless environmental microbes and helminths is fundamentally required for the proper functional maturation of regulatory T-cells 2622. Without this critical immune "education," the body fails to establish appropriate inflammatory tolerance. The modern hyper-sanitized environment, characterized by reduced physical contact with soil, domestic and farm animals, and varied plant microbiomes, contributes to an evolutionary mismatch that drives contemporary epidemics of asthma, allergic rhinitis, and complex autoimmune disorders 226.
Early Life Exposures and Childhood Leukemia
The severe pathological consequences of mismatched immune education are starkly evident in the etiology of childhood acute lymphoblastic leukaemia (ALL). Epidemiological and genetic modeling strongly endorse a dual-hit, multifactorial mechanism for ALL pathogenesis. The first step involves an in utero genetic event - such as a fusion gene formation or hyperdiploidy - that generates a covert, pre-leukaemic clone 43.
The critical second step - the postnatal acquisition of secondary genetic changes (such as RAG and AID-driven copy number alterations) that convert the clone into overt leukemia - is triggered by a delayed infectious exposure 43. In ancestral environments, continuous early-life microbial exposures correctly primed and educated the infant immune system. In modern, highly sanitized societies, behavioral changes (such as delayed daycare attendance or smaller family sizes) restrict early exposure. When the child eventually encounters a common, non-endemic pathogen later in childhood, the unprimed immune system mounts a severely dysregulated inflammatory response. This response inadvertently triggers DNA-altering enzymes that facilitate the secondary oncogenic mutations 43. Consequently, childhood ALL can be viewed as a paradoxical consequence of modernization - a potentially preventable cancer rooted in the evolutionary mismatch of contemporary immune isolation.
Ongoing Biological and Cultural Evolution
While mismatch theory highlights the pathological friction between ancient biology and modern environments, it is a persistent misconception to assume that human biological evolution has ceased. In reality, human evolution is actively progressing, fundamentally steered and accelerated by cultural and technological niche construction 124748.
Genomic Signatures of Recent Selection
Recent large-scale genomic analyses utilizing ancient DNA have overturned the assumption that anatomically modern humans have undergone minimal genetic change since leaving Africa. An unprecedented analysis of 16,000 ancient genomes across West Eurasia demonstrates that directional natural selection has significantly accelerated over the past 10,000 years, particularly following the transition from hunting and gathering to sedentary agriculture 44.
This agricultural shift rapidly altered population densities and dietary profiles, establishing entirely new selective pressures. Genomic scans reveal extensive signals of recent positive selection acting on hundreds of genes - notably those coding for cell surface proteins, immune responses, and metabolic pathways 4546. These hard selective sweeps reflect necessary biological adaptations to the oxidative stress of carbohydrate-rich farming diets and the novel infectious disease loads generated by dense, sedentary communities 846.
Lactase Persistence Dynamics
Lactase persistence (LP) - the continued physiological production of the lactase enzyme into adulthood - serves as the quintessential example of gene-culture coevolution and ongoing biological adaptation 4748. Prior to the Neolithic revolution, all human populations were lactase non-persistent, losing the ability to digest lactose shortly after weaning 4849.
Following the cultural invention of animal husbandry and dairying, specific regulatory mutations upstream of the LCT gene provided a massive fitness advantage in populations reliant on dairy for hydration, calories, and calcium assimilation during periods of resource scarcity 4849. Genetic analysis reveals a pattern of convergent evolution, with the -13.910:C>T variant sweeping through European populations, while distinct mutations (-14.010:G>C) arose independently in African pastoralist groups 4950.
Furthermore, recent comprehensive genomic studies highlight the complexity of this adaptation in other regions. In South Asia, the prevalence of the -13.910*T allele was driven largely by historical Steppe pastoralist-related gene flow. However, intense localized positive selection further amplified its frequency in specific modern pastoralist communities, such as the Toda in South India and the Gujjar in Pakistan, highlighting the intricate, ongoing interplay between human migration, cultural practices, and biological adaptation 51.
Cultural Interventions Replacing Biological Adaptation
As humans increasingly modify their surrounding ecosystems, cultural and technological innovations are rapidly substituting for slow genetic adaptation. Advanced medical technologies, modern sanitation infrastructure, and agricultural engineering now solve biological challenges far more rapidly than genetic mutations can arise and fixate within a broad population 4748.
For example, modern surgical interventions like cesarean sections bypass the stringent selective pressures previously imposed by maternal pelvic constraints and fetal head size, fundamentally altering the trajectory of obstetrical evolution by allowing genes associated with narrow birth canals to persist 47. By buffering humanity against historical environmental pressures, culture itself has become the primary mechanism of human adaptation, shifting the evolutionary burden from the physical genome to shared human institutions and technological systems 1248.
Epistemological and Methodological Caveats
Applying evolutionary theory to modern human health requires rigorous methodological frameworks and philosophical care to avoid misinterpretation and overextending the paradigm.
Rigorous Evaluation of Mismatch Hypotheses
Not every modern affliction is the result of an evolutionary mismatch, and not all cultural evolution is inherently adaptive 12. To rigorously evaluate a mismatch hypothesis, researchers must meet strict evidentiary criteria. They must precisely identify the biological trait, define its functional utility in the ancestral environment (E1), trace its genetic or epigenetic inheritance mechanism, and empirically demonstrate its fitness or health detriments in the modern environment (E2) 12. Furthermore, researchers must isolate true genetic or developmental mismatches from cases where a trait remains completely stable but the novel environment alters its outcome, versus cases where novel environments induce entirely new, uncanalized pathologies that have no ancestral precedent 12.
Avoiding the Naturalistic Fallacy
A frequent peril in the application of evolutionary medicine is the "naturalistic fallacy" - the erroneous philosophical leap of deducing an ethical or prescriptive "ought" from a biological "is" 52. The premise that a specific behavior, diet, or trait is "natural" or evolutionarily ancient does not inherently render it morally good, optimally healthy, or ethically desirable in a modern context 535460.
Natural selection optimizes strictly for reproductive fitness and gene transmission in specific historical environments, not for lifelong physical comfort, moral virtue, or extended post-reproductive longevity 54. For instance, while it was highly advantageous for early human populations to gorge on simple sugars when encountered in the wild, indulging that same "natural" biological impulse in a modern environment leads directly to systemic disease 30. Similarly, the fact that humans may possess ancient biological mechanisms for aggression or tribalism does not justify those behaviors ethically in a modern society.
Understanding evolutionary mismatch provides a uniquely powerful explanatory framework for understanding the origins of chronic human pathology. However, it remains a descriptive biological tool, not a prescriptive moral compass. By carefully separating biological facts from ethical imperatives, researchers and public health officials can utilize evolutionary insights to intelligently redesign modern environments, realigning urban infrastructure, dietary guidelines, and daily routines with the physiological realities of human biology.