Evidence for quantum coherence in biological systems
Introduction to Quantum Biology
The scientific conceptualization of biological systems was historically rooted in classical physics and classical chemistry. The cellular environment - characterized as warm, wet, and highly subject to thermal noise - was universally presumed to induce rapid decoherence, immediately destroying the delicate phase relationships required to sustain quantum mechanical states. Consequently, standard biochemical models utilizing Newtonian mechanics, classical thermodynamics, and classical chemical kinetics were deemed entirely sufficient to explain the molecular machineries of life, ranging from metabolic pathways to sensory transduction 12. The boundaries between the classical realm of biology and the quantum realm of fundamental physics were considered absolute.
However, the rapid development of sophisticated measurement techniques in the late 20th and early 21st centuries - most notably multidimensional ultrafast spectroscopy, cavity ring-down detection, and hybrid quantum mechanics/molecular mechanics (QM/MM) computational simulations - has forced a profound re-evaluation of these boundaries 23. A robust body of empirical evidence indicates that life at the molecular scale does not merely tolerate the quantum realm; in specific contexts, it actively exploits non-trivial quantum phenomena. The emerging discipline of quantum biology systematically investigates how living organisms harness quantum coherence, quantum entanglement, and quantum mechanical tunneling to achieve reaction rates, energy transfer efficiencies, and sensory sensitivities that classical paradigms fundamentally fail to predict 23.
Contemporary quantum biology primarily centers on three distinct phenomena observed in nature: the near-unity quantum efficiency of excitation energy transfer in photosynthetic light-harvesting complexes, the extraordinary sensitivity of avian magnetoreception enabling precise global navigation, and the massive rate accelerations achieved by enzyme-catalyzed chemical reactions 34. Within each of these domains, highly specialized protein architectures have evolved to isolate, stabilize, or manipulate subatomic particles.
Despite the accumulation of experimental data, the field remains characterized by rigorous debate regarding the evolutionary status of these quantum effects. The central theoretical challenge is distinguishing between an "epiphenomenon" - a necessary but functionally incidental byproduct of building complex molecules from atomic constituents - and a "functional necessity" - a quantum mechanism explicitly optimized by natural selection to confer a biological advantage 678. Recent technological breakthroughs throughout 2024 and 2025 have provided unprecedented clarity on this distinction. By leveraging ultra-low temperature assays, isotope substitution, and genetically modified protein analysis, the scientific consensus regarding what quantum biology actually demonstrates has shifted significantly 356.
Photosynthetic Energy Transfer
Photosynthesis represents one of the most fundamental biological processes on Earth, converting solar radiation into chemical energy. Photosynthetic organisms utilize specialized antenna complexes to capture photons and transfer the resulting excitation energy to a central reaction center. This excitation energy transfer (EET) occurs with a quantum efficiency that routinely approaches 100 percent, meaning virtually every absorbed photon successfully reaches the reaction center to drive charge separation 78. The physical mechanisms underlying this extraordinary efficiency, particularly the role of quantum coherence, have been the subject of intensive scrutiny.
Structural Organization of the Fenna-Matthews-Olson Complex
The Fenna-Matthews-Olson (FMO) pigment-protein complex is the simplest and most extensively studied natural light-harvesting complex. Found in green sulfur bacteria, such as Chlorobaculum tepidum and Prosthecochloris aestuarii, the FMO complex operates in extremely low-light environments, necessitating near-perfect energy transfer efficiency 8139. It functions as a molecular wire, mediating the transfer of excitation energy from the massive peripheral light-harvesting chlorosomes to the membrane-embedded bacterial reaction center 89.
Structurally, the FMO protein is a water-soluble homotrimer that exhibits distinct C3 symmetry 139. Each of the three monomeric subunits encapsulates eight bacteriochlorophyll a (BChl a) pigment molecules 6139. These pigments are rigidly fixed within the protein scaffold, bound primarily through the chelation of their central magnesium atoms to specific amino acid residues, most commonly histidine, or via water-bridged oxygen atoms 9. The spatial orientation of the complex dictates its functional flow: specific pigments (BChl 1 and 6, following original crystallographic numbering) are oriented toward the chlorosome to accept energy, while pigments proximal to the reaction center (BChl 3 and 4) act as the terminal emitters 9.
The efficiency of the FMO complex is governed by the specific site energies of the individual BChl a molecules. The surrounding protein environment creates unique, localized electrostatic interactions that finely tune the transition energy of each pigment 79. This arrangement establishes a structured energetic funnel that directs the exciton continuously downhill toward the reaction center 67. Furthermore, because the pigments are densely packed and spatially constrained within a few nanometers of one another, they exhibit strong excitonic coupling. Upon photoexcitation, the absorbed energy does not reside on a single, discrete pigment; instead, it is delocalized across multiple BChl a molecules, creating shared molecular exciton states 610.
The Initial Hypothesis of Electronic Quantum Coherence
For decades, the standard model for excitation energy transfer in photosynthesis was based on Förster Resonance Energy Transfer (FRET). FRET is a semi-classical mechanism that describes the incoherent, step-by-step hopping of energy between weakly coupled donor and acceptor molecules, driven by dipole-dipole interactions 510. However, basic Förster theory struggled to completely account for the extreme speed (often under 200 femtoseconds) and near-perfect efficiency of the transfer within tightly packed chromophore arrays like the FMO complex, where pigments are separated by distances smaller than the theoretical limits of classical FRET models 10.
Beginning in 2007, groundbreaking experiments utilizing two-dimensional electronic spectroscopy (2DES) challenged the classical hopping paradigm. Researchers observed long-lived oscillatory signals, known as quantum beats, in the 2D spectra of the FMO complex when measured at cryogenic temperatures (77 K). These oscillations were interpreted as direct evidence of sustained electronic quantum coherence between excitonic states 581117. Subsequent studies extending into 2010 reported that these coherent beating signals persisted for at least 300 femtoseconds even at physiological ambient temperatures (277 K to 296 K) 81117.
This interpretation had profound implications for biological physics. It suggested that the FMO complex operated akin to a dedicated quantum computer executing a quantum random walk 171213. In this model, the excitation energy existed as a wave-like quantum superposition, simultaneously sampling multiple potential pathways through the pigment network. By exploring all paths concurrently, the system could instantaneously collapse into the most efficient, least dissipative route to the reaction center 31320. This phenomenon, widely termed "environmentally assisted quantum transport," posited that the protein matrix actively protected delicate electronic coherence from thermal degradation, rendering the energy transfer robust against static disorder 8171415.
Two-Dimensional Electronic Spectroscopy Methodologies
The identification of these coherent states relied heavily on the advancement of multidimensional biomolecular spectroscopy. Two-dimensional electronic spectroscopy maps the pathways of light harvesting by applying sequences of ultrashort, femtosecond laser pulses to the sample 1023. By tracking the time-dependent evolution of cross peaks in the resulting spectra, researchers can identify couplings between distinct components of a complex system 10.
Coherent 2D spectra are exquisitely sensitive to oscillations in amplitude arising from superpositions. However, the exact nature of these superpositions - whether they represent electronic coherence (between different exciton states), vibrational coherence (nuclear wavepackets), or vibronic coherence (mixed electronic and vibrational states) - is inherently difficult to untangle because these features overlap significantly in the spectral readouts 1015. Frequency beating maps, recorded using specific phase differences in rephasing and non-rephasing pulse sequences, eventually became the critical tool necessary to distinguish true electronic superpositions from background molecular vibrations 1015.
The Consensus Shift Toward Vibrational Coherence
Despite the paradigm-shifting nature of the initial quantum walk hypothesis, subsequent theoretical advancements and the deployment of more precise diffractive-optics-based 2DES techniques between 2017 and 2025 have forced a fundamental revision of the FMO complex's operational model 52316. A rigorous re-evaluation of the spectroscopic data demonstrated that the remarkably long-lived oscillations observed in earlier experiments were not signatures of electronic coherence. Instead, they were primarily manifestations of vibrational coherence (nuclear motion) in the electronic ground state, or localized vibronic mixing 5915231617.
When research consortiums systematically varied the ambient temperature and carefully parameterized system-bath interactions, they discovered that true electronic coherence between distinct excitonic states is exceedingly fragile in biological environments. In the FMO complex under physiological conditions, electronic coherence is violently damped by surrounding thermal noise, decaying on a timescale of roughly 60 to 100 femtoseconds 5151617. Because the actual physical transfer of energy to the reaction center requires several picoseconds to complete, an electronic coherence lifetime of under 100 fs is mathematically insufficient to functionally govern or optimize the transport dynamics 516.
Rather than relying on long-lived electronic superpositions, the current consensus dictates that nature exploits the intricate interplay between electronic states and molecular vibrations, known as vibronic coupling. High-frequency vibrational modes inherent to the protein scaffold and the pigments themselves (such as heavily studied modes around 180 cm - 1) transiently resonate with the energy gaps between excitonic states 151626. These structural vibrations continuously shake the electronic energy levels, momentarily bridging energy gaps and facilitating a highly efficient, directed flow of energy downhill 1626. Consequently, while the protein scaffold indeed exhibits long-lived, coherent vibrational motions that assist energy transfer, the excitation transfer itself does not depend on a sustained electronic quantum walk 52016.
Markovian Dynamics and Downhill Energy Transfer
To definitively isolate and measure electronic coherence separate from vibrational artifacts, Duan et al. mapped the coherent dynamics of the FMO complex across an extensive temperature gradient, ranging from an ultra-cold 20 K up to physiological room temperatures 2316.
At 20 K, the thermal fluctuations of the surrounding protein bath are sufficiently suppressed to allow transient electronic coherence to persist, marginally surviving up to 200 to 500 fs 2016. However, this coherence diminishes precipitously as the temperature rises. The analysis confirmed that at room temperature, the timescale of optical dephasing is perfectly equivalent to the electronic decoherence timescale. This equivalence reveals that the dynamics of excitation energy transfer in the FMO complex are fully Markovian at physiological temperatures 16. A Markovian process is fundamentally "memoryless," precluding the type of sustained, non-local quantum computation and simultaneous multi-path sampling originally hypothesized 16.
The prevailing biophysical consensus now concludes that the near-unity efficiency of the FMO complex does not arise from functional, long-lived electronic quantum coherence. Rather, it is the result of classical, highly optimized, incoherent Förster-type energy transfer. This efficiency is driven by an evolutionarily perfected arrangement of pigment site energies and strong, structurally fixed system-bath couplings that enforce a unidirectional downhill energy flow, preventing backflow and minimizing dissipation 56.
| Phenomenological Metric | Initial Quantum Walk Hypothesis (2007 - 2015) | Current Consensus Model (2024 - 2025) |
|---|---|---|
| Primary Source of 2DES Oscillations | Electronic quantum coherence (superposition of excitonic states). | Vibrational coherence in the electronic ground state; local vibronic mixing. |
| Electronic Coherence Lifetime (296 K) | > 300 fs (survives long enough to dictate transfer pathway). | < 100 fs (rapidly decoheres prior to transfer completion). |
| Dominant Energy Transfer Mechanism | Wave-like quantum transport; simultaneous sampling of multiple pathways. | Incoherent, structurally optimized downhill Förster Resonance Energy Transfer (FRET). |
| System Memory (Dynamics) | Non-Markovian (retains memory to optimize quantum pathing). | Fully Markovian at physiological temperatures (memoryless). |
| Role of the Protein Scaffold | Actively shields delicate electronic superpositions from thermal decoherence. | Tunes fixed pigment site energies and provides vibrational modes that assist classical hopping. |
Avian Magnetoreception and the Radical Pair Mechanism
While the evidence supporting functional electronic quantum coherence in photosynthesis has been systematically dismantled by recent measurements, the case for active quantum phenomena in avian magnetoreception has grown exceptionally robust. Migratory songbirds, such as the European robin (Erithacus rubecula) and the migratory Japanese quail (Coturnix japonica), navigate thousands of kilometers with remarkable precision 2718291920. They achieve this using an internal biological compass sensitive to the inclination and orientation of the Earth's geomagnetic field 271819.
Behavioral and physiological experiments have long established that this magnetic sense is entirely light-dependent. Birds require specific wavelengths of blue or green light to properly orient; under pure red light or in total darkness, their navigational abilities fail 21. Furthermore, tracing the neural pathways has shown that magnetic information is processed in the visual system and transmitted to the brain via the thalamofugal pathway, confirming that the primary magnetoreceptor must reside within the retina 4273334.
The definitive biophysical explanation for this capability is the Radical Pair Mechanism (RPM), an inherently quantum mechanical process where the final chemical yields of a photoreaction are dictated by the coherent spin dynamics of intermediate, unpaired electrons 27183536.
Cryptochrome 4a as the Primary Magnetosensor
Extensive molecular, genetic, and spectroscopic research has isolated the cryptochrome (Cry) family of proteins as the biological host molecules for the radical pair mechanism 193522. Cryptochromes are blue-light-sensitive flavoproteins. They are evolutionarily homologous to photolyases - enzymes that utilize blue light to repair UV-damaged DNA - but have lost their DNA repair capabilities, evolving instead to perform specialized sensory and signaling functions, including circadian clock regulation and magnetoreception 27232425.
Avian genomes generally encode six specific cryptochrome isoforms: Cry1a, Cry1b, Cry2a, Cry2b, Cry4a, and Cry4b 2226. Among these, Cryptochrome 4a (CRY4a) is universally recognized by the 2024 - 2025 scientific consensus as the primary magnetosensor 182227. CRY4a is localized specifically within the double-cone photoreceptor cells of the retina 271833. Crucially, its expression levels are actively upregulated during migratory seasons, unlike the Cry1 and Cry2 variants which maintain flat, diurnal cycles typical of standard circadian regulators 2628. Recent biochemical characterizations of the Cry4b splice variant revealed that it lacks the ability to bind the essential light-absorbing flavin cofactor, rendering it functionally irrelevant to magnetic sensing and leaving CRY4a as the sole candidate 2226.
Photochemistry of the Tryptophan Tetrad
The radical pair mechanism within CRY4a is strictly initiated by photon absorption. The protein non-covalently binds a flavin adenine dinucleotide (FAD) chromophore deep within its molecular pocket 182529. Upon the absorption of a blue-light photon (approximately 450 nm), the FAD molecule is excited into an unstable, highly reactive state (FAD*), generating an electron vacancy 293031.
To stabilize this vacancy, an electron is rapidly transferred to the FAD from an adjacent electron donor. In avian CRY4a, this donor is the first in a highly conserved, structurally precise chain of four tryptophan amino acid residues, collectively known as the tryptophan tetrad 192332. In the European robin and pigeon models, these residues are sequentially numbered W395 (TrpA), W372 (TrpB), W318 (TrpC), and W369 (TrpD) 1929.
The initial photoexcitation triggers a rapid cascade of sequential electron hops. TrpA donates an electron to the FAD, generating a temporary hole on TrpA. TrpB instantly donates an electron to replenish TrpA; TrpC replenishes TrpB; and finally, the solvent-exposed terminal residue, TrpD, replenishes TrpC 19313248. This rapid, four-step sequential transfer effectively pushes the electron vacancy approximately 20 Å (2.0 nanometers) away from the FAD molecule to the surface of the protein 2233.
This specific 2.0 nm spatial separation is functionally critical for the sensory mechanism. If the radical pair remains too close together, exchange interactions are too strong, and the pair will immediately undergo charge recombination before external magnetic sensing can occur. If the radicals are transferred too far apart, the forward formation rate might be outpaced by ambient spin relaxation 213048. The optimized final configuration creates an isolated, spin-correlated radical pair composed of a flavin semiquinone radical (FAD• - ) and a terminal tryptophan radical (TrpD•+) 193522.
Quantum Spin Dynamics and Singlet-Triplet Interconversion
Once the terminal radical pair [FAD• - TrpD•+] is successfully established, the system enters a delicate, magnetically sensitive quantum state. Because the electron transferred to the FAD and the electron remaining on the tryptophan originally occupied the exact same atomic orbital before excitation, quantum mechanics dictates that their spin states are fundamentally entangled. They initially exist in a spin-correlated, antiparallel configuration known as the "singlet" state 273534.
However, each unpaired electron functions as an infinitesimal magnetic dipole and is therefore subjected to local magnetic forces. The electrons begin to precess around these local fields at specific Larmor frequencies 3351. The total local magnetic environment experienced by each electron is a composite of two distinct forces: the powerful internal magnetic fields generated by the nuclear spins of the surrounding atoms within the protein (hyperfine interactions), and the exceedingly weak external geomagnetic field (the Zeeman effect) 365152.
Because the molecular structure and atomic nuclei of the FAD molecule differ significantly from those of the TrpD residue, their respective hyperfine environments are asymmetric. Consequently, the two entangled electrons precess at different rates. This difference in Larmor precession continuously drives the radical pair to oscillate back and forth between the antiparallel singlet state and the parallel triplet state 273452. Crucially, the external Earth's magnetic field - despite its low intensity of approximately 50 microteslas - is sufficient to alter the extent, timing, and phase of this singlet-triplet interconversion 185152.
The quantum coherence of this oscillating spin state directly determines the macroscopic biochemical fate of the cryptochrome protein. If the radical pair is in the singlet state when the electrons attempt to recombine, back-electron transfer is chemically permitted (adhering to the Pauli exclusion principle), and the cryptochrome rapidly returns to its inactive, oxidized resting state without initiating a signal 2134. Conversely, if the pair is in the triplet state, direct recombination is strictly spin-forbidden 2151. Because the triplet state cannot immediately recombine, the radical pair lives longer. This extended lifetime allows the protein to undergo subsequent structural rearrangements - specifically, the opening of the phosphate-binding loop - which stabilizes the molecule into a biologically active, long-lived signaling state (FADH•) 293132.
By altering the fundamental ratio of singlet to triplet yields, the precise angle of the Earth's magnetic field relative to the bird's retina directly controls the ultimate concentration of the active cryptochrome signaling state. This mechanism flawlessly translates a subatomic quantum physical effect into a continuous biochemical signal 42734.
Evolutionary Conservation and Species-Specific Sensitivities
If the radical pair mechanism in Cryptochrome 4a is indeed a functional necessity selected for precise global navigation, evolutionary biology predicts that the protein should be highly optimized in migratory species compared to non-migratory taxa. Extensive genomic and biochemical studies conducted by international research consortiums in 2024 and 2025 have confirmed this prediction.
Researchers successfully extracted the genetic sequences for CRY4a from the night-migratory European robin, the migratory Japanese/common quail, and the non-migratory chicken. These proteins were synthesized in high volumes using bacterial cell cultures, allowing for comparative in vitro analyses utilizing cavity ring-down and transient absorption spectroscopy 29222733. The empirical data demonstrated that radical pairs formed in the cryptochromes of the migratory robin and quail exhibit significantly higher magnetic sensitivity, slower spin relaxation rates, and highly optimized singlet-triplet dephasing compared to the chicken protein 293033.
Furthermore, a massive phylogenetic analysis comparing over 360 distinct bird genomes revealed that while generic circadian cryptochromes (Cry1, Cry2) remain evolutionarily stagnant across all species, the gene encoding Cry4a has undergone rapid, positive evolutionary selection specifically within the passerine (songbird) order 3536. In distinct clades of birds that do not rely on long-distance magnetic navigation, such as parrots, hummingbirds, and Tyranni (Suboscines), the CRY4 gene has degraded and been entirely lost through evolutionary attrition 3536. This stark contrast strongly indicates that the quantum mechanical sensitivity of CRY4a is not a mere byproduct of flavoprotein chemistry, but a highly targeted sensory adaptation.
| Avian Clade / Species | Primary Ecological Behavior | In Vitro CRY4a Magnetic Sensitivity | Evolutionary Status of CRY4 Gene |
|---|---|---|---|
| European Robin (E. rubecula) | Night-Migratory | Extremely High (Peak relative effect ~21%) | Highly conserved; evidence of positive selection 3336. |
| Common Quail (C. japonica) | Migratory | High (Spectroscopically similar to Robin) | Highly conserved 29. |
| Rock Dove / Pigeon (C. livia) | Homing / Resident | Moderate (Peak relative effect ~17%) | Conserved 33. |
| Chicken (G. gallus) | Non-Migratory | Low (Peak relative effect ~14%) | Present, but features a structurally divergent Trp tetrad 33. |
| Parrots & Hummingbirds | Non-Migratory | N/A | Gene completely lost during evolutionary development 3536. |
Quantum Mechanical Tunneling in Enzyme Catalysis
Enzymes are the ubiquitous biological catalysts of life, capable of accelerating specific biochemical reactions by up to 20 orders of magnitude compared to equivalent uncatalyzed processes in aqueous solution. For decades, the origin of this catalytic power was explained entirely through classical transition state theory. Championed early on by Linus Pauling, the classical model posits that enzymes achieve their remarkable rate accelerations by binding substrates in highly specific geometrical orientations and providing electrostatic stabilization to the high-energy intermediate (the transition state) 3738. By stabilizing this transient structure, the enzyme lowers the activation energy barrier ($ΔH^‡$) that the reactants must physically climb over, via thermal excitation, to transform into stable products 3738.
However, beginning with precision kinetic measurements in the late 1980s and expanding into a vast body of literature over the last two decades, researchers observed severe kinetic anomalies that classical thermodynamics and Arrhenius equations could not reconcile. It is now definitively established that enzymes frequently utilize Quantum Mechanical Tunneling (QMT) to bypass the activation energy barrier entirely. This is particularly prevalent in oxidoreductase reactions involving the transfer of lightweight particles, specifically protons, hydride ions, and hydrogen atoms 33940.
Kinetic Isotope Effects and Tunneling Signatures
The primary empirical methodology for detecting QMT in biological systems is the measurement of Kinetic Isotope Effects (KIE). The KIE is calculated as the ratio of the reaction rate using a standard, light isotope (Hydrogen-1) to the reaction rate using a heavier, substituted isotope (Deuterium or Tritium) 373940.
Because a hydrogen atom is extremely light, its quantum spatial uncertainty - defined by its de Broglie wavelength (approximately 0.1 nm) - is relatively large. According to quantum mechanics, if a potential energy barrier is sufficiently narrow, the wave-like probability distribution of the hydrogen atom will physically extend through the barrier. This allows a portion of the particles to instantly "tunnel" to the product side without possessing the requisite thermal energy to climb over the peak of the barrier 34059. Deuterium, possessing twice the mass of hydrogen, has a substantially shorter de Broglie wavelength, rendering its tunneling probability exponentially lower.
Classical, over-the-barrier transition state theory dictates that the maximum possible KIE for a C-H versus C-D bond cleavage at room temperature should fall strictly between 7 and 10. This classical limit is based entirely on the differences in zero-point vibrational energies between the two isotopes 39. However, in extensively studied enzymatic systems such as soybean lipoxygenase-1, thermophilic alcohol dehydrogenase (ADH), and dihydrofolate reductase (DHFR), researchers routinely observe KIEs ranging from 30 up to 100 4041.
Furthermore, in a purely classical reaction, the KIE must exhibit a strong dependence on ambient temperature, as thermal energy is the sole mechanism for crossing the barrier. In these tunneling-dominant enzymatic systems, the KIE remains remarkably temperature-independent across broad thermal ranges. This temperature independence is a hallmark signature of quantum tunneling, proving that the reaction proceeds entirely "below the barrier" and is uncoupled from ambient thermal activation 34042.
Protein Dynamics and Vibrational Gating
The mathematical probability of quantum tunneling occurring is exponentially dependent on the mass of the transferring particle and the physical width of the energy barrier.
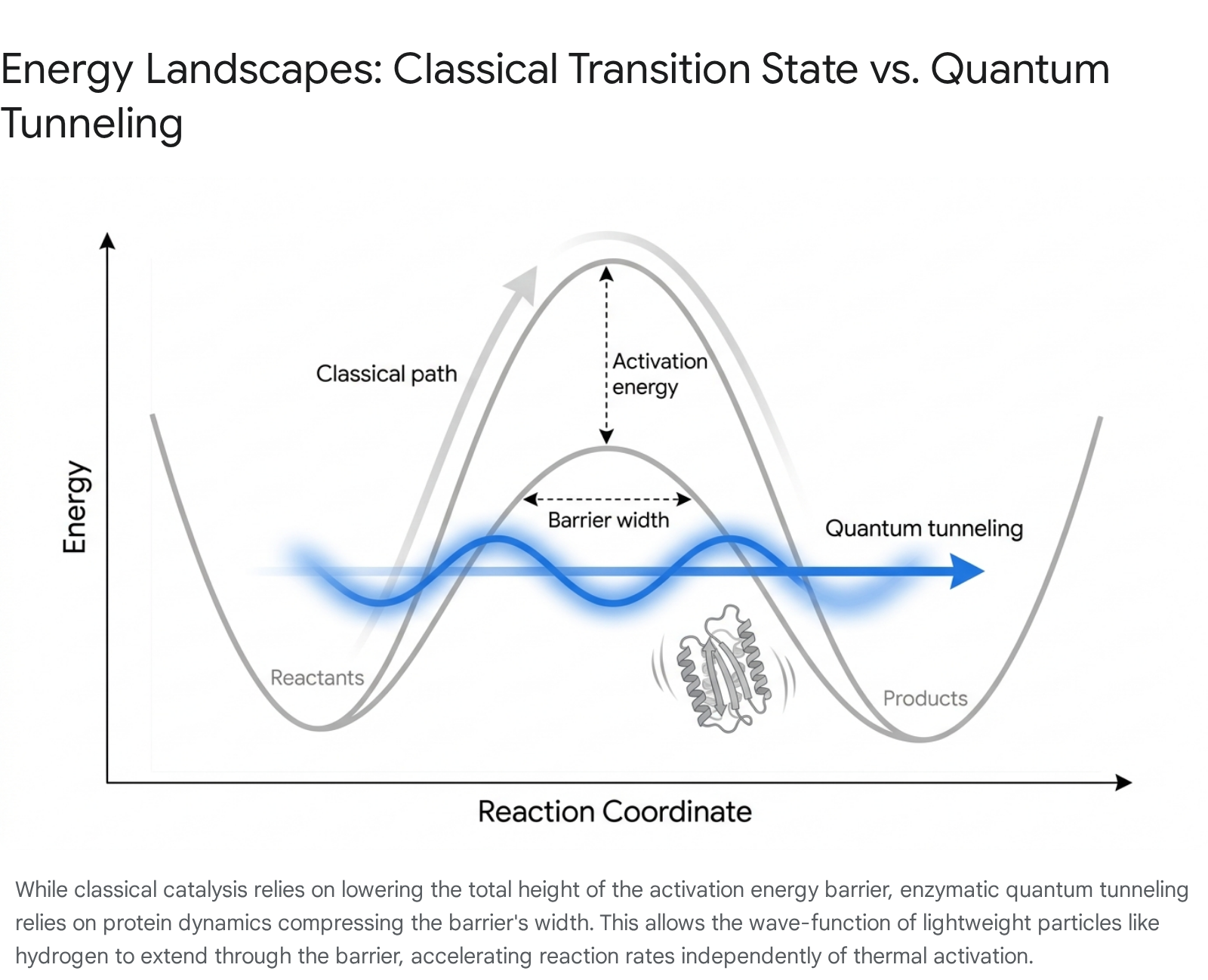
Importantly, it is relatively insensitive to the absolute height of the barrier 340. Because the mass of hydrogen is immutable, the primary physical parameter an enzyme can manipulate to artificially increase the tunneling rate is the barrier width. In a biological context, barrier width corresponds physically to the Donor-Acceptor Distance (DAD) between the reacting molecules 3842.
Advanced computational modeling and multidimensional NMR spectroscopy have established that enzymes are not rigid, static lock-and-key structures. They are highly dynamic nanomachines that constantly fluctuate and deform across a complex hierarchy of timescales 26384344. A phenomenon known as "vibrational gating" occurs when the rapid thermal motions of the protein scaffold - driven by the constant bombardment of surrounding water molecules and internal, pre-organized conformational shifts - momentarily compress the active site. This compression forces the hydrogen donor and acceptor atoms into extremely close proximity, far closer than standard Van der Waals radii would normally permit 4043.
For example, in the study of Escherichia coli dihydrofolate reductase (EcDHFR), which catalyzes the vital transfer of a hydride ion, researchers have identified a highly mobile structural element known as the Met20 loop 42. This loop undergoes a necessary conformational closure over the active site specifically during the chemical step of catalysis. Using hybrid quantum mechanics/molecular mechanics (QM/MM) simulations, scientists mapped distinct networks of amino acid residues that transmit kinetic energy from the solvated surface of the protein directly into the hydrophobic active site 343. These coupled, thermodynamically equilibrated motions effectively squeeze the donor and acceptor molecules together for a fraction of a picosecond. This is just long enough to establish a "tunneling-ready" configuration, drastically reducing the barrier width and causing a massive spike in the probability of QMT 3859.
The Debate Over Functional Necessity
While the existence of hydrogen tunneling in enzymes is undisputed and widely categorized as the "third reactivity paradigm" (alongside thermodynamic and kinetic control), a significant philosophical and biological debate persists regarding its precise evolutionary status: Is QMT merely a physical epiphenomenon, or is it a selected functional necessity? 6739.
The "epiphenomenon" argument posits that because a hydrogen atom is inherently light and naturally exhibits a long de Broglie wavelength, it will invariably tunnel whenever two atoms are forced close together. Proponents of this view argue that enzymes evolved purely to bind substrates tightly in geometrically constrained active sites (classical transition state stabilization). The fact that quantum tunneling happens to occur at these short distances is simply an unavoidable, passive byproduct of atomic physics, not a discrete trait actively selected by Darwinian evolution to enhance function 837.
Conversely, the "functional necessity" argument - which has gained dominant consensus support through recent structural evidence - asserts that enzymes have actively evolved their complex, global dynamic architectures specifically to harness and optimize this tunneling effect. The evidence supporting this view is robust 385945. When researchers study homologous enzymes adapted to different thermal extremes, such as comparing a thermophilic alcohol dehydrogenase (adapted to operate at 65°C) to a mesophilic equivalent (adapted to 25°C), they observe a breakdown in function when conditions are altered. Specifically, the thermophilic enzyme completely loses its tunneling capability when cooled to room temperature, as its dense protein scaffold becomes too rigid to perform the necessary compressive vibrations required to narrow the barrier width 5941.
Furthermore, point mutations introduced distal to the active site - sometimes up to 15 Å away from the reacting hydrogen - can completely abolish the tunneling signature without altering the static geometry or electrostatic environment of the primary binding pocket 4246. These remote mutations physically disrupt the allosteric networks responsible for transmitting vibrational energy, effectively "freezing" the donor-acceptor distance. This demonstrates that the entire macro-architecture of the protein, extending far beyond the immediate catalytic residues, is evolutionarily tuned to sustain the precise molecular dynamics required for quantum mechanical barrier compression 384043. Consequently, harnessing QMT through structural dynamics is now viewed as an evolved necessity for achieving physiological reaction rates.
| Catalytic Mechanism | Primary Method of Barrier Modification | Expected Kinetic Isotope Effect (KIE) | Temperature Dependence of Reaction Rate | Pathway Classification |
|---|---|---|---|---|
| Non-Enzymatic Solution | None (Relies entirely on ambient thermal heat) | Low (~1 to 5) | Highly Dependent | Purely Classical |
| Classical Enzyme Action | Lowers Absolute Barrier Height ($ΔH^‡$) via stabilization | Moderate (Theoretical Maximum of 7-10) | Dependent | Semi-Classical |
| QMT-Optimized Enzyme (e.g., DHFR, ADH) | Compresses Barrier Width (DAD) via global protein dynamics | Anomalously High (Consistently >30 to >100) | Largely Independent (Proceeds below barrier) | Quantum Mechanical Tunneling 394042 |
Conclusions on Quantum Biological Mechanisms
The rigorous pursuit of functional quantum biology over the past two decades has continually refined our understanding of the interface between classical thermodynamics and quantum mechanics in living systems. The expansive evidence accrued through 2025 demonstrates a highly nuanced reality: nature exploits quantum mechanics not as a universal, ubiquitous design language, but as a specialized, precision tool deployed selectively when classical mechanisms fail to meet stringent survival or environmental constraints.
In the realm of photosynthetic excitation energy transfer, the initial allure of discovering sustained, room-temperature electronic quantum computing in biology has been superseded by a more grounded, classical reality. Comprehensive spectroscopic mapping confirms that the FMO complex utilizes highly optimized, classical, Markovian energy hopping. The extremely rapid decoherence of electronic states at physiological temperatures confirms that sustained electronic superpositions are not a functional necessity for efficient light harvesting, relying instead on structural tuning and transient vibrational assistance 516.
Conversely, in the domains of avian magnetoreception and enzymatic catalysis, non-trivial quantum mechanics is foundational to biological function. The radical pair mechanism embedded within Cryptochrome 4a relies fundamentally on the prolonged quantum coherence of entangled electron spins. The exquisite sensitivity of this system to the Earth's weak magnetic field - combined with its definitive evolutionary conservation in migratory species and corresponding degradation in non-migratory species - proves that life can harbor and protect localized quantum phenomena to derive macroscopic sensory data 183336. Similarly, enzyme catalysis utilizes evolved, large-scale protein dynamics to compress energy barriers, making the quantum mechanical tunneling of hydrogen atoms a highly regulated, functional necessity rather than a passive, unavoidable chemical byproduct 33840. Ultimately, the study of quantum biology reveals that living organisms are supremely adaptable architectures, capable of bridging the quantum-classical divide to optimize efficiency, reactivity, and survival in highly specialized niches.