Evidence for Human Pheromones and Chemosignaling
The question of whether humans secrete and detect pheromones has provoked decades of scientific inquiry, characterized by highly contested methodologies, evolutionary debates, and commercial exploitation. While the existence of true human pheromones - defined strictly as species-wide chemical signals that elicit innate, stereotyped physiological or behavioral responses - remains fiercely debated, the broader phenomenon of human chemosignaling is a well-established biological reality. Humans routinely communicate social, emotional, and biological information through olfactory cues, utilizing volatile compounds emitted in sweat, tears, and glandular secretions to modulate aggression, arousal, and social appraisal in receivers.
Anatomical and Genomic Architecture of Human Olfaction
The anatomical and genetic mechanisms of human olfaction provide the critical framework for understanding how chemical signals are processed. In most terrestrial mammals, the detection of social odors and pheromones is strictly bifurcated between two systems: the main olfactory system, which detects volatile environmental odorants, and the accessory olfactory system, centered on the vomeronasal organ, which is primarily responsible for detecting non-volatile pheromones.
The Vestigial Status of the Vomeronasal Organ
In rodents and strepsirrhine primates (such as lemurs), the vomeronasal organ (VNO) is a bilateral, fluid-filled tubular structure located at the base of the nasal septum 12. It houses specialized vomeronasal sensory neurons that express two main families of G protein-coupled receptors: V1Rs (associated with Gαi2 proteins) and V2Rs (associated with G0α proteins). These receptors project to the anterior and posterior accessory olfactory bulb, respectively, and bind specific pheromonal ligands such as major urinary proteins and endocrine gland-secreting peptides 12. This signal transduction cascade relies heavily on the TRPC2 ion channel 23.
In human evolutionary history, the accessory olfactory system underwent massive genomic and anatomical decay. While the VNO develops during the early embryonic period, histological evidence indicates that the sensory cell population diminishes after 12 weeks of gestation, and the structure permanently dissociates from the central nervous system 1. In adult catarrhine primates, which include Old World monkeys, apes, and humans, the VNO is reduced to a non-operational, vestigial epithelial duct 12. Humans lack the vomeronasal nerve and an accessory olfactory bulb to process these signals centrally 13.
Genomic analyses decisively support this anatomical regression. The gene encoding the critical TRPC2 transduction channel is a non-protein-producing pseudogene in humans 23. Similarly, the human genome contains no functional V2R genes, and approximately 95% of the V1R gene repertoire consists of pseudogenes 23. Due to the absence of the anatomical structures, neural pathways, and functional genes required for classic vomeronasal chemoreception, the scientific consensus holds that humans cannot process pheromones through the accessory olfactory system in the manner observed in non-human mammals 345.
Trace Amine-Associated Receptors in the Main Olfactory Epithelium
Despite the regression of the VNO, humans remain highly capable of processing social chemosignals through the main olfactory epithelium (MOE). A small number of functional V1R-like receptors have been identified in the human MOE, suggesting that some rudimentary chemosensory capabilities may have been co-opted by the main olfactory system 67.
More significantly, modern genomic research has identified Trace Amine-Associated Receptors (TAARs) as a secondary class of chemosensory receptors located within the MOE 89. Humans possess five intact and functional TAAR genes (TAAR2, TAAR5, TAAR6, TAAR8, and TAAR9) that recognize volatile primary, secondary, and tertiary amines 89. TAAR5, located on human chromosome 6q23.2, acts as a highly specific receptor for trimethylamine (TMA) and dimethylethylamine (DMEA) 910. Unlike murine TAAR5, which also detects secondary amines, human TAAR5 exhibits distinct species-specific ligand tuning 9.
Unlike classical olfactory receptors, TAARs exhibit unique projection patterns. Transcriptomic and anatomical mapping reveals that TAAR5 is expressed not only in the olfactory epithelium but also ubiquitously throughout limbic brain regions that receive olfactory input. This includes the amygdala, hippocampus, orbitofrontal cortex, entorhinal cortex, and nucleus accumbens 811. Furthermore, human TAAR5 uniquely demonstrates significant inhibition of basal inositol-3-phosphate formation and MAP kinase signaling upon the application of the thyroid hormone derivative 3-iodothyronamine (3-T1AM), indicating complex signal modulation 11. The presence of these receptors in regions of the brain responsible for emotional regulation and social behavior provides a plausible mechanical pathway for how human body odors can subconsciously influence psychological states.
Evaluation of Putative Steroid Pheromones
For over four decades, the search for human pheromones has been dominated by research into a specific class of testosterone-derived steroid molecules found in human sweat. However, the scientific validity of treating these specific molecules as established human pheromones has been heavily criticized.
Androstenes and Copulins in Human Secretions
The primary molecules investigated in the human pheromone literature are the androstene steroids: androstenone (5α-androst-16-en-3-one), androstenol (5α-androst-16-en-3α-ol), androstadienone, and estratetraenol 121213. These compounds are present in human apocrine sweat and exhibit musky odors, though their perception is subject to wide genetic variation; many individuals are entirely anosmic to them 1314. Early studies suggested that exposing women to androstadienone resulted in improved mood, heightened focus, and increased physiological arousal, particularly in specific social contexts 1416.
Another heavily researched class of compounds are "copulins," which consist of a mixture of volatile C2-C5 aliphatic acids secreted in the human vagina 1516. Early research claimed that copulin concentrations peaked during the follicular phase of the menstrual cycle. Subsequent studies reported that exposing men to these acids increased their salivary testosterone levels and simultaneously inhibited their ability to critically evaluate the physical attractiveness of women 1516.
Methodological Constraints and Publication Bias
Despite the volume of literature generated around androstenes and copulins, comprehensive reviews of the field conclude that there is no robust, bioassay-led evidence to classify these molecules as true human pheromones 121718. A rigorous 2015 review demonstrated that the original selection of these molecules was not based on their proven biological relevance to human communication, but rather because androstenone was a known and economically useful sex pheromone in pigs 1213.
The literature supporting androstenes and copulins is characterized by severe methodological shortcomings. Positive results are largely attributed to false positives generated by statistically underpowered studies, small sample sizes, an overestimation of effect sizes, and systemic publication bias 1219. Furthermore, most experiments expose participants to pharmacological doses of these steroids that are orders of magnitude higher than natural human physiological production, rendering the ecological validity of the findings highly questionable 1219.
Commercial Exploitation and Regulatory Scrutiny
The premature designation of these compounds as human pheromones has fueled a lucrative but scientifically unfounded commercial industry. Cosmetic companies market "pheromone perfumes" claiming to guarantee increased sexual attractiveness. The economics of this industry rely heavily on the narrative rather than the chemistry, as the synthetic steroid compounds cost pennies per unit but retail for substantial margins 16.
Historical efforts to commercialize these findings date back to the 1980s. The Athena Institute, founded by Winnifred Cutler, has marketed products like "Athena Pheromone 10X" and "10:13," claiming they were backed by published science and capable of increasing romantic attention and regulating menstrual cycles 222324. However, independent scientific scrutiny has consistently challenged these claims. Critics point out that early formulations were simply DHEA and androstenol, and that the fundamental premise is flawed given the lack of a functional VNO in humans 232520.
This commercialization has recently faced legal and regulatory challenges. Class action lawsuits against brands like Pure Instinct allege that marketing pheromone perfumes as scientifically proven to attract others constitutes false advertising. These legal complaints cite genetic and anatomical evidence that humans lack the necessary receptors to detect these supposedly "human-compatible" pheromones, arguing that the products offer nothing more than conventional fragrance 25. Furthermore, independent analyses of various commercial products reveal that many contain non-human mammalian pheromones (derived from pigs or dogs), which have zero cross-species efficacy, or rely entirely on the placebo effect to boost user confidence 132728.
| Putative Chemosignal | Primary Anatomical Source | Proposed Behavioral / Physiological Effect | Current Scientific Consensus |
|---|---|---|---|
| Androstenes | Male apocrine sweat | Enhances female mood; signals male dominance and sexual viability. | Unsupported. Originally identified in pigs. Human effects are inconsistent and likely artifactual due to unnatural laboratory dosing. 12 |
| Copulins | Vaginal secretions | Increases male testosterone; impairs male judgment of facial attractiveness. | Highly Contested. Frequent replication failures. Effects may be linked to general odor pleasantness rather than specific pheromonal pathways. 151621 |
| MHC-Dissimilar Odors | General body odor | Promotes mate choice favoring genetic dissimilarity; increases relationship satisfaction. | Refuted. Massive genomic meta-analyses show no deviation from random chance in real-world human mate selection. 222324 |
| Hexadecanal (HEX) | Newborn scalp, skin, feces | Triggers reactive aggression in women; blocks reactive aggression in men. | Supported. Replicated physiological and behavioral dissociation with specific neural connectivity mapping. 252627 |
| Emotional Tears | Lacrimal glands (during sadness) | Dampens male physiological arousal and reduces reactive aggression by >40%. | Supported. Proven to activate specific olfactory receptors (in vitro) and downregulate aggression networks. 282930 |
| Areolar Secretions | Montgomery's glands (lactation) | Triggers innate rooting, suckling, and head-turning in all newborns. | Highly Supported. Considered the strongest candidate for a classic, universal human releaser pheromone. 121731 |
The Major Histocompatibility Complex and Mate Selection
The Major Histocompatibility Complex (MHC), known in humans as Human Leukocyte Antigen (HLA), comprises a highly polymorphic group of genes responsible for regulating the adaptive immune system and distinguishing self from non-self proteins 224032. Across various vertebrate taxa, MHC genotype has been shown to influence body odor, serving as a chemical cue that guides mate selection.
Evolutionary Hypotheses of Genetic Compatibility
The evolutionary framework for MHC-associated mate choice rests on two primary hypotheses. The first is "heterozygote advantage," which posits that by selecting a mate with a dissimilar MHC genotype, individuals produce offspring with greater MHC heterozygosity, equipping them to detect and resist a broader spectrum of pathogens. The second is "inbreeding avoidance," wherein extreme allelic diversity acts as a kin-recognition mechanism to prevent incestuous mating and its associated genetic penalties 403242.
In humans, this theory gained widespread traction following a 1995 study by Claus Wedekind, commonly known as the "sweaty T-shirt study." Women who smelled T-shirts worn by anonymous men reported that the body odors of men with dissimilar MHC profiles were more pleasant and reminiscent of former partners 4042. Interestingly, the study noted that women taking oral contraceptives exhibited a reversed preference, favoring MHC-similar odors. This led to hypotheses that hormonal contraceptives could disrupt evolved biological mate selection mechanisms, potentially incurring long-term fertility costs 404243.
Genomic and Meta-Analytical Refutations
Despite the elegance of the MHC heterozygote advantage hypothesis, recent massive technological and statistical analyses have effectively dismantled the notion that MHC dissimilarity drives human mate selection.
A 2020 comprehensive meta-analysis investigated four domains of the MHC hypothesis: genomic mate selection in actual couples, relationship satisfaction, odor preference, and overall combined effects 222344. The analysis of genomic studies - which represent actual reproductive decisions rather than laboratory odor ratings - revealed zero association between MHC dissimilarity and mate choice in real-world couples 2244. Furthermore, the meta-analysis found no significant effect of MHC dissimilarity on relationship satisfaction or odor preferences, noting substantial publication bias in the earlier literature 2244.
These meta-analytical findings are strongly corroborated by modern whole-genome sequencing efforts. A study analyzing 239 mate-pairs of Dutch ancestry using data from the Genome of the Netherlands project investigated whether the genetic content of the MHC influenced partner selection 2433. The results demonstrated that the MHC regions of established couples were no more genetically dissimilar than those of two randomly selected individuals (Qc = 0.829; permutation-based p = 0.703) 24. Limiting the analysis to only common variation or considering the extended MHC region did not alter the findings. Consequently, the scientific consensus has shifted away from the idea that humans utilize MHC-dependent olfactory cues to select mates 2433.
Hexadecanal and the Modulation of Aggression
While the research into sex steroids and genetic matching has faltered, recent investigations into different classes of human body volatiles have yielded profound discoveries regarding how humans communicate emotional and behavioral states chemically. One of the most significant recent discoveries involves the volatile compound hexadecanal (HEX).
Behavioral Dissociation Between Sexes
Hexadecanal is a perceptually odorless chemical emitted from human skin, breath, and feces, and it has been identified as one of the most abundant volatiles emitted from the heads of newborn infants 252746. In 2021, researchers utilized two validated behavioral metrics - the Taylor Aggression Paradigm (TAP) and the Fist-Clench Point Subtraction Aggression Paradigm (FC-PSAP) - to measure how sniffing HEX influences reactive aggression 2647.
The results revealed a remarkable biological dissociation. Exposure to HEX significantly blocked aggressive behavior in men (effect size r = 0.70 in the TAP paradigm) while simultaneously triggering an increase in aggressive behavior in women (effect size r = 0.47) 252648.
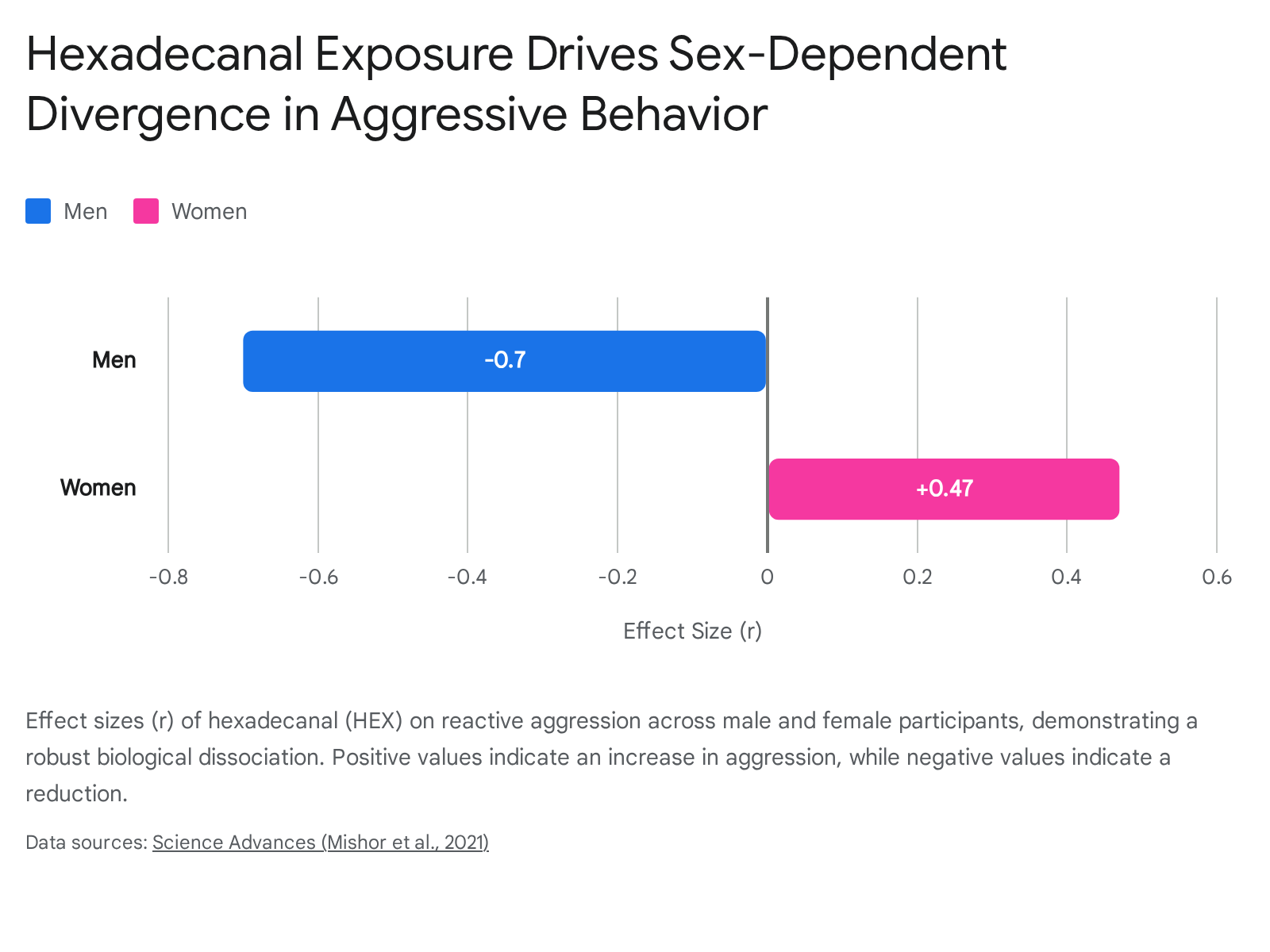
This behavioral shift occurred entirely outside of the participants' conscious awareness, as the addition of HEX to carrier substances like eugenol or mineral oil had no impact on the perceived pleasantness or intensity of the odor, nor did it alter baseline testosterone or cortisol levels 2647.
Neurological Connectivity and Social Appraisal
Functional magnetic resonance imaging (fMRI) mapped the precise neural substrates responsible for this behavioral dissociation. When both male and female participants were provoked while sniffing HEX, brain imaging revealed a significant increase in activity within the left angular gyrus, a region deeply implicated in the integration and perception of social cues 2526.
While the initial activation in the angular gyrus was identical across sexes, the downstream functional connectivity modulated by HEX was highly sex-dependent. In men, sniffing HEX increased functional connectivity between the left angular gyrus and a broader brain network associated with social appraisal and aggressive execution, including the right temporal pole, the bilateral lateral orbitofrontal cortex, and the amygdala 2627. In women, exposure to HEX decreased functional connectivity between these exact same regions 2526. This sex-specific modulation of the brain's aggression network mirrors the behavioral outcomes, suggesting a hardwired neurological response to the volatile cue 2648.
Evolutionary Context in Infant Rearing
The ecological relevance of a chemical signal that simultaneously pacifies men and enrages women is best understood through the evolutionary lens of infant rearing. Because HEX is emitted in abundance from the heads of newborn infants, researchers hypothesize that it functions as a survival mechanism 262747.
In the animal kingdom, maternal aggression (maternal defense behavior) is typically directed outward at intruders to protect offspring, directly improving infant survival rates 26. Conversely, paternal and non-paternal male aggression is frequently directed inward toward the infants themselves, posing a severe threat of infanticide 26. By emitting a single chemical signal that upregulates protective aggression in females while downregulating fatal aggression in males, infants utilize a sophisticated "chemical blanket" to optimize their chances of survival 2627.
Social Chemosignaling Through Emotional Tears
In parallel with the discoveries surrounding HEX, research has uncovered that human emotional tears contain potent, perceptually odorless chemosignals that modulate social behavior. For decades, the biological function of emotional weeping was considered a uniquely human puzzle, distinct from the reflexive tears produced to clear debris from the eye 3034. However, chemosignaling research has aligned human tears with those of other mammals, such as mole rats and mice, which utilize tear-bound peptides to suppress conspecific aggression 29353637.
Reductions in Physiological Arousal and Aggression
Initial studies into human emotional tears demonstrated that sniffing tears collected from women who were watching sad films significantly reduced self-rated sexual arousal and physiological measures of arousal in men, accompanied by a marked drop in salivary testosterone 303637. While these initial findings faced intense scrutiny and failed replications by independent laboratories citing methodological deviations 37383940, subsequent rigorous investigations have shifted focus from sexual arousal to the modulation of aggression.
A landmark 2023 study published in PLOS Biology exposed male participants to either trickled saline or women's emotional tears while engaging in a behavioral game designed to provoke retribution. The men, who could not consciously differentiate the smell of the tears from the saline, exhibited a 43.7% reduction in revenge-seeking aggressive behavior after sniffing the emotional tears 28293435. This massive behavioral suppression carries a nonparametric effect size of rrb = 0.541, indicating a powerful, unconscious biological override of reactive aggression 28.
Receptor Activation and Brain Connectivity
To verify that these behavioral shifts were driven by olfactory processing rather than psychological artifacts, researchers applied the emotional tears to 62 isolated human olfactory receptors in vitro. The tears successfully activated four specific receptors (OR2J2, OR11H6, OR5A1, and OR2AG2) in a dose-dependent manner. This confirms that despite lacking a conscious odor percept, emotional tears contain chemical ligands that directly stimulate the peripheral olfactory system 28293656.
Central brain imaging during these behavioral tasks confirmed the peripheral findings. Sniffing tears significantly dampened neural activity in the brain's aggression networks - specifically the left anterior insula cortex and the bilateral prefrontal cortex - when the subjects were provoked 283541.
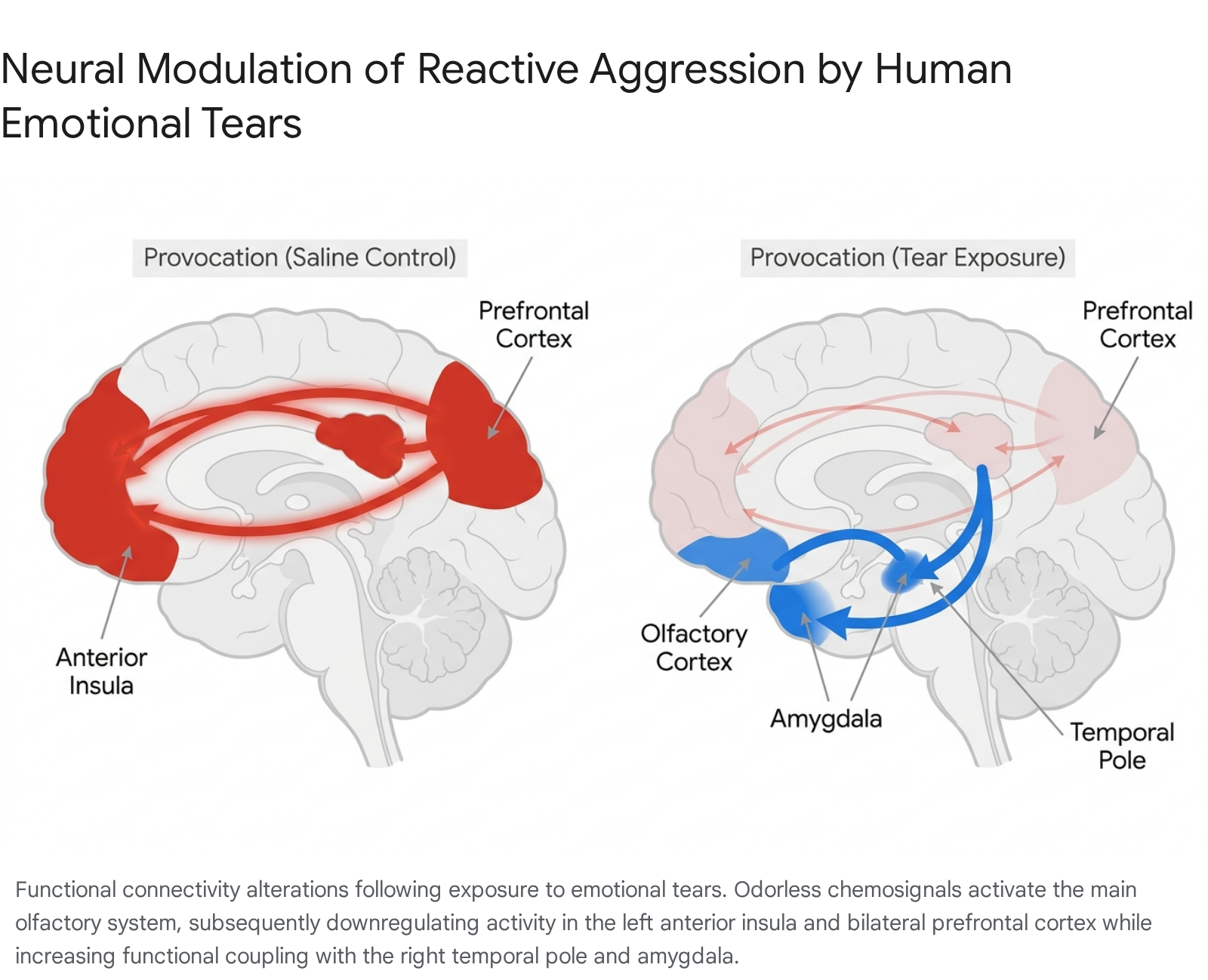
Simultaneously, the tears increased functional connectivity between the left anterior insula and areas associated with olfactory processing, such as the right temporal pole, right amygdala, and piriform cortex 28. This neurological overlap indicates that tears act as a systemic fail-safe, utilizing olfactory circuitry to aggressively downregulate violent retaliation 29.
Replication Controversies and Methodological Rigor
The history of research into emotional tears highlights the rigorous and often contentious nature of replication in the olfactory sciences. Early findings regarding tear-induced testosterone drops faced challenges from laboratories that failed to replicate the effect 3940. Researchers like Gračanin et al. conducted meta-analyses of their own replication attempts, concluding that the effects of female tears on male arousal were weak or non-existent 3940.
However, original researchers noted that these replication attempts frequently deviated from established chemosignaling protocols and utilized varying scope conditions 3738. The subsequent 2023 PLOS Biology findings, which utilized larger sample sizes, independent receptor mapping, and concurrent fMRI scanning, have provided a far more robust, triangulated defense of the chemosignaling properties of tears 2835. This ongoing dialectic underscores the extreme sensitivity of human chemosensory mechanisms to laboratory conditions and contextual variables.
Areolar Gland Secretions as Releaser Candidates
If the strict, classic definition of a pheromone requires a substance to trigger an innate, unlearned, and stereotyped behavioral response universally across the species, the secretions of the human areolar glands remain the only highly probable candidate for a true human pheromone.
Innate Behavioral Triggers in Newborns
During pregnancy and lactation, women develop enlarged sebaceous and lactiferous structures on the areola known as Montgomery's glands, or areolar glands 3142. These glands secrete a lipoid fluid that lubricates the nipple, but they also emit highly volatile odor compounds into the air immediately surrounding the breast 1242.
When these specific secretions are placed under the nose of any sleeping or active newborn baby, they elicit an immediate, stereotyped physical reaction. The infants display autonomic rooting behaviors, turning their heads toward the scent and initiating suckling motions 121242. The olfactory stimulus acts as a state-regulator: it reduces distress and arousal in agitated newborns while simultaneously increasing arousal in sleepy newborns to prepare them for feeding 42. Crucially, this response is entirely independent of learned maternal recognition; any baby will respond instinctually to the areolar secretions of any lactating mother, satisfying the rigorous biological criteria for an innate releaser pheromone 1217.
Implications for Lactation and Neonatal Growth
The physical endowment of these glands has measurable impacts on evolutionary fitness. Morphological studies indicate that the number of functioning areolar glands on a mother's breast correlates directly with the infant's feeding efficiency. Mothers with a higher count of secretory areolar glands have infants who latch faster, suckle more actively, and exhibit significantly faster neonatal weight gain in the critical days immediately following birth 3142. Furthermore, high areolar gland counts are positively related to the faster onset of lactation in first-time mothers 31.
Because the response to areolar secretions can be reliably captured via bioassay and cannot be explained by learned associations - given that the infants react immediately after birth - leading olfactory researchers argue that identifying the exact chemical composition of this secretion represents the single most important frontier in proving the existence of a classic human pheromone 121217.
Systemic Biases in Olfactory Research
The historical turbulence surrounding human chemical communication research is largely a product of poor definitions, methodological hurdles, and profound demographic biases. A clearer understanding of the science requires evaluating the cognitive and cultural context of olfaction.
The Demographic Limitations of WEIRD Samples
The ecological validity of human olfactory research is significantly compromised by systemic demographic biases. A systematic review of the literature reveals that the overwhelming majority of psychological and psychobiological knowledge regarding human olfaction is derived from WEIRD (Western, Educated, Industrialized, Rich, and Democratic) populations 4344.
Within these Western populations, convenience samples of young, undergraduate psychology students are heavily overrepresented 43. Because olfactory perception, odor pleasantness ratings, and the cognitive categorization of smells are heavily influenced by cultural context, diet, and local environments, extrapolating the odor preferences of a Western undergraduate sample to the entire human species is scientifically precarious 4344. Until chemosignaling studies - such as the HEX and tear experiments - are robustly replicated across diverse, non-Western populations, the claim that any specific compound represents a universal human physiological trigger must be calibrated with appropriate scientific uncertainty.
Cognitive Context and Collaborative Olfaction
Olfaction operates differently than vision or audition, relying heavily on implicit memory and unattended, unconscious perception 43. Human olfactory research is frequently complicated by the fact that individuals possess varying metacognitive access to their olfactory judgments. However, recent studies challenge the assumption that humans possess poor olfactory metacognition.
When human pairs (dyads) engage in olfactory discrimination and identification tasks collaboratively, they achieve a "collective benefit." Dyad scores in these tasks are significantly higher than the average of the individuals, proving that humans can effectively communicate their perceptual confidence regarding odors to resolve conflicts 61. The ability to articulate and negotiate olfactory inputs implies that while human chemical communication heavily involves unconscious mechanisms, humans retain sophisticated, contextual processing abilities that elevate olfaction beyond mere rudimentary biological triggers. Furthermore, olfactory cues significantly influence peripheral metabolism and feeding behavior; for instance, prolonged exposure to high-concentration food aromas has been shown to reduce actual food intake by up to 9%, demonstrating the profound integration of olfaction into human physiological regulation 4546.
Conclusions
The science of human chemical communication reveals a system that is far more nuanced than the rigid, instinctual pheromonal networks seen in insects or rodents. The adult human body lacks the functional vomeronasal organ and specific receptor genes required to process pheromones through traditional accessory olfactory pathways. Consequently, the commercialized claims surrounding testosterone-derived sex steroids like androstenol, as well as theories of immune-based mate selection via the Major Histocompatibility Complex, are largely unsupported by rigorous, modern genomic and bioassay-led research.
However, the absence of a classical VNO does not mean humans are chemically blind. The main olfactory epithelium, utilizing trace amine-associated receptors, continuously processes a complex array of social body volatiles that bypass conscious perception to directly modulate the brain's emotional and social centers. Compounds such as hexadecanal and the odorless peptides found in emotional tears demonstrate profound, replicated abilities to downregulate neural aggression networks and alter functional connectivity in a sex-dependent manner. Furthermore, the universal, innate rooting responses triggered by maternal areolar gland secretions provide compelling evidence that humans retain specific, evolutionary chemical triggers designed to ensure infant survival. Ultimately, while the existence of a classical human sex pheromone remains highly improbable, humans are undeniably engaged in a continuous, subconscious dialogue of chemical signaling that subtly but powerfully shapes emotional and social behavior.