Evidence for Age Reversal in Animal Models
The biological processes governing aging have traditionally been viewed as unidirectional, characterized by the progressive, inevitable deterioration of cellular homeostasis, genomic stability, and tissue function. However, the rapid emergence of geroscience has shifted the research paradigm from simple lifespan extension - delaying the onset of age-related decline - to the prospect of age reversal, or systemic physiological rejuvenation. Animal models, ranging from short-lived invertebrates to non-human primates (NHPs), serve as the primary testing grounds for these experimental interventions.
As the volume of aging research accelerates, distinguishing validated physiological rejuvenation from methodological artifacts has become critical. The scientific literature is currently characterized by profound breakthroughs in genetic reprogramming, senolytic clearance, and targeted polypharmacy, running parallel to highly publicized metrics that often obscure the absolute physiological gains. This report evaluates the current state of age reversal in animal models, analyzing the mechanisms of emerging therapies, quantifying their empirical outcomes, and separating substantiated biological realities from uncalibrated hype.
Biological Aging and Measurement Context
To assess whether an intervention actively reverses aging or merely extends baseline survival, researchers rely heavily on biomarkers designed to quantify biological age. The development of these molecular metrics has revolutionized the evaluation of gerotherapeutics, but the fundamental assumptions underlying these measurement tools remain the subject of intense scientific and statistical debate.
The Evolution of Epigenetic Clocks
Epigenetic clocks, primarily based on DNA methylation patterns (DNAm), have become the gold standard for measuring biological age in both animal models and humans. As organisms age, specific genomic regions (CpG sites) undergo predictable hyper- and hypo-methylation, altering gene expression profiles without changing the underlying genetic sequence. The first generation of these tools, such as the Horvath multi-tissue clock and the Hannum blood clock, utilized elastic net regression algorithms optimized to predict chronological age based on these methylation states 123.
Subsequent iterations, frequently referred to as second-generation clocks like PhenoAge and GrimAge, were calibrated against clinical biomarkers of physiological dysregulation, morbidity, and mortality risk rather than chronological age alone 234. In animal models, these tools theoretically allow researchers to evaluate the efficacy of longevity interventions in a fraction of the time required for full lifespan studies. Consequently, when cellular reprogramming or pharmacological interventions are applied to murine models, reductions in DNAm age are frequently cited as primary proof of "age reversal" 45.
Disentangling Damage and Adaptation
Despite their ubiquity in the literature, the assumption that reversing an epigenetic clock is strictly synonymous with whole-organism age reversal remains highly contested. Methodological scrutiny reveals that standard epigenetic clocks may conflate discrete, sometimes opposing, biological processes. Aging induces two distinct categories of methylation changes: Type 1 changes represent the accumulation of stochastic damage and the dysregulation of homeostatic pathways (a shift toward systemic degradation), while Type 2 changes represent adaptive, protective responses orchestrated by the body to mitigate that very damage 678.
Current standard clocks measure the aggregate of both types. Consequently, if a therapeutic intervention artificially suppresses the body's innate repair mechanisms (Type 2 changes), the epigenetic clock may register a "younger" biological age, even though the organism has been deprived of critical damage-control pathways 67. This creates a high risk of misclassifying deleterious interventions as geroprotective, highlighting a significant blind spot in evaluating anti-aging therapies purely through aggregated methylation scores 78.
Analytical Limitations and Covariate Shifts
To address these epistemic uncertainties, next-generation analytical frameworks have emerged. Researchers at Harvard Medical School developed causation-based clocks, termed DamAge and AdaptAge, using epigenome-wide Mendelian Randomization to mathematically separate damage-inducing methylation from adaptive methylation 910. These advanced models correlate DamAge with mortality risk and AdaptAge with longevity, providing a more robust mechanism for evaluating true physiological interventions 9.
Furthermore, recent analyses of DNA methylation profiles during cellular reprogramming suggest that standard algorithms may suffer from severe covariate shifts. The methylation states of artificially reprogrammed cells diverge significantly from the natural aging data used to train the models, injecting high epistemic uncertainty into claims of epigenetic rejuvenation 11. Additional complexities arise in organ perfusion and transplantation studies. Recent research tracking transplanted organs between young and old subjects demonstrates rapid "age assimilation," where a donor organ's epigenetic clock alters to match the recipient's biological age, raising questions about whether epigenetic age is a fixed cellular property or a highly volatile response to systemic circulating factors 1213. Thus, while epigenetic clocks indicate molecular modifications, isolated DNAm reductions in animal models are insufficient to declare physiological age reversal without corroborating functional healthspan metrics.
Comparison of Epigenetic Measurement Modalities
| Clock Generation | Primary Output | Calibration Metric | Key Limitations |
|---|---|---|---|
| First-Generation (e.g., Horvath, Hannum) | Biological Age Estimate | Chronological Age | Conflates damage with protective adaptations; heavily influenced by demographic datasets 12. |
| Second-Generation (e.g., PhenoAge, GrimAge) | Morbidity/Mortality Risk | Clinical Biomarkers & Phenotypes | Still aggregates Type 1 (damage) and Type 2 (repair) methylation changes 34. |
| Causal/Separation Clocks (e.g., DamAge, AdaptAge) | Causative Aging Score | Epigenome-wide Mendelian Randomization | Emerging technology; less longitudinal validation in wide animal cohorts 910. |
Cellular Reprogramming Interventions
Cellular reprogramming relies on the transient, exogenous expression of specific transcription factors - typically Oct4, Sox2, Klf4, and c-Myc (collectively known as OSKM or Yamanaka factors) - to revert specialized somatic cells toward a pluripotent state. Because continuous expression of these factors invariably leads to severe cellular dedifferentiation and the rapid formation of teratomas, contemporary in vivo research focuses exclusively on "partial" reprogramming.
Yamanaka Factors and Partial Reprogramming Fundamentals
By exposing cells to Yamanaka factors cyclically, or by omitting the highly oncogenic c-Myc factor entirely to form an "OSK" cocktail, researchers aim to restore youthful epigenetic profiles while preserving essential cellular identity 1415. The hypothesis posits that cellular identity is highly resilient, whereas age-related epigenetic noise is more labile; a precisely calibrated "pulse" of OSK can erase the aging markers before the cell forgets its functional role 14.
Initial applications of this theory were pioneered by the Salk Institute, utilizing cyclic OSKM expression in transgenic progeroid (prematurely aging) mice. These studies demonstrated extended lifespans and ameliorated classical aging hallmarks without triggering tumorigenesis, providing foundational proof-of-concept 1415. Subsequent experiments transitioned to wild-type mice; cyclic induction from middle age (15 months) to old age (22 months) resulted in rejuvenated epigenetic patterns in renal and dermal tissues, reduced permanent scarring upon injury, and yielded more youthful metabolic profiles in the blood 15.
Murine Lifespan Extension Efficacy and Hype
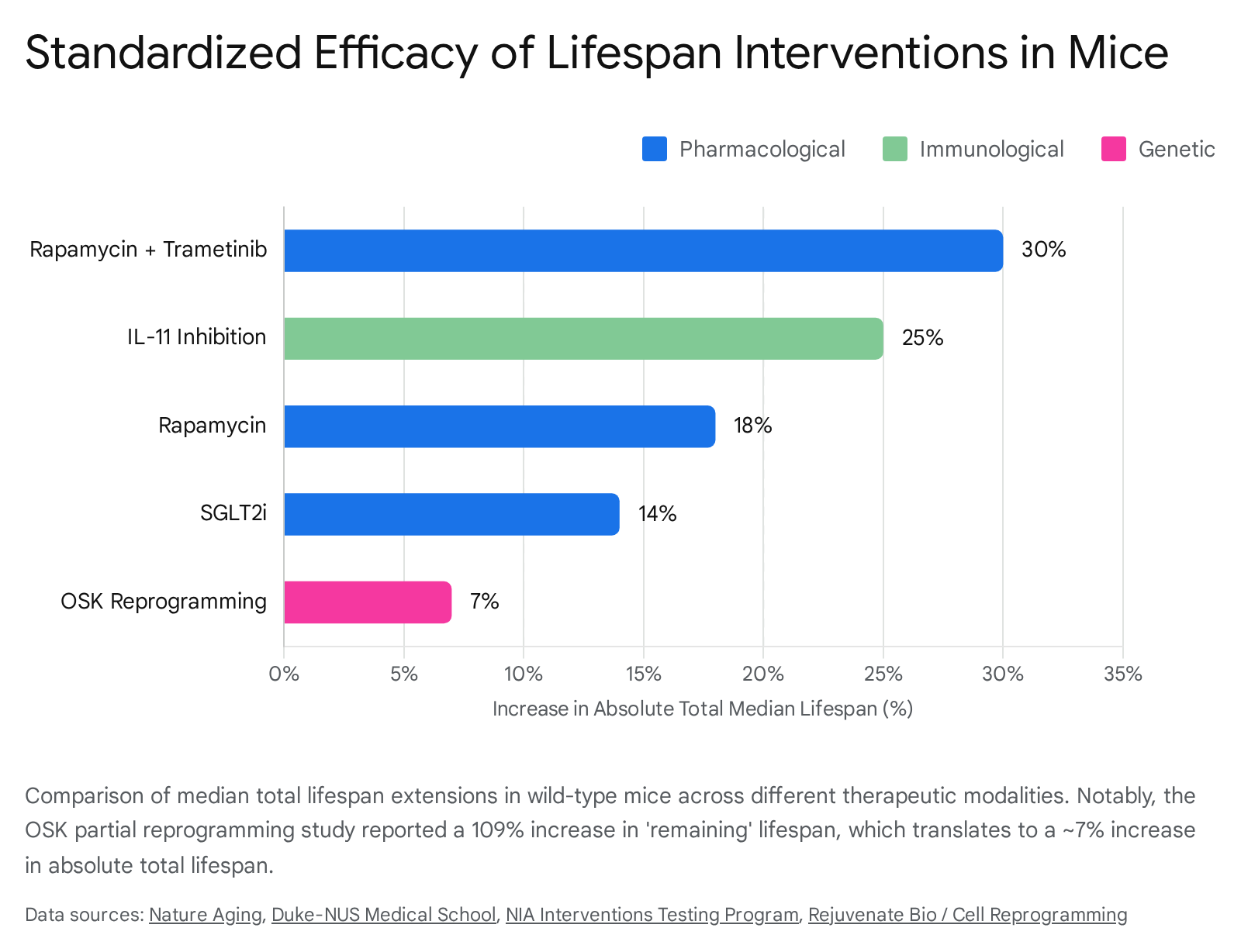
Moving beyond transgenic models, recent research has evaluated absolute lifespan extensions using systemic adeno-associated virus (AAV) delivery of OSK in extremely old, wild-type mice. In a highly publicized 2024 study published in Cellular Reprogramming (Macip et al., Rejuvenate Bio), researchers administered an inducible AAV9-OSK system to 124-week-old C57BL6/J mice - a baseline equivalent to approximately 77 to 80 human years 161717.
The researchers reported a 109% increase in median remaining lifespan alongside improved physical frailty scores and reversed DNAm age in both hepatic and cardiac tissue 1617. However, the reporting and public reception of these results critically exemplify the boundary between biological reality and media hype in the longevity sector. The "109% increase" refers strictly to remaining lifespan measured from the late-stage 124-week baseline 519.
In absolute metrics, the control mice lived approximately 8.8 additional weeks (achieving a median total lifespan of ~133 weeks), while the OSK-treated mice lived 18.5 additional weeks (achieving a median total lifespan of 142.5 weeks) 171720. This translates to a total absolute lifespan extension of roughly 7% 519. Generating an 18-week healthspan and lifespan extension in a subject already at the extreme boundaries of its natural life represents a biologically profound proof-of-concept for the plasticity of aging 2018. Nonetheless, framing the outcome as a "109% extension" in press materials frequently leads to public misinterpretation regarding the sheer magnitude of the physiological effect 1922.
Application to Neural and Cognitive Systems
Beyond systemic delivery, partial reprogramming is actively being evaluated for its capacity to reverse neurodegeneration and cognitive decline. Studies investigating the effects of OSK on the murine brain have demonstrated that partial reprogramming favors the migration of neural stem cells, increases the survival rate of newborn neurons, and improves overall memory retention in young adult mice 19.
When induced prenatally, partial reprogramming led to increased overall brain size (neurogenesis) and enhanced motor and social behaviors 19. Furthermore, targeted partial reprogramming in aged mice has been shown to successfully reverse age-related neural stem cell decline, reinvigorating the production of new neurons. These preclinical signals have prompted biotech firms, such as YouthBio Therapeutics, to secure FDA clearance to begin preparing clinical pathways for gene therapies (e.g., YB002) aimed at applying localized partial reprogramming to combat Alzheimer's disease pathology in humans 19.
Translation to Non-Human Primates
A fundamental limitation of murine models is their physiological and immunological divergence from human biology. Consequently, translating partial reprogramming to non-human primates (NHPs) is an absolute prerequisite for validating clinical feasibility. Due to systemic safety concerns surrounding uncontrollable cell dedifferentiation and tumorigenesis, early NHP trials are heavily restricted to isolated, immune-privileged systems - predominantly the eye 24.
Gene therapy vectors delivering OSK to the retinal ganglion cells of NHPs have demonstrated the ability to mitigate deficits in pattern electroretinogram responses and axon density following non-arteritic anterior ischemic optic neuropathy (NAION) injuries 20. By resetting the epigenetic clocks of aged or injured ocular tissue, the OSK vectors triggered the functional restoration of sight 242021. Histological analysis confirmed tight control of delivery, with OSK expression strictly confined to target perifoveal cells and no evidence of ectopic growth or teratoma formation 20.
This isolated NHP data provides highly controlled, localized evidence that partial reprogramming can safely enact functional, phenotypic rejuvenation in primate nervous tissue. These findings provide the empirical foundation for the first human clinical trials of epigenetic reprogramming (candidate ER-100), currently anticipated to commence in early 2026 for the treatment of severe optic neuropathies 202122.
Targeted Senolytic Therapies
Cellular senescence is a highly regulated stress response wherein cells permanently cease division while remaining metabolically active. While senescence plays a crucial role in embryonic development and tumor suppression in youth, these cells progressively accumulate in aged tissues. As they accumulate, they secrete a toxic, localized mixture of pro-inflammatory cytokines, chemokines, and proteases - collectively known as the senescence-associated secretory phenotype (SASP) 2324.
The failure of the aging immune system to clear these senescent cells accelerates surrounding tissue degradation and drives multimorbidity. The primary therapeutic approach to mitigating this hallmark of aging is the development of senolytics: pharmacological or biological agents designed to selectively induce apoptosis (programmed cell death) in senescent cells while sparing healthy surrounding tissue.
Senolytic Immunization Strategies
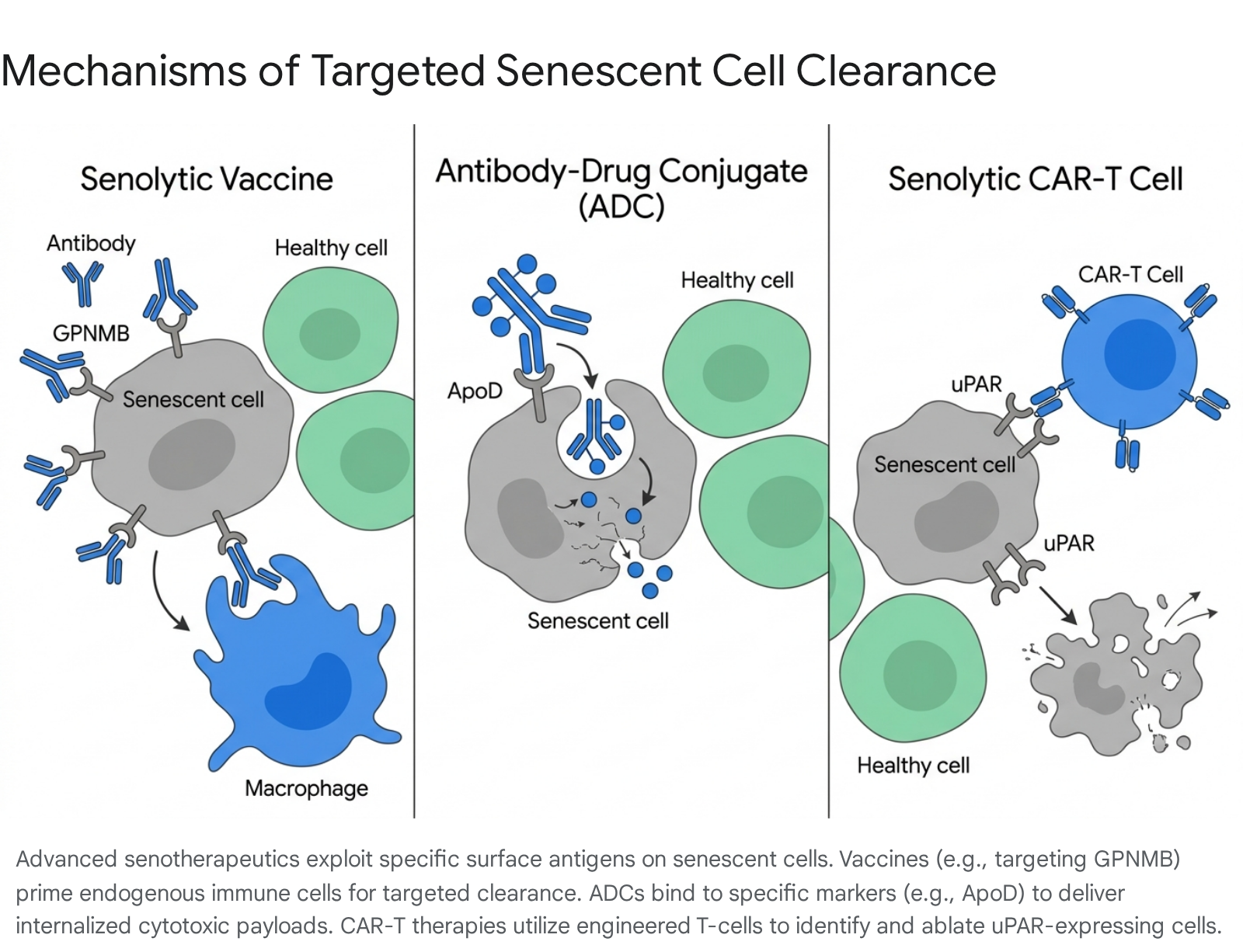
While first-generation small-molecule senolytics (such as the dasatinib and quercetin combination) demonstrated proof-of-concept efficacy, their off-target toxicity, poor bioavailability, and the necessity for continuous administration limited their long-term clinical viability 252627. Recent research has pivoted toward senescence-directed immunization, aiming to leverage the host's own adaptive immune system for sustained, targeted clearance.
Researchers identified glycoprotein nonmetastatic melanoma protein B (GPNMB) as a specific transmembrane antigen highly enriched on the surface of senescent vascular endothelial cells, acting as a viable "seno-antigen" 2427. Utilizing peptides extracted from the extracellular domain of GPNMB, scientists at Juntendo University developed a targeted senolytic vaccine 2333. In progeroid and middle-aged wild-type mouse models subjected to a high-fat diet, the GPNMB vaccine successfully trained the immune system to recognize and eliminate GPNMB-positive senescent cells 333435.
The resulting physiological metrics were highly robust: the vaccine reduced atherosclerotic plaque burden in Apolipoprotein E knockout (ApoE KO) mice, decreased systemic metabolic dysfunction, improved physical speed and movement, and extended the median lifespan in progeroid models 233334.
Parallel studies have utilized peptide vaccines targeting the CD38 protein, whose age-dependent systemic increase is tightly linked to NAD+ decline, mitochondrial dysfunction, and neuroinflammation 28. CD38 vaccination in aged mice successfully reduced the senescent cell burden in solid organs, lowered SASP factor expression, and partially restored youthful NAD+/NADH ratios in both hepatic and neural tissue. This metabolic restoration translated directly to preserved cognitive capabilities and enhanced physical strength in older mice 28.
Engineered Biological Vehicles
Alternative senolytic approaches bypass the active training of the immune system by deploying highly engineered biological vehicles designed to hunt and destroy aging cells. Antibody-Drug Conjugates (ADCs) provide a precise mechanism to deliver cytotoxic payloads directly to specific cellular targets. Recent models have utilized monoclonal antibodies targeting verified senescence markers like apolipoprotein D (ApoD) or beta-2 microglobulin (B2M) 2938.
Upon binding to the senescent cell membrane, the ADC is internalized and degraded by intracellular lysosomes. This degradation process releases a highly toxic pyrrolobenzodiazepine payload that strictly induces apoptosis in the target cell, thereby circumventing collateral damage to adjacent healthy tissue 2938.
Similarly, chimeric antigen receptor (CAR) T-cell therapies have been adapted from their origins in oncology for use as gerotherapeutics. CAR-T cells engineered to target the senescence-associated protein urokinase plasminogen activator receptor (uPAR) have proven highly capable of ablating senescent cells in vivo. A single administration of senolytic CAR-T cells demonstrated long-lasting prophylactic and therapeutic efficacy against age-related metabolic dysfunction in murine models, representing a "one-and-done" approach to senescent cell clearance 25.
Pharmacological Modulators and Polypharmacy
While genetic editing, epigenetic reprogramming, and highly engineered immunotherapy present formidable regulatory, financial, and delivery challenges, pharmacological modulation relies on small molecules to recalibrate the systemic signaling pathways responsible for the hallmarks of aging. The prevailing hyperfunction theory of aging posits that biological aging is not merely passive wear-and-tear, but rather the deleterious consequence of prolonged, hyperactive cellular growth pathways that fail to downregulate after reproductive maturity 30.
Mechanistic Targets of Growth and Stress
Decades of systematic investigation, primarily through the National Institute on Aging's Interventions Testing Program (ITP), have evaluated dozens of molecules for longevity efficacy across independent testing sites 31. To date, rapamycin - an inhibitor of the mechanistic target of rapamycin complex 1 (mTORC1) - has established itself as the single most reliable pharmacological lifespan extender in mammalian models, yielding average lifespan extensions of 15 - 20% by mimicking the cellular effects of nutrient scarcity and upregulating autophagy 3031.
However, the field of longevity research is rapidly transitioning from monotherapies to rational polypharmacy, recognizing that aging is a multifaceted phenomenon that cannot be halted by targeting a single signaling node. Combining compounds that act on separate, complementary pathways is emerging as a superior strategy for extending healthspan.
Synergistic Polypharmacy Outcomes
A landmark 2025 study published in Nature Aging demonstrated striking synergistic age reversal and lifespan extension by combining rapamycin with trametinib, a MEK inhibitor that blocks the Ras - MEK - ERK signaling pathway 3032. While trametinib alone generated only marginal lifespan extensions in mice (5 - 10%), the combined administration of the two drugs produced a powerful additive effect, increasing median absolute lifespan by 34.9% in female mice and 27.4% in male mice 303334.
The combinatorial efficacy stems from complementary pathway suppression. Where rapamycin primarily restores autophagic flux suppressed by hyperactive mTORC1, trametinib independently dampens systemic inflammaging 303234. The dual therapy yielded highly quantifiable physiological outcomes in aged mice, including severely attenuated microglial and astrocyte activation in the striatum (reducing neuroinflammation), normalized brain glucose uptake, a slower decline in cardiac function, and a delayed onset of hepatic and splenic tumors 303233. Furthermore, transcriptomic analysis revealed that the combination uniquely altered gene expression in ways not achieved by either drug in isolation, demonstrating a true systems-level recalibration of the organism's metabolic stress response 34.
Metabolic Repurposing of SGLT2 Inhibitors
Sodium-glucose cotransporter 2 (SGLT2) inhibitors, originally formulated for glycemic control in type 2 diabetes, have unexpectedly emerged as potent, multifaceted geroprotectors. In murine models within the ITP, SGLT2 inhibition extended male median survival by approximately 14% and promoted profound multi-organ healthspan improvements 3135. The intervention induces a fasting-mimicking state through glycosuria, shifting cellular energy utilization toward fat oxidation and the production of beta-hydroxybutyrate (BHB), which subsequently lowers systemic oxidative stress and inhibits the NLRP3 inflammasome 3637.
Recent mechanistic data reveals that SGLT2 inhibitors exert direct senotherapeutic effects. In mice subjected to a highly inflammatory, high-fat diet, short-term administration of SGLT2 inhibitors reduced the senescent cell burden in adipose tissue by up to 50% within 14 days 38. This rapid clearance is linked to the re-empowerment of cytotoxic T-lymphocytes (CTLs); SGLT2 inhibition significantly increases Granzyme B activity, allowing the immune system to overcome the apoptotic resistance typically exhibited by senescent cells 37. Additionally, SGLT2 inhibitors demonstrably cross the blood-brain barrier, exhibiting direct neuroprotective properties by reducing microgliosis and astrogliosis in the hippocampus, thereby preserving youthful cognitive function 38.
Emerging Pharmacological Targets
Parallel to metabolic modulation, targeted inhibition of specific inflammatory cytokines and proteins has yielded dramatic lifespan results. Interleukin-11 (IL-11) has recently been identified as a primary driver of age-related visceral fat accumulation and muscle atrophy (sarcopenia). Preclinical models developed by Duke-NUS Medical School demonstrated that systemic administration of an anti-IL-11 therapy suppressed multiple dysfunctional signaling mechanisms, preventing age-related muscle loss, reducing cardiometabolic disease, and extending absolute lifespan by up to 25% across both sexes 39.
Other compounds are showing targeted efficacy in neural pathways. Researchers in Singapore have demonstrated that Calcium alpha-ketoglutarate (CaAKG) can improve communication between brain cells and restore associative memory in murine models of Alzheimer's disease, repairing synaptic deficits that normally degrade with age 40. In South Korea, researchers at Chung-Ang University identified the drug IU1 as an enhancer of proteostasis; by inhibiting the ubiquitin-specific peptidase 14 (USP14), IU1 simultaneously enhances proteasome and autophagy activity, significantly improving age-related muscle weakness in model organisms 41. Furthermore, studies targeting the High Mobility Group Box 1 (HMGB1) protein have revealed that cellular aging spreads systemically through the bloodstream; blocking the ReHMGB1 isoform with antibodies successfully restored tissue regenerative capacity and physical performance in older mice 42.
Summary of Validated Interventions in Murine Models
| Modality | Intervention | Key Target Pathway | Lifespan Impact (Median) | Validated Physiological Outcomes |
|---|---|---|---|---|
| Polypharmacy | Rapamycin + Trametinib | mTORC1 & Ras-MEK-ERK | + ~30% (absolute) 3033 | Reduced neuroinflammation; delayed tumorigenesis; normalized glucose uptake. |
| Pharmacological | Anti-IL-11 Therapy | Interleukin-11 cytokine | + 25% (absolute) 39 | Reduced visceral fat; prevented age-related sarcopenia (muscle loss). |
| Pharmacological | SGLT2 Inhibitors | Glucose transport / Inflammasome | + 14% (absolute, males) 3135 | Senescent cell clearance; hippocampal neuroprotection; reduced frailty. |
| Gene Therapy | OSK Partial Reprogramming | Epigenome (Yamanaka factors) | + 109% (remaining), ~7% (absolute) 517 | Reversed DNAm age in heart/liver; accelerated wound healing; lowered frailty. |
| Immunotherapy | GPNMB Senolytic Vaccine | GPNMB membrane antigen | Lifespan extension in progeroid mice 2334 | Reduced atherosclerotic plaque; cleared adipose senescent burden. |
Stem Cell and Exosome Rejuvenation Therapies
Traditional stem cell therapies have largely struggled to produce systemic age reversal due to poor viability in aged host environments. The harsh, highly oxidative, and inflammatory microenvironment of elderly tissues - saturated with SASP factors - rapidly degrades transplanted cells before they can enact meaningful tissue repair, severely limiting their clinical benefit 43. Overcoming this biological barrier requires engineering progenitor cells capable of resisting the host's innate toxic environment.
Senescence-Resistant Progenitor Cells
In a seminal 2025 study published in the journal Cell, researchers from the Chinese Academy of Sciences demonstrated sweeping, multi-systemic rejuvenation in cynomolgus macaques utilizing genetically modified stem cells. The research team developed senescence-resistant mesenchymal progenitor cells (SRCs) by utilizing synthetic biology to enhance the expression of FOXO3, a gene deeply associated with longevity, genomic stability, and cellular resilience to severe oxidative stress 4445.
Elderly macaques (physiologically equivalent to humans in their 60s and 70s) received biweekly intravenous infusions of these FOXO3-enhanced SRCs over an intensive 44-week trial, at a dosage of two million cells per kilogram of body weight 4445. Crucially, the protocol demonstrated a pristine safety profile; researchers observed no evidence of tumorigenicity, ectopic tissue growth, or immune rejection throughout the extended administration period 434445.
Multi-Systemic Exosome Mediated Repair
The therapeutic outcomes of this trial represent some of the most comprehensive age-reversal data generated to date in non-human primates. Detailed tissue analyses confirmed robust rejuvenation across 10 major physiological systems and 61 distinct tissue types 4445. The circulating SRCs successfully suppressed chronic tissue inflammation, mitigated age-related periodontal bone loss, reduced fibrosis, and stimulated endogenous progenitor cell populations 4455.
In the central nervous system, MRI and histological analyses revealed lessened thinning of the myelin sheath, increased cortical volume, and enhanced functional connectivity in the hippocampus. These structural improvements directly corresponded to enhanced performance on Wisconsin General Test Apparatus (WGTA) cognitive tasks, with SRC-treated monkeys demonstrating significantly improved memory retention compared to saline-treated controls 444556. Furthermore, the therapy yielded profound rejuvenation of the reproductive system, stimulating sperm production in males and reducing the biological age of ovarian oocytes by approximately five years 4456.
Mechanistically, the researchers discovered that the systemic rejuvenation was not primarily driven by the physical engraftment of the SRCs into host tissues. Instead, the effects were mediated through paracrine signaling. The engineered SRCs secreted vast quantities of exosomes - microvesicles containing highly regenerative molecular payloads - that circulated throughout the body, buffering the host genome, suppressing local SASP signals, and restoring cellular protein balance 434446. When evaluated via multi-tissue biological aging clocks, the exosome-driven therapy effectively rolled back the molecular age of NHP tissues, actively rejuvenating immature neurons by up to seven years 4456.
Standardizing Healthspan Quantification
As interventions demonstrate the capacity to alter the aging process, the geroscience community is grappling with the limitations of evaluating success purely through "time to death." Increased survival does not necessarily equate to delayed senescence if the organism spends the extended lifespan in a state of physiological decline and morbidity 1847.
Evolving Metrics Beyond Survival
To address this, researchers are developing integrative metrics designed to quantify healthspan - the period of life free from disease and severe disability. Recent frameworks have introduced summary statistics such as FAMY (Frailty-Adjusted Mouse Years) and GRAIL (Gauging Robust Aging when Increasing Lifespan) 47. Conceptually similar to Quality-Adjusted Life Years (QALYs) utilized in human clinical trials, these metrics integrate absolute lifespan data with longitudinal measurements of a 31-item clinical frailty index, grip strength, rotarod performance, and glucose tolerance 4748.
The application of tools like GRAIL provides critical nuance to the aging reversal literature. For instance, recent analyses utilizing these metrics revealed that dietary restriction of specific amino acids (like isoleucine) robustly promotes functional healthspan in female HET3 mice without necessarily increasing maximum longevity 47. Moving forward, the routine integration of standardized frailty and healthspan indices is essential to separate interventions that merely prolong end-of-life morbidity from those that genuinely enact age reversal and functional rejuvenation.
Conclusions
Research utilizing varied animal models has definitively proven that biological aging is a highly plastic, malleable condition rather than a predetermined, immutable constant. However, accurately interpreting the vast amounts of data generated by this field requires a strict, critical differentiation between isolated molecular markers and holistic organismal health. While the resetting of epigenetic clocks via partial reprogramming demonstrates the unprecedented feasibility of altering fundamental cellular states, an uncalibrated reliance on these molecular metrics risks amplifying public hype over verified physiological rejuvenation.
The most robust, translatable evidence for systemic age reversal currently relies on targeted, multifaceted interventions that address aging as a systems-level failure. Rational polypharmacy strategies (such as the synergistic combination of rapamycin with MEK inhibitors), highly engineered senescent-directed immunotherapies (including vaccines and ADCs), and exosome-driven stem cell treatments have all generated profound, objectively quantifiable improvements in murine and primate healthspans. As these advanced therapeutic modalities mature, the primary focus of the geroscience community must shift from isolated cellular demonstrations toward the rigorous evaluation of safety, dosing regimens, and comprehensive biomarker validation required to successfully translate these interventions from animal models to human clinical trials.