Epigenetic noise and the information theory of aging
Theoretical Foundations of Epigenetic Aging
The physiological and functional deterioration of organisms over time has historically been attributed to the accumulation of random molecular damage, most notably genetic mutations. However, the Information Theory of Aging (ITOA), formalized prominently by David Sinclair and colleagues, posits a fundamentally different primary driver: the progressive loss of epigenetic information 1. The theory is grounded in a conceptual distinction between genetic and epigenetic information storage. While the genome stores information in a highly stable, digital format consisting of nucleic acid sequences, the epigenome stores information in a digital-analog format comprising chemical modifications to DNA and histone proteins 1. This analog layer regulates gene expression patterns, endowing cells with their specific identities and functions. However, because it is an analog system, the epigenome is inherently highly susceptible to alterations induced by environmental signals, metabolic fluctuations, and cellular damage 1.
According to the ITOA, the primary driver of biological aging is the stochastic accumulation of "epigenetic noise," which degrades the precision of cellular differentiation 23. As this noise accumulates, the epigenetic landscape - often conceptualized as a Waddington landscape - begins to flatten. Cells progressively lose their specialized identities in a process sometimes referred to as ex-differentiation, beginning to inappropriately express genes characteristic of other cellular lineages, which ultimately leads to systemic tissue dysfunction, cellular senescence, and organismal frailty 34.
Mechanisms of Epigenetic Drift and Noise Accumulation
At the core of the ITOA is the specific molecular mechanism by which cellular identity erodes in response to genomic stress. Throughout an organism's life, cells experience continuous exposure to endogenous and exogenous stressors, such as reactive oxygen species, cosmic rays, and metabolic byproducts, that cause DNA double-strand breaks (DSBs) 567. The repair of these DSBs is a complex biological process requiring the temporary relocalization of chromatin-modifying proteins from their standard regulatory positions in the genome to the specific sites of DNA damage 257.
Prominent among these epigenetic regulators are members of the sirtuin family (such as SIRT1 in mammals and Sir2 in yeast) 28. Under normal, youthful conditions, sirtuins and other chromatin modifiers return to their original loci after successfully coordinating DNA repair, thereby restoring the cell's precise epigenomic landscape 57. However, the ITOA hypothesizes that over repeated cycles of damage and repair throughout a lifetime, this homing process becomes increasingly inefficient. Chromatin modifiers fail to perfectly re-establish the original epigenetic state, leaving behind slight alterations in DNA methylation and histone acetylation patterns 27. Over decades, these minute errors compound, resulting in a disorganized epigenome that can no longer accurately read the underlying genetic code 2.
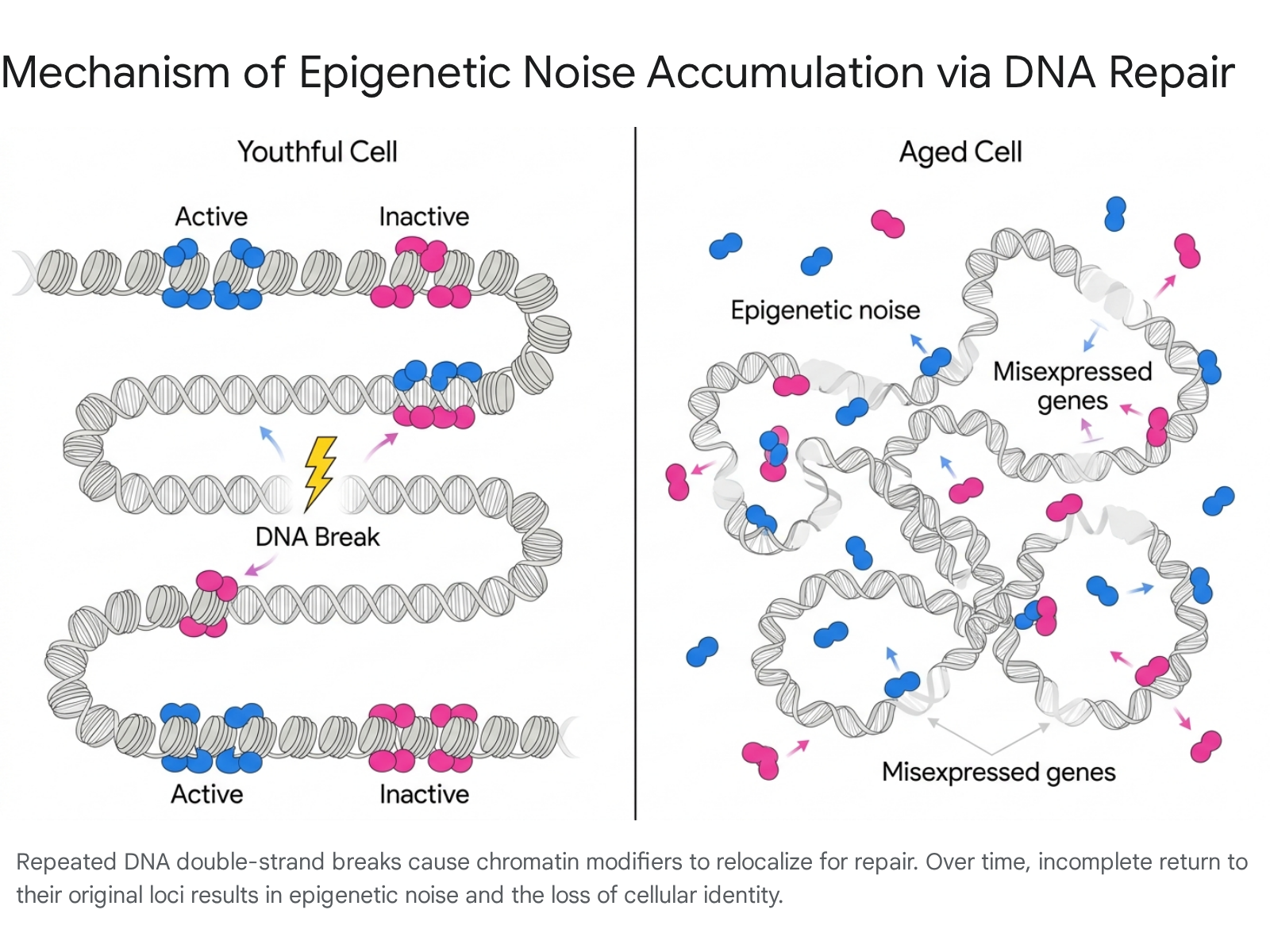
Conceptualization of Reversibility via Cellular Reprogramming
A defining feature separating the ITOA from traditional mutation-centric theories of aging is the theoretical potential for reversibility. Genetic mutations are permanent structural changes to the digital genomic code; correcting them across trillions of cells in an adult organism is currently unfeasible 346. Conversely, if aging is primarily an analog epigenetic problem, the original digital code remains intact. The ITOA suggests that a preserved "backup copy" of youthful epigenetic information exists within the cell and that cellular identity can be restored if the epigenome is appropriately accessed and rebooted 346.
This theoretical framework relies heavily on the application of the Yamanaka factors (Oct4, Sox2, Klf4, and c-Myc, collectively OSKM), which are known to reprogram adult somatic cells back to a pluripotent embryonic state 910. The ITOA proposes that a modified, partial application of these factors - often omitting the oncogene c-Myc to reduce cancer risk, leaving the OSK cocktail - can induce epigenetic rejuvenation without pushing the cell all the way back to pluripotency 346. This transient partial reprogramming aims to erase age-related epigenetic noise, restore cellular identity, and reverse the biological markers of aging while preserving the specific functional lineage of the cell 3910.
Empirical Testing via the ICE Mouse Model
To empirically isolate epigenetic noise from genetic mutation as the primary driver of aging, Sinclair and colleagues developed the Inducible Changes to the Epigenome (ICE) mouse model, which was extensively detailed in a 2023 publication in the journal Cell by Yang et al. 571211. The goal of the ICE model was to simulate an accelerated lifetime of faithful DNA double-strand breaks and repair, thereby generating pure epigenetic noise in the absence of significant mutational burden 511.
Design and Mechanics of the Inducible Epigenome Model
The ICE system utilizes a sophisticated transgenic approach. The mice were engineered to carry two separate genetic cassettes inserted into their embryos: an "ERT2-HA-I-PpoI-IRES" cassette and a "Cre-ERT2" cassette 12. The critical component is I-PpoI, a highly specific restriction endonuclease that recognizes and cuts a specific 15-base-pair DNA sequence 1211. In the mouse genome, there are exactly 19 canonical recognition sites for I-PpoI, the vast majority of which are located in non-coding regions, specifically within the 28S ribosomal DNA (rDNA) 121112. Because the cuts occur primarily in non-coding regions and create predictable "sticky" overhangs, the resulting DSBs are generally repaired faithfully with a very low mutagenic potential 1112.
The expression of I-PpoI is strictly temporal. Post-developmentally, when the mice reach adulthood, researchers administer low concentrations of tamoxifen in their diet. Tamoxifen activates the Cre recombinase, which removes a STOP sequence, leading to the translation of I-PpoI and its subsequent transient localization into the cell nucleus 1211. The enzyme creates targeted DNA breaks, triggering a massive cellular DNA damage response (DDR) and forcing the repeated relocalization of chromatin-modifying enzymes to facilitate repair 511.
Physiological and Molecular Phenotypes of ICE Mice
Following a short induction phase, the tamoxifen is removed, and the mice are monitored over several months. The Yang et al. study reported that the ICE mice exhibited profound physiological, cognitive, and molecular changes that closely phenocopied natural mammalian aging 5711. After ten months post-treatment, the ICE mice displayed accelerated physical deterioration characteristics of much older wild-type animals, including alopecia (hair loss), loss of pigment on the extremities, reduced body weight and fat mass, kyphosis (curvature of the spine), and decreased spontaneous ambulatory motion during dark cycles 415.
At the molecular level, researchers observed a widespread erosion of the epigenomic landscape. Specifically, the model showed a global disruption of the H3K27ac histone modification, a critical marker of active super-enhancers that dictates cell fate and identity 11. ICE cells demonstrated cellular ex-differentiation, losing their specialized transcriptomic signatures 67. Furthermore, the ICE mice exhibited accelerated aging according to DNA methylation (DNAm) clocks specifically adapted for mouse tissue (such as blood and muscle clocks), registering significantly older biological ages than their chronological counterparts 511.
To test the reversibility hypothesis central to the ITOA, the researchers subjected the prematurely aged ICE mice to viral gene therapy delivering the OSK reprogramming factors 457. The study reported that the OSK therapy successfully accessed the preserved epigenetic information, reversing multiple physiological and molecular markers of aging and improving tissue function, most notably rejuvenating transcriptomic profiles in retinal ganglion cells 457.
Methodological and Theoretical Critiques
Despite the significant attention the ITOA and the ICE mouse model have garnered in both academic literature and popular media, the framework remains highly controversial within the broader biogerontology community. Critics argue that the experimental designs conflate severe genotoxic stress with natural aging and that the premise oversimplifies the multifactorial nature of biological senescence.
Cytotoxicity and the p53 Damage Response
A primary critique of the ICE model, articulated prominently by researchers such as Charles Brenner, focuses on the inherent cytotoxicity of the I-PpoI restriction enzyme 81213. While Yang et al. contend that the DNA breaks are non-mutagenic and purely drive epigenetic noise, critics highlight that inducing organism-wide DNA double-strand breaks constitutes a severe traumatic insult rather than a subtle simulation of natural wear-and-tear 13.
Critics argue that the forced expression of I-PpoI triggers a robust p53-mediated DNA damage response 51213. The p53 protein is a critical tumor suppressor that, when heavily activated by catastrophic DNA damage, initiates either apoptosis (programmed cell death) or cellular senescence (irreversible cell cycle arrest) to prevent malignant transformation 517. A previous study involving the corresponding authors of the ICE paper, utilizing an "iDSB" (induced double-strand break) mouse model targeting I-PpoI specifically to epidermal stem cells, demonstrated that the intervention resulted in a massive p53 response and the near-total elimination of the targeted cells within a month 13. Critics of the ICE paper note that Yang et al. did not present extensive systemic analysis during the critical immediate one-month post-induction window, potentially masking acute toxicity and cell death 13.
Senescence and the Confounding Role of Inflammaging
The downstream consequence of a massive p53 response is the accumulation of senescent cells. Senescent cells undergo a profound phenotypic shift, secreting a complex mixture of pro-inflammatory cytokines, chemokines, proteases, and growth factors collectively known as the senescence-associated secretory phenotype (SASP) 1718.
Critics suggest that the premature aging observed in ICE mice is not driven by the abstract loss of epigenetic information or subtle "epigenetic noise," but rather by the massive, forced accumulation of highly inflammatory senescent cells resulting from genotoxic trauma 513. The chronic sterile inflammation caused by this intervention - a phenomenon broadly termed "inflammaging" - is a well-established and potent secondary driver of systemic aging 514. Thus, the ICE phenotype may simply model accelerated DNA damage toxicity and SASP-induced physical deterioration rather than the natural, progressive erosion of epigenetic states 513.
Stem Cell Attrition as an Alternative Explanatory Mechanism
Further critiques target the methodological completeness of the Yang et al. study and offer alternative explanations for the physical frailty of the ICE mice. Reviewers on post-publication peer review platforms like PubPeer have noted a lack of deep single-cell transcriptomic data in the original study, which makes it difficult to conclusively prove that individual cells are progressively losing their unique identity ("ex-differentiation") rather than the tissue simply experiencing a shift in the proportional representation of different cell types due to selective cell death 11.
A compelling alternative explanation for the rapid aging in ICE mice is adult stem cell exhaustion 517. Severe DNA damage responses are known to cause the rapid depletion of adult stem cell niches, either through apoptosis or forced premature differentiation to repair immediate tissue damage 517. The depletion of regenerative stem cell pools across the organism could fully explain the observed phenotypes - such as frailty, alopecia, and organ dysfunction - without requiring the invocation of a novel "information loss" paradigm 513.
Skepticism Regarding Sirtuins and Resveratrol Efficacy
The ITOA is deeply intertwined with the function of sirtuins, which act as both epigenetic regulators and primary DNA repair responders. David Sinclair's earlier research heavily positioned sirtuins as universally conserved longevity genes and identified small molecules like resveratrol as potent direct activators of these pathways 2811.
This positioning has faced sustained pushback over the past decade. Several independent laboratories and large-scale pharmaceutical trials have failed to replicate early claims that sirtuin overexpression extends lifespan in standard model organisms such as Caenorhabditis elegans (worms) and Drosophila melanogaster (flies) 8. In yeast, some studies have even suggested that sirtuin overexpression can be detrimental to chronological lifespan under certain conditions 8. Furthermore, Charles Brenner and other biochemists assert that resveratrol acts as an assay artifact rather than a true direct allosteric activator of SIRT1, noting that massive pharmaceutical investments into resveratrol analogs failed to produce viable longevity therapeutics 8. Consequently, researchers like Matt Kaeberlein argue against viewing aging predominantly through the lens of a single pathway (e.g., sirtuin-mediated epigenetic noise) when basic biology indicates that aging encompasses a highly complex, polygenic deterioration process spanning numerous interacting systems 201516.
Epigenetic Clocks and the Measurement of Biological Age
The discourse surrounding the Information Theory of Aging frequently intersects with the rapidly expanding literature on epigenetic clocks. However, a critical conceptual distinction must be drawn between abstract "epigenetic noise" (the stochastic degradation of cellular identity proposed by the ITOA) and the precisely measured, directional DNA methylation changes utilized by modern epigenetic clocks.
The Evolution of DNA Methylation Algorithms
Epigenetic clocks are sophisticated bioinformatics models that evaluate the methylation status of specific cytosine-guanine dinucleotide (CpG) sites across the genome to estimate an organism's biological age 171819. Since their introduction, these tools have evolved through three distinct generations, each serving different clinical, diagnostic, and research imperatives.
| Clock Generation | Primary Training Objective | Representative Models | Strengths and Applications | Core Limitations |
|---|---|---|---|---|
| First Generation | Predict chronological age using cross-sectional datasets. | Horvath Multi-tissue Clock, Hannum Clock 172021 | High accuracy in estimating strict chronological time. Useful for broad tissue-specific benchmarking. | Limited predictive power for adverse health outcomes, mortality, or disease risk. Captures normal developmental drift better than pathology 172122. |
| Second Generation | Predict physiological "state," healthspan, and mortality risk. | PhenoAge, GrimAge 17202123 | Incorporates clinical biomarkers (e.g., glucose, C-reactive protein) during training. Excellent for stratifying disease risk and all-cause mortality 21222324. | Output values are complex composites of various pathways (inflammation, metabolism), making pure molecular mechanistic interpretations difficult 1721. |
| Third Generation | Quantify the dynamic "pace" of aging in real-time. | DunedinPACE 1721 | Based on longitudinal data. Highly sensitive to short-term changes; ideal as an endpoint for interventional clinical trials 1721. | Requires massive longitudinal datasets for validation; long-term predictive value for decadal health outcomes is still accumulating 1721. |
Distinguishing Stochastic Epigenetic Noise from Deterministic Clocks
While the ITOA assumes that aging is a consequence of accumulating epigenetic disorder, conventional epigenetic clocks do not actually measure entropy or noise. First- and second-generation clocks rely on linear regression to capture consistent, directional, and predictable changes in methylation that occur across a population 17. As researchers have pointed out, pure random noise cannot act as a reliable timekeeping mechanism precisely because of its stochastic nature.
Some biogerontologists propose that the reliable "ticking" of epigenetic clocks represents the continuous unfolding of developmental programs rather than the accumulation of damage. According to the developmental theory of epigenetic aging, early-life ontogenesis transitions into late-life "maturo-developmental" programs 25. In this view, the highly specific clock CpG sites (many of which are located near developmental genes like those of the Hox and polycomb classes) track a highly coordinated, deterministic biological process, whereas "epigenetic noise" tracks uncoordinated decay 1725.
Incorporating Biological Resilience into Age Measurement
Current standard methylation clocks measure the pace and state of systemic decay but generally fail to quantify the system's underlying biological resilience - the capacity to respond to, reorganize, and maintain homeostasis under stress 17. Two individuals with the exact same epigenetic "biological age" reading may possess vastly different capacities to rebound from a stressor, such as surgery or severe infection 17.
Newer mechanistic frameworks, such as the EpiAge-R (Epigenetic Age with Resilience) metric, are being developed to explicitly model the dynamic equilibrium between damage (noise/entropy) and adaptive compensatory changes (repair capacity) 17. This shift represents an attempt to transition the field of epigenetic measurement from the passive correlation of CpG sites to the active assessment of a tissue's reprogramming and rejuvenation potential, bridging the gap between statistical clocks and mechanistic aging biology 17.
Contextualizing Information Loss Within the Hallmarks of Aging
The ITOA is largely framed as a singular, unified cause of aging, proposing that epigenetic information loss sits upstream of most other aging phenotypes 2. However, the global consensus among biogerontologists strongly favors a multifactorial model that views aging as a deeply interconnected web of failure points.
Integration with the Expanded Twelve Hallmarks
In 2013, and subsequently expanded in 2023, the scientific community codified the "Hallmarks of Aging" to establish a systematic, integrative framework for understanding cellular decline. A biological process is considered a hallmark if it fulfills three criteria: it manifests during normal aging, it accelerates aging when aggravated experimentally, and it slows aging when therapeutically ameliorated 172627.
The 2023 update recognizes twelve distinct hallmarks categorized into primary (initiating triggers that directly cause damage), antagonistic (context-dependent compensatory responses that become toxic over time), and integrative (downstream consequences that occur when compensatory mechanisms fail) groups 172627. The Information Theory of Aging maps directly onto only a subset of these processes, suggesting it may not serve as the universal upstream driver it claims to be.
| Category | Hallmark of Aging (2023 Update) | Biological Description | Intersection with the Information Theory of Aging (ITOA) |
|---|---|---|---|
| Primary | Genomic Instability | Accumulation of DNA damage, chromosomal rearrangements, and mutations over time 172728. | ITOA relies on DNA double-strand breaks to trigger chromatin modifier relocalization; however, ITOA explicitly argues that the epigenetic consequence, not the structural mutation, is the true driver of decline 25. |
| Primary | Telomere Attrition | Shortening of protective chromosome caps with each cell division, leading to replicative senescence 1728. | Viewed by the ITOA primarily as a parallel or downstream effect of disorganized chromatin architecture and the failure of epigenetic silencing mechanisms 2. |
| Primary | Epigenetic Alterations | Changes to DNA methylation, histone modifications, and broad chromatin folding 1728. | The central pillar of the ITOA. Represents the specific "loss of information" and "epigenetic noise" that obscures cellular identity and drives the aging phenotype 2417. |
| Primary | Loss of Proteostasis | Inability to correctly fold, traffic, and degrade proteins, leading to toxic cellular aggregates 1728. | Claimed by the ITOA to be a downstream consequence of corrupted epigenetic gene expression programs failing to produce necessary chaperone proteins 2. |
| Primary | Disabled Macroautophagy | Decline in the cellular recycling of large organelles and aggregated biomolecules 1736. | Similar to proteostasis; autophagic dysregulation is viewed as secondary to transcriptomic noise. |
| Antagonistic | Deregulated Nutrient Sensing | Alterations in insulin/IGF-1, mTOR, AMPK, and sirtuin metabolic pathways 1728. | High overlap. Sirtuins are the proposed physical link between cellular energy sensing and the maintenance of the epigenetic landscape 219. |
| Antagonistic | Mitochondrial Dysfunction | Decline in oxidative phosphorylation and increased oxidative stress 1728. | Sirtuin depletion (due to DNA repair duties) allegedly reduces mitochondrial gene expression, linking epigenetic noise directly to metabolic decline 229. |
| Antagonistic | Cellular Senescence | Irreversible cell cycle arrest accompanied by the pro-inflammatory SASP 1728. | A critical point of contention. Critics argue senescent cell accumulation (not abstract noise) is the true cause of rapid aging in models like the ICE mouse 513. |
| Integrative | Stem Cell Exhaustion | Depletion of regenerative stem cell pools necessary for tissue maintenance 1728. | ITOA suggests epigenetic drift causes loss of stem cell identity; critics argue severe DNA damage leads to outright stem cell death independent of epigenetic noise 528. |
| Integrative | Altered Intercellular Communication | Breakdown in signaling between cells (e.g., endocrine, paracrine) 1728. | Viewed as a secondary effect resulting from cellular ex-differentiation and disorganized secretion profiles. |
| Integrative | Chronic Inflammation | "Inflammaging" - sterile, low-grade systemic inflammation 171436. | Driven heavily by the SASP and immunosenescence. Represents systemic noise and intercellular disruption rather than purely intracellular epigenetic noise 1436. |
| Integrative | Dysbiosis | Imbalance of the host microbiome (e.g., gut flora) 1736. | An environmental and systemic factor largely independent of cellular epigenetic reprogramming models. |
Interconnected Drivers and Downstream Consequences
The interdependence of the aging hallmarks means that the experimental accentuation or attenuation of one specific hallmark usually affects multiple others 26. While Sinclair's ITOA provides a compelling narrative for how genomic instability leads to epigenetic alterations, it struggles to fully encompass non-nuclear drivers of aging, such as disabled macroautophagy or dysbiosis. Treating epigenetic alterations as the sole root cause risks ignoring the complex feedback loops - for instance, how mitochondrial dysfunction generates reactive oxygen species that in turn cause the DNA damage that initiates epigenetic drift in the first place 1719.
Alternative Paradigms of Biological Aging
Outside of the Information Theory of Aging, several other dominant theoretical frameworks offer competing explanations for the biological origins of senescence. These theories reject the idea that aging is primarily a passive loss of epigenetic information.
The Hyperfunction Theory and Quasi-Programmed Senescence
The Hyperfunction Theory, pioneered by Mikhail Blagosklonny, suggests that aging is not a consequence of accumulated molecular damage, but rather the aimless, quasi-programmed continuation of developmental growth programs 303132.
According to this view, metabolic and genetic pathways that are absolutely essential for early-life growth, development, and reproduction - most notably the Mechanistic Target of Rapamycin (mTOR) pathway - become hyperfunctional in post-reproductive life 303132. Because evolution does not select against traits that only become detrimental after an organism has reproduced, these growth pathways remain active, driving cellular hypertrophy, hyperfunction, and eventually cellular senescence 303133. The robust efficacy of rapamycin (a specific mTOR inhibitor) in extending healthspan and lifespan across multiple species - including yeast, mice, and currently being tested in companion dogs by researchers like Matt Kaeberlein - provides strong empirical support for the Hyperfunction Theory 30313443. This paradigm suggests that muting overactive growth programs is more critical for extending lifespan than attempting to restore lost epigenetic information 31.
The Reliability Theory and Systems Failure
The Reliability Theory approaches the aging organism from an engineering and mathematical systems biology perspective. Originating from the work of Leonid Gavrilov and Natalia Gavrilova, it posits that aging is defined by the progressive stochastic failure of redundant systems 353637.
In this model, complex multicellular systems face an intrinsic reliability problem where the machinery maintaining structural order degrades over predictable timescales 3637. While the ITOA focuses narrowly on the specific substrate of the epigenome as an information store, a layered reliability view suggests that dysfunction arises from state encoding and feedback failures distributed across multiple substrates with vastly different turnover timescales 363747. This has profound therapeutic consequences: if a fast-turnover encoding layer (like the epigenome) is therapeutically reset via reprogramming, but slower structural or extracellular matrices have permanently degraded, the slow substrates will simply re-specify the fast ones, pulling the cellular system rapidly back toward dysfunction 3637.
Evolutionary Theories of Aging
Underpinning all mechanistic theories are the classic evolutionary theories of aging, which broadly posit that aging is a non-adaptive byproduct resulting from a decline in the intensity of natural selection with chronological age 3839.
- Mutation Accumulation (MA): Proposes that late-acting deleterious mutations accumulate in a population because natural selection is too weak to weed them out after the organism has passed its reproductive peak 3338.
- Antagonistic Pleiotropy (AP): Suggests that genes offering early-life reproductive advantages are strongly selected for, even if they have highly detrimental, pleiotropic effects late in life (a concept that aligns closely with the Hyperfunction Theory) 3338.
- Disposable Soma (DS): Framed around resource allocation, positing that organisms have a finite energy budget. Evolution favors allocating resources toward rapid growth and reproduction rather than indefinite somatic maintenance and cellular repair, guaranteeing eventual bodily decay 3338.
| Theory Category | Specific Theory | Primary Driver of Aging | Therapeutic Implication |
|---|---|---|---|
| Information Loss | Information Theory of Aging (ITOA) | Accumulation of epigenetic noise due to continuous DNA repair; loss of cellular identity. | Reversible. Treat via partial epigenetic reprogramming (OSK) to restore youthful analog states 23. |
| Overactivation | Hyperfunction Theory | Aimless continuation of developmental growth programs (e.g., mTOR) causing cellular hypertrophy and senescence. | Modifiable. Treat via pharmacological inhibitors of growth pathways (e.g., Rapamycin) to suppress hyperfunction 3031. |
| Systems Engineering | Reliability Theory | Progressive, stochastic failure of redundant biological components across multiple tissue layers and timescales. | Difficult to reverse universally. Requires multi-layered interventions addressing both fast-turnover cells and slow-turnover extracellular matrices 3637. |
| Evolutionary Resource Allocation | Disposable Soma Theory | Finite biological energy prioritized for reproduction over long-term somatic DNA repair and maintenance. | Preventive. Suggests that increasing maintenance efficiency or reducing metabolic load (e.g., caloric restriction) delays decay 3338. |
Clinical Translation of Epigenetic Reprogramming
Despite robust theoretical debates regarding its status as the absolute root cause of aging, the concept of utilizing epigenetic manipulation to reverse tissue decline has advanced rapidly toward clinical translation. The therapeutic premise of partial reprogramming is to expose somatic cells to the Yamanaka factors (OSK) just long enough to erase accumulated epigenetic noise and restore youthful transcriptomic profiles, without crossing the critical threshold into dedifferentiation and pluripotency 910.
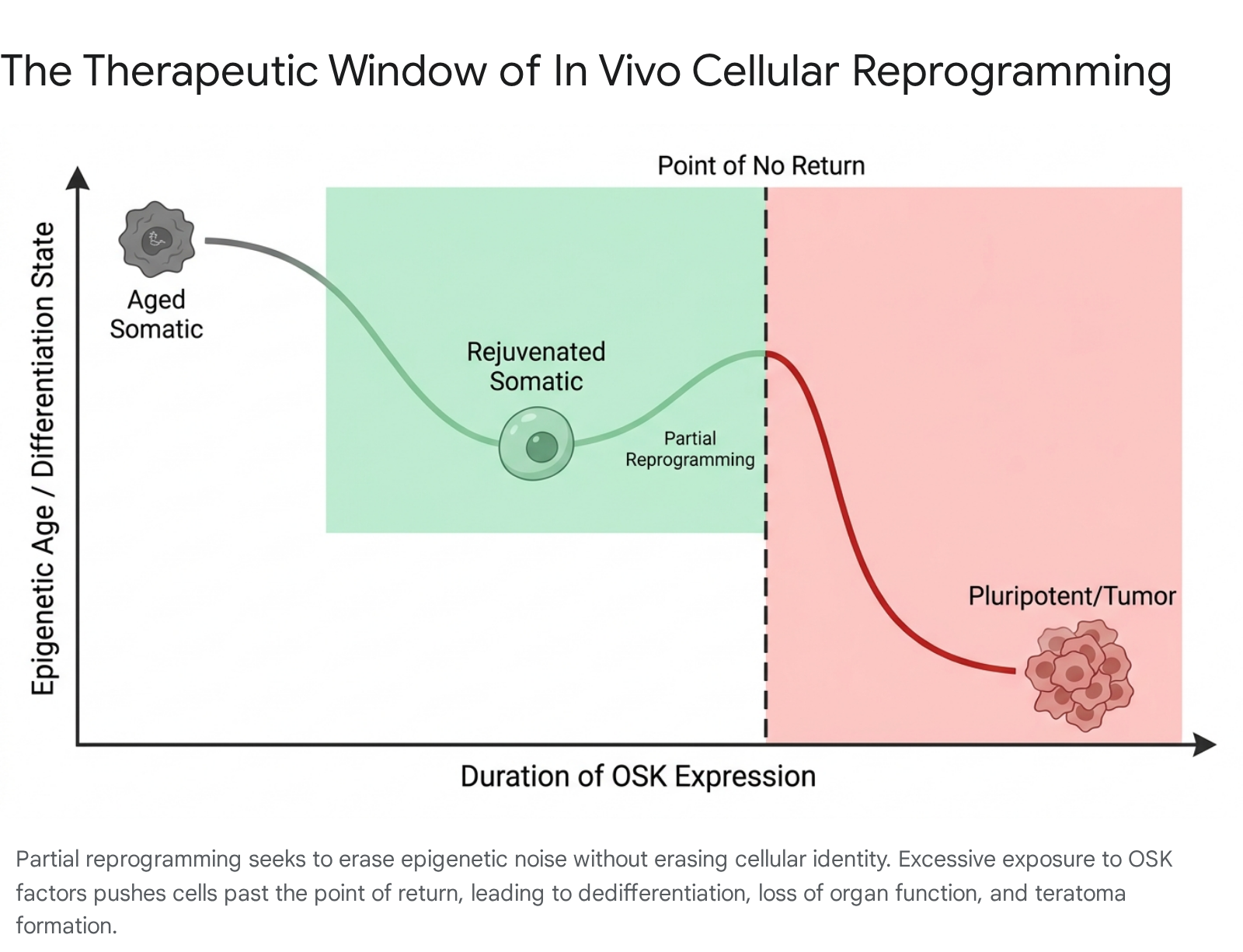
Navigating the Teratoma Risk in Somatic Tissues
In vivo partial reprogramming presents extraordinary safety and engineering challenges. If somatic cells are exposed to pluripotency factors for too long, or at too high a dosage, they lose their somatic identity entirely. This dedifferentiation can lead to the formation of teratomas - aggressive, fast-growing tumors comprising multiple unorganized tissue types 9104041.
Preclinical studies have consistently shown that continuous, unchecked induction of OSKM in murine models results in severe weight loss, catastrophic organ failure, and high mortality within a matter of days 4041. The tissue microenvironment heavily influences this delicate balance; highly proliferative tissues or those under constant inflammatory stress (such as the liver and intestines) are particularly sensitive to reprogramming factors and are highly prone to tumorigenesis 10. Consequently, researchers have focused on identifying highly precise dosing schedules, utilizing cyclic or highly transient expression regimens, and investigating non-integrating delivery methods like adenoviral vectors (AAVs) or chemical reprogramming approaches to strictly control the temporal window of OSK activation 94041.
Ocular Neuropathies as an Initial Clinical Target
Because systemic administration of OSK poses significant off-target oncogenic risks, initial clinical applications have prudently targeted highly compartmentalized organs. The eye serves as an ideal candidate for early human intervention due to its immune privilege, the post-mitotic nature of its neurons (such as retinal ganglion cells), the ability to administer precise local dosages via intravitreal injection, and the exceptionally low risk of the viral vector escaping into systemic circulation 5242.
The Life Biosciences ER-100 Clinical Trial
In a landmark milestone for the field of longevity medicine, Life Biosciences received Investigational New Drug (IND) clearance from the U.S. Food and Drug Administration (FDA) in January 2026 to initiate a Phase 1 human clinical trial (NCT07290244) for their partial epigenetic reprogramming therapy, designated ER-100 524344. This marks the first time an OSK-based cellular rejuvenation therapy has entered human clinical testing 4244.
The ER-100 therapy utilizes a modified adeno-associated virus (AAV) vector to deliver the OCT-4, SOX-2, and KLF-4 genes directly to the retina 5657. Crucially, the system incorporates a doxycycline-inducible switch, allowing clinicians to precisely control the activation and, if necessary, the rapid deactivation of the reprogramming factors by administering or withdrawing systemic doxycycline 5257.
The Phase 1 trial focuses on patients suffering from severe, age-related optic nerve disorders, specifically open-angle glaucoma (OAG) and non-arteritic anterior ischemic optic neuropathy (NAION), a condition often described as a "stroke of the eye" that causes sudden blindness 52445658. Preclinical data in non-human primate models demonstrated that ER-100 successfully targeted retinal ganglion cells, restored youthful DNA methylation patterns, and measurably improved visual function following optic nerve injury without inducing tumor formation 424458.
By framing the intervention strictly as a treatment for distinct, recognized ocular pathologies rather than targeting "aging" as a broad systemic condition - which the FDA does not currently classify as a disease - Life Biosciences successfully navigated the existing regulatory framework 4359. This strategic approach provides a viable clinical pathway to establish the basic safety and mechanistic proof-of-concept for epigenetic rejuvenation in humans. If successful, it could pave the way for treatments targeting more complex, systemic age-related conditions, such as metabolic dysfunction-associated steatohepatitis (MASH), for which the company is already generating preclinical proof-of-concept data 424358.
Global Advances in Gerontological Geography and Epigenetics
Simultaneously, alternative research avenues globally are dramatically expanding the scope of epigenetic and gerontological understanding, suggesting that aging markers vary significantly across different tissues and populations.
Spatial Transcriptomic Mapping of Aging
In China, researchers from the Chinese Academy of Sciences (CAS) and BGI Research have pioneered high-precision spatial transcriptomic mapping, introducing the concept of "Gerontological Geography" (GG) 4561. By analyzing millions of spatial spots across various organs, this research seeks to map the specific epicenters of aging 45. A major recent finding from this collaboration, led by researchers including Guanghui Liu and Jing Qu, revealed that the accumulation of immunoglobulins, specifically IgG, directly drives tissue structural disorder, loss of cellular identity, and cellular senescence across multiple organs 4561. This identifies IgG not just as a biomarker, but as a potent inducer of systemic aging, notably triggering inflammatory factor release in macrophages and microglia 45.
Further work by Liu and Qu has focused on the molecular mechanisms of aging driven by the resurrection of endogenous retroviruses (ERVs) 46. Their research indicates that epigenetic derepression during aging allows these dormant viral sequences (which make up a significant portion of the human genome) to be transcribed and translated, leading to the accumulation of viral-like particles that reinforce cellular senescence and tissue aging 46. This highlights how the loss of epigenetic regulation can activate deeply embedded genomic threats, providing another layer of complexity to the ITOA.
Organ-Specific Aging Models and Future Directions
Recognizing that genetic and epigenetic regulation of aging differs vastly across human organs, Chinese research teams have also established new models to predict the aging of specific organs, identifying distinct genetic loci associated with multi-organ versus isolated organ aging 47. To address demographic specificities, the Aging Biomarker Consortium (ABC) launched the X-Age Project to construct a comprehensive aging evaluation system and composite aging clocks tailored specifically to the Chinese population, acknowledging that current epigenetic clocks trained predominantly on European or Hispanic ancestries may lack accuracy across diverse global cohorts 244849.
These global advancements underscore that while the Information Theory of Aging provides a profound conceptual leap in understanding how cells lose their identity through epigenetic noise, the physical reality of aging remains an immensely complex interplay of spatial transcriptomics, immune hyperfunction, viral resurrection, and systemic metabolic decline.