Epidemiology and Mechanisms of Sleep and Human Longevity
Epidemiological Associations Between Sleep Duration and Mortality
The Non-Linear Dose-Response Mortality Curve
Extensive epidemiological data establishes a definitive, non-linear relationship between sleep duration and lifespan. Aggregate data from global prospective cohort studies consistently demonstrate a U-shaped or J-shaped dose-response curve regarding sleep duration and all-cause mortality 12.
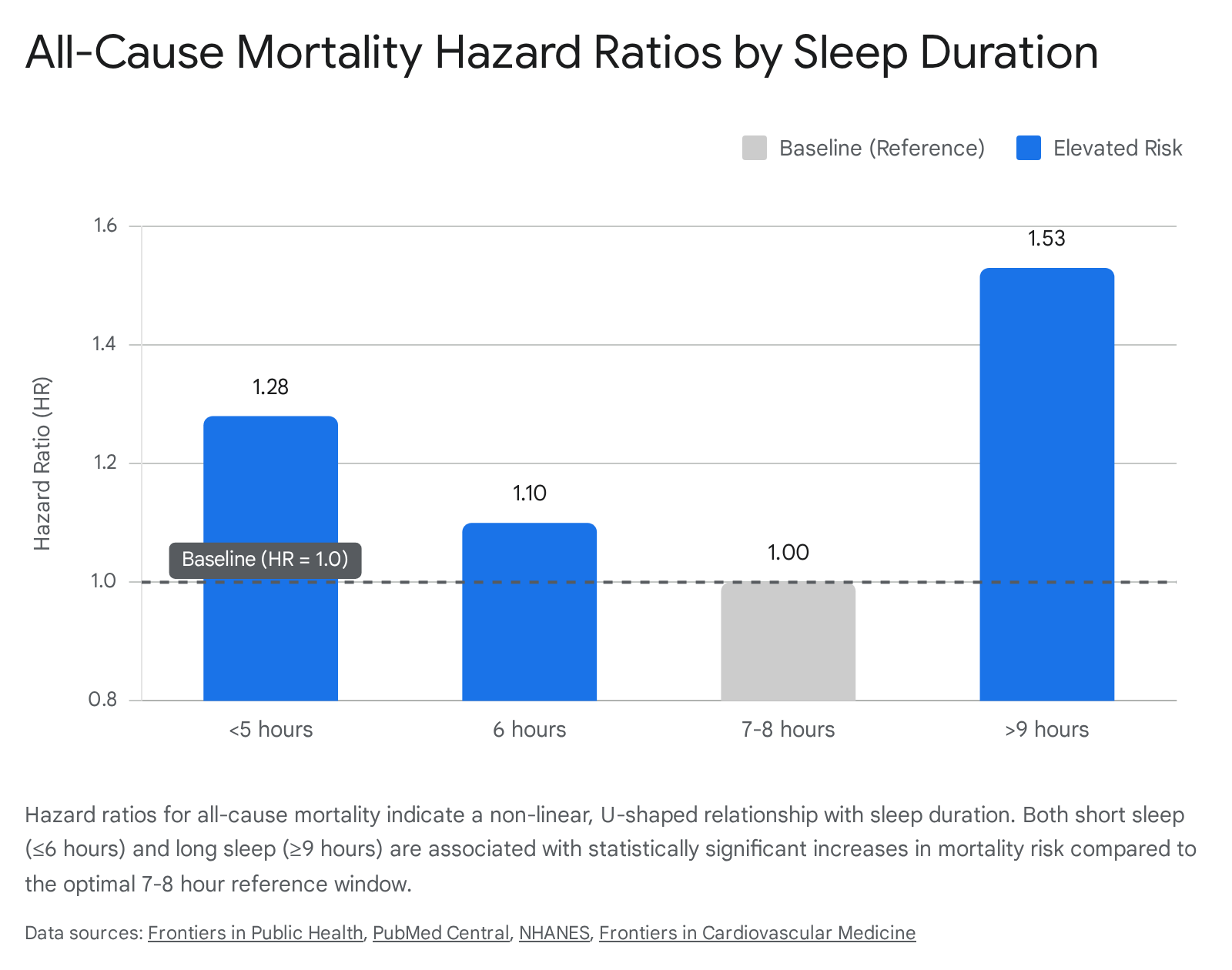
The nadir of this mortality curve - representing the lowest risk of death and major cardiovascular events - is reliably observed at approximately seven to eight hours of sleep per night for the general adult population 23. Deviations from this optimal window in either direction are associated with measurable reductions in life expectancy and increased incidences of cardiometabolic and neurodegenerative diseases 45.
Short sleep duration, generally defined as fewer than six to seven hours per night, carries an elevated mortality risk that scales with the severity of sleep deprivation. Meta-analyses encompassing diverse populations - including robust non-Western cohorts - indicate that sleep durations of five hours or less are associated with hazard ratios (HR) for all-cause mortality ranging from 1.25 to 1.30 when compared to the optimal baseline 467. The physiological detriments of short sleep are highly dose-dependent; researchers have observed that a one-hour reduction in sleep below the recommended threshold corresponds to a 3% to 11% increased risk for all-cause mortality, coronary heart disease, and type 2 diabetes mellitus 4.
Long sleep duration, typically defined as nine or more hours per night, exhibits an equally robust, and often steeper, association with mortality in observational cohorts. In multiple large-scale analyses, participants sleeping more than nine hours demonstrated an all-cause mortality hazard ratio between 1.24 and 1.50 compared to the seven-to-eight-hour reference group 689. Furthermore, excessive sleep duration has been linked to specific outcomes, such as a 34% increased risk in cardiovascular disease (CVD) mortality and elevated risks for cancer-related mortality 510.
Sleep Trajectories and Regularity Over Time
While cross-sectional average sleep duration provides a foundational metric for population health, recent longitudinal research emphasizes sleep trajectories and night-to-night sleep regularity as critical, independent predictors of mortality. In observational cohorts tracking the sleep habits of adults over multi-year periods, maintaining a consistent trajectory of seven to nine hours of sleep is associated with the absolute lowest mortality health risks 1011.
Individuals who transition between sleep categories over time - such as shifting from a healthy duration to a short duration, or vice versa - face significantly higher risks than those maintaining a stable, albeit suboptimal, routine. For instance, transitioning from a short sleep duration to a long sleep duration yields an all-cause mortality hazard ratio of 1.29 to 1.33, while transitioning from long to short sleep substantially increases cardiovascular mortality risk specifically (HR 1.34) 1011. These trajectories suggest that the instability of sleep architecture may inflict distinct metabolic and cardiovascular damages above and beyond the absolute hours slept.
Irregular sleep patterns, defined by high night-to-night variability in sleep onset and duration, severely compound the risks associated with suboptimal duration. When analyzing objectively measured sleep via accelerometry - which removes the bias of self-reported questionnaires - irregular sleep habits combined with suboptimal duration exacerbate mortality outcomes. Studies separating populations by sleep regularity indicate that individuals with less than seven hours of irregular sleep, or more than eight hours of regular sleep, both face an approximately 26% to 28% increased risk of all-cause mortality 12. This compounding evidence has prompted major medical organizations, including the American Heart Association, to explicitly recognize sleep regularity alongside total duration as a core, modifiable metric of cardiometabolic health 5.
Mortality Risk Disparities in Specific Cohorts
The universal applicability of the seven-to-eight-hour optimal sleep window requires nuanced calibration when examining specific demographic subgroups, particularly the oldest-old populations. While middle-aged cohorts strictly adhere to the U-shaped curve centered at seven hours, data from longitudinal surveys of populations aged 80 and older suggest a slight rightward shift in optimal duration. In surveys of the oldest-old, such as the Chinese Longitudinal Healthy Longevity Survey involving over 15,000 individuals aged 80 and above, sleep duration maintained a U-shaped association with all-cause mortality, but the absolute minimum risk of death was centered closer to eight hours 2. Furthermore, in these advanced age brackets, sleeping a little longer (between eight and nine hours) was optimal for both overall survival and maintaining a high quality of life near the end of life 2.
Sex-specific analyses also reveal divergent risk profiles regarding sleep architecture. Meta-analyses indicate that the increased risk of all-cause mortality associated with long sleep duration is statistically significant and highly pronounced in women (HR 1.48), whereas the risk associated with short sleep duration reaches clearer statistical significance in men (HR 1.13) 8. These variations underscore the necessity of moving beyond generalized public health guidelines toward precision-based sleep recommendations that account for age, sex, and baseline physiological health.
| Demographic / Behavioral Cohort | Optimal Sleep Duration | Key Mortality Risk Observations |
|---|---|---|
| General Adult Population | 7 - 8 hours | U-shaped risk curve; HR 1.25 for <7 hours; HR 1.28-1.50 for >9 hours 69. |
| Oldest-Old (Age 80+) | ~8 hours | Slight rightward shift in optimal duration; >9 hours remains a risk factor 2. |
| Irregular Sleepers | N/A (Highly Variable) | Irregularity compounds duration risks; HR 1.28 for short/irregular sleep 12. |
| Trajectory Shifters | Maintained 7 - 9 hours | Transitioning from short to long sleep yields highest all-cause mortality risk (HR 1.33) 10. |
Evaluating Causality Through Mendelian Randomization
Addressing Reverse Causality in Observational Data
A central limitation of observational sleep epidemiology is the inherent inability to distinguish whether extreme sleep durations directly cause physiological decline, or whether underlying, unmeasured illnesses cause extreme sleep patterns. This phenomenon, known as reverse causality, is particularly scrutinized regarding the association between long sleep duration and mortality. Early analyses suggested that long sleep might merely serve as a diagnostic marker for subclinical morbidities - such as undiagnosed cancer, depression, systemic inflammation, or cancer-related fatigue - rather than acting as a direct biological driver of death 113.
To isolate causal effects and bypass the limitations of observational confounding, modern epidemiological research utilizes Mendelian Randomization (MR). MR functions conceptually similarly to a randomized controlled trial by utilizing genetic variants - specifically single nucleotide polymorphisms (SNPs) that dictate a genetic predisposition to certain sleep traits - as instrumental variables. Because genotypes are assigned randomly at conception and cannot be altered by the subsequent onset of disease or lifestyle factors, MR effectively eliminates reverse causality and environmental confounding 141516.
Genetic Predisposition to Short Sleep and Cardiovascular Risk
Mendelian Randomization studies analyzing massive genomic datasets, such as the UK Biobank and the FinnGen study, have provided nuanced, and sometimes conflicting, insights into the true causality of sleep duration. Regarding short sleep, genetic evidence strongly supports a direct causal pathway to systemic disease. MR analyses confirm that genetically predicted short sleep duration directly causes hypertension, coronary artery disease, and an elevated risk for various broad cardiovascular diseases 5. In meta-analyses of MR studies, a genetic predisposition to shorter sleep durations is associated with a drastically increased odds ratio (OR) for circulatory conditions, reaching as high as 3.05 for overall cardiovascular diseases 14. This solidifies the consensus that sleep deprivation is not merely a correlate of poor cardiometabolic health, but a primary, modifiable instigator of arterial and cardiac dysfunction.
Genetic Evidence Regarding Long Sleep and Specific Pathologies
The genetic findings surrounding long sleep duration are significantly more complex and contested. When examining overall all-cause mortality, some extensive MR analyses find no causal association between genetically predicted long sleep duration and lifespan reduction, heavily reinforcing the reverse-causality hypothesis that underlying illness drives the statistical association seen in observational studies 31717. In these models, long sleep acts primarily as a symptom rather than a pathogen.
However, when mortality and morbidity outcomes are subdivided by specific organ systems, MR indicates that long sleep does possess intrinsic causal detriments. Genetically inferred prolonged sleep has been shown to directly cause an increased risk of specific neurodegenerative diseases (OR 2.55) and certain cardiovascular outcomes (OR 2.71) 14. Furthermore, recent MR investigations have linked long sleep duration to specific renal pathologies. A genetic predisposition to prolonged nighttime sleep significantly elevates Kidney Injury Molecule-1 (KIM-1) levels and causes renal tubular damage, suggesting that extreme sleep duration directly precipitates renal stress 16.
Additionally, MR studies investigating daytime napping - often a corollary behavior to long or fragmented nighttime sleep - show a potential causal association with an increased risk of all-cause mortality in older adults. This napping-mortality link appears to be largely driven by an increased genetic risk for cancer-related mortality 317. This highlights the physiological necessity of distinguishing between brief, restorative circadian naps and prolonged daytime sleepiness driven by genetic or biological dysregulation.
Molecular and Cellular Mechanisms of Sleep-Driven Longevity
Epigenetic Age Acceleration and DNA Methylation
Sleep health fundamentally influences the molecular clocks that dictate biological aging. Epigenetic clocks, which measure DNA methylation patterns at specific CpG sites across the genome to estimate biological age versus chronological age, offer highly precise metrics of cellular degradation. Severe sleep disturbances and suboptimal sleep durations are independently linked to epigenetic age acceleration (EAA), providing a molecular explanation for the mortality risks observed in epidemiology 1820.
Recent studies utilizing advanced, second-generation methylation metrics - specifically the GrimAge clock and the DunedinPACE (Pace of Aging Computed from the Epigenome) algorithm - demonstrate that poor sleep quality actively drives biological aging. DunedinPACE is a highly sensitive longitudinal biomarker trained to track the speed of multi-system decline across 19 different organ system biomarkers; it effectively measures the current "speedometer" of aging 192021. Analysis using DunedinPACE reveals that individuals suffering from insomnia and short sleep (fewer than six hours) age at a significantly faster biological rate than undisturbed sleepers 1822.
In clinical cohorts, the deterioration of subjective sleep quality, quantified via the Pittsburgh Sleep Quality Index (PSQI), correlates positively and significantly with an accelerated pace of aging and the onset of metabolic syndrome 18. Notably, sleep's impact on epigenetic aging is most pronounced in populations already facing cognitive vulnerabilities; older adults with Alzheimer's disease who exhibit poor sleep show the highest rates of epigenetic age acceleration across multiple distinct clock algorithms, suggesting a vicious cycle between neurodegeneration and molecular aging 20.
Telomere Maintenance and Cellular Senescence
At the cellular level, the physical toll of inadequate sleep manifests through the accelerated attrition of telomeres and the premature onset of cellular senescence. Telomeres, the protective repetitive nucleoprotein caps at the ends of linear chromosomes, naturally shorten with each cycle of cell division. When telomeres reach a critical minimum length, they trigger a profound DNA damage response that halts the cell cycle, pushing the cell into a state of irreversible growth arrest known as replicative senescence 2324.
Crucially, senescent cells do not undergo apoptosis; instead, they remain metabolically active and adopt a senescence-associated secretory phenotype (SASP). The SASP is characterized by the widespread secretion of pro-inflammatory cytokines, chemokines, and tissue-remodeling matrix metalloproteinases. This chronic, sterile, low-grade inflammation - often termed "inflammaging" - drives the broader systemic aging process, creates a pro-tumorigenic microenvironment, and contributes to the progressive dysfunction of adjacent healthy tissues 2324.
While subjective "sleep quality" alone has shown mixed correlations with telomere length in general meta-analyses, objective physiological extremes - such as severe short sleep duration, chronic insomnia, and obstructive sleep apnea - are significantly and consistently associated with shorter leukocyte telomeres, marking them as primary environmental drivers of cellular aging 25. The precise physiological transition states between sleep stages may also play a role in maintaining cellular homeostasis. Research identifies the Intermediate State (IS) occurring between Slow Wave Sleep (SWS) and Rapid Eye Movement (REM) sleep as a period of profound autonomic and neurobiological reorganization, though the exact impacts of these micro-architectural sleep stages on telomerase expression and maintenance require further longitudinal study 26.
Endocrine Regulation During Slow Wave Sleep
Sleep operates as a master regulator of systemic endocrine function and metabolic clearance, independent of neurological waste removal. Short sleep duration reliably and rapidly impairs glucose metabolism and insulin sensitivity. Randomized crossover trials demonstrate that restricting sleep by merely 1.5 hours per night for six weeks significantly elevates fasting insulin and increases systemic insulin resistance (measured via HOMA-IR) in healthy adult women, entirely independent of dietary changes, caloric intake, or body weight fluctuations 5. Based on this robust physiological evidence, the American Diabetes Association formally recognizes sleep duration as a critical, modifiable risk factor for type 2 diabetes, with extreme sleep durations raising disease risk by up to 50% 5.
Deep Non-Rapid Eye Movement (NREM) sleep - specifically Slow Wave Sleep (SWS) - is the primary biological window for endocrine restoration. The pituitary gland releases the vast majority of its daily growth hormone in distinct, concentrated pulses during SWS 2728. Growth hormone is essential for adult homeostasis; it facilitates tissue repair, maintains bone density, regulates muscle mass, and supports cognitive alertness 27. Chronic sleep curtailment, advancing age, or sleep fragmentation selectively deprives the body of SWS. This deprivation blunt-forces a severe reduction in growth hormone secretion, shifting the body from an anabolic state of repair to a catabolic state of accelerated physical aging.
The Glymphatic System and Neurobiological Clearance
Fluid Dynamics in Non-Rapid Eye Movement Sleep
While sleep dictates metabolic and epigenetic health globally, its role in neurobiology is heavily defined by the mechanical actions of the glymphatic system. The glymphatic network is a macroscopic, brain-wide waste clearance pathway that utilizes cerebrospinal fluid (CSF) to actively flush metabolic byproducts from the brain's interstitial spaces 2930.
Glymphatic function is highly governed by circadian rhythms and is fundamentally dependent on the sleep state. During Slow Wave Sleep (SWS) in the NREM phase, the brain's interstitial space expands by an astonishing 40% to 60%. This massive volumetric expansion is accompanied by a concurrent contraction of intracellular volume, radically reducing physical resistance to fluid flow 2930. CSF is driven deep into the brain parenchyma along perivascular spaces by the mechanical force of arterial pulsations and brain-wide vasomotor waves 30. Conversely, transitions into REM sleep - which is characterized by widespread brain activation and a sharp decrease in slow-wave electroencephalogram (EEG) activity - are associated with severe decreases in glymphatic transport and a rapid reduction in total brain water content 30.
Recent advancements in neuroimaging, specifically intrathecal gadolinium-enhanced dynamic contrast-enhanced MRI (DCE-MRI) and AI-based velocimetry algorithms deployed in 2023 and 2024, have allowed for the real-time quantification and mapping of human glymphatic transport 29. These high-dimensional computational fluid dynamics studies confirm that sleep loss and sleep fragmentation severely disrupt the amplitude of CSF pulsatility and diminish vascular compliance, resulting in the stagnation of interstitial fluid 29.
Blood-Brain Barrier Integrity and Amyloid Clearance
The primary evolutionary function of this hydraulic clearance system is the removal of neurotoxic waste, most notably amyloid-beta and tau proteins. The accumulation of these misfolded proteins is the primary pathological hallmark of Alzheimer's disease and broader neurodegenerative cascades 530.
Over decades, the failure to secure adequate SWS directly causes the pathogenic deposition of amyloid-beta. A 2024 study tracking 24-hour activity rhythms demonstrated that disrupted sleep patterns actively predict subsequent amyloid-beta deposition, an effect that is highly magnified in individuals carrying the APOE4 genetic risk variant 5. Furthermore, modern updates to glymphatic research indicate that chronic sleep deprivation inflicts structural damage beyond mere clearance failure. Prolonged sleep loss actively damages the integrity of the blood-brain barrier, triggering local inflammatory activation and allowing peripheral immune cells to infiltrate the central nervous system, thereby exacerbating the neurodegenerative decline 29.
Longevity Phenotypes in Centenarian Populations
Sleep Architecture in the Blue Zones
Observations of centenarian populations in designated "Blue Zones" - regions characterized by exceptional geographic longevity such as Okinawa (Japan), Sardinia (Italy), and Loma Linda (California) - provide invaluable real-world insight into the integration of healthy sleep architectures within a complete lifestyle matrix. Epidemiological studies of Okinawan and Sardinian elders, who exhibit some of the highest life expectancies globally and significantly lower baseline rates of cardiovascular disease, cancer, and dementia, reveal distinct sleep preservation 3132333435.
Centenarians in these regions generally adhere closely to the seven-to-eight-hour sleep target, naturally aligning their sleep-wake cycles with circadian light patterns 3238. Their sleeping environments often emphasize minimalism and natural materials - such as Okinawan futons on tatami mats - which are hypothesized to provide firm support while limiting the circadian disruption caused by the artificial light and electronic prevalence endemic to Western sleep spaces 3236.
Synergistic Lifestyle Factors Affecting Sleep
In Blue Zone populations, sleep health is deeply intertwined with daily physical activity, communal social structures, and specific dietary patterns. Actigraphy studies tracking the physical activities of Okinawan elderly populations (e.g., in the village of Ogimi) show that individuals with the highest objective sleep quality metrics consistently engage in daytime walking, maintain highly regular eating habits over decades, and frequently take short, restorative daytime naps 3137.
Dietary inputs specific to these regions also chemically support sleep architecture. The traditional Okinawan diet is rich in soy, which contains high levels of the amino acid tryptophan - a direct biochemical precursor to melatonin and serotonin 27. Additionally, the high consumption of fish and seaweed provides omega-3 fatty acids and essential vitamins that facilitate the processing of homocysteine, reducing vascular inflammation and supporting the deep SWS required for optimal growth hormone release 2737.
While napping is a prominent feature in several long-lived populations (such as the traditional "siesta" in Ikaria or regular short naps in Okinawa), its health benefits appear highly dependent on duration and context. Brief naps serve to blunt cortisol spikes and reduce cardiovascular stress; however, excessive daytime sleepiness or prolonged napping in non-centenarian cohorts is frequently flagged as a marker of underlying systemic morbidity or nocturnal sleep fragmentation 33836.
| Sleep/Lifestyle Element | General Western Populations | Blue Zone Centenarian Populations |
|---|---|---|
| Average Duration | High variance; 1 in 3 adults sleep fewer than 7 hours 38. | Highly consistent; averages 7 to 9 hours 3238. |
| Circadian Regularity | High irregularity; shift work and social jetlag are common 10. | Aligned with natural solar light cycles (high circadian stability) 36. |
| Daytime Napping | Often a compensatory response to chronic nocturnal sleep deprivation 3. | Integrated as a brief, intentional stress-reducing daily routine 3637. |
| Physical Integration | Often sedentary; exercise is episodic 34. | Constant, low-intensity natural movement (e.g., gardening, walking) 34. |
Clinical Interventions and Their Impact on Lifespan
Iatrogenic Risks of Sedative-Hypnotic Medications
Given the dire biological consequences of sleep deprivation, clinical interventions are frequently deployed to manage insomnia. However, the specific modality of intervention has profound implications for a patient's ultimate longevity. The widespread pharmacological prescription of sedative-hypnotics - specifically benzodiazepines (BZDs) and non-benzodiazepine receptor agonists (Z-drugs like zolpidem, zopiclone, and eszopiclone) - introduces severe iatrogenic risks, particularly to aging populations 3839.
Epidemiological and meta-analytic data consistently link prescription sleep medications to drastically increased all-cause mortality and rapid cognitive decline. Z-drug use is associated with a 60% increased risk of overall mortality (HR 1.60) compared to non-users, a risk that persists across various demographics and follow-up durations 383940. Similarly, traditional benzodiazepines carry severe, dose-dependent mortality hazards; daily users exhibit an all-cause mortality HR of 1.37 to 1.67, alongside a parallel increase in cardiovascular-specific mortality 41. Benzodiazepine cotreatment with opioids further escalates this risk, yielding a twofold increase in all-cause mortality 42.
The neurological risks of pharmacological sleep aids are equally substantial and devastating to longevity. In longitudinal cohorts of older adults, frequent use of sleep medications increases the risk of incident dementia by 30% to 79% 4344. Short-to-intermediate half-life benzodiazepines and Z-drugs elevate dementia risk by roughly 11% to 20%, with higher cumulative lifetime doses (>180 defined daily doses) pushing the hazard ratio up to 1.49 45.
Notably, demographic disparities exist in these pharmacological risks. The Health, Aging and Body Composition study found that white older adults who frequently used sleep medications faced a 79% higher risk of developing dementia, whereas black older adults - who generally utilized markedly lower quantities of these drugs - did not exhibit a similar statistical increase in dementia risk 4346. Beyond cognitive decline, the physical side effects of these drugs are lethal to the elderly; higher doses of Z-drugs prescribed to patients with existing dementia drastically increase the risk of ischemic stroke (HR 1.88), falls (HR 1.33), and devastating hip fractures (HR 1.96) 47.
Efficacy of Cognitive Behavioral Therapy for Insomnia
Because of the profound adverse events tied to pharmacological agents, behavioral interventions are universally recognized as the clinical gold standard for preserving sleep-dependent longevity. Cognitive Behavioral Therapy for Insomnia (CBT-I) is a highly structured, multimodal intervention that addresses the behavioral conditioning, poor sleep hygiene, and cognitive distortions underlying chronic sleep fragmentation without the use of central nervous system depressants 4849.
Meta-analyses spanning dozens of randomized controlled trials and thousands of patients indicate that CBT-I yields robust, long-lasting improvements in sleep architecture. Across populations suffering from complex chronic diseases (such as cancer, chronic pain, and cardiovascular disease), CBT-I demonstrates large effect sizes for reducing overall insomnia severity (Hedges' g = 0.98) and moderate-to-large effects for improving sleep efficiency (g = 0.77) and reducing sleep onset latency (g = 0.64) 50.
Crucially, CBT-I entirely avoids the neurotoxic, anticholinergic, and dependency-forming side effects of sleep medications. Population health economic models project that actively shifting older adults away from prescription sleep medications and toward CBT-I protocols would significantly reduce the lifetime incidence of cognitive impairment by 2.1% and physical falls by 8.5%. At a population scale, this behavioral shift is calculated to increase average life expectancy by 1.3 months per individual - yielding a massive 1.7 million life-years gained across the aging population, the vast majority of which would be spent in good health 51.
Exercise as a Non-Pharmacological Sleep Modulator
Complementary to structured cognitive therapies, physical exercise serves as a potent non-pharmacological modulator of sleep quality. Meta-analyses of randomized controlled trials targeting older adults demonstrate that exercise interventions significantly improve subjective sleep quality (measured via PSQI), yielding massive effect sizes (SMD = -2.46) 5253.
Interestingly, the relationship between exercise dose and sleep improvement is not strictly linear but follows a U-shaped dose-response curve. The most profound benefits to sleep architecture in elderly populations are observed with relatively low-barrier interventions: session durations of 30 minutes or less, low-to-moderate physical intensity, and frequencies of just twice a week 5253. The optimal effect is achieved at approximately 527 MET-minutes per week. This indicates that exhausting, high-intensity regimens are not required to unlock the longevity and sleep benefits of movement; rather, consistent, manageable physical activity - mirroring the natural movement patterns seen in Blue Zone populations - optimizes the physiological conditions necessary for restorative sleep and extended human lifespan.
| Intervention Category | Specific Modality / Agents | Efficacy for Sleep Architecture | Impact on Longevity and Disease Risk |
|---|---|---|---|
| Pharmacological | Benzodiazepines (e.g., Halcion, Restoril) | Short-term efficacy; high risk of biological tolerance and dependence. | Increases all-cause mortality (HR ~1.31-1.60). Increases dementia risk by up to 79% in frequent users. Elevated risk of lethal falls 414345. |
| Pharmacological | Z-Drugs (e.g., Zolpidem, Zopiclone) | Short-term efficacy; originally believed to be safer than traditional BZDs. | Increases all-cause mortality (HR ~1.60). Significantly increases risk of hip fractures and ischemic stroke in elderly patients 3847. |
| Behavioral | CBT-I (Cognitive Behavioral Therapy for Insomnia) | High long-term efficacy (Hedges' g = 0.98 for insomnia severity) across chronic disease cohorts 50. | Eliminates chemical side effects. Reduces lifetime incidence of cognitive impairment by 2.1% and actively increases life expectancy 51. |
| Behavioral | Structured Exercise & Physical Activity | Moderate-to-high efficacy; optimal at low-frequency, low-intensity for older adults 52. | Modulates endocrine function, mitigates cardiometabolic disease, and improves PSQI scores significantly 5254. |