Environmental drivers of HFRS expansion in Eurasia
Epidemiological Context and Pathogen Virology
Hemorrhagic Fever with Renal Syndrome (HFRS) represents a significant and intensifying public health threat across the Eurasian continent. The shifting epidemiology of this disease is driven by a complex interplay of environmental, climatic, and anthropogenic factors that dictate the distribution and density of its rodent reservoirs 12. HFRS is an acute viral zoonosis characterized by a clinical progression that includes fever, hemorrhage, headache, back pain, and varying degrees of renal failure. The disease typically progresses through five distinct clinical phases: febrile (toxic), hypotensive, oliguric (low urine production), diuretic (high urine production), and convalescent 3. Globally, it is estimated that between 10,000 and 100,000 hantavirus infections occur annually, with the vast majority of the disease burden concentrated in Europe and Asia 45.
The etiological agents of HFRS are members of the Orthohantavirus genus within the Hantaviridae family (order Bunyavirales). These pathogens possess a segmented, negative-sense, single-stranded RNA genome consisting of three segments: the small (S) segment (approximately 1,828 nucleotides) encoding the viral nucleoprotein; the medium (M) segment (approximately 3,650 nucleotides) encoding the viral envelope glycoproteins Gn and Gc; and the large (L) segment (approximately 6,550 nucleotides) encoding the viral RNA-dependent RNA polymerase 678910.
Unlike the New World hantaviruses (such as the Sin Nombre and Andes viruses) that cause Hantavirus Cardiopulmonary Syndrome (HCPS) with case fatality rates approaching 50%, the Old World hantaviruses responsible for HFRS generally exhibit lower mortality rates, ranging from less than 1% to 15% depending on the specific viral strain 3111112. Notably, while the Andes virus in South America has demonstrated limited human-to-human transmission, Eurasian hantaviruses are transmitted almost exclusively through the inhalation of aerosolized rodent excreta, with no documented human-to-human transmission to date 5111413.
The geographic distribution and expansion of HFRS are intrinsically linked to the population dynamics and range shifts of specific rodent reservoir hosts. Each hantavirus species is highly adapted to a primary host species, establishing a persistent, asymptomatic infection wherein the virus is shed continuously in saliva, urine, and feces 6714.
| Virus Species | Primary Rodent Reservoir | Primary Geographic Endemicity | Clinical Presentation & Severity | Estimated Case Fatality Rate |
|---|---|---|---|---|
| Puumala virus (PUUV) | Bank vole (Myodes glareolus) | Northern, Central, and Eastern Europe; Russia | Nephropathia epidemica (mild HFRS) | < 1% 7811 |
| Dobrava-Belgrade virus (DOBV) | Yellow-necked mouse (Apodemus flavicollis) | Balkans, Southern and Central Europe | Severe HFRS | 10% - 12% 614 |
| Hantaan virus (HTNV) | Striped field mouse (Apodemus agrarius) | East Asia (China, South Korea), Far Eastern Russia | Severe HFRS | 1% - 15% 111415 |
| Seoul virus (SEOV) | Brown rat (Rattus norvegicus) | Worldwide (urban and port centers) | Moderate HFRS | ~1% 316 |
In recent years, epidemiological surveillance has recorded notable geographic expansions and intensified baseline transmission of these viruses across Eurasia. Understanding this expansion requires an exhaustive analysis of the environmental drivers modulating host ecology, viral environmental stability, and the human-animal interface.
Reservoir Ecology and Population Dynamics
The geographic expansion of HFRS in Eurasia is fundamentally a reflection of the changing ecology of its rodent hosts, primarily the bank vole (Myodes glareolus) for PUUV and various Apodemus species for DOBV and HTNV 71416. The density and distribution of these rodents directly dictate the environmental viral load and, consequently, the risk of human exposure.
Masting Cycles in Temperate Forests
In the temperate forest ecosystems of Central and Western Europe, bank vole population dynamics are heavily regulated by resource availability, specifically the masting of deciduous trees. Mast seeding is the synchronous and highly variable overproduction of seeds by perennial plants, an evolutionary strategy that profoundly impacts trophic ecosystems and species interactions 17. Studies of broadleaf forests in Germany and neighboring regions indicate that heavy masting of beech (Fagus spp.) and oak (Quercus spp.) provides an overabundance of high-quality winter forage for bank voles 1819.
The environmental transmission cycle of Eurasian hantaviruses is driven directly by these climatic inputs. Changes in temperature and precipitation trigger mast seeding in temperate forests and alter snowpack dynamics in boreal regions. These ecological shifts jointly facilitate rodent population booms and subsequent behavioral adaptations. Following a mast year, winter mortality among bank voles drops significantly, allowing the rodents to commence breeding earlier in the spring with a larger baseline population 20. This flora response leads to explosive vector population growth the following summer and autumn. The density-dependent nature of hantavirus transmission means that these population peaks result in intensified intra-species transmission, rapidly increasing the prevalence of infectious virus within the rodent community and enhancing viral aerosolization in the environment. Ultimately, this cascading chain of events leads to direct human exposure through the inhalation of aerosolized excreta in localized dwellings and forest environments 717182021.
Predictive models analyzing PUUV outbreaks in Germany have demonstrated that the intensity of beech flowering and subsequent masting serves as a powerful leading indicator of HFRS risk. A recent modeling approach using support vector machine classifiers linked fluctuations in human infections to weather and phenology parameters driving host population growth. The model accurately identified high-risk zones, predicting that 41 additional districts in Central and Southern Germany will face a high outbreak risk based on flowering intensity metrics, demonstrating the dominance of extrinsic environmental parameters in forecasting disease expansion 20.
Predator-Driven Cycles in Boreal Regions
In contrast to the resource-driven cycles in temperate zones, bank vole populations in the boreal and subarctic environments of Fennoscandia and northern Russia exhibit multiannual fluctuations primarily driven by predator-prey dynamics, specifically interactions with specialist predators like mustelids 72223. These cycles historically operate on a 3- to 4-year periodicity, progressing through increase, peak, and crash phases.
During the peak phase, which often occurs in late autumn, the prevalence of PUUV within the bank vole population reaches its maximum 723. High population densities not only increase the absolute number of infected animals but also heighten stress and territorial conflicts among voles, leading to increased shedding of the virus. Climate change is currently disrupting these cycles. Warmer, more variable winters can dampen the amplitude of these cyclic crashes, leading to prolonged periods of high baseline rodent density. When bank voles remain relatively widespread in the landscape during what should be cyclic minima, PUUV can spread from a higher baseline, culminating in sustained risk of PUUV transmission across broader geographic expanses 2324.
Vegetation Response and Nutritional Availability
The impact of changing winter conditions extends beyond tree masting to understory vegetation. Experimental simulations of snowpack removal and alteration in high-altitude and alpine environments (such as those observed in the Qinghai-Tibet Plateau) demonstrate that snowpack reduction significantly affects the resource allocation strategies of perennial plants 25. Reductions in snow cover lead to decreased biomass in vegetative organs of plant species (such as Saussurea nigrescens and Anaphalis lactea), altering the availability of forage for ground-dwelling herbivores and omnivores 25.
Furthermore, changes in snowpack influence soil nitrogen and phosphorus contents, which dictate the nutritional quality of the flora 25. For rodent populations, these shifts in understory plant reproductive traits and biomass allocation directly impact carrying capacity. As climate shifts alter local plant productivity, energy flow across trophic levels is modified, frequently favoring adaptable, generalist species like bank voles and field mice that can exploit shifting nutrient availability faster than specialist competitors 26.
Snowpack Modulations and Viral Environmental Persistence
A critical driver of HFRS geographic expansion involves the alteration of seasonal snow cover across mid-latitude and boreal regions. Climate warming is driving reductions in snow depth, extent, and duration 212728. The downstream abiotic and biotic impacts of these reductions profoundly influence both the persistence of hantaviruses in the environment and the behavior of their rodent hosts.
Soil Thermodynamics and Freeze-Thaw Variability
Snow acts as a highly effective thermal insulator. An adequate snowpack decouples soil temperatures from extreme cold air temperatures, maintaining a stable, relatively warm environment at the soil surface 2125. A thinner snowpack, or its complete absence during winter warming anomalies, removes this insulation. Paradoxically, this greater exposure to cold air can result in "colder soils in a warmer world" 21.
This phenomenon significantly increases the amplitude of soil temperature fluctuations and the frequency of freeze-thaw cycles 21. Hantaviruses are lipid-enveloped viruses, and their environmental stability is highly sensitive to external conditions. While the virus can persist for up to two weeks in dried excretions and is generally preserved by stable cold, moist conditions, repeated freeze-thaw cycles degrade the viral lipid envelope, reducing environmental viability 82429.
The complex physics of snowmelt further complicates this dynamic. The release of water from snowpack depends on overcoming a "Runoff Energy Hurdle" (REH), which groups the cold content of the snow and the latent heat required to fill interconnected networks of snow grains with liquid water to residual saturation 30. During warm and low-snow accumulation years, the snowpack is activated numerous times throughout the winter, accelerating solute infiltration rates and dramatically altering soil moisture profiles 2130. Soil microbial dynamics are also highly responsive to these shifts; snow removal experiments demonstrate complex patterns of initial decreases in microbial carbon and nitrogen biomass followed by sharp increases during premature snowmelt periods 21.
Across Western Europe and Scandinavia, predictive models indicate that PUUV risk aligns with colder winters, as stable cold conditions favor environmental viral persistence 24. However, the precise impact of changing soil thermodynamics on viral stability is geographically dependent, highlighting a complex threshold effect where temperature shifts may either facilitate or hinder viral survival outside the host depending on the specific microclimate.
Subnivean Habitat and Human-Rodent Interface
Beyond viral stability, snowpack modulations directly alter rodent behavior. In boreal ecosystems, bank voles rely on the subnivean space - the insulated gap between the soil and the bottom of the snowpack - for winter foraging and survival 72130. When the snowpack is reduced, or when mid-winter rain-on-snow events create impermeable ice layers, the subnivean habitat is degraded or entirely eliminated.
Deprived of their natural winter refuge and facing harsher surface conditions, bank voles and Apodemus mice are driven to seek alternative shelter. This routinely forces them into anthropogenic structures, including rural homes, barns, garages, and forestry cabins 72933. Consequently, milder winters in Northern and Arctic settings coincide with significantly higher HFRS incidence, primarily due to this behavioral shift that forces infected rodents into close proximity with humans, radically increasing the probability of indoor exposure to aerosolized viral particles 24.
Anthropogenic Land Use and Ecotone Expansion
While climatic variables modulate the temporal risk of HFRS, anthropogenic land-use changes act as spatial amplifiers, establishing new zones of endemicity. Deforestation, agricultural expansion, and the sprawl of suburban developments into previously continuous wildlands alter host dynamics and pathogen transmission pathways 21531.
Bank voles and striped field mice thrive in ecotones - the transitional areas between two biomes, such as the edge between a forest and an agricultural field. Human-driven habitat fragmentation maximizes these edge environments, artificially supporting higher carrying capacities for generalist rodent species while simultaneously reducing broader biodiversity 2231. The "dilution effect" hypothesis suggests that robust biodiversity typically suppresses the transmission of zoonotic pathogens by interspersing incompetent hosts among competent reservoirs; conversely, biodiversity loss in fragmented landscapes often favors dominant, highly competent reservoir species 31.
In recent global risk assessments utilizing machine learning to map epidemic-prone diseases, population density emerged as the single strongest driver of outbreak risk, outweighing individual environmental factors 31. Demographic shifts toward peri-urban living and the widespread cultural practice in Eastern Europe and the Russian Federation of maintaining rural garden plots (dachas) significantly increase human occupational and recreational exposure to these edge habitats. During HFRS outbreaks, health surveillance frequently identifies high infection rates among urban residents who contract the virus while engaging in seasonal activities (e.g., harvesting, cleaning sheds, winterizing cabins) in these fragmented rural settings 3532. In the 2025 Russian outbreak, for example, an unusual epidemiological pattern emerged where urban residents accounted for 86% of HFRS patients, despite the disease being traditionally associated with rural occupational exposure, underscoring the risk generated by temporary human migration into rodent-dense ecotones 3532.
Geographic Expansion Vectors in Eurasia
The synthesis of these climatic and anthropogenic pressures is resulting in detectable geographic shifts, evolutionary adaptations, and intensifications of HFRS across multiple Eurasian territories.
Northern Europe and Fennoscandia
Finland currently reports the highest incidence of hantavirus infections globally, with 1,000 to 3,000 cases of nephropathia epidemica recorded annually, driven by robust, cyclical bank vole populations 73733. The 2008 epidemic in Finland remains the largest documented annual European outbreak, with over 3,250 confirmed cases, and recent surveillance through 2026 continues to note moderate seasonal increases aligned with high-circulation periods 3733.
However, the most significant geographic expansion signal in Northern Europe originates from Sweden. Historically, PUUV endemicity in Sweden was sharply demarcated, restricted primarily to the boreal forests north of the Dalälven river, with the southern, more densely populated regions remaining largely free of the virus 34. Recent epidemiological and molecular investigations, including a seminal 2024 study published in Emerging Infectious Diseases, have confirmed the emergence of PUUV causing nephropathia epidemica in bank voles in Scania, in the far south of Sweden 33. Genotyping indicates that these lineages are consistent with Finnish variants, suggesting a definitive southward ecological expansion of the pathogen into new host sub-populations. This movement into the more densely populated regions of central and southern Sweden represents a material escalation in public health risk 34.
Central and Southern Europe
In Central Europe, Germany serves as the primary endemic focus, frequently accounting for the majority of EU/EEA cases alongside Finland 3335. PUUV is predominantly entrenched in the western and southern regions of Germany (e.g., Baden-Württemberg and Bavaria), a distribution strongly correlated with the presence of the Western evolutionary lineage of the bank vole 3336. The dynamics here are distinguished by extreme interannual volatility dictated by the aforementioned beech mast cycles, where incidence rates can spike to over 10 cases per 100,000 inhabitants during favorable years 18. While the virus currently remains largely absent from northern and eastern Germany, public health authorities have expanded monitoring to evaluate for further northeastern expansion 36.
Concurrent with the PUUV threat, the Dobrava-Belgrade virus is extending its range across Central and Southern Europe. DOBV exhibits significant genetic diversity, and its expansion is facilitated by the specific evolutionary adaptations of its sub-lineages.
| DOBV Genotype | Primary Rodent Host | Geographic Distribution | Clinical Severity & Fatality Rate |
|---|---|---|---|
| Dobrava | Yellow-necked mouse (Apodemus flavicollis) | Slovenia, Serbia, Greece, Czechia, Slovakia, Hungary | Moderate-to-severe (10 - 12% CFR) 614 |
| Sochi | Black Sea field mouse (Apodemus ponticus) | Black Sea region, Eastern Russia | Moderate-to-severe (≥6% CFR) 637 |
| Kurkino | Striped field mouse (Apodemus agrarius) | Germany, Slovakia, Russia, Hungary, Slovenia, Estonia | Mild-to-moderate (0.3 - 0.9% CFR) 61638 |
| Saaremaa | Striped field mouse (Apodemus agrarius) | Saaremaa island (Estonia) | Not definitively associated with mortality 6 |
The most common method of hantavirus evolution is through individual nucleotide mutations, but due to their segmented genomes, recombination and reassortment of segments frequently occur 6. The Saaremaa genotype, for instance, is likely a reassortant virus 6. The expansion of Apodemus mice beyond their historical ranges in the Balkans into Central Europe is introducing the more virulent DOBV lineages into overlapping territories with PUUV, creating complex epidemiological landscapes where multiple HFRS pathogens circulate simultaneously 614.
Russian Federation and Asian Dynamics
The Russian Federation bears the highest absolute burden of HFRS globally. Between 2000 and 2022, Russia reported 164,580 HFRS cases, with a massive concentration of these occurring in European Russia 16.
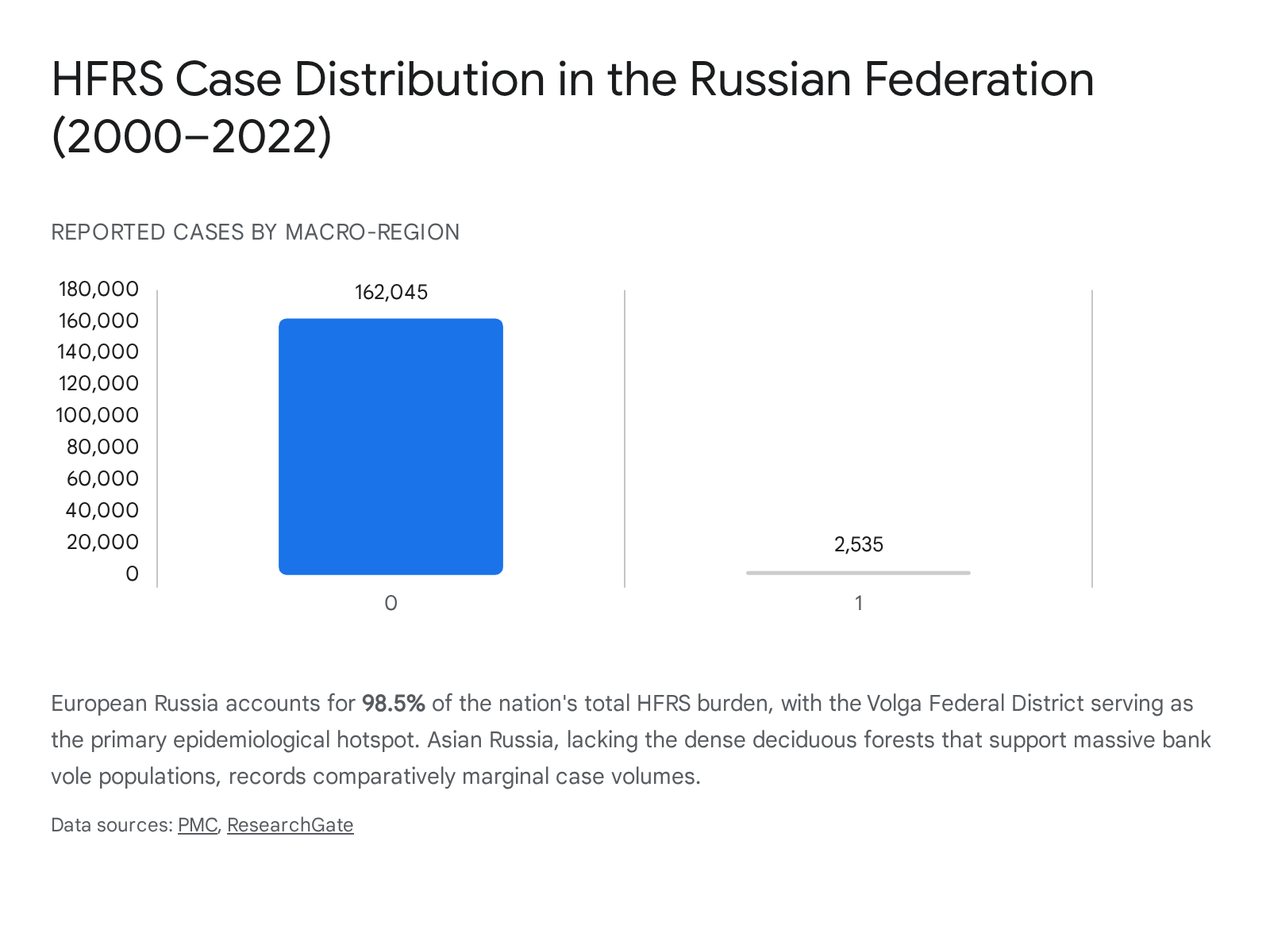
The pathogen landscape is heavily dominated by PUUV (responsible for 97.7% of cases), though HTNV causes severe morbidity in the Far East (Khabarovsk and Vladivostok regions), and DOBV variants (such as the Kurkino and Sochi genotypes) are active in the central and southern oblasts 16353738.
The Volga Federal District serves as the primary endemic hotspot, recording extreme incidence rates. For instance, the Udmurtia and Bashkiria regions recorded average annual incidence rates of 57.6 and 44.5 cases per 100,000 population, respectively 38. The environment here provides optimal conditions for bank voles. Epidemiological forecasting indicates a tense and escalating situation; in 2025, incidence surged by 50% year-over-year in certain regions, reaching over 3,300 cases across 54 regions between January and October alone 3532. This surge aligns with cyclical multi-year peaks driven by favorable environmental conditions 35.
A critical risk signal for geographic expansion within Russia lies in the 15 administrative regions that have historically never reported an HFRS case 35. Given the broad distribution of the bank vole and changing climate parameters that are increasing the habitability of higher latitudes, continuous epizootiological monitoring is required by agencies such as Rospotrebnadzor. Any detection of human cases in these previously naive regions would signal a fundamental shift in the ecology of Eurasian hantaviruses 35. Furthermore, genetic analysis indicates that reassortment events among PUUV lineages (such as the Russian and Finnish sublineages) are occurring frequently in these high-incidence areas, indicating that viral evolution is actively proceeding alongside geographic and environmental shifts 910.
Surveillance Bias Versus Ecological Shift
When assessing the geographic expansion of any zoonotic pathogen, it is necessary to apply calibrated uncertainty: does the increase in reported cases represent a true biological invasion of the virus into new territories, or is it an artifact of enhanced surveillance, superior diagnostic assays, and increased clinical awareness?
Methodological reviews of ecological analysis highlight the risk of "sampling fraction bias," where the intensity of data collection artificially skews the perceived distribution of a phenomenon, leading to significant underestimation or overestimation of true relationships when using aggregate measures from multiple datasets 44. Modern epidemiological infrastructure, governed by the European Centre for Disease Prevention and Control (ECDC) and national health agencies, is detecting milder HFRS infections that would have previously gone undiagnosed 3735. For example, 2023 saw 1,885 reported cases of hantavirus in the EU/EEA, with Finland and Germany accounting for 60.5% of the total dataset - a figure that may reflect robust local reporting practices as much as absolute ecological risk 1133.
Furthermore, applying rigid terms like "invasion" to complex ecological shifts must be done cautiously, as changes in disease distribution are often nuanced responses to broader environmental restructuring rather than unidirectional biological invasions 45. To better understand these complex system shifts, researchers have begun employing advanced machine-learning models to predict abrupt socio-ecological transitions, demonstrating that predictive power can be extracted even from highly complex, seemingly random datasets 39.
Despite these caveats regarding surveillance bias, the consensus drawn from molecular evidence - such as the isolation of PUUV genotypes in southern Sweden and the identification of novel DOBV natural hosts (e.g., Apodemus ponticus carrying the Sochi genotype) - confirms that genuine geographic expansion and host-switching are occurring 333437. The physical footprint of the virus is actively widening, independent of surveillance artifacts.
Conclusion
The geographic expansion of Hemorrhagic Fever with Renal Syndrome across Eurasia is not a localized anomaly but a widespread ecological consequence of a changing environment. It is driven by the synergistic effects of climate change - manifesting as altered snowpack dynamics, variable freeze-thaw cycles, and intensified tree masting events - combined with anthropogenic land-use changes that expand ecotone habitats.
These factors manipulate the population densities, geographic range, and wintering behaviors of reservoir species like the bank vole, yellow-necked mouse, and striped field mouse. As these rodents expand their territories and are forced into closer proximity with human settlements due to environmental stressors such as the loss of subnivean habitats, the interface for aerosolized pathogen spillover grows exponentially. Consequently, regions previously considered low-risk or entirely naive to HFRS, from southern Scandinavia to unexposed districts in the Russian Federation, must adapt their public health monitoring strategies. The integration of meteorological forecasting, forestry data, and robust wildlife pathogen surveillance will be paramount in anticipating and mitigating the future trajectory of Eurasian hantaviruses.