Enteric nervous system and vagus nerve communication with the brain
The gastrointestinal tract and the central nervous system (CNS) engage in a continuous, bidirectional dialogue that regulates physical homeostasis, orchestrates digestive processes, and profoundly influences cognition, interoception, and emotional affect. Historically partitioned into isolated fields of physiology and neurology, modern neurogastroenterology recognizes the anatomical, functional, and neurochemical continuity between the brain and the gut. Driven by the enteric nervous system (ENS) - a vast, autonomous neural network embedded within the gastrointestinal wall - and mediated by the vagus nerve, this integrated system functions as an expansive sensory organ. The emergence of high-resolution transcriptomics, optogenetics, and advanced neuroimaging has dismantled the classical view that gut-brain communication relies exclusively on slow, hormone-mediated endocrine pathways. Instead, recent discoveries highlight rapid, millisecond-scale synaptic transmissions originating directly in the intestinal epithelium.
This report provides an exhaustive analysis of the structural anatomy, cellular mediators, and neurotransmitter dynamics that characterize the gut-brain axis. It delineates the mechanisms of interoception, tracing how subliminal visceral signals ascend the vagus nerve to shape conscious affective states and intuitive decision-making. Furthermore, it examines the modulatory role of the human microbiome, clinical developments in neuromodulation, and the translational limitations of preclinical animal models in contemporary neurogastroenterology research.
Structural Anatomy of the Gut-Brain Axis
The architecture of gut-brain communication is defined by two primary neural components: the intrinsically active enteric nervous system and the extrinsic vagal and spinal pathways that physically tether the gastrointestinal tract to the brainstem and spinal cord.
The Enteric Nervous System Architecture
The enteric nervous system constitutes the largest accumulation of neural tissue outside the central nervous system. Originating from pre-enteric (rhombencephalon) and sacral neural crest cells during embryogenesis, the human ENS contains an estimated 100 million to 600 million neurons, forming a complex, mesh-like web along the entire length of the gastrointestinal tract 123. Due to its sheer magnitude, neurochemical complexity, and unique capacity for autonomous reflex activity, the ENS is frequently designated as the "second brain" 1345.
Structurally, the ENS is organized into two primary ganglionated plexuses: the myenteric (Auerbach's) plexus, which is situated between the longitudinal and circular smooth muscle layers and governs gastrointestinal motility, and the submucosal (Meissner's) plexus, which regulates mucosal secretion, fluid exchange, and local blood flow 1678. The ENS operates with a remarkable degree of autonomy. It contains intrinsic primary afferent neurons (IPANs) that detect mechanical distension and chemical alterations within the lumen, interneurons that process these signals, and secretomotor neurons that execute the appropriate physiological responses 128. While the ENS can direct peristalsis and secretion independently of CNS input - as demonstrated in models where the vagus nerve is surgically severed - optimal functioning and integration with systemic homeostatic demands require continuous, bidirectional central modulation 129.
Extrinsic Pathways and the Vagus Nerve
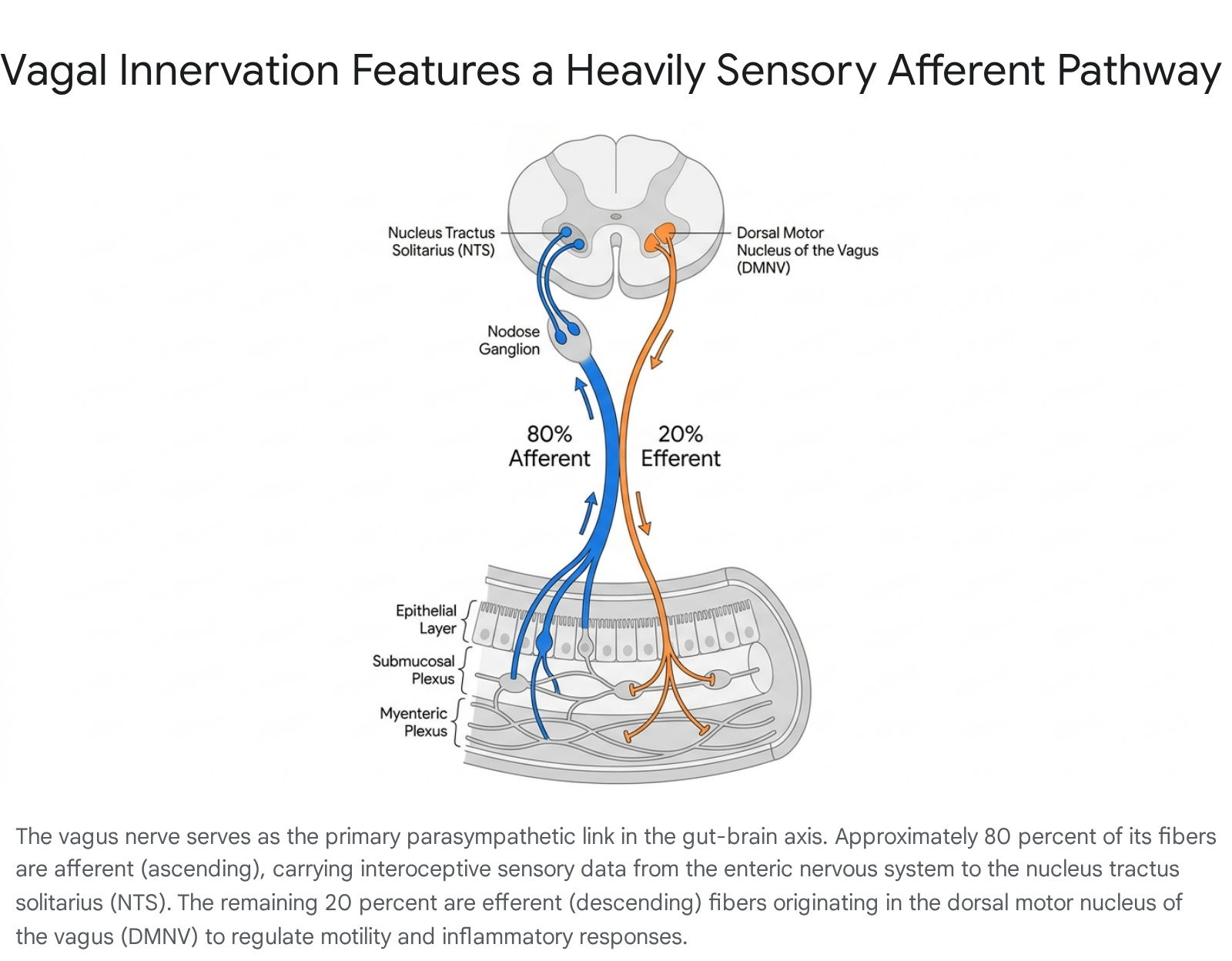
The primary conduit for extrinsic communication between the ENS and the CNS is the vagus nerve (Cranial Nerve X). As the principal component of the parasympathetic nervous system, the vagus nerve innervates major visceral organs, extending from the brainstem down to the transverse colon 101112. The structural composition of the vagus nerve heavily favors ascending sensory transmission over descending motor command.
Quantitative anatomical analyses demonstrate that approximately 80% to 90% of vagal nerve fibers are afferent, dedicated to conveying continuous interoceptive feedback from the viscera to the brain 11121314151617. The remaining 10% to 20% are efferent fibers transmitting parasympathetic motor signals. The right and left branches of the vagus exhibit distinct anatomical and functional asymmetries. The right cervical vagus nerve contains an average of 105,000 fibers and primarily regulates the sinus node of the heart, significantly affecting heart rate. The left vagus nerve, containing roughly 87,000 fibers, plays a more specialized role in the contractile function of the myocardium 1114. Recent micro-CT and immunohistochemistry studies have also identified cross-connecting branches between the sympathetic trunk and the cervical vagus nerve, indicating that sympathetic nerve fibers frequently run within the vagus, complicating traditional paradigms of strictly parasympathetic vagal stimulation 1418.
The cell bodies of the visceral afferent fibers reside in two primary ganglia: the superior (jugular) ganglion, which innervates the auricular and meningeal branches, and the inferior (nodose) ganglion, which houses the afferents for the visceral organs 81113. From the nodose ganglion, sensory signals project primarily into the nucleus tractus solitarius (NTS) and the area postrema in the medullary brainstem, establishing the foundation for vago-vagal reflexes and higher-order cortical processing 410151617. Conversely, the efferent fibers originate predominantly from the dorsal motor nucleus of the vagus (DMNV) and the nucleus ambiguus, descending to regulate gastric acid secretion, control the pace of intestinal transit, and mediate the cholinergic anti-inflammatory pathway 1011121516.
Signal Processing and Fascicular Dynamics
Signal processing within the vagus nerve is not a simple, passive relay mechanism. Because the vast majority - ranging from 85% in the cervical region to nearly 99% in the abdominal region - of vagal fibers are unmyelinated, they exhibit unique biophysical properties during signal ascent 15. Microanatomical evidence indicates that vagal fibers undergo a "split and merge" process, reorganizing themselves between fascicles roughly every 560 micrometers along the nerve trunk 15.
This structural reorganization suggests that signal integration begins well before the impulses reach the brainstem. Furthermore, the dense packing of these unmyelinated fibers facilitates ephaptic coupling - a mechanism of non-synaptic transmission where the electrical field generated by one active axon alters the excitability of its immediate neighbors 15. Within the nerve trunk, ascending sensory fibers and descending motor fibers often intermix within the same fascicles, allowing for local sensorimotor integration. By the time these high-dimensional arrays of mechanical, osmotic, and chemical signals reach the NTS, they have been heavily compressed and summarized, allowing the brain to process broad interoceptive states rather than disparate raw data points 15.
Cellular Mediators of Enteric Sensation
Historically, the scientific consensus posited that the intestinal epithelium communicated with the nervous system almost exclusively through endocrine mechanisms. Enteroendocrine cells (EECs), scattered among the absorptive enterocytes, were understood to sense intraluminal contents and release peptide hormones into the bloodstream or local lamina propria, eventually stimulating vagal nerve endings on a timescale of minutes 8181920. However, advances over the last decade have revolutionized this paradigm, revealing a secondary, instantaneous pathway of neuroepithelial gut-brain communication.
Enteroendocrine and Neuropod Cell Biology
Researchers have identified a specialized subset of enteroendocrine cells characterized by distinct basolateral extensions resembling axons. Coined "neuropod cells" in 2018, these electrically excitable epithelial cells form direct physical synapses with primary afferent nerve fibers, including vagal nodose neurons and spinal dorsal root ganglia 18192122. This unique anatomical configuration establishes a single-synapse neuroepithelial circuit connecting the intestinal lumen directly to the brainstem.
Unlike traditional EECs that rely on the slow diffusion of peptide hormones, neuropod cells straddle the line between endocrine cells and neurons. They possess the presynaptic and postsynaptic machinery necessary to release rapid-acting classical neurotransmitters 192122. Experimental optogenetic and rabies virus-tracing models demonstrate that neuropod cells transmit nutritive and noxious signals to the vagus nerve within milliseconds 192123.
Modality-Specific Neurotransmission
Neuropod cells display a sophisticated capacity for modality-specific neurotransmission, altering their signaling outputs based on the specific intraluminal stimulus detected. When exposed to sugars like sucrose, neuropod cells (enriched with T1R3 taste receptors and SGLT1 glucose transporters) utilize glutamate as an excitatory neurotransmitter to instantly fire action potentials in vagal neurons, driving taste preference and reward 192122. Conversely, when exposed to artificial sweeteners such as sucralose, these cells shift their transmission dynamics, releasing ATP instead 202122.
In contexts of visceral pain and hypersensitivity, neuropod cells utilize distinct receptor profiles. Recent findings demonstrate that guanylyl cyclase C (GUCY2C) is heavily expressed in neuropod cells, where it regulates visceral pain neuromodulation 21. Mice lacking GUCY2C specifically in their neuropod cells are unresponsive to the analgesic effects of the drug linaclotide, underscoring the critical role these cells play in pain transduction. Furthermore, certain enterochromaffin cells exhibiting neuropod-like properties sense chemical irritants and mechanical stretch, releasing synaptic serotonin directly onto dorsal root ganglion sensory neurons to modulate nociception and induce anxiety-like behaviors 2122.
Real-Time Microbial Sensing Mechanisms
The sensory repertoire of neuropod cells extends beyond dietary nutrients to include the direct surveillance of the gut microbiome. Research published in 2025 has elucidated a "neurobiotic sense," demonstrating that the brain can detect microbial activity in the colon in real time without relying on secondary inflammatory or immune cascades 23242526.
This sensory mechanism is mediated by flagellin, an ancient structural protein shed by the flagella of motile intestinal bacteria during periods of active feeding 2324. Neuropod cells located in the colonic epithelium are equipped with Toll-like receptor 5 (TLR5), an innate immune receptor that recognizes this bacterial flagellin 232425. Upon binding flagellin, neuropod cells rapidly transduce the microbial signal into a neural impulse via the vagus nerve, immediately signaling satiety and curbing appetite in the host brain 242526. The identification of this TLR5-mediated pathway provides direct evidence that the microbiome actively shapes host feeding behavior and decision-making on a moment-to-moment basis.
Neurotransmitter Dynamics and Neural Intersections
The physiological similarities between the enteric and central nervous systems are profoundly evident in their shared neurochemical architecture. The ENS utilizes over 30 distinct neurotransmitters, matching the CNS in chemical complexity 12. However, the concentrations, cellular origins, and functional roles of these neurotransmitters differ significantly depending on the system in which they operate. Modulating these neurochemicals within the gut alters local motility and systemic mood, highlighting the therapeutic overlap between neurogastroenterology and psychiatry 927.
Comparative Neurotransmitter Profiles
The table below summarizes the divergent origins, concentrations, and physiological roles of key neurotransmitters across the ENS and CNS.
| Neurotransmitter | Origin & Role in the Enteric Nervous System (ENS) | Origin & Role in the Central Nervous System (CNS) | Systemic & Pathological Implications |
|---|---|---|---|
| Serotonin (5-HT) | ~90% of total body concentration. Synthesized primarily by enterochromaffin cells and enteric neurons. Initiates peristaltic and secretory reflexes (via 5-HT4) and regulates mucosal immunity and inflammation 15792728. | Synthesized in the brainstem (raphe nuclei). Regulates mood, sleep-wake cycles, appetite, and cognitive function. Can be converted to melatonin 7312930. | Imbalances are causally linked to Irritable Bowel Syndrome (IBS), functional dyspepsia, clinical depression, and anxiety disorders 92730. |
| Dopamine (DA) | ~50% of total body concentration. Produced by tyrosine hydroxylase-positive (TH+) myenteric neurons. Acts primarily to inhibit smooth muscle contraction, modulating gastric motility and fluid absorption 1292729. | Synthesized in the substantia nigra and ventral tegmental area. Governs motor control, reward-seeking behavior, motivation, and executive function 312930. | Enteric dopaminergic neuron loss occurs early in Parkinson's disease, leading to chronic constipation that often precedes CNS motor symptoms by decades 293132. |
| Gamma-Aminobutyric Acid (GABA) | Synthesized endogenously by enteric neurons and produced by gut microbiota. Acts as a critical signaling molecule for maintaining intestinal immune homeostasis and relaxing smooth muscle via nitric oxide 72829. | Functions as the primary inhibitory neurotransmitter of the cerebral cortex. Reduces neuronal excitability, promotes relaxation, and limits autonomic stress responses 31293033. | Deficits correlate with central anxiety disorders, insomnia, epilepsy, and heightened visceral hypersensitivity in the gut 3033. |
| Acetylcholine (ACh) | Serves as the primary excitatory neurotransmitter of the ENS parasympathetic pathways. Drives smooth muscle contraction (peristalsis) and stimulates digestive enzyme and mucus secretion 7893129. | Modulates attention, learning, memory consolidation, and neuroplasticity across the cerebral cortex and hippocampus 312933. | Cholinesterase inhibitors are routinely used to treat GI motility disorders (e.g., gastroparesis) and to mitigate cognitive decline in Alzheimer's disease 933. |
Enteric Neurotransmitter Synthesis and Function
Serotonin is the paramount signaling molecule of the gut-brain axis. Approximately 90% of the body's serotonin is synthesized within the gastrointestinal tract, largely by enterochromaffin cells in response to mechanical pressure, chemical stimuli, or microbial metabolites 1928. Within the gut, serotonin acts simultaneously as a classical neurotransmitter, a paracrine factor, and a neuroprotective hormone. By binding to 5-HT3 and 5-HT4 receptors, it triggers intrinsic sensory nerve cells that initiate propulsive motor activity and mucosal secretion 57. Furthermore, mucosal serotonin promotes inflammatory responses necessary to neutralize invading pathogens, while neuronal serotonin protects the ENS from collateral inflammatory damage, acting simultaneously as both the "sword and shield of the bowel" 5.
Dopamine, representing approximately 50% of the body's total supply, operates distinctly in the ENS compared to its role in the CNS 1. While central dopamine is synonymous with reward circuitry and voluntary movement, enteric dopamine primarily modulates motility by antagonizing colonic muscle contractility and regulating electrolyte absorption 227. The vulnerability of these TH-positive dopaminergic enteric neurons is a major focal point of neurodegenerative research. In Parkinson's disease, MPTP toxicity selectively targets dopaminergic neurons in the myenteric ganglia just as it does in the central substantia nigra 232.
Gamma-Aminobutyric Acid (GABA) serves as a potent inhibitory transmitter in both systems. In the ENS, GABAergic neurons function as interneurons that facilitate smooth muscle relaxation through the activation of nitrergic pathways and help preserve mucosal barrier integrity 72834. Uniquely, because its chemical structure requires no metabolic conversion, the ENS can directly utilize dietary GABA derived from fermented foods and germinated grains, making it an actionable target for nutritional psychiatry 728. Acetylcholine, acting as the principal excitatory neurotransmitter of the parasympathetic nervous system, binds to nicotinic and muscarinic receptors on smooth muscle and enteroendocrine cells to forcefully drive peristaltic waves and glandular secretions 789.
Neuroimmune Crosstalk and Macrophage Regulation
The ENS does not operate in isolation; it integrates seamlessly with the mucosal immune system. An intricate crosstalk exists between enteric neurons and muscular macrophages (MMs) residing in the smooth muscle layers. MMs alter the patterns of smooth muscle contractions by secreting bone morphogenetic protein 2 (BMP2) 634. BMP2 activates the bone morphogenetic protein receptor (BMPR) expressed on the surface of enteric neurons 6. In response to this activation, the enteric neurons secrete colony stimulatory factor 1 (CSF1), an essential growth factor required for the survival and development of the MMs 634.
This symbiotic relationship is fundamentally dependent on constant immunological stimulation from the gut microbiome. Lipopolysaccharides (LPS) generated by commensal intestinal bacteria regulate the expression of BMP2 by the macrophages and the reciprocal expression of CSF1 by the enteric neurons 634. Furthermore, during inflammatory states, enteric glial cells interact with neuronal P2X7 receptors, which induces ATP release and activates glial P2Y1 receptors 35. This activation triggers downstream signaling that produces inducible nitric oxide synthase (iNOS) and nitric oxide, driving context-dependent neuroinflammation 35.
The Microbiota-Gut-Brain Axis and Metabolite Signaling
The intersection of neurobiology and microbiology has established the microbiota-gut-brain axis (MGBA) as a central determinant of systemic neural health. The estimated trillions of microbes residing in the human lower gastrointestinal tract do not exist merely as passive commensals; they form a biochemically active ecosystem that directly interfaces with the ENS, the vagus nerve, and the central nervous system 835.
Microbial Endocrinology and Epigenetic Modulation
Gut bacteria actively participate in neurochemical signaling, a field increasingly referred to as "microbial endocrinology" 6. Specific microbial strains synthesize human-identical neurotransmitters, including GABA, norepinephrine, dopamine, and serotonin, which can target local intestinal elements or enter systemic circulation 6827. Beyond direct neurotransmitter synthesis, the microbiome influences host gene expression through epigenetic mechanisms, including the production and regulation of host microRNAs that alter neuroimmune responses 8.
Metabolite Receptor Activation
The metabolic byproducts of microbial activity are potent neuromodulators. The fermentation of dietary fibers produces short-chain fatty acids (SCFAs) such as butyrate, propionate, and acetate 8102835. SCFAs maintain optimal motility by activating enteric G-protein-coupled receptors (GPR41 and GPR43) and can induce the release of peptide YY (PYY) to suppress colonic motor complexes 2835. Furthermore, butyrate protects the intestinal barrier by upregulating the tight junction protein claudin-1 and directly stimulating vagal afferents, thereby bypassing the epithelium to communicate directly with the brainstem 10. Similarly, primary bile acids transformed by microbiota into secondary bile acids bind to TGR5 receptors on enteric neurons and enteroendocrine cells, stimulating the release of GLP-1 to regulate glucose homeostasis, inhibit appetite, and decelerate intestinal peristalsis 835.
Pathological Microbiome Signatures
Dysfunction within the MGBA is increasingly implicated in the pathophysiology of central neurological and psychiatric disorders. Through Mendelian randomization and animal models, researchers have identified disease-specific microbial signatures 36. For example, the elevation of the Desulfovibrionaceae family has been robustly linked to an increased risk of Alzheimer's disease and vascular dementia, independent of traditional risk factors like body mass index 36. Similarly, the attenuation of Butyricimonas is associated with Parkinson's dementia risk 36.
In models of epilepsy, extended administration of antibiotics induces dysbiosis - characterized by increased Akkermansia and Bacteroides abundance - which enhances seizure susceptibility 3640. Fecal microbiota transplantation (FMT) has been shown to reverse this microbial dysbiosis and attenuate pro-epileptogenic effects, likely by restoring serum indole-3-propionic acid levels and suppressing neuroinflammation 36. In psychiatric models, baseline microbial profiles (such as the Firmicutes/Bacteroidetes ratio) modulate responses to antidepressant treatments, and interventions like electroacupuncture have been shown to correct gut dysbiosis, lower brain lipid peroxidation, and alleviate depressive-like behaviors 36.
Interoception and Cortical Representation
The scientific understanding of "gut feelings" extends beyond metaphor; it is firmly rooted in the physiological process of interoception. Interoception is defined as the nervous system's capacity to sense, interpret, and integrate signals originating from within the body to maintain homeostasis and guide complex behavior 1317374238.
Ascending Neural Pathways
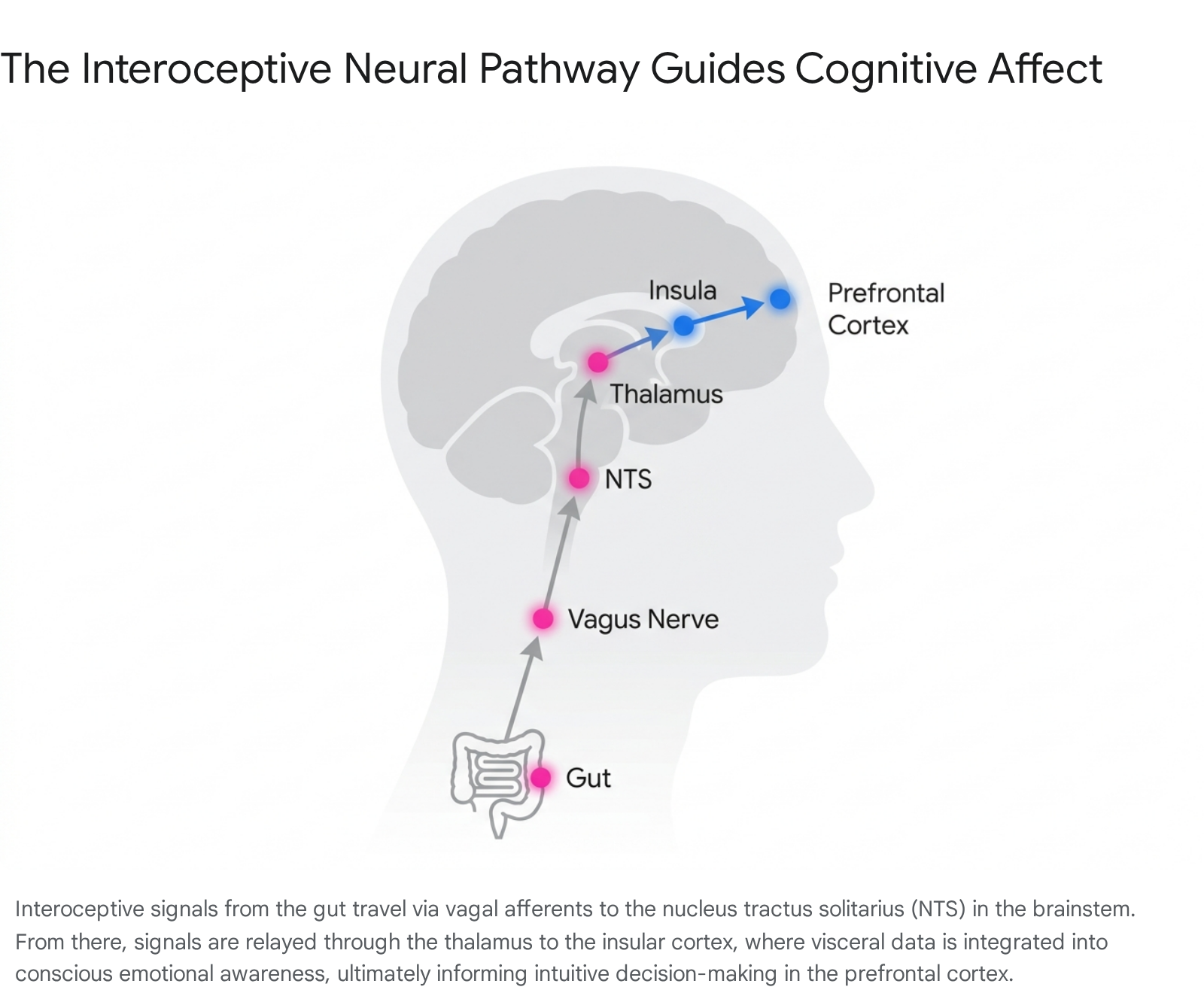
The continuous stream of mechanical, chemical, and metabolic data originating in the gastrointestinal tract rarely reaches conscious awareness. Under homeostatic conditions, these subliminal signals regulate autonomic functions automatically 4842. However, this massive influx of visceral data is continuously routed through the vagus nerve and spinal afferents to the brainstem's NTS 4173940. From the NTS, signals project to the parabrachial nucleus and the area postrema - a chemoreceptive zone lacking a traditional blood-brain barrier - before ascending to the thalamus 1517.
The thalamus acts as a central relay, transmitting these signals to a distributed network of cortical and subcortical structures, including the anterior cingulate cortex (ACC), the prefrontal cortex, the amygdala, and the insular cortex 41739.
Ultra-High Precision Cortical Mapping
The insular cortex, specifically the anterior insula (aINS), serves as the primary interoceptive cortex in humans. Recent ultra-high precision 7-Tesla functional magnetic resonance imaging (fMRI) studies have mapped the functional connectivity of this region in unprecedented detail. This research identifies a large-scale allostatic-interoceptive system in the human brain, characterized by dense intrinsic connectivity between the dorsal mid and posterior insula and key visceromotor cortical regions 46.
These regions include the anterior midcingulate cortex (aMCC), the pregenual and subgenual anterior cingulate cortices (pACC and sgACC), the ventral anterior insula (vaIns), and the dorsal sector of the amygdala 46. In the insula, gustatory, olfactory, and viscerosensory signals from the gut are synthesized with exteroceptive (external) stimuli and top-down cognitive predictions to generate a high-resolution meta-representation of the body's internal state 4817. The presence of von Economo neurons - specialized, fast-conducting spindle cells localized primarily in the fronto-insular and anterior limbic cortices - is believed to facilitate the rapid transmission of these complex interoceptive states to brain regions responsible for emotional arousal and behavioral execution, such as the orbitofrontal cortex (OFC) 4817.
Translation into Conscious Affect and Intuition
The popular conceptualization of "intuition" - knowing something without conscious analytical reasoning - is heavily reliant on this interoceptive network. The brain does not passively receive reality; it continuously generates predictive models of future events 43941. When assessing a complex or uncertain scenario (such as a novel social interaction or a sudden environmental threat), the brain rapidly queries interoceptive memories associated with similar past experiences 441.
If a past scenario was deleterious, the brain initiates a subtle, non-conscious visceromotor response, sending a descending signal back to the gut. The resultant physical sensation - a slight drop in gastric motility, a constriction of blood vessels, or a tension in the abdominal wall - is relayed back to the insular cortex via the vagus 424142. This physical "somatic marker" is interpreted emotionally as discomfort, unease, or a "gut feeling" that warns the individual against a certain decision before the slower, analytical cortices can fully process the data 41. While these intuitive shortcuts are highly efficient evolutionary adaptations, they are contingent upon the accuracy of past associations. They can lead to biased, stereotyped, or generalized decision-making if interoceptive signaling is distorted by chronic stress, visceral pain, or localized gut inflammation 4142.
Clinical Translability and Treatment Paradigms
The expanding understanding of the gut-brain axis has catalyzed the development of novel diagnostic criteria and targeted therapeutics. Clinical gastroenterology is increasingly adopting a multidimensional approach that treats functional gastrointestinal disorders as disorders of gut-brain interaction (DGBI).
Functional Dyspepsia and Regional Guidelines
Functional dyspepsia (FD) exemplifies a disorder characterized by impaired gut-brain communication, typically presenting as chronic epigastric pain, postprandial fullness, and early satiety in the absence of an organic cause 43. The 2025 Asian Consensus Report on Functional Dyspepsia, developed jointly by the Asian Neurogastroenterology and Motility Association (ANMA) and the Asian Pacific Association of Gastroenterology, highlights several recent breakthroughs in the management of this condition.
The consensus recommends prioritizing the eradication of Helicobacter pylori in all confirmed patients, regardless of immediate symptom benefit, to mitigate long-term complications 43. Furthermore, recent epidemiological data indicates a potential link between COVID-19 and the subsequent development or deterioration of FD symptoms, expanding the definition of post-infectious functional disorders 43. In terms of pharmacotherapy, while traditional proton pump inhibitors (PPIs) remain a staple, the emergence of potassium-competitive acid blockers (P-CABs) such as tegoprazan and vonoprazan shows significant promise for patients with refractory GERD and overlapping FD, offering rapid onset and sustained acid suppression 434445. Additionally, emerging evidence suggests that specific probiotic strains (e.g., Bifidobacterium animalis subsp. lactis BL-99 and Bacillus coagulans) may improve symptoms by directly modulating the intestinal flora, although the consensus advises that further large-scale validation is required 43.
Vagus Nerve Stimulation Mechanisms
To address the neurological components of DGBI and central neurodegenerative diseases, neuromodulation techniques are gaining traction. Vagus nerve stimulation (VNS) has long been utilized via implantable devices to treat refractory epilepsy and depression 1846. More recently, non-invasive transcutaneous auricular vagus nerve stimulation (taVNS) has emerged as a promising intervention for both gastrointestinal and psychiatric applications.
Studies from 2024 demonstrate that taVNS directly modulates the functional connectivity (FC) between the insula and the medial prefrontal cortex (MPFC) 40. By weakening pathological negative connectivity between these regions, taVNS enhances the patient's ability to flexibly balance interoceptive awareness and cognitive experiences, offering therapeutic potential for anxiety, insomnia, and functional visceral pain 40. Furthermore, preliminary research published in 2026 suggests that VNS may help protect cognitive function in the early stages of Alzheimer's disease 47. By regulating the firing pace of neurons in the locus coeruleus - a brainstem region that produces norepinephrine and is uniquely susceptible to early tau tangle accumulation - VNS acts as an effective neural regulator, establishing optimal activity levels and potentially slowing neurodegenerative decline 47.
Limitations of Preclinical Models in Neurogastroenterology
While the foundational principles of gut-brain communication have been established through robust in vivo animal studies, the clinical translation of novel neurogastroenterological therapeutics suffers from remarkably high attrition rates. Over 90% of central nervous system and gastrointestinal drug candidates that demonstrate efficacy and safety in rodent models ultimately fail in human clinical trials 484950. This translational gap is driven by profound anatomical, molecular, and environmental divergences between species.
Interspecies Structural and Cellular Divergence
The human enteric nervous system is vastly more complex than its murine counterpart. While an adult mouse possesses roughly 2.5 million enteric neurons, the human ENS contains approximately 168 million 51. Furthermore, the cellular composition supporting these networks differs dramatically. Humans exhibit an enteric glia-to-neuron ratio of 7:1, compared to a 1:1 ratio observed in mice 51. Given that enteric glia play a critical role in neuroinflammation, barrier maintenance, and synaptic regulation, this severe structural discrepancy limits the predictive validity of murine models in studying human enteric neuropathies and visceral hypersensitivity 51.
Animal models of neurodegenerative diseases also fall short of establishing construct validity. For example, most Alzheimer's disease models rely on transgenic mice that overexpress mutant forms of human proteins, which fails to recapitulate the complex, multi-factorial pathological features and cognitive deficits seen in naturally occurring human cases 49. Similarly, the symptoms of Parkinson's disease, such as severe constipation, often present distinct functional differences in human myenteric and submucous plexuses that are not accurately mirrored in neurotoxin-induced rodent models 51.
Receptor Discrepancies and Translational Attrition
Molecularly, the distribution and function of specific neuroreceptors diverge across species, significantly impacting pharmacodynamics. For example, the mouse ENS is exquisitely sensitive to histamine, where H3 receptors exert a pronounced influence on transmitter release and directly alter smooth muscle tone and gut motor function 51. In humans, histamine's role is largely modulatory, acting indirectly on the system 51. Similar species-specific discrepancies have been identified in purinergic (P2X) receptor subtypes, complicating the pharmacological targeting of functional dyspepsia and irritable bowel syndrome based solely on rodent data 951. These differences in drug metabolism and toxicity can be catastrophic; an infamous example is the therapeutic antibody TGN1412, which was safe in animal models but caused a near-fatal immune response in human clinical trials 4952.
Alternative Methodologies
In addition to genetic and structural variations, traditional laboratory models fail to capture the complex environmental variables that drive human disorders. Laboratory mice are housed in highly controlled, pathogen-free environments, fed uniform diets, and subjected to artificial stressors (e.g., water-avoidance stress) that do not adequately mirror the chronic psychosocial stress, diverse dietary inputs, and heterogeneous microbiome profiles characteristic of human patient populations 525354.
Consequently, researchers and regulatory bodies - prompted by legislation such as the FDA Modernization Act 3.0 - are increasingly advocating for the integration of New Approach Methodologies (NAMs) 48. Advanced frameworks, such as human-derived intestinal organoids, "first in man" patch-clamp recordings on isolated human myenteric ganglia, and in silico computational modeling, are required to bridge the translational divide 48495051. By utilizing non-invasive techniques like ingestible vibrating mechanosensory capsules, future research can more accurately decode how the human brain processes gut signals, paving the way for targeted, human-relevant therapeutics in neurogastroenterology 374651.