Endothelial glycocalyx in vascular aging
Introduction to the Endothelial Glycocalyx
The vascular endothelium constitutes a dynamic, single-cell layer lining the entire expanse of the circulatory system. Spanning an estimated surface area equivalent to six tennis courts, this continuous cellular interface represents the human body's largest organ, serving a critical role in regulating systemic blood pressure, localized immunity, and the coagulation cascade 12. Coating the luminal surface of these endothelial cells is the endothelial glycocalyx, an extensively hydrated, gel-like macromolecular meshwork composed predominantly of proteoglycan polymers, glycoproteins, and glycosaminoglycans 3456. Ranging in physical thickness from 0.2 to 5.0 micrometers - depending heavily on the specific vascular bed, local hemodynamics, and species - this carbohydrate-rich matrix acts as the primary vanguard between the flowing blood and the delicate vascular wall 678.
Historically, scientific consensus characterized the endothelial glycocalyx merely as a passive, inert structural barrier. However, contemporary cardiovascular research has redefined the glycocalyx as an active, mechanosensory organelle that is absolutely critical for the maintenance of vascular homeostasis. The structure is characterized by dynamic equilibrium, constantly remodeling itself to balance the shear-stress-induced shedding of its surface components with the robust de novo synthesis of its structural matrix by the underlying endothelial cells 69.
The physiological functionality of the endothelial glycocalyx is entirely dependent upon its structural integrity. Functionally, it operates as a selective permeability barrier, rigorously limiting the extravasation of plasma macromolecules, such as albumin, and fluids into the interstitial space. Consequently, the glycocalyx is now recognized as the definitive structural determinant of the oncotic gradient across the microvasculature, necessitating a fundamental revision of the traditional Starling principle of fluid dynamics 101112. Furthermore, the glycocalyx serves as a highly sensitive mechanotransducer. It detects the biomechanical forces exerted by fluid shear stress from flowing blood and transmits these physical signals into the endothelial actin cytoskeleton. This mechanotransduction prompts the activation of endothelial nitric oxide synthase (eNOS) and facilitates the subsequent localized release of nitric oxide (NO), a potent molecular mediator of vasodilation 3613. Additionally, the dense, negatively charged steric matrix of the intact glycocalyx physically masks critical endothelial adhesion molecules under resting, homeostatic conditions. By physically obstructing access to intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and various selectins, the glycocalyx actively prevents the untoward adhesion and transmigration of circulating leukocytes and platelets 21415.
With advancing chronological age and the progressive onset of metabolic disease, the delicate homeostatic equilibrium of the glycocalyx is persistently disrupted. Research indicates that the structural deterioration of the glycocalyx significantly precedes traditional clinical manifestations of vascular aging, including large-artery stiffness, hypertension, and impaired flow-mediated dilation. This positions continuous glycocalyx degradation as a primary, foundational instigator of age-related cardiovascular and microvascular pathologies, shifting the endothelium from a vasoprotective state to one that promotes leukocyte adhesion, thrombosis, and adverse vascular remodeling 31617.
Structural Composition and Molecular Scaffolding
To comprehend the mechanisms underlying glycocalyx degradation, it is necessary to first delineate its highly specific, non-random biochemical architecture. The matrix is assembled from distinct classes of biological macromolecules that self-organize into a three-dimensional, brush-like surface layer.
Glycosaminoglycans
Glycosaminoglycans represent the most abundant molecular constituent of the endothelial glycocalyx by volume. These linear, unbranched carbohydrate polymers consist of repeating disaccharide units that are extensively modified by highly specific, enzymatically driven sulfation patterns. The three major glycosaminoglycans resident in the glycocalyx are heparan sulfate, chondroitin sulfate, and hyaluronan 31819.
Heparan sulfate comprises the vast majority of the functional glycosaminoglycan pool within the endothelial glycocalyx and plays the dominant role in binding circulating plasma proteins, growth factors, and regulatory chemokines 1220. The biosynthesis of the heparan sulfate chain initiates with the generation of a core tetrasaccharide linkage region (GlcAβ1-3Galβ1-3Galβ1-4Xylβ1-Ser) attached to a serine residue of a core protein. This is followed by the stepwise addition and subsequent modification of repeating disaccharide units 1920. Its high degree of sulfation provides the intense negative electrostatic charge required to electrostatically repel circulating erythrocytes and selectively bind antithrombin III, thereby conferring inherent anticoagulant properties to the healthy endothelium 1219.
Hyaluronan, or hyaluronic acid, contrasts with heparan sulfate in that it is an entirely unsulfated glycosaminoglycan. Instead of being synthesized in the Golgi apparatus and attached to a core protein, hyaluronan is synthesized continuously at the inner surface of the plasma membrane by specific hyaluronan synthases - primarily the HAS2 isoform in the vascular endothelium - and extruded directly into the extracellular space 17. High-molecular-weight hyaluronan intertwines heavily with the surrounding proteoglycan network, acting as a primary structural stabilizer. Because of its massive capacity to bind water molecules, hyaluronan contributes significantly to the hydration, volume, and gel-like biomechanical properties of the overarching glycocalyx barrier. The hyaluronan matrix also associates non-covalently with specific membrane receptors, most notably CD44, to further anchor the layer 1721.
Proteoglycans and Glycoproteins
Proteoglycans are complex macromolecules consisting of a designated core protein to which one or more glycosaminoglycan chains are covalently attached 19. The syndecan family, comprising syndecans 1 through 4, consists of single-pass transmembrane proteins that firmly anchor the glycocalyx matrix to the internal endothelial actin cytoskeleton, facilitating direct inside-out and outside-in mechanotransduction signaling 102021. Syndecan-1 is universally recognized as the primary structural pillar of the glycocalyx and serves as the most extensively studied biomarker of glycocalyx injury 141822. Glypicans, particularly glypican-1, represent another major proteoglycan family in this microenvironment; unlike syndecans, glypicans lack a transmembrane domain and are instead anchored to the outer leaflet of the plasma membrane via glycosylphosphatidylinositol linkages 621.
The glycoprotein fraction of the glycocalyx consists predominantly of endothelial cell adhesion molecules - including selectins, integrins, and members of the immunoglobulin superfamily - as well as specialized receptors involved in the coagulation and fibrinolysis pathways. While these glycoproteins are physically shorter than the overarching proteoglycan matrix and are normally coated and shielded by it, they play a critical pathophysiological role when the protective glycosaminoglycan shielding is enzymatically stripped away during acute inflammatory states, facilitating robust immune cell adhesion and tissue infiltration 1518.
| Component Category | Primary Molecules | Structural Role and Functional Significance |
|---|---|---|
| Proteoglycans | Syndecan-1, Syndecan-4, Glypican-1 | Transmembrane and GPI-anchored core proteins that firmly attach the extracellular matrix to the endothelial cell cytoskeleton. Critical for active mechanotransduction and robust structural anchoring 6102021. |
| Sulfated Glycosaminoglycans | Heparan Sulfate, Chondroitin Sulfate | Highly negatively charged, linear polymers that bind antithrombin III, electrostatically repel red blood cells, and sequester circulating growth factors and chemokines. Crucial for the maintenance of the permeability barrier 121920. |
| Unsulfated Glycosaminoglycans | Hyaluronan (Hyaluronic Acid) | Synthesized directly at the plasma membrane by HAS2. Provides massive hydration capacity, overall volume, and structural stability to the complex glycocalyx meshwork. Associates with CD44 receptors 1721. |
| Glycoproteins | ICAM-1, VCAM-1, E-selectin | Shorter surface adhesion molecules normally shielded by the intact, hydrated glycocalyx. Exposed during pathological glycocalyx shedding to facilitate leukocyte tethering and diapedesis 215. |
Mechanisms of Glycocalyx Degradation and Shedding
The degradation of the endothelial glycocalyx, commonly referred to in the literature as "shedding," is not a random process of physical erosion or simple mechanical weathering. It is a highly regulated, stepwise biochemical cascade. Under standard physiological conditions, the processes of localized shedding and biosynthesis occur at equivalent rates to maintain a stable, homeostatic layer 56. However, under pathological stressors - such as ischemia/reperfusion injury, profound systemic sepsis, acute trauma, and advanced vascular aging - the enzymatic shedding process is markedly accelerated, rapidly overwhelming the endothelial capacity for regeneration and leading to a denuded vascular wall 6121824.
The Stepwise Enzymatic Cascade
The pathological degradation of the glycocalyx is primarily orchestrated by three classes of sheddase enzymes: heparanase, matrix metalloproteinases, and hyaluronidases. The activation of these enzymes is typically triggered by an influx of reactive oxygen species and pro-inflammatory cytokines, most notably tumor necrosis factor-alpha, interleukin-1beta, and interleukin-6 21223.
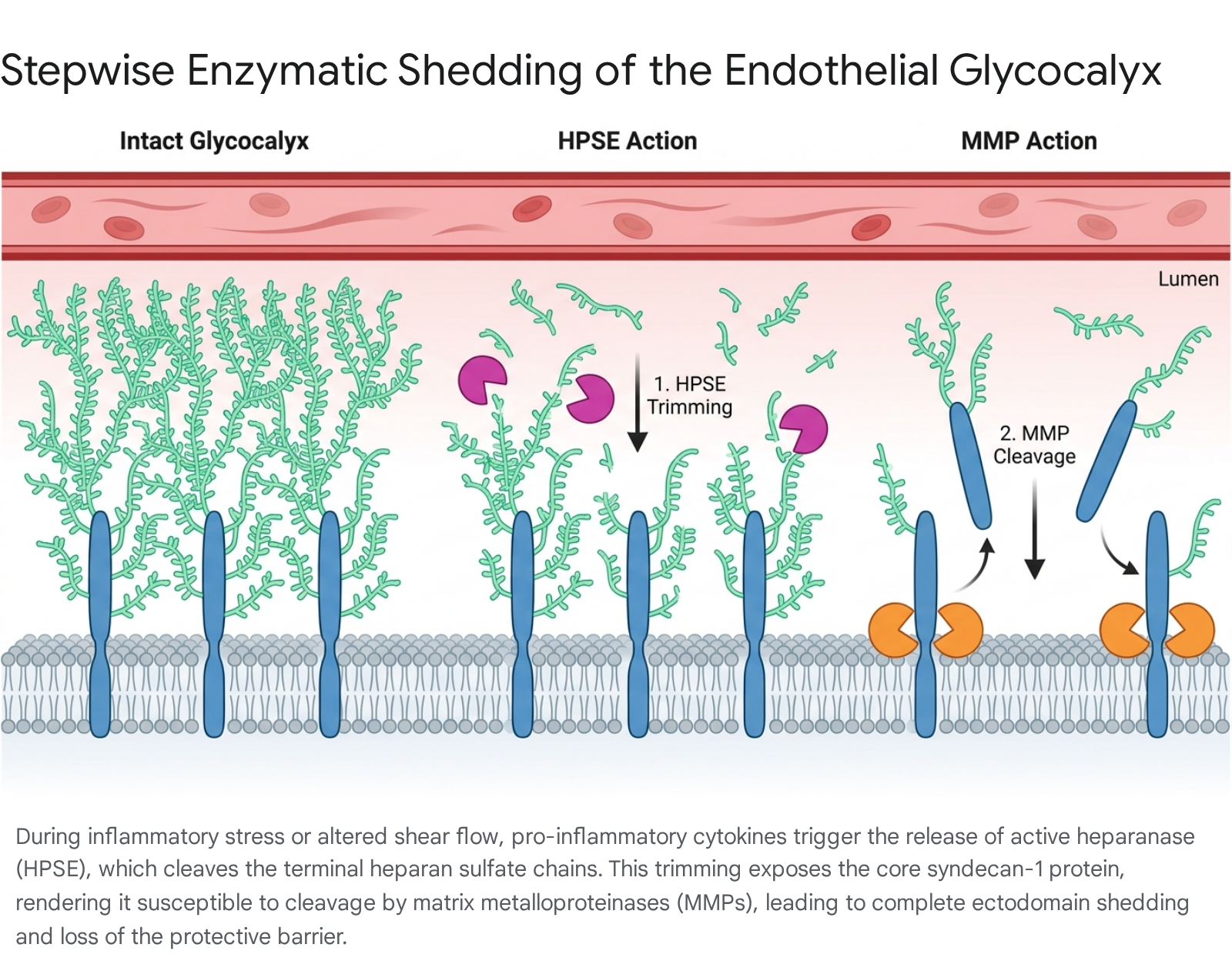
The shedding of syndecan-1, the primary structural biomarker of glycocalyx injury, occurs via a precise and sequential molecular mechanism: First, latent heparanase is released by innate immune cells, platelets, and the endothelial cells themselves during the response to infection or physical injury. This pro-enzyme is subsequently internalized, processed in lysosomes into its enzymatically active form, and secreted back into the extracellular space 142024. Once active, heparanase selectively cleaves the bulky heparan sulfate glycosaminoglycan chains from the outer limits of the glycocalyx matrix. This initial trimming substantially reduces both the physical thickness and the dense negative charge of the barrier 1421.
The removal of the heparan sulfate chains unmasks specific, previously hidden proteolytic cleavage sites on the core syndecan-1 protein. This exposure renders the core proteoglycan highly susceptible to enzymatic degradation by matrix metalloproteinases, particularly the zinc-containing endopeptidases MMP-2, MMP-7, and MMP-9, as well as the disintegrin and metalloproteinase ADAM17 510142021. Additionally, circulating proteases such as thrombin participate in cleaving the syndecan-3 and syndecan-4 ectodomains directly 10. Finally, the matrix metalloproteinases cleave the syndecan-1 ectodomain close to the plasma membrane. The shed syndecan-1 fragments, alongside the free heparan sulfate chains, are released completely into the systemic circulation, where they function as detectable, quantitative biomarkers of severe endothelial injury 142021.
Flow Dynamics and Endothelial Autophagy
Beyond chemical inflammatory triggers, the physical hemodynamics of local blood flow strictly dictate glycocalyx health. Physiological, unidirectional shear stress exerted by healthy laminar blood flow promotes the active biosynthesis of glycocalyx components, ensuring that the layer is maintained and can rapidly adapt to flow changes 3612. Conversely, pathological low shear stress or oscillatory flow patterns - commonly found at arterial bifurcations where atherosclerotic plaques preferentially develop - aggressively disrupt this synthetic balance 1217.
Recent research models indicate that low shear stress directly induces endothelial autophagy. Experimental models utilizing human umbilical vein endothelial cells subjected to low shear stress demonstrate an upregulation of autophagic markers, specifically increased expression of LC3II/LC3I ratios and Beclin-1, alongside decreased levels of p62 23. This flow-induced autophagic state directly increases the expression of MMP-2 and MMP-9, leading to the accelerated degradation of syndecan-1 and heparan sulfate 23. Consequently, the localized loss of mechanotransduction via the degraded glycocalyx results in a rapid decline in eNOS activity, the uncoupling of nitric oxide production, and the transition of the endothelium into a pro-inflammatory, pro-thrombotic state characterized by uncontrolled vasoconstriction 3624. The structural deterioration of the glycocalyx thus operates both as a consequence of poor flow dynamics and a primary driver of subsequent vascular pathology.
Biochemical Distinction: Glycocalyx Proteoglycans versus Advanced Glycation End-Products
In broader scientific and popular literature, both the endothelial glycocalyx and the pathogenic molecules associated with diabetes are frequently discussed using colloquial sugar-related terminology. The glycocalyx is often referred to metaphorically as the "sugar coating" of the blood vessels, given its dense polysaccharide composition 25. However, this linguistic overlap frequently creates semantic and conceptual confusion, improperly conflating the highly protective glycocalyx with Advanced Glycation End-Products (AGEs). The two represent entirely opposing biochemical entities, origins, and functional outcomes.
The Functional Polysaccharide Matrix
The endothelial glycocalyx is a functional, genetically encoded, and enzymatically synthesized structural organelle. Its glycosaminoglycans, or sugar chains, are deliberately and precisely assembled in the endoplasmic reticulum and Golgi apparatus by specific transferase enzymes, such as galactosyltransferases and glucuronosyltransferases 1925. These polysaccharides are structurally integrated to serve highly specific physiological roles, including fluid barrier maintenance, mechanotransduction, and the sequestration of vital growth factors. The "sugar" in this anatomical context refers to functional, complex carbohydrates that strictly preserve vascular health and integrity 319.
The Pathology of Advanced Glycation End-Products
In stark biochemical contrast, Advanced Glycation End-Products are a heterogeneous group of toxic, irreversible adducts that form spontaneously and completely independently of enzymatic regulation 2627. When reducing sugars, such as circulating glucose, are present in abnormally high concentrations over prolonged periods - as universally observed in chronic hyperglycemia and unmanaged diabetes mellitus - they spontaneously react with the free amino groups of circulating and tissue proteins, particularly the lysine and arginine residues abundant on collagen and elastin matrices 2728.
This spontaneous, non-enzymatic process, formally known as the Maillard reaction, begins with the formation of unstable Schiff bases and Amadori products. Under states of oxidative stress, these unstable intermediates undergo irreversible glycoxidation. This reaction creates complex, permanent, cross-linked structures such as carboxymethyllysine, carboxyethyllysine, pentosidine, and glucosepane 2729.
Rather than acting as a protective physiological barrier, advanced glycation end-products directly assault the cardiovascular system via two primary mechanisms. First, they form rigid, pathological crosslinks within extracellular matrix proteins, destroying the flexibility of collagen and elastin, which leads to severe arterial stiffness and reduced tissue compliance 262728. Second, advanced glycation end-products act as specific, high-affinity ligands for a cell surface signal transduction receptor known as the Receptor for Advanced Glycation End-products (RAGE) 262830.
The activation of RAGE by circulating advanced glycation end-products triggers a devastating intracellular signaling cascade. It activates NADPH oxidase and specific kinases, profoundly elevating reactive oxygen species levels, and stimulates the master inflammatory transcription factor NF-κB 2628. This activation results in the massive localized release of pro-inflammatory cytokines, including tumor necrosis factor-alpha and interleukin-6 2830. Paradoxically, these are the exact cytokines that activate the matrix metalloproteinases and heparanase enzymes responsible for degrading the protective endothelial glycocalyx 242830. In summary, the non-enzymatic, pathogenic "sugars" (AGEs) systematically destroy the protective, functional "sugar coating" (the glycocalyx).
| Feature | Endothelial Glycocalyx | Advanced Glycation End-Products |
|---|---|---|
| Biochemical Origin | Enzymatically synthesized in the ER and Golgi apparatus by highly specific transferase enzymes 19. | Non-enzymatically generated via the Maillard reaction from circulating reducing sugars under oxidative stress 2627. |
| Molecular Nature | Highly organized network of proteoglycans, glycoproteins, and glycosaminoglycans 3. | Irreversible, heterogeneous glycoxidized adducts and structural crosslinks (e.g., carboxymethyllysine, pentosidine) on host proteins 27. |
| Physiological Role | Vasoprotective. Actively regulates vascular permeability, transduces shear stress, and repels immune cells 36. | Pathogenic. Induces severe arterial stiffness, intracellular oxidative stress, and systemic inflammation 262730. |
| Interaction Dynamics | Serves as the primary physical and electrostatic barrier protecting the underlying endothelial cell layer 2531. | Binds to RAGE receptors on endothelial cells, triggering NF-κB, inhibiting eNOS, and actively driving glycocalyx shedding 3032. |
The Glycocalyx in Vascular Aging and Chronic Pathology
Chronological aging remains the dominant, independent prognostic risk factor for the development of virtually all cardiovascular diseases. Extensive molecular evidence indicates that endothelial dysfunction - driven primarily by the progressive, uncompensated degradation of the glycocalyx - is the initiating mechanism underlying age-related vascular decline 1333.
The Senescent Endothelium and Inflammaging
During the biological aging process, endothelial cells undergo profound phenotypic shifts. This transition is characterized by irreversible cell cycle arrest, telomere shortening, increased expression of beta-galactosidase activity, and widespread mitochondrial dysfunction 34. This cellular senescence directly contributes to the progressive decline in nitric oxide bioavailability and widespread vascular stiffness. Furthermore, senescent endothelial cells adopt what is known as a senescence-associated secretory phenotype. Rather than remaining quiescent, these aged cells release a continuous, unregulated stream of low-grade inflammatory cytokines - a systemic process frequently termed "inflammaging" 16.
This chronic state of low-grade inflammation shifts the carefully maintained balance of glycocalyx homeostasis heavily toward continuous degradation. The perpetual activation of NF-κB and the subsequent persistent production of tumor necrosis factor-alpha maintain a steady state of activation for heparanase and matrix metalloproteinases. This ensures that the glycocalyx remains chronically thinned and degraded in older populations 16. Without this protective gel-like layer, aging blood vessels fundamentally lose their ability to properly sense flow-mediated shear stress, impairing necessary vasodilation. This leaves the bare, unprotected endothelial cells highly vulnerable to lipid deposition and leukocyte diapedesis, initiating the early stages of atherosclerosis 1316.
Genetic Polymorphisms Influencing Vascular Aging
The inherent susceptibility of the endothelial glycocalyx to age-related degradation is not uniform across populations; it is significantly influenced by genetic variations, specifically single-nucleotide polymorphisms, within the genes encoding key synthetic and degradative enzymes.
The hyaluronan synthase 2 gene plays a pivotal role in maintaining the structural integrity of the glycocalyx. It is responsible for synthesizing high-molecular-weight hyaluronan. Transcriptomic studies of human venous tissues and aging arterial models demonstrate that hyaluronan synthase 2 expression is significantly downregulated with advancing age 335. The inherited or age-related deficiency in this specific synthase results in a profound lack of structural hyaluronan, fatally compromising glycocalyx volume and precipitating age-related vascular hyperpermeability 31735.
Polymorphisms in the endothelial nitric oxide synthase gene also severely impact the endothelium's ability to maintain vascular tone in the presence of a degraded glycocalyx. The T786C polymorphism, specifically located in the promoter region of the gene, fundamentally reduces transcription initiation, causing an approximately 50 percent reduction in overall promoter activity 3436. Clinical cohort analyses reveal that individuals homozygous for the TT genotype exhibit altered minimum serum nitric oxide levels, directly linking genetic nitric oxide deficiency to a higher susceptibility to coronary artery disease and accelerated vascular aging 36.
Furthermore, epigenetic modifications dictate the aggressiveness of the enzymatic shedding cascade. Hypomethylation of the heparanase promoter, coupled with the corresponding upregulation by the transcription factor EGR1, leads to excessive, unchecked heparanase expression. This hyperactive state contributes heavily to the rapid, severe degradation of the glycocalyx observed in acute inflammatory events and chronic age-related fibrosis 2437.
Diagnostic Modalities: Sublingual Videomicroscopy and Biomarkers
The historical limitation of glycocalyx research was the extreme difficulty of measuring this fragile, highly hydrated structure in vivo. Routine histological tissue processing dehydrates and physically collapses the glycocalyx, rendering standard tissue biopsies and traditional electron microscopy highly unreliable for precise volumetric measurement 5. The advent of handheld incident dark-field and sidestream dark-field videomicroscopy has revolutionized clinical assessment, allowing for the rapid, non-invasive visualization of the sublingual microcirculation at the bedside 33839.
Videomicroscopy and the Perfused Boundary Region
The most prominent commercial analytical system for sublingual glycocalyx assessment is GlycoCheck. This software utilizes a handheld camera to directly image the sublingual capillary bed, focusing on vessels between 4 and 25 micrometers in diameter. Because the glycocalyx matrix itself is virtually transparent to standard light microscopy, the system relies on tracking the physical behavior of red blood cells to infer glycocalyx health 3140.
An intact, healthy, thick glycocalyx restricts red blood cells strictly to the central lumen of the microvessel. When the glycocalyx is degraded, the barrier is compromised, and red blood cells penetrate laterally, moving deeper toward the endothelial cell surface 4041. The software automatically detects this dynamic lateral movement and calculates a metric known as the Perfused Boundary Region, expressed in micrometers. A higher Perfused Boundary Region clearly indicates deeper red blood cell penetration, serving as an inverse marker of glycocalyx thickness (i.e., a higher Perfused Boundary Region equates to a thinner, damaged glycocalyx) 384042. Alongside the boundary region, these advanced imaging systems continuously measure the Microvascular Flow Index, Total Vessel Density, and the Proportion of Perfused Vessels 43.
Reproducibility and Rigorous Examination Conditions
While sublingual videomicroscopy provides an unprecedented window into systemic microvascular health, extensive debate persists regarding its clinical reproducibility and technical limitations.
Clinical reproducibility studies of the GlycoCheck system exhibit mixed outcomes that depend heavily on the rigorousness of the specific data collection protocol. In a robust, controlled study evaluating healthy volunteers, the intra-class correlation for a single, isolated measurement of the Perfused Boundary Region was shown to be relatively modest, calculated at an intra-class correlation of 0.30 44. However, the analytical reproducibility improves significantly when multiple measurements are aggregated; analyzing the pooled means of five consecutive sublingual measurements yields a much more reliable intra-class correlation of 0.58 44.
Furthermore, the sublingual microcirculation is extraordinarily sensitive to acute environmental and dietary variables. Studies demonstrate that smoking a single cigarette, consuming a high-calorie meal, or drinking a cup of coffee significantly alters both the Perfused Boundary Region and microvascular perfusion rates. These acute physiological effects peak immediately following consumption and persist with statistical significance for up to 180 minutes 3145. Consequently, strict standardization of clinical protocols - specifically enforcing a minimum 180-minute abstinence from these routine triggers - is absolutely mandatory for accurate, reliable data collection 3145.
The Clinical Utility Gap: Predictive Value versus Diagnostic Specificity
The clinical application of sublingual videomicroscopy and circulating glycocalyx biomarkers currently presents a distinct paradox: these tools possess exceptional prognostic value in acute critical illness but demonstrably lack diagnostic specificity for chronic, stable cardiovascular disease.
In the highly acute settings of the emergency department and the intensive care unit, glycocalyx shedding functions as a highly reliable predictor of patient mortality and morbidity. Systematic reviews and exhaustive meta-analyses confirm that circulating plasma levels of syndecan-1 and heparan sulfate routinely increase 3- to 4-fold during severe sepsis and major trauma 846. Correspondingly, sublingual microcirculation assessments reveal that patients presenting with an elevated Perfused Boundary Region, significantly reduced capillary density, and a low Proportion of Perfused Vessels at initial admission are at a substantially higher risk for progressing to multiple organ dysfunction syndrome, sepsis-associated encephalopathy, and ultimately death 49474849.
For example, the prospective NOSTRADAMUS study evaluating pediatric sepsis identified three distinct microvascular phenotypes using factorial analysis: Preserved, Partially Compensated, and Decompensated 4950. Patients falling into the Decompensated microvascular phenotype exhibited profound global microvascular failure and universal mechanical ventilation requirements 50. Similarly, the EDGE study tracking 299 emergency department patients with suspected sepsis demonstrated that combined glycocalyx damage and reduced capillary density more than doubled the odds of in-hospital mortality 48. The persistence of these microvascular derangements after 24 to 72 hours reliably identifies patients who are failing to respond to standard fluid and vasopressor resuscitation, granting the technology significant real-time prognostic power 4950.
Conversely, the utility of sublingual videomicroscopy for screening primary cardiovascular risk in stable, chronic outpatients remains highly debated and largely unsubstantiated. While observational studies demonstrate that individuals with established coronary artery disease exhibit lower microvascular density and higher boundary regions compared to perfectly healthy cohorts, the absolute numerical values show massive, confounding overlap between the two populations 38. After statistically adjusting for standard demographics and established cardiovascular risk factors such as hypertension, hyperlipidemia, and diabetes, the isolated presence of obstructive coronary artery disease is frequently not independently associated with sublingual microvascular parameters 3843. Because glycocalyx shedding is a ubiquitous, generalized physiological response to almost all forms of acute and chronic systemic inflammation - lacking any sensitivity or specificity for a single distinct etiology - sublingual imaging currently struggles to meaningfully reclassify cardiovascular risk beyond the capabilities of traditional, established scoring systems like the Framingham or European Heart SCORE models 83846.
| Sublingual Microcirculation Parameter | Description | Clinical Significance |
|---|---|---|
| Perfused Boundary Region | Measures lateral penetration of red blood cells into the glycocalyx matrix 4041. | Inverse marker of glycocalyx thickness. Elevated levels indicate structural shedding and barrier dysfunction 3842. |
| Microvascular Flow Index | Semiquantitative assessment of predominant blood flow velocity in small vessels 4347. | Reduced index correlates strongly with impaired tissue perfusion, sepsis severity, and mortality 4749. |
| Total Vessel Density | Total length of all visualized microvessels per specific mucosal area 4349. | Indicates overall capillary anatomical reserve. Frequently diminished in advanced age and chronic disease 4349. |
| Proportion of Perfused Vessels | Percentage of the total vessel density that exhibits continuous active red blood cell flow 4349. | Highly predictive value for sepsis mortality; indicates functional capillary recruitment failure 4849. |
Population Diversity and Ethnic Disparities in Glycocalyx Data
A major structural obstacle to establishing universal, reliable clinical reference ranges for glycocalyx thickness is the significant biological variability of vascular metrics across geographically and genetically diverse human populations, a problem critically compounded by unequal representation in major clinical research trials.
Observational studies assessing foundational endothelial characteristics frequently reveal baseline differences across diverse ethnic groups. For instance, cohort studies such as the South African Eye Study, which examined ocular microvascular parameters across 402 participants, documented distinct baseline variations in central corneal thickness between individuals of African, European, and ethnically diverse descent 51. The African population presented with a mean central corneal thickness of roughly 514 micrometers compared to nearly 550 micrometers in the European cohort 51. While adjusting for age mitigated some disparities in endothelial cell counts, the fundamental structural baselines differed 51.
Furthermore, broad analyses comparing brachial flow-mediated dilation and circulating endothelial biomarkers across five distinct racial and ethnic groups demonstrate highly varied physiological responses to altered metabolic states. For example, the paradoxical clinical association where impaired fasting glucose corresponds with higher flow-mediated dilation was observed predominantly in White populations of normal body mass index, reflecting potential, unmapped ethnic differences in endothelial insulin signaling pathways between the microvasculature and skeletal muscle 52.
Despite this inherent biological and genetic diversity, major clinical networks evaluating microvascular pathologies consistently fail to capture a representative sample. Data from the DRCR Retina Network trials evaluating diabetic macular edema - a condition heavily driven by microvascular hyperpermeability - revealed a massive overrepresentation of White and Black cohorts while significantly underrepresenting Asian, Hispanic, and multiracial populations compared to broader census baselines 53. Until massive, globally diverse clinical registries are established to accurately map baseline sublingual perfused boundary regions and systemic syndecan-1 levels across distinct genetic populations, the broad diagnostic thresholding of what constitutes pathological glycocalyx degradation will remain inherently compromised.
Therapeutic Interventions for Glycocalyx Restoration
Recognizing that vascular aging and endothelial dysfunction are fundamentally driven by glycocalyx perturbation, modern pharmacological strategies have shifted toward structurally restoring this critical barrier rather than merely targeting downstream systemic inflammation or localized lipid accumulation. Two major clinical interventions currently exhibit strong, peer-reviewed evidence for modifying glycocalyx health: Sulodexide and high-dose Omega-3 fatty acids.
Sulodexide: A Direct Glycosaminoglycan Precursor
Sulodexide is a highly purified, naturally derived mixture of specific glycosaminoglycans composed of approximately 80 percent fast-moving heparin fraction and 20 percent dermatan sulfate 754. Because its molecular structure closely mimics the endogenous glycosaminoglycans natively present in the glycocalyx, it acts as a highly effective synthetic precursor. By providing the necessary molecular building blocks directly to the endothelium, it facilitates the physical rebuilding of the degraded matrix 155.
The precise molecular mechanisms by which Sulodexide restores the endothelial glycocalyx are multifold: First, direct matrix restoration is evident. In vivo balloon-injury surgical models demonstrate that sulodexide therapy physically rebuilds the glycocalyx lining over denuded arteries, rapidly restoring clear cytoarchitecture, normalizing endothelial cell spacing, and attenuating pathological endothelial hyperplasia 55. Second, sulodexide exerts potent, direct anti-heparanase activity. It actively and competitively inhibits the heparanase-mediated degradation of syndecan-1 core proteins and heparan sulfate chains, thus pharmacologically halting the primary initial step of the entire shedding cascade 5657. Third, barrier function recovery is accelerated. In both severe sepsis models and human diabetes trials, systemic sulodexide treatment upregulates essential tight junction proteins, specifically Zonula Occludens-1, significantly reversing vascular hyperpermeability and restoring normal fluid dynamics 115658.
In targeted human clinical trials, an eight-week oral administration of sulodexide in patients with chronic Type 2 diabetes successfully and measurably restored both sublingual and retinal glycocalyx physical dimensions 58. Furthermore, this treatment protocol significantly decreased systemic plasma hyaluronidase activity and demonstrated a strong statistical trend toward normalizing the transcapillary escape rate of albumin, directly confirming the restoration of the microvascular permeability barrier 58. While not yet adopted as a universal standard of care globally, sulodexide represents the most direct, targeted pharmacological agent currently available for comprehensive glycocalyx repair.
Omega-3 Fatty Acids
Omega-3 polyunsaturated fatty acids, specifically eicosapentaenoic acid and docosahexaenoic acid, have long been studied extensively for their cardioprotective effects. Their primary physiological influence on the endothelial glycocalyx is largely indirect, operating fundamentally through the potent, systemic suppression of background inflammation and oxidative stress. By actively reducing the transcription and release of pro-inflammatory cytokines, Omega-3 fatty acids dramatically reduce the chemical stimuli that activate the devastating heparanase and matrix metalloproteinase shedding cascades 5960.
Recent comprehensive, high-powered meta-analyses of numerous randomized controlled trials - involving over 134,000 global participants - confirm definitively that continuous Omega-3 supplementation significantly reduces the absolute risk of major adverse cardiac events, spontaneous myocardial infarction, and overall cardiovascular death 6061. Interestingly, granular subgroup analyses from these trials demonstrate that protocols administering high-dose eicosapentaenoic acid alone yield a statistically superior reduction in cardiovascular events and the subsequent need for coronary revascularization compared to traditional formulations combining eicosapentaenoic acid with docosahexaenoic acid 606162. Physiologically, optimal doses of Omega-3s - calculated at approximately 1,650 milligrams of eicosapentaenoic acid and 750 milligrams of docosahexaenoic acid daily - measurably enhance flow-mediated dilation and effectively reduce the augmentation index 63. These metrics indicate a profound restoration of normal endothelial mechanotransduction, reflecting improved glycocalyx structural integrity and a corresponding increase in functional nitric oxide bioavailability 63.
Conclusion
The endothelial glycocalyx serves as the fundamental, irreplaceable gatekeeper of comprehensive vascular health. Operating as an actively regulated, highly hydrated matrix of complex proteoglycans and glycosaminoglycans, it strictly dictates systemic vascular permeability, modulates localized inflammatory responses, and serves as the primary mechanosensor for shear-stress-mediated vasodilation. The process of vascular aging, critically exacerbated by metabolic syndromes and the toxic accumulation of non-enzymatic Advanced Glycation End-Products, accelerates a precise, devastating enzymatic degradation cascade driven primarily by heparanase and matrix metalloproteinases. This systematic pathological shedding strips the vascular endothelium of its essential protective shield, initiating the chronic inflammation, cellular senescence, and arterial stiffness that hallmark cardiovascular disease.
While advanced diagnostic technologies, such as sublingual incident and sidestream dark-field videomicroscopy, provide profound, highly accurate prognostic insights in acute settings like sepsis and severe trauma, their broad utility as a primary screening tool for chronic, stable cardiovascular disease remains significantly constrained. The high physiological overlap between patient populations, coupled with extreme sensitivity to routine environmental variables and a glaring lack of diverse population baselines, necessitates further refinement of these diagnostic parameters. However, future advancements in targeted, structure-modifying therapeutics - such as the direct glycosaminoglycan matrix restoration achieved with Sulodexide or the potent inflammatory modulation provided by high-dose eicosapentaenoic acid - hold immense promise. These interventions offer a pathway to move beyond mere symptom management, focusing instead on repairing the fundamental structural foundation of the vascular wall and actively reversing the trajectory of vascular aging.