Dietary protein and mTOR signaling in muscle mass and longevity
The intersection of dietary protein intake, the mechanistic target of rapamycin (mTOR) signaling pathway, and skeletal muscle maintenance represents a complex frontier in modern geroscience. At the core of this discussion lies the "protein paradox." Experimental biology and epidemiological studies frequently demonstrate that reducing overall protein intake and inhibiting nutrient-sensing pathways like mTOR extends lifespan and delays age-related pathology. Conversely, clinical geriatrics emphasizes that high protein intake and episodic mTOR activation are required to prevent sarcopenia, physical frailty, and premature mortality in older populations 12. Designing a diet that optimally balances these competing physiological priorities requires a nuanced understanding of molecular signaling, age-stratified metabolic needs, and the specific amino acid compositions of dietary proteins.
The Protein Paradox in Nutritional Science
Dietary protein provides the essential amino acids required for the synthesis of cellular structures, enzymes, hormones, and skeletal muscle. The standard public health recommendation of 0.8 grams of protein per kilogram of body weight per day (g/kg/d) represents the minimum required to maintain nitrogen balance in healthy adults, but researchers increasingly view this metric as insufficient for optimizing long-term healthspan 134.
Epidemiological Evidence on Dietary Protein
Long-term population data reveal that the relationship between protein intake and mortality is highly context-dependent. Western-style obesity-promoting diets are characterized by high caloric density, heavy reliance on animal proteins, and increased systemic inflammation, correlating strongly with higher disease incidence 23. Analysis of the Rotterdam Study, which tracked 7,786 participants, alongside meta-analyses of prospective cohort studies, indicates that total dietary protein intake exhibits varying mortality risks depending on the primary protein source 4. High consumption of red and processed meats is consistently associated with an elevated risk of colorectal, colon, rectal, and lung cancers, as well as cardiovascular mortality 5.
Part of the morbidity associated with high animal protein consumption is linked to the generation of exogenous dietary advanced glycation end-products (dAGEs). Modern Western dietary patterns frequently involve cooking high-protein and high-fat foods at high, dry heat (e.g., grilling or frying). This thermal processing introduces massive dAGE loads into the systemic circulation, which bind to the Receptor for AGEs (RAGE), directly compromising tissue integrity through irreversible structural cross-linking and inducing chronic oxidative stress and inflammation 3.
In contrast, plant-based diets, which naturally feature lower total protein and restricted levels of specific amino acids, are associated with improved metabolic profiles and lower cardiovascular incidence 2. The traditional diets of populations in "Blue Zones" (such as Sardinia and Okinawa) typically feature protein intakes averaging around 18.5% of total energy, predominantly sourced from plants, legumes, and occasional aquatic sources 910. However, the longevity data from these regions are heavily debated; demographic analyses (such as those by Newman, 2024) have demonstrated that exceptional longevity records in some of these hotspots correlate strongly with high regional poverty and poor birth record-keeping, complicating the direct attribution of longevity solely to these dietary patterns 26.
The Age-Stratified Protein Requirement
The epidemiological contradiction surrounding protein is most clearly resolved when stratifying populations by age. Evidence from the NHANES cohort and longitudinal studies indicates that high protein intake in middle age (50 - 65 years) correlates with higher overall mortality and cancer risk, an effect largely mediated by elevated insulin-like growth factor 1 (IGF-1) levels and chronic cellular proliferation signaling 2612.
However, in populations over the age of 65, the physiological paradigm shifts completely. For older adults, higher protein consumption is associated with a reduction in all-cause mortality by up to 28% 26. This inflection point is driven by "anabolic resistance," a phenomenon wherein aging skeletal muscle becomes progressively less responsive to the anabolic stimuli provided by circulating amino acids and insulin 14. While young individuals can stimulate muscle protein synthesis with relatively low doses of amino acids, older adults require significantly higher concentrations of high-quality protein to breach the anabolic threshold, combat sarcopenia, and prevent the rapid decline of metabolic stability and bone density 137. Maintaining a low-protein diet (0.8 g/kg/d) into late life exacerbates the loss of lean tissue mass, rendering the individual highly vulnerable to frailty and insulin resistance 1158.
Expert Paradigms in Dietary Design
The clinical translation of the protein paradox is evident in the divergent dietary frameworks proposed by leading longevity experts. These frameworks prioritize different mechanisms of aging, emphasizing either the reduction of molecular growth signals or the preservation of physical performance metrics.
Longevity and Caloric Restriction Protocols
Dr. Valter Longo's paradigm is rooted in molecular gerontology, prioritizing the minimization of chronic mTOR and IGF-1 signaling to delay cellular aging and prevent unregulated tissue growth (e.g., carcinogenesis). For adults under 65, Longo advocates a low-protein, predominantly plant-based diet providing approximately 0.31 to 0.36 grams of protein per pound of body weight (roughly 0.8 g/kg/d) 1718. This equates to approximately 40 to 47 grams of daily protein for a 130-pound individual.
This approach restricts animal-derived proteins, specifically limiting branched-chain amino acids (BCAAs) to maintain low basal mTOR signaling 19. Protein sources in this framework rely heavily on legumes (beans, chickpeas, lentils) alongside high quantities of complex carbohydrates and healthy fats from olive oil and nuts, mimicking the macronutrient distribution of historically long-lived populations 1718. Longo acknowledges the physiological shift that occurs around age 65, at which point he recommends increasing protein intake by approximately 1.0 g/kg/d to prevent sarcopenia, shifting the diet to incorporate more low-mercury fish, eggs, and dairy products derived from goats and sheep 1517. For individuals who are obese or overweight, protein needs in this framework are calculated against an ideal body weight (estimated via body fat percentage) rather than total body weight, further restricting total amino acid load 15.
Metabolic Resilience and Muscle Hypertrophy Protocols
In contrast, physician Dr. Peter Attia approaches longevity through the lens of metabolic capacity and physical reserve. Attia highlights that skeletal muscle acts as the body's primary sink for glucose disposal, making it a critical buffer against hyperinsulinemia and metabolic syndrome 139. Furthermore, midlife grip strength, appendicular lean mass, and high VO2 max are among the strongest independent predictors of all-cause survival 13.
Because the recommended dietary allowance (RDA) of 0.8 g/kg/d is merely a survival minimum for nitrogen balance, Attia asserts it is profoundly inadequate for building or preserving functional tissue 341011. Attia recommends roughly 2.2 g/kg/d (1.0 g per pound of ideal body weight) for adults of all ages engaged in exercise 41112. Rather than fearing mTOR activation, Attia characterizes post-exercise mTOR stimulation via complete animal proteins (such as whey, eggs, and venison) as a transient, necessary physiological event 19. In this view, episodic mTOR activation for muscle protein synthesis does not carry the same pathological risks as the chronic, systemic mTOR hyperactivation seen in sedentary, hypercaloric populations.
| Parameter | Caloric Restriction Paradigm (Valter Longo) | Metabolic Resilience Paradigm (Peter Attia) |
|---|---|---|
| Core Philosophy | Minimize cellular aging and proliferation by restricting IGF-1 and chronic mTOR signaling. | Maximize physical reserve, glucose disposal capacity, and cardiovascular fitness. |
| Target Protein Intake (<65 Years) | ~0.8 g/kg/d (Calculated on ideal lean weight). | ~2.2 g/kg/d (~1.0 g/lb of ideal body weight). |
| Target Protein Intake (>65 Years) | ~1.0 g/kg/d (Modest increase to prevent frailty). | ~2.2 g/kg/d (Maintained or slightly increased due to anabolic resistance). |
| Preferred Protein Sources | Legumes, plant sources, specific low-mercury aquatic species (salmon, anchovies). | High-quality animal proteins, venison, whey, eggs, dairy. |
| View on mTOR Activation | High BCAA intake accelerates aging pathways; activation should be minimized. | Transient, pulsatile activation is protective; chronic activation via overeating is pathological. |
| Primary Clinical Metrics | BMI, systemic inflammation, IGF-1 levels. | VO2 max, grip strength, DEXA (lean mass), ApoB, HOMA-IR. |
Molecular Architecture of mTOR Signaling
Understanding how dietary protein selectively drives hypertrophy or accelerates aging requires a deep biochemical mapping of the mechanistic target of rapamycin (mTOR). mTOR is an atypical serine/threonine kinase that functions as the central node integrating intra- and extracellular stimuli - including growth factors, amino acids, stress signals, and cellular ATP levels - to dictate whether a cell engages in anabolic growth or catabolic recycling 1314. mTOR operates within two structurally and functionally distinct multiprotein complexes: mTOR Complex 1 (mTORC1) and mTOR Complex 2 (mTORC2).
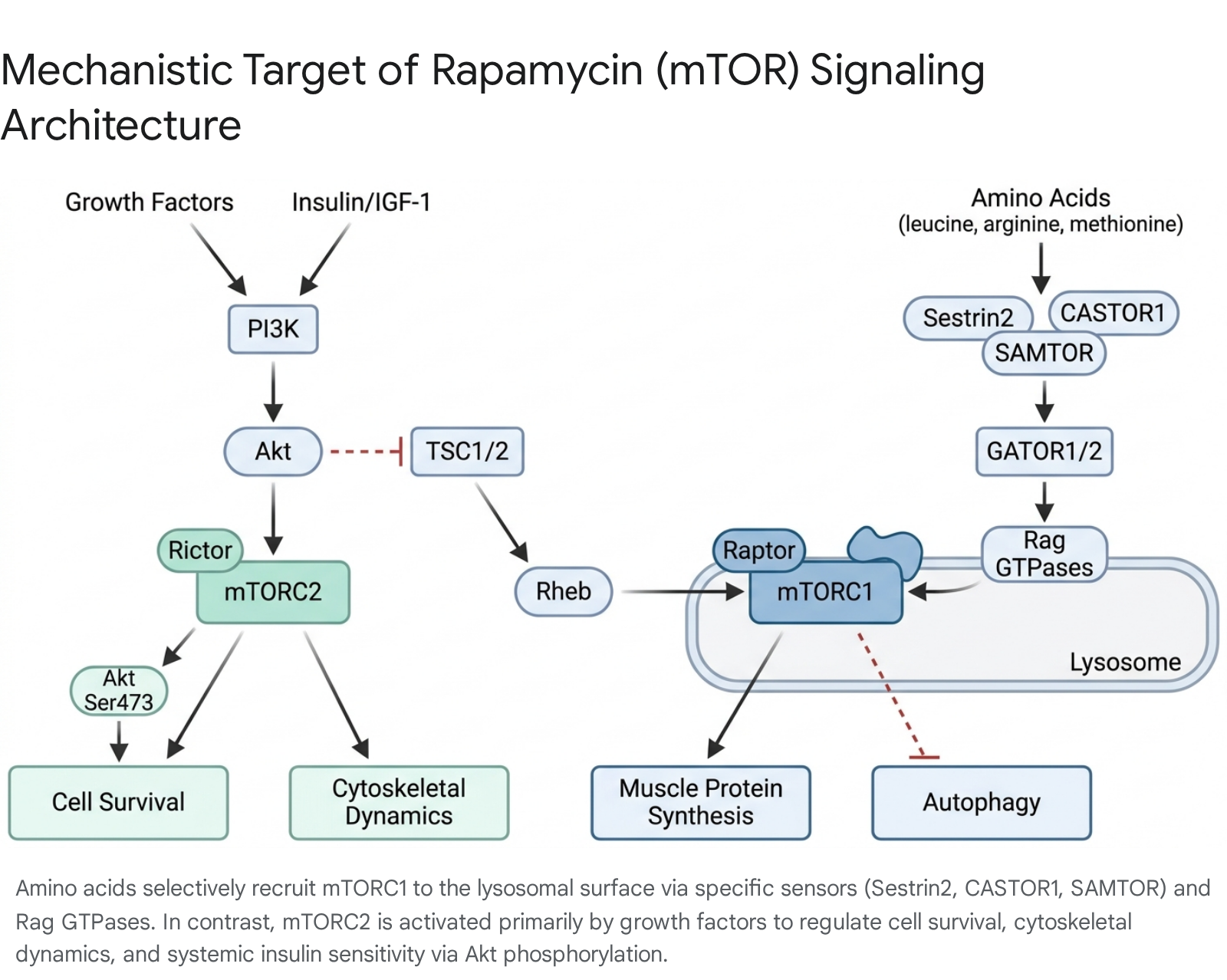
mTOR Complex 1 Activation and Amino Acid Sensing
mTORC1 is the master regulator of cell growth, responsible for upregulating mRNA translation, lipid synthesis, and glycolysis, while simultaneously suppressing lysosomal biogenesis and autophagy 1415. The complex comprises the mTOR kinase, the scaffolding protein Raptor (which recruits specific substrates to the complex), GβL (mLST8), PRAS40, and DEPTOR 141516.
Maximal activation of mTORC1 requires the convergence of two distinct signaling arms: growth factors and amino acid availability.
Growth factors, such as insulin and insulin-like growth factors (IGF), stimulate surface receptors to activate phosphoinositide 3-kinase (PI3K) and the kinase Akt. Akt phosphorylates and inhibits the Tuberous Sclerosis Complex (TSC1/2). Under fasting conditions, TSC1/2 functions as a GTPase-activating protein (GAP) that maintains the small G-protein Rheb in an inactive, GDP-bound state 1314. When insulin signaling inhibits TSC1/2, Rheb accumulates in its active, GTP-bound form. Active Rheb is an essential, potent stimulator of mTORC1 kinase activity 14.
However, active Rheb alone is insufficient if mTORC1 is physically separated from it. Rheb resides exclusively on the surface of the lysosome. The translocation of mTORC1 to the lysosome is entirely dependent on intracellular amino acid levels, functioning independently of the insulin/PI3K axis 14. The cell utilizes precise amino acid sensors to detect nutritional abundance. Sestrin2 functions as the cytosolic sensor for leucine. Under conditions of leucine deprivation, Sestrin2 binds to and inhibits GATOR2. When dietary leucine enters the cell, it binds Sestrin2, causing it to dissociate from GATOR2. Liberated GATOR2 then inhibits the GATOR1 complex 17. Concurrently, CASTOR1 operates as a sensor for arginine, and SAMTOR detects S-adenosylmethionine (a proxy for methionine availability), utilizing similar disinhibition mechanisms via the GATOR complexes and the KICSTOR complex 17.
The ultimate target of these sensing mechanisms is a group of small molecular switches known as the Rag GTPases (RagA, B, C, and D). Anchored to the lysosome by the Ragulator complex and the v-ATPase proton pump, active Rag GTPases physically recruit the Raptor subunit of mTORC1 from the cytoplasm to the lysosomal membrane 131417. Once localized at the lysosome, mTORC1 contacts active Rheb and initiates downstream anabolism, prominently phosphorylating S6K1 and 4E-BP1 to drive massive increases in skeletal muscle protein synthesis 131518.
mTOR Complex 2 and Systemic Insulin Sensitivity
While mTORC1 controls cellular growth, mTORC2 primarily regulates cellular survival, glucose metabolism, and cytoskeletal remodeling. mTORC2 is defined by the scaffolding protein Rictor, functioning alongside mTOR, GβL, Sin1, PRR5/Protor-1, and DEPTOR 1419. Unlike mTORC1, mTORC2 is generally insensitive to acute nutrient and amino acid fluctuations. Its activation is driven predominantly by growth factor stimulation via the PI3K/PIP3 signaling axis 152021.
The most critical metabolic function of mTORC2 is the phosphorylation of Akt at Serine 473 (in the hydrophobic motif) and Threonine 450 (in the turn motif). This phosphorylation is absolutely required for maximal Akt activity, which subsequently governs hepatic gluconeogenesis, glucose transport, and whole-body insulin sensitivity 151821. Furthermore, mTORC2 coordinates adrenergic signaling and regulates ion transport via SGK1 142022.
Cross-Talk and Feedback Inhibition
The balance between muscle growth and healthy longevity relies heavily on the cross-talk between these two complexes. Constant grazing on high-protein, calorie-dense foods leads to chronic, unremitting activation of mTORC1. This hyperactivation initiates a pathological feedback loop: activated mTORC1 and its downstream effector S6K1 directly phosphorylate Insulin Receptor Substrate 1 and 2 (IRS1/2), promoting their proteasomal degradation. mTORC1 also activates Growth-Factor-Bound Protein 10 (Grb10), which further disrupts the interaction between the insulin receptor and IRS1/2 1520.
By degrading IRS1/2, chronic mTORC1 activity essentially severs the insulin signaling cascade, preventing the normal activation of PI3K and directly downregulating mTORC2. This mTORC1-mediated dampening of mTORC2 abrogates Akt signaling, leading to impaired glucose uptake and profound systemic insulin resistance 152022. Consequently, dietary protocols that allow mTORC1 to periodically return to a basal state (via fasting or protein cycling) are imperative to preserve mTORC2 functionality and maintain metabolic health.
Specific Amino Acid Restriction
Recognizing the consequences of chronic mTORC1 activation, researchers have sought to replicate the longevity benefits of total protein restriction without inducing the muscle wasting associated with low nitrogen intake. This is achieved by restricting specific essential amino acids that serve as the primary triggers for nutrient-sensing pathways, notably the branched-chain amino acids (BCAAs) and the sulfur-containing amino acid methionine.
Mechanisms of Branched-Chain Amino Acid Catabolism
The BCAAs - leucine, isoleucine, and valine - are critical for stimulating muscle protein synthesis, but elevated circulating levels are strongly correlated with insulin resistance, obesity, and cardiovascular risk in humans 232425. In skeletal muscle, excessive BCAA oxidation during insulin-resistant states exacerbates metabolic disturbances. However, reducing dietary BCAAs by 50% to 70% in murine models yields significant improvements in cardiometabolic parameters, reducing weight gain and promoting glucose tolerance despite an increase in hyperphagia (food consumption), a phenomenon driven by increased uncoupled respiration and energy expenditure 2325.
Short-term human clinical trials demonstrate that diets restricted in BCAAs lower postprandial insulin secretion, improve glucose sensitivity, and increase circulating levels of Fibroblast Growth Factor 21 (FGF21) by approximately 20% 23. FGF21 is an insulin-sensitizing, liver-derived hormone heavily implicated in stress adaptation and metabolic health, suggesting a primary mechanism by which amino acid modulation improves systemic phenotypes 23.
Isoleucine Restriction Efficacy
Recent mechanistic dissections reveal that the physiological impacts of the three BCAAs are not uniform. While leucine is the most potent agonist of mTORC1, lifelong dietary restriction of leucine yields negligible metabolic benefits and actually increases adiposity in aged subjects 2627. In stark contrast, restriction of isoleucine has emerged as both necessary and sufficient to replicate the metabolic benefits of broader protein restriction.
In genetically heterogeneous UM-HET3 mice and C57BL/6J models, a 67% reduction in dietary isoleucine alone - without calorie restriction - drastically improves glucose tolerance, reduces fasting blood glucose, and decreases adiposity through the beiging of inguinal white adipose tissue 252829. Importantly, these benefits are maintained even when the intervention is initiated in late life. Aged mice subjected to an isoleucine-restricted diet demonstrate rejuvenated hepatic gene expression profiles, enhanced uncoupled energy expenditure, and substantial lifespan extension, achieving up to a 33% increase in median survival in males 243031.
Translational evidence in humans is highly promising. A 2024 randomized controlled trial in older adults demonstrated that a 50% reduction in dietary isoleucine, without reducing total calories, significantly improved postprandial glucose control, enhanced fat oxidation, and upregulated mitochondrial gene expression 32.
However, manipulating individual amino acids carries sexually dimorphic complexities. While isoleucine restriction improves metabolic parameters in both sexes, it has been shown to induce a rapid loss of lean mass and reduce grip strength in aged mice 31. Furthermore, extreme cellular deprivation of isoleucine forces the aminoacyl-tRNA synthetase IARS1 to misacylate tRNA with valine, generating unprogrammed amino acid substitutions (I > V) in cellular proteins, raising questions about extreme long-term restriction 33. Additionally, in Alzheimer's disease models (3xTg mice), while restriction of isoleucine provides the greatest cognitive and memory improvements in males, restriction of valine confers superior cognitive benefits to females, underscoring the need for highly personalized amino acid targeting 26.
Continuous and Intermittent Methionine Restriction
Methionine is essential for cellular metabolism and serves as the precursor for glutathione, a master cellular antioxidant. However, because cancer cells have an aberrantly high requirement for methionine (the "Hoffman effect"), restricting this amino acid has been extensively explored as an anti-tumor and life-extending intervention 3034. Continuous methionine restriction (MR) extends the lifespan of rodents by up to 45%, improves glycemic control, entirely protects against diet-induced hepatosteatosis, and reduces levels of insulin, IGF-1, and leptin 323536.
In translational studies involving humans with overweight or obesity, 4-to-12-week trials of sulfur amino acid restriction (SAAR) resulted in significant loss of fat mass, lowered total and LDL cholesterol, and improved metabolic flexibility 303437. At the molecular level, MR downregulates the SAMTOR/mTORC1 axis and favorably alters the Nrf2 signaling pathway, though tissue-specific glutathione depletion must be managed 1738.
Despite its profound metabolic benefits, continuous MR poses severe challenges for long-term human adherence due to diet palatability and serious physiological side effects. Chronic MR in mammals induces a loss of musculoskeletal mass, reduces bone mineral density, and radically increases marrow adipogenesis (fat accumulation within the bone), leading to structurally weaker bones 3639.
To capture the longevity benefits while circumventing musculoskeletal damage, researchers have developed Intermittent Methionine Restriction (IMR) protocols. By restricting methionine for only three days per week, murine subjects achieved metabolic health benefits identical to continuous restriction 3639. Crucially, IMR prevented the accumulation of marrow fat, preserved both trabecular and cortical bone mass by increasing osteoblast numbers, and maintained the mechanical strength of the skeleton 3536. Furthermore, IMR demonstrated protective effects against diet-induced chronic inflammation and conditions such as ulcerative dermatitis 39. The success of IMR strongly supports cyclical, rather than continuous, dietary restriction protocols in human dietary design.
| Restriction Modality | Primary Metabolic Benefits | Potential Drawbacks / Considerations | Clinical Outlook |
|---|---|---|---|
| Total Protein Restriction | Lowers circulating IGF-1, reduces cancer risk, promotes autophagy. | Sarcopenia, functional decline, delayed wound healing in adults >65. | Best applied in mid-life (50-65 years); detrimental in advanced age. |
| Isoleucine Restriction | Increases energy expenditure, improves glucose tolerance, reduces ectopic adiposity. | May reduce grip strength and specific lean mass in older populations. | Highly promising. 2024 human RCTs confirm improved fat oxidation and glycemic control. |
| Continuous Methionine Restriction | Up to 45% lifespan extension in rodents, robust anti-obesity effects, hepatoprotection. | Loss of bone mineral density, marrow fat accumulation, highly unpalatable diet. | Medically challenging for long-term adherence in humans. |
| Intermittent Methionine Restriction | Matches metabolic benefits of continuous MR while preserving bone density and strength. | Requires rigid cycling of amino acid intake. | Highly translatable framework for balancing longevity with structural integrity. |
Pharmacological Interventions: The PEARL Clinical Trial
Given the difficulty of adhering to highly specific amino acid restrictions, pharmacological mimetics that directly inhibit the mTOR pathway are actively researched. Rapamycin, an allosteric inhibitor that complexes with FKBP12 to bind and inhibit mTORC1, is the most robust pharmacological lifespan-extender in preclinical models 1440. However, assessing its efficacy and safety as a gerotherapeutic in healthy, normative-aging humans has been hindered by a lack of clinical trials.
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial, published in 2024 and 2025, represents a landmark double-blind, randomized, placebo-controlled study designed to evaluate intermittent rapamycin use in humans 41424344. The trial followed 114 generally healthy older adults (average age ~60) over 48 weeks. Participants were randomized to receive a placebo, 5 mg of compounded rapamycin weekly, or 10 mg of compounded rapamycin weekly 414244.
A critical pharmacological note from the trial is that the compounded rapamycin utilized yielded only about one-third of the blood concentration normally achieved by commercially available formulations, meaning the effective dosages were significantly lower than the stated 5 mg and 10 mg targets 45.
Trial Outcomes and Sex-Specific Responses
The PEARL trial established that low-dose, intermittent rapamycin administration is highly tolerable and relatively safe for normative-aging adults. Researchers detected no significant differences in moderate to severe adverse events between the rapamycin cohorts and the placebo group 434445. Standard blood biomarkers largely remained within normal ranges, alleviating concerns that intermittent dosing would induce the severe hyperlipidemia, immunosuppression, or mouth sores often associated with chronic, daily rapamycin therapy in transplant patients 134144. Minor shifts included increased red blood cell counts in the 5 mg group and slight increases in blood urea nitrogen (BUN) and minor gut dysbiosis indices in males in the 10 mg group 45.
While the primary endpoint - a reduction in visceral adiposity measured via DEXA scan - did not achieve statistical significance, the trial uncovered profound sex-specific improvements in healthspan markers 42444546: * Female Participants: Women in the 10 mg/week group experienced a statistically significant 4.5% increase in lean tissue mass, alongside a marked reduction in self-reported pain and osteoarthritis symptoms 41424344. * Male Participants: Men did not demonstrate significant changes in lean mass but did show a modest 1.4% average increase in bone mineral content (BMC) in the 10 mg/week cohort 4143. * General Health: Participants receiving the 5 mg/week dose exhibited significant improvements in self-reported general health and emotional well-being, though the placebo group also showed improvements in emotional parameters 424445.
The significant gain in lean muscle mass among older women taking an mTOR inhibitor initially appears counterintuitive to the protein paradox (which requires mTOR activation for growth). However, in aged populations characterized by chronic, low-grade inflammation (inflammaging) and hyperactive basal mTORC1, dampening this overactivity with rapamycin may reduce catabolic inflammatory signaling and restore cellular sensitivity to acute anabolic stimuli (like exercise and dietary protein), thereby facilitating muscle preservation 314245.
| Metric Assessed | 5 mg Weekly Rapamycin | 10 mg Weekly Rapamycin | Placebo |
|---|---|---|---|
| Lean Tissue Mass | No significant change. | +4.5% significant increase in female participants. | Baseline |
| Bone Mineral Content | Potential minor decrease in BMD noted in some analyses. | +1.4% average increase in male participants. | Baseline |
| Self-Reported Pain | No significant effect. | Significant reduction in female participants (WOMAC scale). | Baseline |
| General Health & Mood | Significant improvements in emotional well-being and general health. | Improvements in overall QoL and social functioning. | Baseline |
| Visceral Adiposity | No statistically significant change. | No statistically significant change. | Baseline |
While the PEARL trial relied heavily on self-reported quality-of-life metrics and did not prove lifespan extension in humans, it validates the safety profile of intermittent mTOR inhibition and justifies larger, longitudinal studies utilizing diverse dosing ranges 414244.
Mitochondrial Health and Macronutrient Intake
Beyond muscle mass and systemic insulin sensitivity, the balance of amino acid intake directly modulates the structural integrity and functional capacity of the mitochondrial network. Mitochondria are central hubs of cellular metabolism, and their dysfunction is a primary hallmark of both chronological aging and cardiometabolic diseases like Type 2 Diabetes Mellitus (T2DM).
BCAA Catabolism and Mitochondrial Biogenesis
Branched-chain amino acids are uniquely essential for promoting mitochondrial biogenesis in skeletal and cardiac muscle 47. BCAAs are catabolized within the mitochondria by the branched-chain α-keto acid dehydrogenase complex. The resulting metabolites enter the tricarboxylic acid (TCA) cycle, directly feeding ATP production and enhancing oxidative phosphorylation efficiency under stress 4748.
However, the efficacy of mitochondrial energy generation is heavily dependent on sirtuin activity. SIRT3, a primary mitochondrial deacetylase, maintains basal ATP levels by deacetylating and activating several electron transport chain targets, including NDUFA9 in Complex I and succinate dehydrogenase in Complex II 47. SIRT3 also mitigates cardiac hypertrophy by augmenting antioxidant defense mechanisms through FoxO3a 47. Diets that excessively over-activate mTORC1 suppress these mitochondrial maintenance pathways, accelerating the accumulation of damaged organelles through the suppression of mitophagy 47.
Mitochondrial DNA Methylation and Disease Pathology
Pathological nutrient overload disrupts the epigenetic landscape of the mitochondrial genome. Differential methylation of mitochondrial DNA (mtDNA) - particularly within the D-loop region, which controls mtDNA replication and transcription - is increasingly identified as a biomarker for cardiometabolic disease 4849.
In patients with T2DM and severe cardiovascular disease, hypermethylation of the D-loop and the ND6 regions correlates with significant alterations in mitochondrial gene expression, reduced oxidative phosphorylation capacity, and lowered overall mtDNA copy number 4849. This decline in mtDNA content is inversely associated with ectopic visceral adiposity 48. Furthermore, mitochondrial genetic dysfunction and altered BCAA metabolism have been implicated in driving chronic inflammatory states and immune modulation in diseases such as endometriosis 5051. Consequently, dietary interventions that restrict specific amino acids (like isoleucine) play a critical role in restoring mitochondrial gene expression patterns and rescuing the epigenetic stability of the mitochondrial network 4751.
Dietary Strategies for Sarcopenia Prevention
Synthesizing the molecular biology of mTOR, the clinical necessity of maintaining physical reserve, and emerging data on selective amino acid restriction leads to actionable frameworks for dietary design. Designing a diet that balances longevity with muscle mass relies on strategically manipulating protein sources, absolute daily volume, and the temporal distribution of meals.
Plant-Based Protein Substitution
The primary vector by which individuals can modify their amino acid profile without restricting total protein volume is by shifting the source of their macronutrients. Animal-derived proteins (meat, dairy, eggs) possess exceptionally high Digestible Indispensable Amino Acid Scores (DIAAS) and are dense in leucine, isoleucine, and methionine 13252. While highly efficient at stimulating muscle protein synthesis, the chronic consumption of red meat drives prolonged mTORC1 activation and systemic inflammation 23253.
Plant-based proteins - such as legumes, chickpeas, brown rice, and pea protein - are naturally lower in isoleucine and methionine. Transitioning toward a plant-heavy dietary pattern inadvertently mimics the longevity-promoting effects of specific amino acid restriction 1732. A comprehensive 2023 meta-analysis confirmed that substituting just 3% of daily energy from animal protein with plant-based alternatives is associated with profound cardiometabolic improvements and a 10% reduction in all-cause mortality 12.
Importantly, relying on plant-based proteins does not sentence an individual to sarcopenia. Clinical trials utilizing high-protein vegan diets demonstrate that consuming a diverse profile of plant proteins (e.g., mycoprotein or blended pea/rice isolates) supports daily myofibrillar protein synthesis rates equivalent to those achieved on isonitrogenous omnivorous diets, provided the total protein volume is adequate 3254. For individuals requiring maximal intervention against sarcopenia, multi-ingredient protein supplements combining high-quality milk proteins (whey/casein) with creatine monohydrate and Vitamin D3 offer superior hypertrophic outcomes during resistance training regimens, with a physiological ceiling effect appearing around 1.5 to 1.7 g/kg/d 55.
Timing and Pulsatile Dosing Mechanisms
To resolve the protein paradox, researchers advocate abandoning the modern pattern of constant caloric grazing in favor of strategic, pulsatile dosing. Rather than maintaining a low-grade, chronic activation of mTORC1 throughout the day, dietary protein should be leveraged as an acute therapeutic trigger.
Muscle protein synthesis requires crossing a specific "leucine threshold" during a meal. While younger adults may achieve this threshold with 20 grams of high-quality protein, the blunted sensitivity of anabolic resistance means that older adults require between 30 and 40 grams of protein in a single sitting to effectively flip the molecular switch for muscle building 127. Distributing protein symmetrically across distinct meals - for example, consuming ~35 grams at breakfast, lunch, and dinner - has been shown to generate up to a 25% higher 24-hour muscle protein synthesis response compared to the standard Western pattern of consuming negligible protein early in the day and a massive bolus at dinner 7.
Crucially, this pulsatile dosing must be paired with periods of metabolic silence. Implementing daily overnight fasting protocols or time-restricted eating (e.g., a 12-hour feeding window) ensures that circulating amino acids are cleared and insulin levels drop 291856. This prolonged fasting state deprives the Rag GTPases and PI3K pathways of their activating signals, allowing mTORC1 activity to return to baseline. In this deactivated state, the cell lifts the inhibition on autophagy, clearing misfolded proteins, and restores the sensitivity of the mTORC2/Akt signaling axis to ensure efficient glucose disposal during the next feeding cycle 1521.
Conclusion
The biological tension between longevity and muscle hypertrophy requires abandoning monolithic dietary guidelines in favor of precision nutrition. Experimental and clinical data indicate that the "protein paradox" is navigated by manipulating age-specific protein targets, the amino acid profile of the food source, and the timing of consumption. During mid-life, individuals benefit from moderate protein intake heavily skewed toward diverse plant sources to restrict circulating isoleucine and methionine, thereby minimizing the chronic mTORC1 signaling that drives cellular aging and oncogenesis.
As individuals transition into late-life, combating the onset of anabolic resistance and subsequent frailty demands an elevation in high-quality protein intake (1.2 - 2.2 g/kg/d) alongside vigorous resistance training. By administering protein in concentrated, pulsatile doses to breach the leucine threshold, and pairing these meals with adequate fasting windows, individuals can selectively harness the transient anabolic power of mTOR to maintain vital physical reserves. This dynamic orchestration of nutrient sensing pathways preserves systemic insulin sensitivity, protects mitochondrial integrity, and ultimately aligns the preservation of physical function with the extension of maximum human healthspan.