Default mode network and self-referential thought in depression
Neuroanatomical Architecture of the Default Mode Network
The default mode network (DMN) is a large-scale, intrinsically organized brain system characterized by highly synchronized, low-frequency oscillations that emerge when an individual is not engaged in externally directed, attention-demanding tasks. Initially identified through positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) as a network that reliably "deactivates" during goal-directed cognitive paradigms, the DMN is now recognized as the fundamental neurological substrate of internal cognition 123. Its evolutionary persistence, with homologous structures identified in rodents and non-human primates, suggests a critical adaptive role in mammalian neurobiology. Across primate evolution, an expansion of the frontotemporal subnetwork and a transition toward a dorsal frontoparietal pathway point to the DMN's specialized capacity for integrating past experiences, simulating future outcomes, and navigating complex social hierarchies 3.
Network Subsystems and Nodal Interactions
The functional anatomy of the DMN is not a monolithic entity; rather, it is best conceptualized as a centralized hub system interfacing with multiple interacting sub-networks. The structural integrity and functional connectivity of these nodes dictate the network's capacity to orchestrate complex internal mental simulations 14. The DMN comprises three primary functional subdivisions: a midline core, a dorsal medial prefrontal cortex (dMPFC) subsystem, and a medial temporal lobe (MTL) subsystem 4.
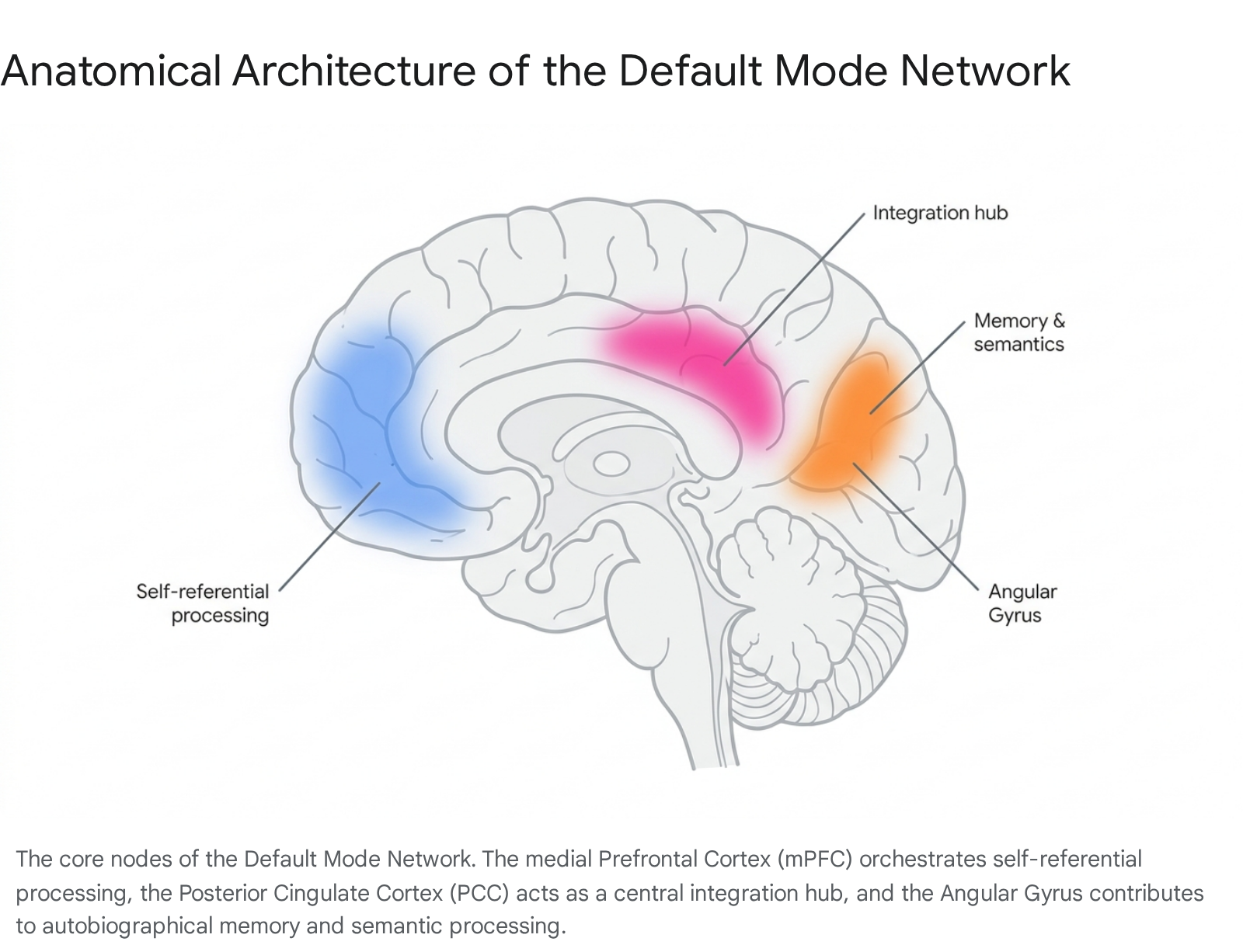
The midline core consists of the anterior medial prefrontal cortex (mPFC) and the posterior cingulate cortex (PCC), extending into the precuneus. The mPFC is deeply involved in the generation of self-referential schemas, integrating affective and autonomic states into a coherent sense of self 2. The PCC functions as a highly connected integration hub, facilitating metabolic communication across different subsystems and tracking the continuous updating of environmental and internal changes 12.
The MTL subsystem, encompassing the hippocampus, parahippocampal cortex, and retrosplenial cortex, provides the raw mnemonic information - such as autobiographical memories and contextual associations - that serves as the fundamental building blocks for mental simulation and prospection 1. Finally, the dMPFC subsystem, which includes the temporoparietal junction (TPJ), the angular gyrus (inferior parietal lobule), and the lateral temporal cortex, is heavily implicated in social cognition, mentalizing (Theory of Mind), and semantic processing 235.
Structural Covariance and Network Maturation
The anatomical foundation of the DMN is further elucidated through structural covariance networks (SCNs), which measure the correlated variations in gray matter morphology across spatially distributed brain regions. Source-based morphometry reveals that spatial patterns of structural covariance closely overlap with functional connectivity patterns observed via resting-state fMRI 6. The maturation of these structural networks is not static. Throughout neurodevelopment, the strength of DMN functional connectivity follows an inverted U-shape trajectory. The network is relatively immature and weakly connected in childhood, reaches peak intrinsic connectivity and robustness during young adulthood, and gradually exhibits diminished connectivity in the elderly 78.
Furthermore, demographic factors introduce normal variance in DMN architecture. Neuroimaging meta-analyses indicate that biological sex plays a role in baseline connectivity, with healthy females generally exhibiting stronger intra-network connectivity within the DMN compared to males 78. Defining this normal comparative base is paramount for clinical neuroscience, as it provides the critical benchmark required to identify when DMN alterations cross the threshold into psychiatric pathology.
The Default Mode Network in Healthy Cognition
In healthy individuals, the DMN governs a state of "wakeful rest" and stimulus-independent thought. When the brain is released from the constraints of immediate external task demands, the DMN enables spontaneous cognition, often characterized as "mind-wandering" 2. Rather than representing mere metabolic noise, this default activity fulfills essential psychological functions that maintain cognitive health and emotional stability.
Self-Referential Processing and Mental Time Travel
The primary cognitive domain supported by the DMN is self-referential processing. The network is consistently activated when individuals evaluate their own traits, reflect on their emotional states, or construct a coherent internal narrative 129. This self-referential capacity is deeply intertwined with autobiographical memory retrieval. By drawing upon the medial temporal lobe subsystem, the DMN allows individuals to access past experiences and contextualize them within their current identity 1.
Beyond historical recall, the DMN facilitates "mental time travel." The same neural architecture used to remember the past is recruited to project the self into the future 1. This capacity for prospection enables individuals to anticipate future events, construct long-term plans, simulate alternative scenarios, and evaluate the emotional valence of potential outcomes. By simulating future social interactions or personal challenges, the healthy DMN provides a safe cognitive testing ground that maximizes the utility of moments when the organism is not actively engaged by the external world 13.
Social Cognition and Environmental Assessment
The DMN is also heavily recruited during social processing, particularly in tasks that require inferring the mental states, beliefs, and intentions of others - a capability known as Theory of Mind or mentalizing 23. The dMPFC and the angular gyrus are critical for adopting the perspective of another individual and understanding social narratives 12. In a healthy state, the DMN remains dynamically flexible. It sustains self-reflection and social assessment but rapidly and efficiently deactivates when a sudden environmental stimulus demands focused, exogenously directed attention. This flexibility is the hallmark of healthy cognitive function and is directly positively correlated with overall cognitive performance 78.
| DMN Sub-node | Healthy State Functional Role | Major Depressive Alteration |
|---|---|---|
| Medial Prefrontal Cortex (mPFC) | Central to self-referential processing, emotional regulation, and the integration of social and affective information. Critical for introspection and mental projection into the past, present, and future 2. | Exhibits hyperconnectivity with other DMN regions. Correlates with rumination traits, excessive negative self-reflection, and failure to down-regulate internal focus 210. |
| Posterior Cingulate Cortex (PCC) | Functions as a highly connected hub facilitating communication between DMN subsystems. Monitors internal and external stimuli and integrates autobiographical data 211. | Characterized by aberrant hyperactivity and hyperconnectivity with limbic regions (e.g., hippocampus), contributing to inescapable self-criticism and recurrent negative thoughts 212. |
| Angular Gyrus (Inferior Parietal Lobule) | Key region for higher cognitive processes, including introspection, spatial cognition, autobiographical memory, and social cognition 25. | Shows intensified functional connectivity in unhappy or depressed individuals. Hyperconnectivity here is positively related to incubation and maladaptive reflection 25. |
The Triple Network Model of Brain Organization
To comprehend the DMN's role in psychiatric pathology, it must be analyzed not in isolation, but as part of a dynamic, interacting system of large-scale neurocognitive networks. The Triple Network Model (TNM), proposed by Menon, provides a unifying neurobiological framework for understanding how the human brain dynamically allocates computational resources between internal mental processes and external environmental demands 131415.
Central Executive and Salience Networks
The TNM posits that high-level human cognition relies on the precise, time-locked coordination of three core networks: the Default Mode Network (DMN), the Salience Network (SN), and the Central Executive Network (CEN, frequently referred to as the Frontoparietal Network or FPN) 1314.
The CEN is anchored in the dorsolateral prefrontal cortex (dlPFC) and the posterior parietal cortex. It operates as the brain's primary engine for high-level cognitive functions, including working memory, active problem-solving, and the manipulation of information during goal-directed, externally oriented tasks 1416. In a healthy neurological state, the CEN and the DMN operate in a strictly anticorrelated manner. When cognitive resources are demanded by an external task, the CEN upregulates its activity, while the DMN is actively and proportionately suppressed 1718.
The Salience Network acts as the critical regulatory mediator between the DMN and the CEN. Anchored by the anterior insula (AI) and the dorsal anterior cingulate cortex (dACC), the SN is tasked with continuously monitoring internal interoceptive states (e.g., heart rate, autonomic arousal) and external sensory inputs to detect biologically or cognitively salient events 131419.
Mechanisms of Network Integration and Segregation
When the SN detects a highly salient stimulus - such as a sudden environmental threat, a socially relevant cue, or an immediate cognitive demand - the anterior insula functions as a dynamic neural switch 1319. It generates a control signal that engages the CEN to handle the external demand while simultaneously disengaging the DMN to halt internally directed mind-wandering 19.
The proper functioning of the human brain requires an optimal, fluid balance of network segregation (the ability of distinct networks to operate independently without pathological cross-talk) and network integration (the ability to share information across global systems when required) 2021. The SN's capacity to efficiently toggle between the internally focused DMN and the externally focused CEN represents healthy network segregation. The breakdown of this switching mechanism, leading to aberrant integration or rigid, inflexible connectivity patterns, is increasingly recognized as a primary transdiagnostic biomarker for major psychiatric conditions 141519.
Network Dysregulation in Major Depressive Disorder
Major depressive disorder (MDD) is characterized clinically by profound alterations in mood, anhedonia, psychomotor retardation, and persistent negative self-referential thought 14. Neuroimaging studies spanning fMRI, PET, magnetoencephalography (MEG), and EEG consistently demonstrate that MDD is fundamentally a disorder of large-scale brain network dysregulation, with the DMN occupying a central pathological role 162223.
Intrinsic Hyperconnectivity and the Subgenual Cingulate
The most robust initial finding in the neuroimaging literature of depression is the presence of intrinsic hyperconnectivity within the DMN during resting states 161823. Specifically, patients with MDD exhibit abnormally elevated functional coupling between the midline core nodes - the mPFC and the PCC 410. This hyperconnectivity is frequently accompanied by increased functional connectivity between the DMN and subcortical limbic structures, notably the subgenual anterior cingulate cortex (sgACC) and the amygdala 1724.
Functionally, this hyper-integrated state traps the patient in a cycle of persistent, internally directed cognition. Because the DMN is the substrate for self-referential thought, an overactive DMN biases the patient's cognitive baseline toward an inescapable internal environment of negative self-evaluation and autobiographical rumination 225. Furthermore, during tasks that normally require attention to external stimuli, patients with MDD fail to adequately deactivate or suppress the DMN 17. This failure of task-induced deactivation leads to a persistent interference of internal negative thoughts during attempts to engage with the external world, providing a neurological explanation for the profound cognitive deficits and concentration difficulties clinically observed in depression 1726.
Altered Inter-Network Dynamics and Causal Influences
The Triple Network Model reveals systemic dysfunction in MDD that extends beyond isolated DMN hyperactivity. Depression is characterized by aberrant interactions between the DMN, SN, and CEN, effectively paralyzing the brain's ability to switch cognitive modes 1519.
First, MDD is associated with abnormal hyperconnectivity between the DMN and the SN, particularly between the medial prefrontal nodes of the DMN and the insular and dACC nodes of the SN 1327. This specific dysconnectivity suggests a pathological feedback loop: internally generated negative thoughts (originating in the DMN) are inappropriately tagged as highly salient and threatening by the SN. This causes the brain to allocate excessive, unwarranted attentional resources to depressive ruminations rather than external environmental stimuli 1319.
Second, patients with MDD exhibit decreased functional connectivity between the SN and the CEN, alongside general hypoactivation within the CEN itself 131927. The reduction in CEN-SN coupling indicates a severely impaired top-down cognitive control mechanism. Consequently, the patient cannot recruit the central executive resources necessary to willfully disengage from negative mood states, shift attention to goal-directed behavior, or suppress the overactive DMN 1328. In studies of adolescents with early-onset depression, the PCC demonstrates inflexibly elevated connectivity with the subcallosal cingulate during both resting and task states, indicating that early disease onset adversely impacts the normal developmental segregation of these networks 17.
| Network | Core Anatomical Regions | Primary Healthy Function | Major Depressive Alteration |
|---|---|---|---|
| Default Mode (DMN) | mPFC, PCC, Precuneus, Angular Gyrus | Self-referential thought, episodic memory, prospection 1. | Intrinsic hyperconnectivity; failure to deactivate during external tasks; hyper-coupling with limbic regions (sgACC) 171824. |
| Salience (SN) | Anterior Insula, Dorsal ACC, Amygdala | Stimulus detection, assigning biological relevance, network switching 19. | Hyperconnectivity with DMN; aberrant tagging of internal negative affect as highly salient 1319. |
| Central Executive (CEN) | dlPFC, Posterior Parietal Cortex | Working memory, problem-solving, cognitive control 14. | Hypoactivation; hypoconnectivity with SN; impaired top-down suppression of the DMN 1319. |
Frequency-Specific Effective Connectivity
Advanced effective connectivity (EC) analyses demonstrate that these triple-network causal dynamics are highly frequency-specific, suggesting complex, oscillatory disruptions in information flow. Research utilizing exact low-resolution electromagnetic tomography and magnetoencephalography reveals that causal influence from the SN to the DMN is significantly decreased in specific higher-frequency bands (e.g., 0.12 - 0.18 Hz), while the mean effective connectivity from the CEN to the SN increases abnormally in lower sub-frequency bands (0.04 - 0.08 Hz) 1528. This frequency-dependent disruption indicates that the brain's timing mechanisms for network recruitment are fundamentally desynchronized in MDD, contributing to both the affective and psychomotor symptoms characteristic of the disorder.
Large-Scale Global Neuroimaging Consortia Findings
The landscape of psychiatric neuroimaging has historically been hindered by small sample sizes, methodological variations, and low statistical power, leading to replication failures and an overestimation of effect sizes 2930. To establish definitive biomarkers of depression, the field has recently pivoted to large-scale, global data-sharing consortia. These massive datasets provide the statistical rigor required to map the structural and functional topology of MDD across diverse populations.
The ENIGMA Major Depressive Disorder Working Group
The Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) MDD Working Group comprises structural and functional MRI data from over 5,000 MDD patients and 9,000 healthy controls sourced from 45 cohorts across 14 countries and six continents 313233. ENIGMA analyses have identified highly robust structural alterations that correlate with DMN dysfunction.
Specifically, the consortium confirmed a replicable reduction in hippocampal volume and cortical thinning in the medial orbitofrontal cortex (mOFC) - both crucial nodes that interact intimately with the DMN and SN 313234. These structural deficits are highly correlated with illness duration and recurrence. Notably, ENIGMA analyses revealed that while small-scale studies frequently report DMN hyperconnectivity, meta-analyses of recurrent MDD patients actually demonstrate significantly decreased within-network DMN functional connectivity compared to both healthy controls and first-episode MDD patients 431. This pivotal finding suggests that DMN connectivity fluctuates dynamically over the disease course: acute, initial depressive episodes may drive hyperconnectivity through active rumination, while chronic, recurrent depression may eventually induce structural degradation and functional exhaustion within the network, resulting in hypoconnectivity and profound emotional blunting 41831.
The REST-meta-MDD Project and Cross-Cultural Insights
Concurrently, the REST-meta-MDD project, established by the Depression Imaging REsearch ConsorTium (DIRECT) in China, has pooled resting-state fMRI data from over 1,300 MDD patients and 1,128 controls across 25 cohorts 30. This consortium focuses heavily on functional brain metrics and topological properties.
Analyses of voxel-mirrored homotopic connectivity (VMHC) - a measure of interhemispheric functional integration - reveal significant reductions in the fusiform gyrus for mild to moderate depression, expanding into extensive reductions across the insula, postcentral gyrus, and angular gyrus in severe depression 35. Furthermore, REST-meta-MDD data has uncovered profound sex-related differences in functional connectivity. Male patients exhibit increased functional connectivity between the DMN and the frontoparietal network compared to female patients. Transcriptomic-neuroimaging analyses mapped these sex-specific connectivity differences to the expression of 1,777 genes, primarily those governing ion channel activity, synaptic plasticity, and neuronal differentiation 36.
The integration of these massive datasets into deep neural network (DNN) models has demonstrated immense predictive potential. Machine learning models utilizing frontolimbic and DMN dysconnectivity features have achieved up to 89% accuracy in diagnosing MDD and 78% sensitivity in predicting which at-risk individuals will develop the disorder over a two-year follow-up period 37.
Neurological Markers of Ruminative Thought
The clinical manifestation of DMN and Triple Network dysregulation is most directly observable through the psychological phenomenon of "rumination." Rumination is recognized as a central, transdiagnostic vulnerability factor in psychopathology, characterized by a repetitive, passive, and inescapable focus on one's negative emotions, their causes, and their consequences 383940.
The Response Styles Theory of Rumination
Susan Nolen-Hoeksema's foundational Response Styles Theory (RST) originally conceptualized rumination as a maladaptive response to distress. According to RST, when individuals face a discrepancy between their current state and a desired goal, or when exposed to uncontrollable stressors, they naturally reflect on the cause of their negative affect 404142. If this reflection leads to actionable problem-solving, the individual engages in active coping and the negative mood dissipates. However, in vulnerable individuals, this cognitive process becomes perseverative 41. The brain fails to transition from internal evaluation to external instrumental action. Instead, rumination prolongs depressive episodes by enhancing negative mood-congruent thinking, impairing executive problem-solving, and eroding social support networks 383941.
Maladaptive Brooding Versus Adaptive Reflection
Subsequent factor analyses of the Ruminative Responses Scale (RRS) refined RST by partitioning rumination into two distinct cognitive components with divergent clinical outcomes: brooding and reflection 434445.
Ruminative Brooding is a passive, self-absorbed process involving a fatalistic comparison of one's current situation with unachieved standards. It involves dwelling on the negative consequences of depression without formulating solutions. Brooding is strictly maladaptive; longitudinal studies confirm it consistently predicts the onset, severity, and duration of depressive episodes, as well as significantly elevating the risk for suicidal ideation 434446.
Ruminative Reflection, conversely, is a purposeful, self-distanced cognitive process aimed at understanding distress to facilitate problem-solving. In non-clinical, healthy populations, reflection serves as an adaptive emotion-regulation strategy that precedes active coping and predicts subsequent reductions in dysphoria 434647. However, in populations currently experiencing a major depressive episode, the distinction between brooding and reflection blurs. Because of the systemic deficits in the Central Executive Network, depressed patients attempting adaptive reflection quickly devolve into maladaptive brooding 4348.
Neural Substrates of Brooding and Reflection
Neuroimaging explicitly differentiates the neural substrates of brooding and reflection, linking these psychological constructs directly to the Triple Network Model 4349.
Brooding relies heavily on the hyper-integration of the DMN and the Salience Network. Elevated brooding scores correlate strongly with increased resting-state functional connectivity between the core nodes of the DMN (mPFC and PCC) and between the anterior insula (SN) and the parahippocampal gyrus 124349. In adolescent populations, heightened SN coherence serves as a neurodevelopmental marker that mediates the relationship between brooding and internalizing symptoms, particularly in early pubertal females 49. Furthermore, brooding involves abnormal connectivity between the amygdala and the temporal pole, reinforcing negative affective states in the absence of cognitive oversight 43.
Reflective rumination, in contrast, relies heavily on the coupling between the DMN and the Frontoparietal Network (CEN/FPN). While the DMN is required to direct attention internally to evaluate the self, the Central Executive Network must be robustly engaged to maintain cognitive distance, organize thoughts instrumentally, and prevent a descent into passive despair 43. Reflection is characterized by enhanced functional connectivity between the insula and the dlPFC, indicating active, top-down executive control over internal emotional representations 43. In depression, the hypoconnectivity and hypoactivation of the CEN effectively eliminate the patient's biological capacity for reflection, leaving the pathological brooding pathway as the default response to distress 2843.
Network Neuromodulation and Therapeutic Interventions
Because major depressive disorder is fundamentally driven by network rigidity and aberrant functional connectivity, successful therapeutic interventions must induce large-scale network reorganization, effectively "resetting" the communication between the DMN, SN, and CEN.
Conventional Pharmacotherapy and Electroconvulsive Therapy
Traditional interventions selectively modulate specific network interactions. Standard selective serotonin reuptake inhibitors (SSRIs), such as escitalopram, appear to act via network dampening. Response to escitalopram is associated with decreased linear connectivity within the DMN and Salience networks, and reduced connectivity between the DMN and the Ventral Attention Network (VAN) 50. By dampening self-referential processing and salience tagging, SSRIs effectively blunt the intensity of depressive rumination, though they often leave patients reporting emotional flattening 50.
Electroconvulsive therapy (ECT), highly effective for treatment-resistant depression (TRD), induces more profound structural and functional network resets. Source-based morphometry and resting-state fMRI reveal that ECT rapidly elevates previously suppressed Salience Network structural connectivity and restores dynamic functional connectivity between the dmPFC and the PCC 5152. The ability of ECT to forcibly reorganize DMN-SN-CEN causal dynamics correlates strongly with the remission of severe affective and psychomotor symptoms 5152.
Psychedelic Interventions and Network Desegregation
The most significant recent paradigm shift in treating network dysregulation involves classic psychedelics (e.g., psilocybin, LSD, DMT). Psychedelics exert their primary pharmacological effects via agonism of the serotonin 5-HT2A receptor. These receptors are densely expressed on layer 5 pyramidal neurons in the transmodal association cortex - precisely the anatomical substrate of the Default Mode Network 2154.
During the acute psychedelic state, 5-HT2A agonism results in asynchronous glutamate release and a subsequent loss of synchronous gain across neural ensembles 21. This massively disrupts the hierarchical organization of the brain 5354. The disruption manifests as a profound "desegregation" of established brain networks: within-network connectivity (the rigid, rumination-driving coupling of the DMN) drops precipitously, while between-network connectivity (communication across normally distinct networks) increases globally 5556. The brain transitions into an "entropic," highly flexible state, characterized by a loss of modularity and the complete dissolution of the rigid boundaries separating the DMN, SN, and CEN 215758.
Recent precision functional mapping using longitudinal fMRI demonstrates that psilocybin produces neurobiological changes that significantly outlast the acute experience 5357. Specifically, psilocybin induces a persistent decrease in functional connectivity between the anterior hippocampus and the DMN, an effect that remains highly significant for weeks following a single high dose (25mg) 535459. This persistent decoupling of the hippocampus from the DMN represents a neuroanatomical correlate for the drug's antidepressant effects, effectively uncoupling the brain's raw episodic memory storage from the pathological rumination engine, allowing patients to access autobiographical memories without triggering the automatic, rigid descent into brooding 5460.
Clinical Efficacy of Psilocybin
These network-level alterations translate to significant clinical efficacy. Clinical trials evaluating synthetic psilocybin (COMP360) for Treatment-Resistant Depression demonstrate that a single, high dose can induce rapid and durable remission.
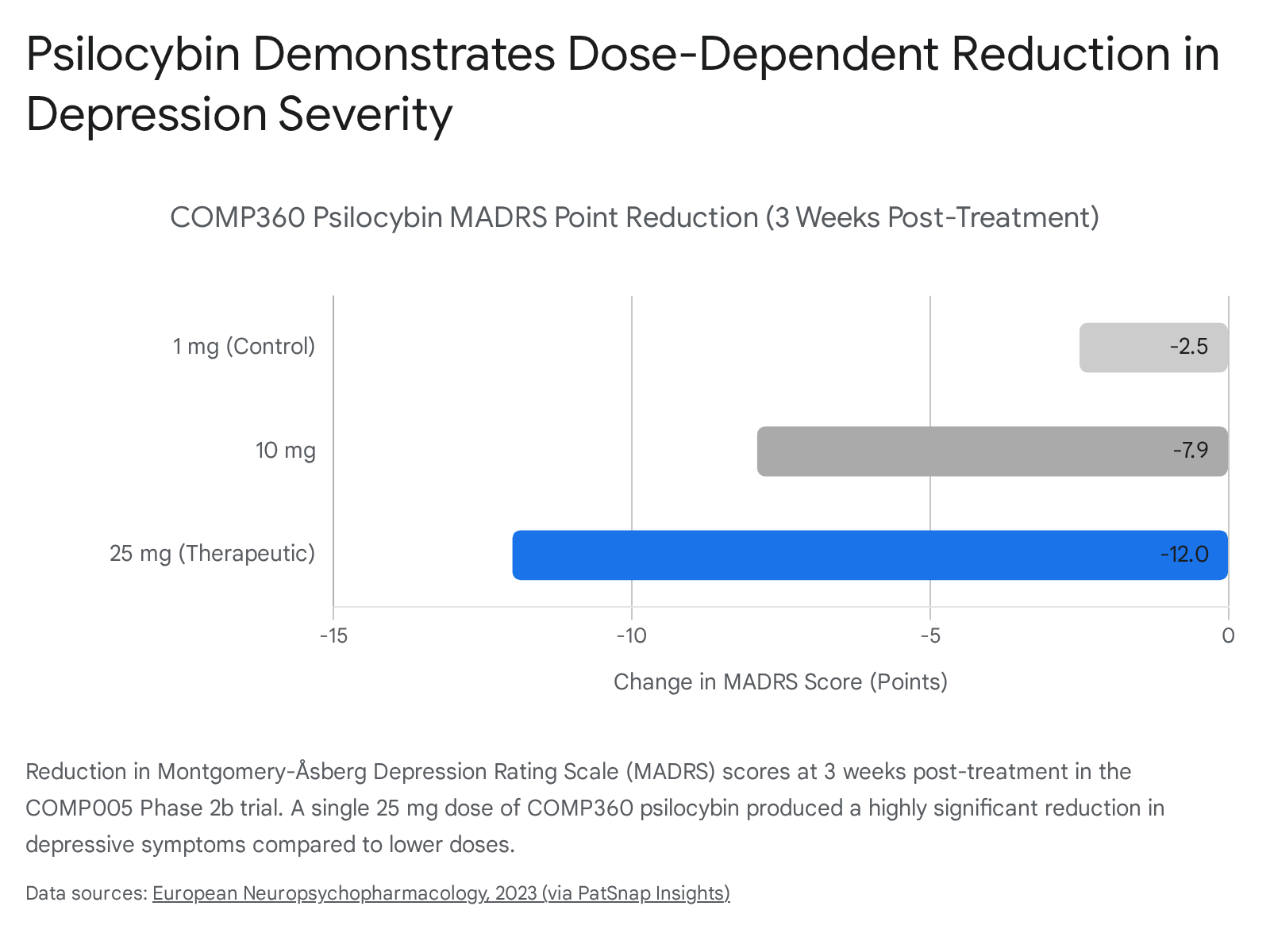
In the COMP006 Phase 3 trial encompassing 581 participants, a 25 mg dose administered three weeks apart demonstrated a highly statistically significant reduction in symptom severity compared to a 1 mg control, with rapid onset observed the day following administration and durable effects maintained through six weeks 6162. The ability of psilocybin to temporarily collapse the modularity of the DMN correlates directly with patient self-reports of "ego dissolution" and psychological flexibility, providing a window of opportunity to revise entrenched patterns of thought 5763.
The Neuroplasticity and Subjective Experience Debate
A major frontier in contemporary psychiatric neuroscience is the ongoing debate regarding the exact mechanism of psychedelic efficacy: is the subjective, entropic experience (the "trip") strictly necessary for therapeutic benefit, or is structural neuroplasticity the sole underlying driver? 6465.
The classical neurocognitive view, heavily influenced by predictive processing models, posits that the acute psychological experience - specifically the dissolution of the ego (a phenomenological correlate of DMN collapse) and the induction of a "mystical" state - is required to flatten the brain's free-energy landscape and break the overly precise, rigid cognitive priors of the depressed brain 216364. Studies indicate that the degree of acute cortical signal entropy (measured via EEG) directly predicts the magnitude of psychological well-being improvements one month later 63.
Conversely, the "psychoplastogen" hypothesis argues that the trip may be an epiphenomenon. Preclinical research demonstrates that psychedelics rapidly promote structural neuroplasticity (e.g., spinogenesis, dendritogenesis) and reopen critical periods of social learning by modulating extracellular matrix integrity and restoring neuronal sensitivity to oxytocin 6566. This occurs even in animal models incapable of human-like abstract rumination. If structural plasticity is the primary mechanism of action, novel engineered non-hallucinogenic analogues (such as tabernanthalog) could provide the network-resetting benefits of classical psychedelics without inducing intense hallucinogenic states or requiring extensive, costly psychological monitoring 64.
Ultimately, decoding the intricate dynamics of the Default Mode Network not only elucidates the pathophysiology of major depressive disorder but also provides profound insights into the neurological basis of human self-awareness. By targeting the dysfunctional interactions between the networks that govern our internal reality and our engagement with the external world, precision psychiatric therapies promise to restore the cognitive flexibility fundamentally lost in depression.