Compression of morbidity in longevity science
Theoretical Frameworks of Population Health Dynamics
The primary objective of modern public health and applied longevity science extends beyond the mere prolongation of chronological lifespan; the focus is increasingly on the maximization of healthspan. Healthspan is defined as the period of life spent free from chronic disease and debilitating functional disability. This paradigm is conceptually anchored in the "compression of morbidity" hypothesis, first articulated by James Fries in 1980 12. The hypothesis proposes that if the onset of chronic infirmity can be delayed at a faster rate than the increase in total life expectancy, the cumulative lifetime burden of illness will be compressed into a shorter, highly predictable period immediately preceding death 23.
The Fries hypothesis stands in direct theoretical contrast to alternative models of population health dynamics that emerged during the late twentieth century. The "expansion of morbidity" theory, posited by Gruenberg in 1977, argued that medical and technological advancements primarily function to prevent acute death from chronic and infectious diseases without necessarily curing the underlying pathologies 14. According to this framework, individuals survive longer but spend those additional chronological years in states of prolonged chronic illness, thereby expanding the population's overall "sickspan" 34.
A third theoretical framework, the "dynamic equilibrium" model introduced by Kenneth Manton in 1982, offers a nuanced synthesis of these extremes. Manton suggested that while the absolute prevalence of chronic morbidities may rise as populations age, the severity of these diseases and their deterministic linkage to severe functional disability may systematically weaken 14. Medical interventions, earlier diagnostic capabilities, and chronic disease management protocols reduce the rate of disease progression. This results in an equilibrium state where individuals live longer with chronic conditions, but their proportion of life spent with severe functional disability remains constant or actively declines 15.
Methodological Approaches to Measuring Survival and Morbidity
To empirically evaluate these theories, researchers employ complex mathematical modeling and survival analysis techniques. The Kaplan-Meier estimator is a foundational statistical approach used to graphically represent the survival function of a population over time 67. In a standard Kaplan-Meier curve, time is plotted on the horizontal x-axis, and the probability of survival is plotted on the vertical y-axis 786. At time zero, the survival probability is 1.0 (100%), and the curve descends as mortality events occur 8.
In the context of morbidity compression, the shape of the survival curve is highly indicative of healthspan outcomes. Theoretical models, such as the saturating removal (SR) model of aging, demonstrate that interventions which "steepen" the survival curve - creating a flatter top that drops sharply at advanced ages, often referred to as the "squaring of the survival curve" - effectively compress morbidity in relative terms 310. Conversely, interventions that merely shift the survival curve to the right without changing its fundamental shape exhibit "scaling." Scaling extends both lifespan and healthspan proportionally, failing to compress the period of morbidity in absolute or relative terms 310. Longitudinal tracking in model organisms, such as Caenorhabditis elegans and Drosophila melanogaster, frequently reveals scaling effects rather than true compression, underscoring the biological difficulty of restricting the sickspan 310.
Evaluating the Evidence in Human Populations
Recent empirical analyses of human population data reveal a complex reality that does not uniformly support a single theoretical framework. A 2023 multidimensional analysis of United States birth cohorts spanning from 1998 to 2018 demonstrated patterns that closely align with Manton's dynamic equilibrium theory 1. Researchers found that across all age groups, the years of life expected to be entirely free from morbidity declined significantly, indicating that the prevalence of chronic diseases has expanded 1. However, disability-free life expectancy remained remarkably stable across successive cohorts 1. This suggests that advancements in healthcare have successfully weakened the deterministic link between morbidity and disability, allowing older adults to maintain functional independence despite harboring chronic conditions 15.
Conversely, studies measuring subjective health metrics, such as pain morbidity, provide strong evidence for the expansion of morbidity. Analysis of the US Health and Retirement Study (1993 - 2018) found that for adults in their 70s and 80s, gains in total life expectancy were predominantly driven by increases in years lived with nonlimiting and limiting pain, while the absolute number of pain-free years remained stagnant or declined 5. For example, a 70-year-old man in 1993 could expect 81.2% of his remaining life to be pain-free, but by 2016, this figure had dropped to 60.5% 5. This divergence underscores that while severe functional disability may be held in check (equilibrium), subjective quality-of-life impairments are expanding alongside lifespan 5.
Global Trends in Health-Adjusted Life Expectancy
To quantify population-level healthspan and compare international progress toward morbidity compression, the World Health Organization (WHO) utilizes the Health-Adjusted Life Expectancy (HALE) metric. HALE integrates both mortality data and the burden of non-fatal health outcomes, providing a summary measure of the average number of years a person can expect to live in "full health," adjusted for time spent in states of less-than-full health due to disease or injury 78.
Pre-Pandemic Baselines and the Morbidity Gap
Prior to the systemic shock of the COVID-19 pandemic, global health metrics exhibited a continuous, century-long upward trajectory. Between 2000 and 2019, global life expectancy at birth increased by 6.4 years, rising from 66.8 years to 73.1 years 910. Concurrently, global HALE increased by 5.3 years, moving from 58.1 to 63.5 years 9.
However, a critical analysis of this data reveals an underlying expansion of morbidity at the global level. Because the absolute gains in total life expectancy (6.4 years) outpaced the gains in healthy life expectancy (5.3 years), the global mean healthspan-lifespan gap - the absolute period of life characterized by morbidity - widened 911. In 2000, the global gap was 8.5 years; by 2019, it had expanded to 9.6 years 11. The gains in HALE were primarily driven by declining mortality rates across all age groups rather than a substantive reduction in years lived with disability 9.
Impact of the COVID-19 Pandemic on Global Longevity
The COVID-19 pandemic caused an unprecedented disruption to global longevity trends, exposing the fragility of public health progress. According to the 2024 WHO World Health Statistics report, the pandemic effectively wiped out nearly a decade of progress in just two years 12. By 2021, global life expectancy had plummeted by 1.8 years to 71.4 years, returning the global average to 2012 levels 12. Similarly, global HALE dropped by 1.5 years to 61.9 years 12.
The impact was distributed highly unequally across global regions. The WHO regions for the Americas and South-East Asia experienced the most severe healthspan contractions, with life expectancy dropping by approximately 3 years and HALE by 2.5 years between 2019 and 2021 12. In contrast, the Western Pacific Region absorbed the shock more effectively, with minimal losses of less than 0.1 years in life expectancy and 0.2 years in HALE 12. While noncommunicable diseases (NCDs) remained the predominant global killer - accounting for 74% of all deaths pre-pandemic and 78% of non-COVID deaths during the pandemic - COVID-19 rapidly emerged to become the leading cause of death in the Americas for both 2020 and 2021 12.
Comparative Healthspan Across Benchmark Nations
The realization of morbidity compression is highly uneven when examining specific national contexts. The discrepancy between total life expectancy and HALE illustrates the localized realities of disease burden, healthcare efficacy, and lifestyle factors.
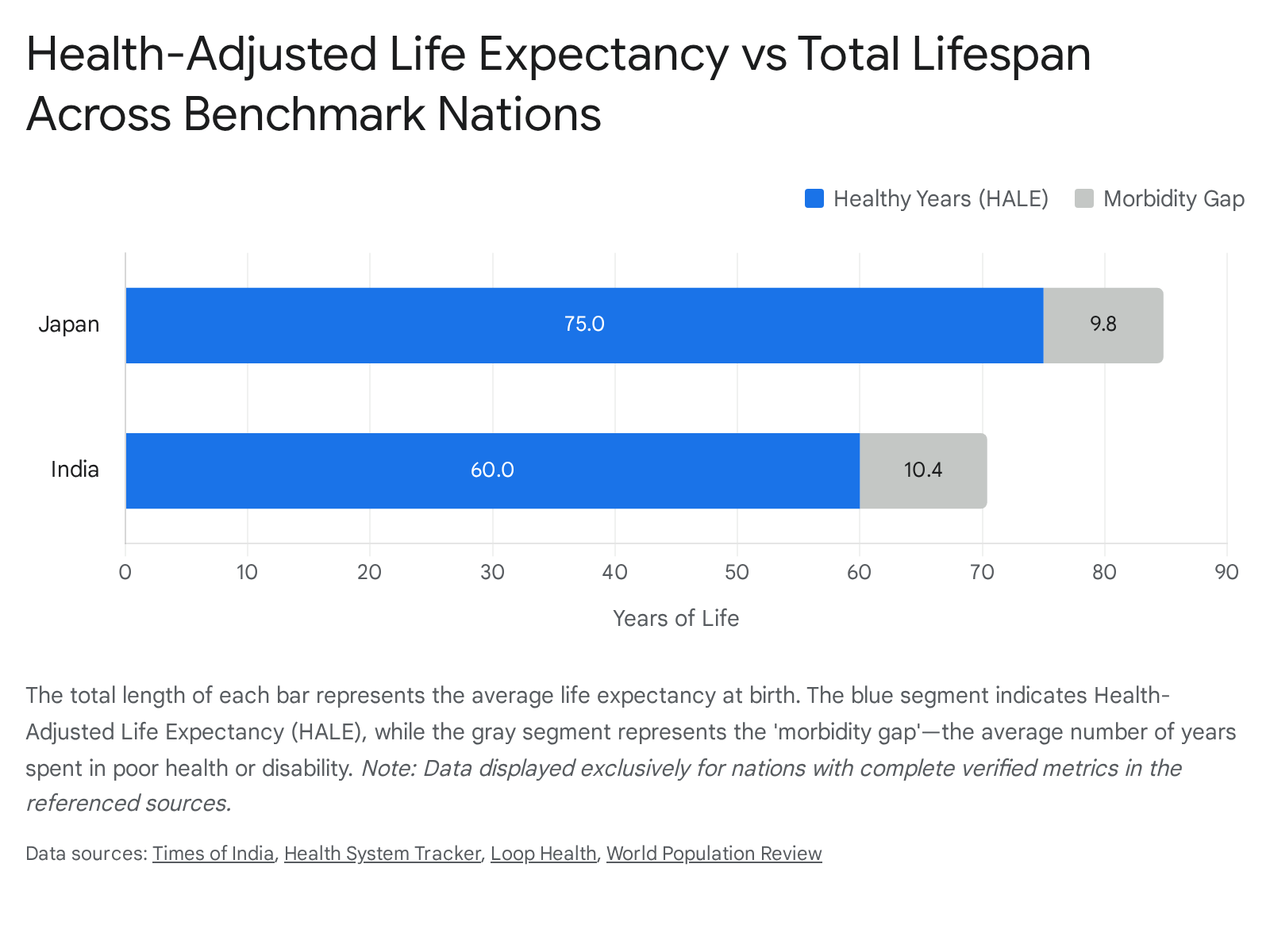
| Country | Estimated Life Expectancy (LE) | Estimated HALE | Approximate Morbidity Gap | Public Health Context |
|---|---|---|---|---|
| Japan | 84.8 years | 75.4 years | 9.4 years | High LE and highly compressed morbidity. Driven by universal healthcare, low obesity, and dietary factors 131415. |
| United States | 79.0 years | 66.6 years | 12.4 years | Highest OECD healthcare spending, yet significant morbidity gap driven by chronic diseases, obesity, and unequal access 1116. |
| India | 73.0 years | 60.0 years | 13.0 years | Sharp decline in HALE after age 60 (only 11 healthy years remaining) driven by noncommunicable diseases 1722. |
| Nigeria | 54.6 years | 47.0 years | 7.6 years | Low absolute LE due to communicable diseases, high infant mortality, and infrastructural health deficits 15. |
Japan represents the current demographic zenith of morbidity compression. With a life expectancy approaching 85 years, the Japanese population benefits from high functional independence in later life 131415. This is sustained by a combination of universal healthcare coverage, high GDP per capita, culturally integrated physical activity, and diets low in ultra-processed foods 1315.
In stark contrast, the United States exhibits an elevated healthspan-lifespan gap of approximately 12.4 years, which is 29% higher than the global mean 11. Despite spending nearly twice as much per capita on healthcare as comparable OECD peers, US life expectancy in 2024 (79.0 years) remained 3.7 years below the comparable country average (82.7 years) 16. The American healthcare system's orientation toward acute disease management rather than preventative geroscience has resulted in a population that survives fatal acute events but lives for over a decade with high burdens of chronic noncommunicable diseases, exacerbated by high obesity rates and systemic inequalities in care access 161718. Furthermore, the US exhibits a persistent gender gap; life expectancy for American women is 4.9 years longer than for men, slightly wider than the 4.2-year gap seen in peer countries 16.
In emerging economies like India, the burden of morbidity strikes much earlier in the life course. While Indian life expectancy has seen massive gains - increasing by 27 years since 1965 to reach over 70 years today - HALE at birth remains at approximately 60 years 1722. More critically, HALE at age 60 drops to just 11 years, meaning the average Indian adult faces the onset of chronic illness, disability, or reduced physical function by age 71 22. This compression failure is heavily driven by a rising prevalence of working-age NCDs, including cardiovascular disease, diabetes, and musculoskeletal disorders, resulting in a disease burden 2.5 to 3 times higher than global healthspan leaders 22. Sub-Saharan African nations, such as Nigeria, face a dual burden. While dealing with rising NCDs due to rapid urbanization and dietary shifts, their absolute life expectancy is still severely depressed (approx. 54.6 years) due to high infant mortality and persistent communicable diseases 1519.
Socioeconomic Stratification of Morbidity Compression
The theoretical debate over whether morbidity is universally compressing or expanding often overlooks a crucial, deterministic variable: socioeconomic status (SES). Global and national data overwhelmingly indicate that the compression of morbidity is currently a highly stratified phenomenon. It is realized primarily by affluent and highly educated cohorts, while lower-income populations experience a severe expansion of morbidity, deep lifespan inequality, and premature mortality 1202122.
Income Disparities and Lifespan Inequality
In the United States, analyses of geographic and income data expose stark mortality divides that are rapidly widening. A 2025 congressional report utilizing median household income data revealed that individuals residing in the top 1% of US counties live an average of 84.3 years, whereas those in the bottom 50% average just 77.4 years - a sweeping seven-year geographic penalty 22. When examining specific demographic extremes, the gap is even more severe. Men in the top income decile outlive those in the bottom decile by 15 years, and women in the top decile outlive their poorest counterparts by 10 years 2324.
The Southern Community Cohort Study, tracking nearly 86,000 participants in the US, found a strong dose-response relationship between household income and mortality 25. Individuals in the lowest income group (under $15,000/year) exhibited a 3.3-fold increased risk of all-cause mortality compared to those earning over $50,000 25. Life expectancy was more than 10 years shorter for the lowest income group 25. Crucially, low-income cohorts are not only dying younger, but they also spend a substantially larger fraction of their shorter lives in poor health 21. Data from Great Britain indicates that the gap in healthy life expectancy between the most and least deprived areas is approximately 19 years 21.
On a global scale, the United Nations World Population Prospects 2024 data highlights extreme lifespan inequality. In 2023, the global average age at death for the youngest 20% of deaths was just 18 years, while the oldest 20% died at an average age of 89 32. In 35 low- and lower-middle-income countries, the average age at death for the lowest quintile remains under 1 year of age 32. In these environments, there is zero mortality compression; deaths occur uniformly across the lifespan rather than being concentrated in old age 32.
Educational Attainment and Multimorbidity Accumulation
Educational attainment has emerged as a predictor of morbidity compression that rivals or exceeds demographic indicators like race. In the US, holding a bachelor's degree significantly delays the onset of functional disability, allowing highly educated cohorts to spend more years without chronic morbidities 1. By 2020, educational attainment had become a stronger predictor of life expectancy than race in the US .
High social and material deprivation correlates directly with a faster accumulation of multimorbidity (the presence of two or more chronic conditions) 26. A 2024 analysis of community-based health center patients in the US found that individuals residing in socially deprived areas had higher initial chronic disease counts 26. Interestingly, patients who were continuously or discontinuously insured had higher recorded chronic disease counts than the continuously uninsured, highlighting a dangerous "reservoir" of undiagnosed chronic disease among those lacking coverage 26. Factors driving this disparity include systemic barriers to healthcare access, reliance on low-quality diets, higher environmental stress, and lower health literacy 193427. For low-income patients, survival often becomes a financial negotiation, leading to delayed medical care and the exacerbation of preventable complications 27.
Geographic Isolation and Rurality Indices
Geographic isolation acts as an independent socioeconomic variable that actively prevents morbidity compression. In the US, small rural counties with median household incomes of $30,000 exhibit average life expectancies of 71.7 years, presenting a 10-year deficit compared to affluent urban and suburban regions earning $100,000 22. A mere $10,000 increase in median annual household income in a rural county is associated with an additional 2.6 years of life expectancy 22.
Similar spatial inequalities are observed even in nations with robust universal healthcare. In Japan, researchers utilizing the Rurality Index for Japan (RIJ) analyzed 1,897 municipalities covering 126 million people 2829. They found that greater rurality directly correlates with higher standardized mortality ratios (SMRs) for cerebrovascular diseases and male suicide 282930. A distinct dose-response relationship was established between rural isolation and these specific mortality outcomes 29. The inability to access rapid emergency interventions, advanced preventative care, and specialized psychiatric services in rural areas restricts these populations from achieving the morbidity compression seen in Japan's major urban centers 2830.
Clinical Progress in Longevity Interventions
While public health infrastructure and socioeconomic policy represent the macro-strategy for achieving morbidity compression, molecular longevity science - or geroscience - represents the micro-strategy. Geroscience posits a fundamental paradigm shift: rather than treating age-related chronic diseases (e.g., cancer, Alzheimer's disease, cardiovascular dysfunction) sequentially and reactively, medicine must target the underlying biological "hallmarks of aging" 1839. By modifying fundamental mechanisms like cellular senescence, epigenetic alterations, mitochondrial dysfunction, and deregulated nutrient sensing, interventions aim to delay the onset of all age-related comorbidities simultaneously 183132.
Recent years (2024 - 2026) have witnessed the critical transition of longevity therapeutics from academic animal models to active human clinical trials, offering the first empirical evidence of biological morbidity compression in humans 3343.
Senolytics and Cellular Clearance Mechanisms
Cellular senescence occurs when cells permanently cease to divide due to stress or telomere attrition, yet resist apoptosis 3934. These dormant cells accumulate with age and secrete a toxic, pro-inflammatory cocktail of cytokines, chemokines, and proteases known as the senescence-associated secretory phenotype (SASP) 393234. Senolytic therapies aim to selectively induce apoptosis in these senescent cells, thereby reducing systemic tissue inflammation and restoring native regenerative capacity 3243.
Human clinical trials of first-generation senolytics have demonstrated highly promising results. In pilot studies, the intermittent oral administration of Dasatinib (a tyrosine kinase inhibitor) combined with Quercetin (a plant flavonoid) - commonly referred to as D+Q - reduced the burden of p16-positive senescent cells in adipose tissue and skin within three months 3932. In patients with diabetic kidney disease and idiopathic pulmonary fibrosis, D+Q lowered circulating SASP factors and measurably improved physical function 3932. Another natural flavonoid, Fisetin, has shown potential in preserving cognitive function and reducing neuroinflammation in models of Alzheimer's disease, occasionally outperforming D+Q in specific genotypes 32.
Furthermore, established pharmaceutical agents are being repurposed via the geroscience lens. SGLT2 inhibitors, traditionally utilized for glucose control in diabetics, have recently been found to reduce senescence markers in humans 45. This discovery links their well-documented cardiovascular and renal protective effects directly to aging biology, suggesting their benefits extend far beyond glycemic regulation to the fundamental mechanisms of morbidity compression 45.
mTOR Inhibition and Nutrient Sensing Modulation
The mechanistic target of rapamycin (mTOR) pathway serves as a central biological regulator of cellular metabolism, growth, and autophagy (the cellular waste-clearance process) 3243. Hyperactive mTOR signaling in later life is intrinsically linked to a loss of proteostasis and deregulated nutrient sensing, driving age-related decline 32. Inhibiting mTOR complex 1 (mTORC1) with rapamycin, or its analogs (rapalogs) like everolimus, has consistently extended both lifespan and healthspan across multiple model organisms 1834.
Recent human translational research has focused on intermittent, low-dose administration protocols to secure the geroprotective benefits while avoiding the severe immunosuppressive side effects observed when high doses are used for organ transplant recipients 1834. A 2025 pilot clinical study involving healthy older men demonstrated that an 8-week regimen of 1 mg daily rapamycin significantly improved cardiovascular and endothelial function 45. The cardiovascular improvements materialized within months, much faster than could be attributed to metabolic shifts alone 45. Similarly, a clinical trial involving 218 adults over age 65 found that a 6-week course of everolimus safely and significantly enhanced immune responses to the influenza vaccination 18. These early human indicators suggest that mTOR inhibitors possess potent senomorphic capabilities - meaning they can suppress SASP secretions and delay functional decline without necessarily inducing cell death 3234.
This mirrors the fundamental biological effects of caloric restriction, the most robust, non-pharmacological life-extension intervention. The CALERIE trial, which randomized non-obese adults to a 2-year caloric restriction intervention, demonstrated that reduced caloric intake successfully upregulated autophagy and DNA repair mechanisms while downregulating inflammatory responses, confirming the plasticity of nutrient-sensing pathways in humans 18.
GLP-1 Receptor Agonists and Metabolic Health
Presently, the most globally impactful pharmaceutical agents driving incidental morbidity compression are glucagon-like peptide-1 (GLP-1) and dual GLP-1/GIP receptor agonists, such as semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound) 353637. Originally developed to stimulate insulin release and slow gastric emptying for glycemic control in type 2 diabetes, their profound secondary effects on weight regulation and systemic metabolic health have unmasked a cascading series of healthspan benefits 353649.
Extensive 2024 and 2025 clinical evidence reveals that these therapies actively compress morbidity across multiple organ systems. Beyond reducing major adverse cardiovascular events (MACE) and slowing chronic kidney disease (CKD) progression, real-world observational studies have highlighted striking secondary preventative effects 3549. For example, GLP-1 users experience a roughly 50% reduction in the risk of obesity-related cancers compared to matched cohorts undergoing usual care, a protective effect that appears even stronger than traditional bariatric surgery 3738.
The SURMOUNT trials demonstrated that dual-action agonists like tirzepatide yield extraordinary weight reduction (averaging 20.2% body weight loss) and improved cardiometabolic profiles 49. Furthermore, these agents have demonstrated unexpected efficacy in mitigating obstructive sleep apnea symptoms, lowering rates of vertebral and lower-extremity fractures, and even reducing alcohol consumption in patients with alcohol use disorder 3738. By systematically resolving the metabolic dysregulation that underpins systemic inflammation, GLP-1 agonists represent the most widely deployed, clinically validated tools for extending the healthy period of late mid-life 3536.
Epigenetic Reprogramming and Future Horizons
The most ambitious frontier of longevity science lies in epigenetic reprogramming, a theoretical approach that seeks to revert aged cells to a younger functional state by resetting their DNA methylation patterns without altering the underlying genetic code 393339. Pioneered via the introduction of transcription factors (Yamanaka factors), partial reprogramming has successfully reversed aging phenotypes and restored youthful tissue function in murine models 3952.
In a significant regulatory milestone, the FDA greenlit the first human clinical trials for a gene therapy aimed at epigenetic cellular age reversal in early 2026 3952. Led by biotech firms utilizing advanced delivery mechanisms, these trials will test whether epigenetic programming therapies can safely restore human cells to a youthful state without inducing oncogenesis (cancer formation) 3952. If successful, epigenetic reprogramming offers the potential to transcend morbidity compression entirely, moving the field into active morbidity reversal 39.
Systemic Barriers to Scaling Longevity Therapeutics
Despite extraordinary scientific optimism and expanding venture capital investment (which reached $8.5 billion in 2024), the broad population-level realization of morbidity compression faces severe translational, regulatory, and systemic hurdles 393353.
First, regulatory frameworks remain an active impediment. Global health authorities, including the FDA, do not classify "aging" as a curable disease or a valid primary indication for drug approval 3339. Consequently, longevity therapeutics must be trialed against specific, isolated age-related diseases - such as osteoarthritis, macular degeneration, or specific cancers - rather than being evaluated for their holistic, preventative capacity to extend overall healthspan 3233. This forces researchers to conduct fragmented, highly specific trials rather than broad preventative longevity studies.
Second, the validation of reliable, real-time biological markers is incomplete. While "aging clocks" (epigenetic methylation panels), telomere length measurements, and multi-omic profiling offer unprecedented insights, their sensitivity to short-term interventions and their precise predictive correlation to long-term clinical morbidity in humans remain highly debated 3133. Without robust, universally accepted, and standardized biomarkers of aging, proving the efficacy of a preventative longevity drug in a reasonable clinical timeframe is prohibitively difficult, as researchers cannot wait decades for natural mortality endpoints 3132. Additionally, the complex manufacturing protocols for advanced therapies like stem-cell derived exosomes face immense hurdles in batch-to-batch consistency and scalable purification 31.
Finally, the economics of longevity science risk exacerbating the profound socioeconomic stratification discussed earlier. The US healthcare workforce is already facing a critical shortage, with an estimated deficit of 121,900 physicians by 2030, and only 7,400 practicing geriatricians available compared to the 30,000 required to manage the aging demographic . Advanced longevity therapies - such as individualized gene therapies, continuous biomarker tracking via proprietary wearables, and precision senolytics - are likely to be highly expensive upon initial commercialization 3133. If access is restricted to high-income populations with premium health coverage, the primary public health outcome will not be universal morbidity compression. Instead, it will catalyze a hyper-stratified society where the wealthy experience dynamic equilibrium and radical healthspan extension, while lower socioeconomic tiers continue to suffer from the expansion of multi-morbidities and premature mortality 24252740.
Conclusion
The compression of morbidity remains the ultimate objective of both traditional public health infrastructure and vanguard longevity science. Current global health statistics indicate that at a macro level, absolute chronological lifespan has historically expanded faster than healthspan, leading to an absolute expansion of time spent with chronic diseases. However, the severity of these diseases is increasingly managed by advanced pharmacology, driving a state of dynamic equilibrium where populations live longer with illness but without commensurate increases in severe, life-limiting functional disability.
Crucially, the success of morbidity compression is profoundly unequal, heavily concentrated among high-income, highly educated populations in developed nations. While the clinical validation of senolytics, mTOR inhibitors, and GLP-1 agonists suggests that targeted biological intervention can effectively delay age-related decline, the true test of longevity science will not only be its molecular efficacy, but its economic scalability. Achieving a population-wide compression of morbidity will require integrating these biotechnological advances with systemic public health initiatives that address the fundamental socioeconomic determinants of health, ensuring that the final decades of life are characterized by vitality rather than prolonged medical management.