Comparison of lifespan and healthspan
Introduction
Over the past century, public health initiatives, sanitation improvements, and the advent of modern medicine have generated an unprecedented extension of the human lifespan. Global life expectancy at birth has increased by decades, fundamentally altering the demographic structure of populations worldwide. However, the systematic conquest of acute, infectious, and parasitic diseases has unveiled a complex epidemiological paradox: the additional years of life are predominantly characterized by chronic, degenerative conditions. As modern medicine continues to excel at delaying mortality, it increasingly prolongs the period of late-life morbidity.
This divergence between lifespan - the absolute number of years lived - and healthspan - the duration of life spent in robust health, free from chronic disease and functional disability - has emerged as the defining clinical and socio-economic challenge of the twenty-first century. A growing consensus within epidemiology and geroscience suggests that the primary metric of medical progress must shift from extending survival to compressing morbidity. The pursuit of longevity without a proportional increase in functional capacity threatens to overwhelm healthcare infrastructure, drain economic resources, and significantly diminish the quality of life for aging populations. This report examines the exhaustive epidemiological data surrounding the healthspan-lifespan gap, evaluates the theoretical frameworks governing morbidity trends, and explores emerging functional metrics and pharmacological interventions designed to optimize the final decades of human life.
The Epidemiological Landscape of Aging
Global Life Expectancy Trends
The Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2023 provides the most comprehensive and updated evaluation of mortality and morbidity trends worldwide. Despite the massive, acute disruptions caused by the COVID-19 pandemic - which resulted in 18 million excess deaths between 2019 and 2023 - demographic data indicate that global life expectancy (LE) largely rebounded by 2023 123. Global life expectancy returned to pre-pandemic levels, reaching 76.3 years for females and 71.5 years for males 24. This represents an aggregate increase of more than 20 years compared to 1950, driven by a remarkable 66.6% decline in the global age-standardized all-cause mortality rate over the same period 2.
The global all-cause mean age at death increased significantly from 46.8 years in 1990 to 63.4 years in 2023 5. For males, the mean age at death increased from 45.4 years to 61.2 years, and for females, it increased from 48.5 years to 65.9 years 5. In high-income super-regions, the mean age at death for females reached 80.9 years, and 74.8 years for males in 2023, reflecting the peak of modern survival trajectories 5. Total global deaths in 2023 reached 60.1 million, an absolute increase driven by considerable population growth and aging, despite the sharp declines in standardized mortality rates 2.
The Healthspan Discrepancy
While mortality rates have plummeted, metrics tracking the quality of those additional years reveal a concerning lag. Healthy Life Expectancy (HALE) - a metric utilized by the World Health Organization to summarize the expected number of years lived in full health, adjusted for disease severity and disability - has not increased at the same velocity as raw life expectancy 678. Current estimates suggest that the global average individual spends approximately 20% of their life in states of poor health or functional limitation 9.
Between 1995 and 2017, global average LE increased from 66.20 to 72.98 years, while global average HALE increased from 57.59 to 63.25 years 10.
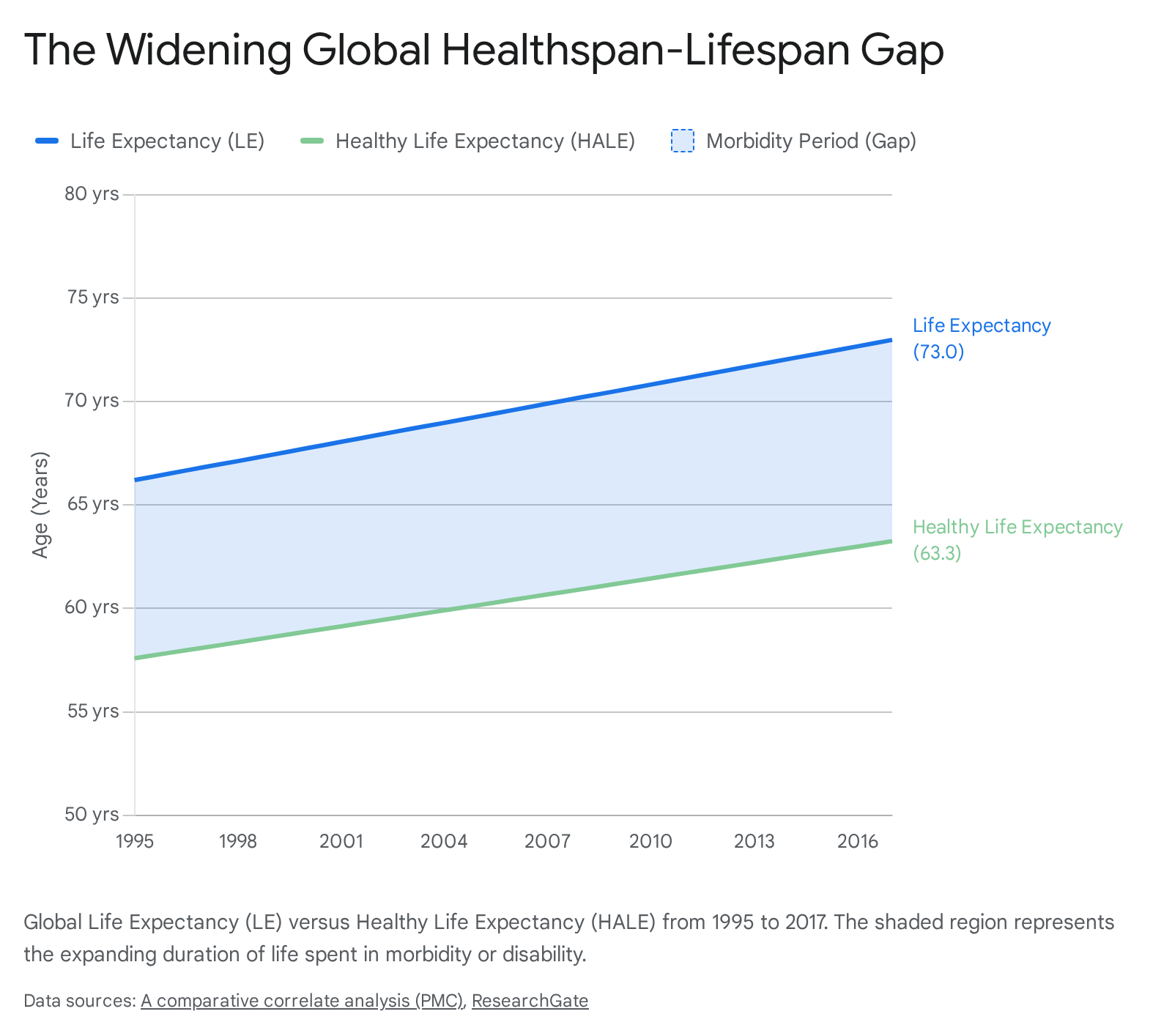
Consequently, the absolute "gap" between lifespan and healthspan expanded globally from 8.62 years to 9.72 years over that two-decade period 1011.
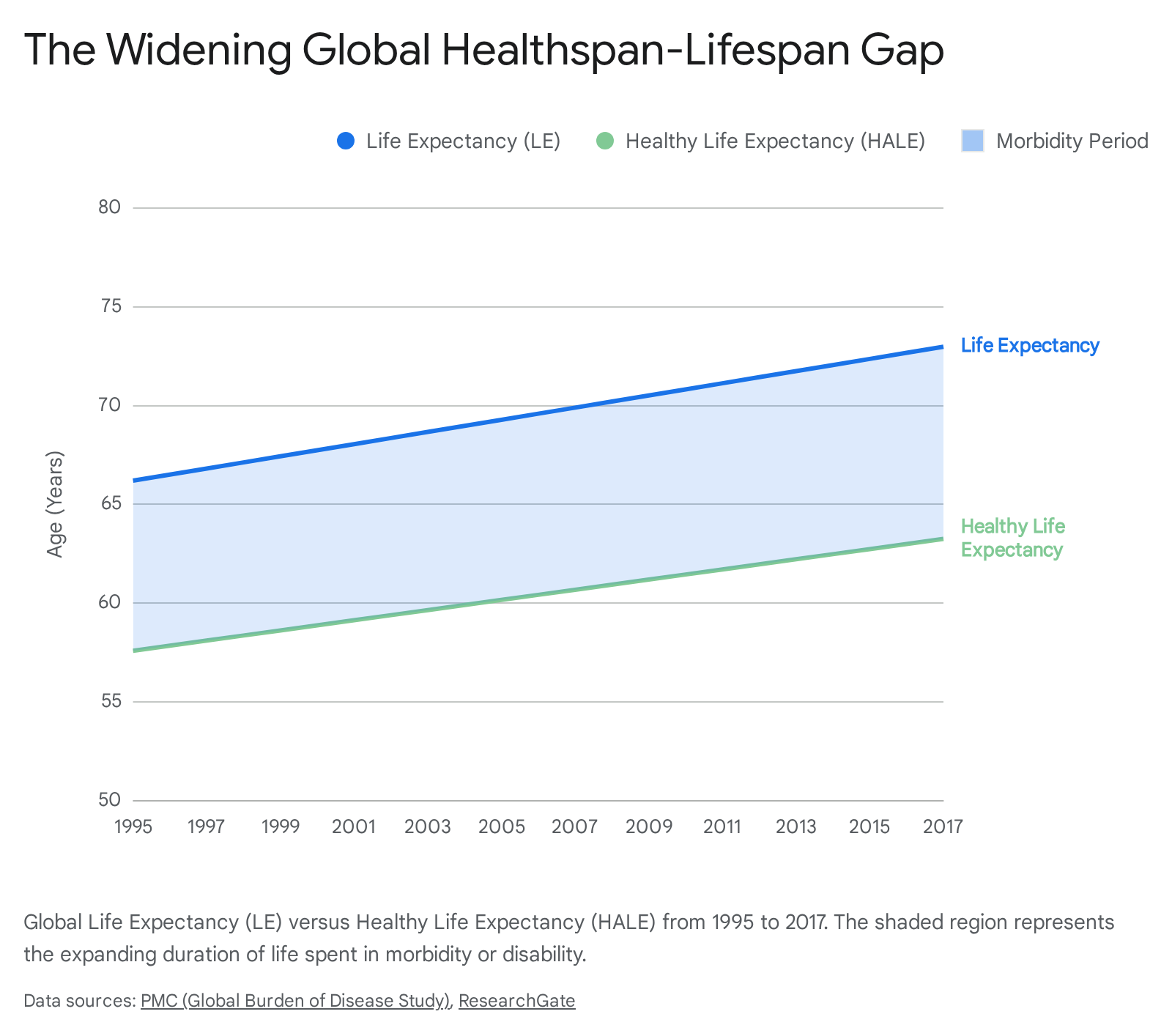
The shift driving this gap is rooted in an epidemiological transition. Communicable, maternal, neonatal, and nutritional (CMNN) diseases have seen remarkable reductions; DALYs (Disability-Adjusted Life Years) due to CMNN diseases fell by 25.8% globally from 2010 to 2023, led by steep declines in diarrheal diseases (49.1%), HIV/AIDS (42.9%), and tuberculosis (42.2%) 12. Conversely, non-communicable diseases (NCDs) have surged, contributing 1.80 billion global DALYs in 2023, up from 1.45 billion in 2010 12. As infectious diseases recede, human populations survive to experience the protracted morbidity of cardiovascular disease, cancer, metabolic syndromes, and neurological decline 1213.
Regional and Socio-Demographic Disparities
The aggregate global data masks severe regional stratification. As of 2017, the five nations with the highest life expectancies were Singapore (84.79 years), Japan, Switzerland, Italy, and Kuwait 10. In contrast, the five nations with the lowest life expectancies were the Central African Republic (51.87 years), Lesotho, Papua New Guinea, Mozambique, and Somalia 10. This creates a massive 32.92-year variance in total survival time across the globe 1011.
Healthy life expectancy follows a similar, stratified pattern. Singapore leads globally with a HALE of 74.22 years, while the Central African Republic reports a HALE of only 44.75 years - a variance of 29.47 years 10. Generally, high-income and upper-middle-income countries possess a life expectancy exceeding 65 years and a HALE above 55 years, whereas low-income nations frequently fall below these thresholds 10.
Furthermore, some regions have experienced localized mortality crises amidst the global improvement. Between 2000 and 2023, the Caribbean region showed increases in death rates across 40 distinct age-sex-cause cohorts, particularly driven by a 52.6% increase in NCD deaths among females aged 30 - 34 5. In high-income North America, life expectancy gains have stalled or reversed in specific cohorts due to the rising incidence of drug-use disorders, which have heavily impacted younger adult mortality over the past 15 years 4.
The Gender Healthspan Gap
The divergence between lifespan and healthspan exhibits profound demographic disparities across gender lines. While women consistently outlive men globally - averaging a lifespan of 27,083 days compared to 25,477 days for men - they spend an estimated 25% more of their lives in poor health 1414.
According to exhaustive analyses published by the McKinsey Health Institute in 2024, this gender health gap translates to 75 million DALYs lost annually . This equates to approximately seven days of lost healthy life per woman every single year, or roughly 500 days of compounded functional impairment over an average female lifetime . Despite lower absolute mortality rates, women collectively spend 26% more years living with disabilities (YLDs) than men 15.
The roots of this disparity are multifaceted and systemic. Historically, medical research governing women's health has been simplified to exclusively target sexual and reproductive conditions, systematically underrepresenting the unique ways that broad, chronic diseases - such as cardiovascular disease, autoimmune disorders, and neurological conditions - manifest in female populations 14. For instance, women face significantly higher burdens of morbidity from autoimmune diseases and osteoporosis, conditions characterized by long-term functional decline rather than immediate mortality 1416.
Furthermore, pharmaceutical investment has traditionally prioritized diseases with high mortality over those generating long-term disability. For example, debilitating gynecological conditions like endometriosis receive merely a tenth of the research pipeline volume compared to high-mortality cancers, despite endometriosis affecting between 24 million and 190 million women worldwide and severely degrading daily functional capacity . Addressing the female healthspan gap represents an enormous public health and economic opportunity; economic models project that closing this gender-specific healthspan gap could yield a $1 trillion annual boost to the global GDP by 2040, equivalent to returning 137 million women to full-time workforce capacity .
| Metric | Male Population (Global Average) | Female Population (Global Average) | Implication |
|---|---|---|---|
| Total Lifespan (Days) | ~25,477 days | ~27,083 days | Women possess a distinct survival advantage. |
| Time Spent in Poor Health | Baseline | +25% relative to men | Women spend a larger proportion of life functionally impaired. |
| Total DALYs Lost Annually | Baseline | 75 million additional DALYs | The gap severely impacts global female economic productivity. |
| Years Lived with Disability (YLDs) | Baseline | +26% relative to men | Late-life morbidity disproportionately affects women. |
Theoretical Frameworks of Morbidity and Aging
To interpret the epidemiological divergence of lifespan and healthspan, demographers, public health experts, and gerontologists rely on three predominant theoretical frameworks. Originally formulated in the late twentieth century, these theories attempt to describe the evolving mathematical and physiological relationship between mortality declines and the prevalence of late-life morbidity 17.
Expansion of Morbidity
The "expansion of morbidity" hypothesis, primarily attributed to Ernest Gruenberg's 1977 concept of the "failures of success," posits that modern medicine effectively prevents fatal complications of chronic diseases without actually curing the underlying physiological decline 1819. As medical interventions successfully decrease case fatality rates for acute clinical events - such as myocardial infarctions, strokes, and aggressive malignancies - individuals who previously would have died instead survive into advanced age 19.
However, because these individuals survive with their underlying pathologies intact, they accumulate multiple chronic conditions and functional disabilities. Consequently, while total life expectancy increases, both the absolute and relative duration of life spent in a state of illness expands 1719.
Extensive evidence supporting this theory is visible in longitudinal studies tracking multimorbidity. Analyses of data from the U.S. Health and Retirement Study (HRS), which tracked thousands of individuals over a decade, found that the average age of onset for an individual's first chronic disease is 61.3 years 20. Following this initial diagnosis, individuals accumulate additional diseases at an average rate of 0.1 diseases per year 20. Because medical interventions prolong survival despite this accumulation, individuals are living longer but spending a much larger percentage of their total lifespans with clinically evident disease, resulting in an unprecedented burden of multimorbidity on healthcare systems 20. A comprehensive analysis of successive cohorts by Payne (2024) confirmed a generalized expansion with respect to chronic disease incidence 18.
Compression of Morbidity
Proposed by James Fries in 1980, the "compression of morbidity" hypothesis offers a more optimistic outcome. Fries argued that human lifespan has an inherent, natural limit, and that public health must focus on optimizing the quality of life leading up to that limit 1719. He posited that if the onset of chronic illness can be delayed through preventive measures, aggressive lifestyle modifications, and early behavioral interventions at a rate faster than the increase in overall life expectancy, the period of morbidity will be "compressed" into a narrow, brief window just prior to death 182122.
Under this scenario, the ideal human life course resembles a "squared" survival curve: a prolonged, robust healthspan followed by a rapid, synchronous failure of physiological systems 1723. Some empirical data do support this theory, particularly regarding severe disabilities. Certain studies tracking U.S. populations - such as the National Long Term Care Study - and specific European cohorts in Austria and Denmark have noted a gradual decline in the prevalence of severe, end-stage functional limitations 181922. This suggests that while minor chronic disease diagnoses may be rising, the period of extreme, debilitating morbidity requiring institutionalization is being compressed in some high-income populations 1824.
Dynamic Equilibrium
Serving as a synthesis of the expansion and compression models, the "dynamic equilibrium" theory was introduced by Kenneth Manton. This framework suggests that as total life expectancy increases, the proportion of life spent with morbidity remains relatively stable, but the severity profile of that morbidity shifts dramatically 17.
According to dynamic equilibrium, while the absolute prevalence of chronic diseases increases (due to higher survival rates), aggressive secondary medical management, pharmaceutical interventions, and environmental adaptations render these conditions less severely disabling 1719. A comprehensive longitudinal evaluation of functional limitations in New Zealand meticulously tracked this phenomenon. The study found that while the absolute expectation of life with mobility and agility limitations increased significantly between 1981 and 1996, the changes were primarily driven by a massive shift from major, institutionalizing disabilities to moderate, manageable limitations 25. The researchers concluded that dynamic equilibrium provides the most accurate fit for modern population health data, where increased disease burden is offset by reduced disease severity 25.
Comparative Analysis of Morbidity Theories
| Theoretical Framework | Core Premise and Mechanism | Implication for Healthspan | Primary Medical Drivers |
|---|---|---|---|
| Expansion of Morbidity | Decreased mortality from fatal diseases leads to prolonged survival with intact chronic illness. | Healthspan remains stagnant while lifespan grows; the "sickspan" expands absolutely and relatively. | Medical "rescue" technologies (e.g., ICU care, advanced surgical cardiovascular interventions). |
| Compression of Morbidity | The onset of chronic disease is delayed at a rate faster than the delay in overall mortality. | Healthspan increases rapidly; total morbidity is compressed into a short window at the end of life. | Primary prevention, aggressive lifestyle optimization, behavioral risk reduction. |
| Dynamic Equilibrium | Increased chronic disease prevalence is offset by decreased disease severity and functional impact. | Relative healthspan remains stable; populations live longer with manageable, milder limitations. | Secondary prevention, advanced chronic disease management, assistive technologies. |
The Economic and Societal Burden of Expanding Morbidity
The failure of global health systems to perfectly synchronize lifespan extension with healthspan preservation generates profound structural and economic consequences. As societies undergo rapid demographic transitions - with the global population aged 65 and over projected to more than double from 0.7 billion in 2019 to 1.5 billion by 2050 - the structural paradox of expanding morbidity threatens to overwhelm fiscal sustainability and social infrastructure 16.
Direct Medical and Long-Term Care Projections
Current econometric models indicate that long-term care (LTC) demands will escalate dramatically over the coming decades. In industrialized nations, episodic hospital care, polypharmacy, and highly specialized medical treatments during the final years of life completely dominate health system expenditures 16. Based on current population aging trajectories and the growing prevalence of non-fatal functional disability, expenditures for LTC across OECD countries are projected to multiply by 2.5 times by 2050 26.
If nations attempt to improve the generosity of these LTC systems to shield their aging populations from devastating out-of-pocket costs and poverty, models suggest total LTC expenditures could quadruple by mid-century 26. In the United States, the direct LTC cost as a percentage of GDP is projected to increase from 0.4% in 2020 to a staggering 4.2% in 2050 27. Similarly, the UK is projected to spend more than a fifth of its entire national output on services for the elderly by 2050, up from just 0.8% of GDP allocated to public spending on elderly care in recent years 28.
The Silent Crisis of Unpaid Caregiving
Beyond direct medical spending, the macroeconomic burden of caregiving is simultaneously expanding. The economic cost of unpaid family caregiving - driven by lost wages, reduced workforce participation, and disrupted career trajectories - is projected to double by 2050 29. In 2013, the estimated cost per caregiver due to lost wages was $5,251 annually; projections indicate this cost could escalate to $6,323 by 2050 29.
Caregivers, who are disproportionately female, frequently face the harsh necessity of reducing work hours or abandoning careers entirely to support aging relatives 29. Research indicates that women aged 51 to 70 work three to ten hours less per week when forced into caregiving roles 29. The broader economic implication is severe: a shrinking workforce must support a rapidly growing, medically dependent population. Falling birthrates combined with rising chronic illness create a paradox that existing frameworks of economic and social organization are fundamentally unable to assimilate without radical restructuring 1329.
The Acute Cost of Cognitive Decline
Neurodegenerative diseases represent the most profound and catastrophic manifestation of the healthspan-lifespan gap, as they relentlessly degrade cognitive capacity while physiological survival may continue unabated for a decade or more. Global spending on dementia care alone is currently estimated at $263 billion, but demographic and incidence models project this figure to reach an unsustainable $1.6 trillion by 2050 30. Without fundamental medical breakthroughs that compress cognitive morbidity, the extended lifespans achieved by modern cardiovascular and oncological medicine risk becoming an extended period of profound dependency, devastating families and bankrupting municipal health systems 30.
Clinical Assessment of Biological and Functional Age
Shifting the clinical paradigm from merely extending lifespan to aggressively optimizing healthspan requires the adoption of new metrics. Traditional age relies on chronological time, but biological and functional age metrics capture true vitality and capacity 3132. Accurate measurement allows clinicians to identify the trajectory toward the "marginal decade" - the final ten years of an individual's life - decades before irreversible decline occurs 3334.
Molecular and Blood-Based Biomarkers
Traditional clinical endpoints (e.g., disease diagnosis, emergency hospitalization, or mortality) are structurally inadequate for longevity medicine because chronic diseases develop silently over decades 33. Atherosclerosis, systemic insulin resistance, and neurodegeneration frequently exhibit preclinical, asymptomatic phases lasting 10 to 20 years 3335. By measuring specific blood-based biomarkers, clinicians can detect the systemic molecular damage driving the aging process long before a diagnosis is rendered.
Extensive longitudinal epidemiological analyses have identified specific clinical biomarkers that are highly predictive of the end of healthspan. Elevated glycemic markers (HbA1c and fasting blood glucose), systemic inflammatory markers (C-reactive protein [CRP]), and specific lipid profiles (ApoB, triglycerides) correlate strongly with a significantly increased risk of chronic disease onset 3637. For example, a massive 16-year longitudinal study of over 12,000 Swedish individuals aged 47 - 94 found that a single standard-deviation increase in HbA1c was associated with a 29% increased risk of healthspan termination (defined as the onset of any major chronic disease) 3638. Conversely, elevated high-density lipoprotein (HDL) related markers (such as ApoA1) showed strong protective effects against morbidity 3639.
Other critical functional markers include Cystatin-C, which provides a far more accurate measure of true kidney filtration rate and biological renal aging than standard creatinine tests 374041. Composite scoring algorithms, such as the PhenoAge system, aggregate these standard blood panels to estimate cumulative functional organ decline and metabolic dysregulation, allowing for rapid, actionable feedback in clinical longevity settings 3740.
Epigenetic and Multi-Omic Clocks
Beyond standard blood chemistry, the frontier of aging measurement involves tracking cellular alterations. Epigenetic clocks assess DNA methylation patterns to determine biological age. Third-generation tools, such as the DunedinPACE clock, represent a paradigm shift: rather than providing a static biological age, they measure the current pace of aging 37. Developed from a longitudinal cohort study tracking 19 biomarkers over decades, DunedinPACE identifies the methylation signature associated with the rate of functional decline. A score of 1.0 indicates aging at one biological year per calendar year; a score of 0.85 indicates the individual is aging 15% slower than average 37.
Other specialized analyses include GlycanAge, which measures the glycosylation patterns of immunoglobulin G (IgG) to accurately assess systemic immune and inflammatory aging, responding to lifestyle interventions faster than traditional epigenetic clocks 37. Notably, while telomere length was previously popularized as an aging metric, longevity clinicians increasingly view it as possessing weak test-retest reliability and high genetic variability, rendering it less actionable than metabolic or epigenetic profiling 3237.
Physical Performance and Cardiorespiratory Fitness
While blood biomarkers reveal molecular and metabolic aging, functional performance metrics determine whether cellular decline has translated into real-world, physical limitations. As longevity researchers note, a patient with poor metabolic markers but exceptional physical strength may possess a significantly lower risk of near-term mortality and disability than an individual with perfect blood chemistry but severe physical frailty 31.
Cardiorespiratory fitness, specifically measured via VO2 max (the maximum rate of oxygen consumption during incremental exercise), is widely considered the single most powerful predictor of longevity and healthspan 314248. High VO2 max values correlate inversely with all-cause mortality, reflecting the integrated efficiency of the pulmonary, cardiovascular, and muscular systems working under stress 3448. A 2018 study analyzing over 120,000 adults confirmed that elite VO2 max levels confer significant survival advantages, maintaining strong predictive power even after controlling for weight, age, and traditional cardiovascular risk factors 48.
Similarly, grip strength and gait speed serve as vital, easily administered proxies for overall neurological and musculoskeletal resilience. A landmark pooled analysis of nine cohort studies involving over 34,000 older adults found that gait speed predicted survival as accurately as age, sex, chronic conditions, and smoking history combined, earning it the moniker of the "sixth vital sign" 31. Body composition, specifically analyzed via DEXA scan, is utilized to track lean muscle mass, bone mineral density, and dangerous visceral fat, all of which predict mobility and metabolic stability in later decades 4142.
The Fried Frailty Phenotype
In geriatric medicine, frailty is recognized as a distinct clinical syndrome characterized by a severe decline in multiple physiological systems, resulting in an increased vulnerability to minor stressors 43. The most widely validated operational definition is the Fried Frailty Phenotype, developed by Dr. Linda Fried, which evaluates physical decline to distinguish pathological frailty from normal chronological aging 4445. An individual is classified as frail if they exhibit three or more of the following five criteria 4346:
- Unintentional Weight Loss: Self-reported or documented loss of 10 lbs or >5% of total body weight in the prior year 4346.
- Weakness (Grip Strength): Measured via a handheld dynamometer. Clinical cut-offs are strictly adjusted for sex and BMI. Generally, weakness is defined as a maximum isometric grip below 21 kg for women (depending on BMI) and below 31 kg for men 434446.
- Slowness (Gait Speed): The time required to walk 15 feet (4.5 meters), stratified by sex and height. Typically, taking greater than 7 seconds to complete the walk indicates pathological slowness 4346.
- Exhaustion: Self-reported lack of energy or endurance, usually assessed via the CES-D depression scale (e.g., indicating that "everything was an effort" or "I could not get going" for a moderate to severe amount of time in the past week) 4346.
- Low Physical Activity: Energy expenditure falling below specific weekly thresholds (<383 kcals/week for men, <270 kcals/week for women) 4346.
| Assessment Modality | Primary Metrics | Biological / Functional Mechanism Measured | Clinical Utility for Healthspan |
|---|---|---|---|
| Metabolic & Blood Biomarkers | ApoB, HbA1c, CRP, Cystatin-C | Subclinical molecular damage, inflammation, cardiovascular risk, and organ filtration efficiency. | Highly actionable; responds to dietary, pharmacological, and exercise interventions in weeks. |
| Epigenetic & Multi-omic Clocks | DunedinPACE, GlycanAge, DNA methylation | Rate of biological aging at the cellular and DNA expression level; immune system degradation. | Tracks the ongoing dynamic "pace of aging" rather than absolute accumulated damage. |
| Cardiorespiratory Metrics | VO2 Max, CPET | Efficiency of heart, lungs, and cellular mitochondria under physical stress. | Strongest overall predictor of delayed mortality, extended survival, and functional independence. |
| Musculoskeletal & Frailty Metrics | Grip strength, DEXA scan, Gait speed | Sarcopenia, bone mineral density, neuromuscular integration, and progression toward the Fried Frailty Phenotype. | Accurately predicts frailty, fall risk, hospitalization, and loss of independent living capacity. |
Preventive Paradigms and the Marginal Decade
Moving Beyond the Medicalization of Aging
The traditional healthcare model, frequently termed "Medicine 2.0," excels at acute rescue - treating severe infections, responding to traumatic injuries, and managing late-stage symptomatic disease 1435. However, indiscriminately applying this reactive framework to the chronic conditions of aging has resulted in the "medicalization of normal aging." This phenomenon occurs when natural physiological shifts (e.g., mild cognitive changes, reduced stamina, normal hormonal fluctuations) are heavily pathologized 4748.
When ordinary aspects of biological transition are labeled as diseases requiring pharmaceutical correction, it leads to over-testing, severe healthy anxiety, polypharmacy, and interventions that carry substantial iatrogenic risk but yield minimal improvements to the actual quality of life 4748. Focusing purely on suppressing individual age-related symptoms - treating the numbers rather than the patient - without addressing systemic vitality risks exacerbating the expansion of morbidity 4748.
In stark contrast, longevity researchers and progressive clinicians advocate for an accelerated transition to "Medicine 3.0" 1435. This paradigm shifts the clinical focus entirely from reactive disease management to proactive health optimization and human performance. It prioritizes the aggressive, highly personalized, and absurdly early prevention of the "four horsemen" of chronic disease: atherosclerotic cardiovascular disease, cancer, neurodegenerative disease, and metabolic dysfunction 143549. Because these conditions gestate silently over decades, Medicine 3.0 relies on advanced diagnostics and intensive lifestyle interventions initiated in a patient's 30s or 40s, long before clinical symptoms necessitate "rescue" care 3542.
Training for the Centenarian Decathlon
A cornerstone of the proactive longevity approach is preparing the physical body for the "Marginal Decade" - the final ten years of an individual's life 344850. If a person's physical capacity declines along a standard, unmanaged trajectory, the marginal decade is characterized by extreme frailty, loss of mobility, forced dependency, and the inability to perform basic activities of daily living 48.
To combat this physiological cliff, longevity protocols utilize a strategy of backward planning. Patients establish the specific physical capabilities they wish to retain at age 85 (e.g., picking up a grandchild, carrying groceries, hiking), and calculate the physiological reserve required at age 50 or 60 to guarantee that outcome, given the inevitable biological decay of aging 3448. To avoid crossing the disability threshold, individuals engage in targeted physical training, functioning akin to a "Centenarian Decathlon" 50.
Clinical targets for this level of longevity readiness are significantly more demanding than standard public health guidelines. Key benchmarks include 34505152: * High-Endurance Aerobic Capacity: Achieving a VO2 max in the 75th percentile or higher for one's age and gender group, often assessed via rigorous lab testing or field tests like the 12-minute Cooper run 3451. * Functional Strength and Grip: The ability to perform a dead hang from a pull-up bar for up to two minutes (for men in their 40s) or 90 seconds (for women), ensuring vital grip strength and shoulder mobility 5152. * Heavy Load Carriage: Performing a farmer's carry holding a substantial percentage of body weight (up to 100% for men, 75% for women) for one to two minutes, mimicking the functional strength required for daily independence 505152. * Mobility, Power, and Stability: The capacity to lift a 30-pound weight from the floor, perform an air squat hold for two minutes, execute a hex bar deadlift of body weight for five to ten repetitions, and stand from a seated position on the floor using only one point of support 345051.
Through rigorous strength training, continuous Zone 2 aerobic conditioning, and dedicated stability work, individuals build a massive physical "reserve." This reserve delays the onset of frailty, effectively forcing a structural compression of morbidity into the absolute final months of life 4850.
Pharmacological Interventions and Geroscience
While lifestyle modifications, elite physical conditioning, and rigorous sleep hygiene form the undeniable foundation of healthspan extension, the "geroscience hypothesis" posits that the fundamental biological mechanisms of aging can be directly targeted and modulated through pharmacology 4753.
Traditional medicine approaches aging by treating individual diseases sequentially (e.g., treating hypertension, then later treating diabetes, then treating arthritis). Geroscience argues that underlying physiological decay - such as cellular senescence, mitochondrial dysfunction, stem cell exhaustion, and epigenetic alterations - serves as the universal root cause of all these age-related chronic conditions 475360. Gerotherapeutics aim to safely alter these core mechanisms, thereby delaying the onset of multimorbidity universally rather than specifically.
Metformin and the TAME Trial
One of the most foundational and highly publicized efforts to validate the geroscience hypothesis in humans is the Targeting Aging with Metformin (TAME) trial 6154. Metformin is an FDA-approved, inexpensive, generic medication utilized globally to treat type 2 diabetes for over six decades 54. Beyond simple glycemic control, extensive observational data and animal models suggest metformin exerts pleiotropic effects, modulating metabolic and cellular processes deeply associated with aging 54. Retrospective epidemiological studies have frequently observed that diabetics taking metformin surprisingly experience lower rates of cancer, cardiovascular disease, and cognitive decline compared to non-diabetics who are not taking the drug 55.
The TAME trial is designed as a nationwide, six-year clinical trial spanning 14 leading research institutions, aiming to enroll over 3,000 adults aged 65 to 79 who do not have diabetes 5354. The trial will test whether prophylactic metformin administration can delay the composite incidence of major age-related diseases (specifically myocardial infarction, congestive heart failure, stroke, cancer, dementia, and death) 5354.
Crucially, the broader strategic goal of the TAME trial is to force the FDA to grant an indication for "aging" itself, establishing a regulatory pathway that proves aging can be treated as a modifiable condition, thereby opening the floodgates for future gerotherapeutic drug development 6054. However, the trial has faced significant systemic obstacles. Because metformin is a generic, off-patent drug, pharmaceutical companies have zero financial incentive to fund the estimated $45 to $70 million required for the study, leaving it chronically underfunded and reliant on philanthropic pledges 60. Furthermore, some prominent longevity researchers actively question metformin's potency as a true geroprotector in non-diabetic, healthy individuals, suggesting its benefits may be strictly limited to correcting existing metabolic dysfunction rather than fundamentally altering the aging process in optimized patients 6061.
SGLT2 Inhibitors and Rapamycin
As longevity research aggressively progresses, newer pharmaceutical candidates have shown remarkably robust potential to extend healthspan. Sodium-glucose cotransporter-2 (SGLT2) inhibitors (e.g., canagliflozin, dapagliflozin), originally developed solely for diabetes management, have yielded extraordinary, unexpected clinical results. Large-scale landmark trials, such as the CANVAS study, demonstrated massive reductions in all-cause mortality, cardiovascular events (14% reduction), and renal decline (40% reduction in eGFR decline), notably extending these benefits to non-diabetic populations suffering from heart failure or kidney disease 646556.
Beyond systemic organ protection, emerging molecular data suggest that SGLT2 inhibitors directly impact the fundamental hallmarks of aging. In experimental models, short-term administration of SGLT2 inhibitors for as little as 7 to 14 days reduced markers of cellular senescence by up to 50%, while also improving endothelial function, enhancing mitochondrial health, and activating longevity-linked pathways like AMPK and SIRT1 6465. Because of their profound, multi-systemic protective effects and established safety profiles, researchers and clinicians are increasingly viewing SGLT2 inhibitors as potent geroprotectors capable of actively compressing morbidity 6456.
Simultaneously, rapamycin - an mTOR inhibitor traditionally used clinically as an immunosuppressant to prevent organ transplant rejection - remains the most biologically validated lifespan-extending compound in mammalian models, consistently increasing longevity in laboratory mice by 15-20% across varying ages and sexes 6456. Translating these profound animal findings safely to humans is the focus of intense emerging research. The upcoming $38 million VITAL-H study is set to investigate whether low doses of rapamycin, dapagliflozin, or semaglutide can measurably improve "intrinsic capacity" - a composite regulatory endpoint capturing both physical and cognitive functionality - in over 700 healthy adults in their 60s 57.
Survival Curve Dynamics and Morbidity Compression
Despite the massive theoretical promise of lifestyle interventions and emerging gerotherapeutics, rigorous mathematical modeling of biological aging suggests researchers must proceed with extreme caution to avoid unintended consequences. Researchers applying the saturating-removal mathematical model of aging to longitudinal health data have demonstrated that not all life-extending interventions successfully compress morbidity 5869.
Scaling Versus Steepening the Curve
In experimental gerontology, a survival curve plots the fraction of organisms surviving as a function of age. Interventions that simply extend the mean lifespan while preserving the original, gradual downward shape of the survival curve - such as chronic caloric restriction observed in certain mouse models - do not compress the sickspan relative to the lifespan 58.
Instead, these interventions exhibit "scaling"; they proportionally stretch both the healthspan and the period of morbidity 58. In absolute terms, this inadvertently expands the total duration of illness and frailty an organism experiences before death, representing a catastrophic outcome if applied to human populations 5859. Data from Caenorhabditis elegans (nematodes), Drosophila melanogaster (fruit flies), and mice repeatedly confirm that many celebrated longevity interventions extend the sickspan proportionally without compressing morbidity in absolute or relative terms 58.
To achieve genuine, desirable compression of morbidity, an intervention must actively steepen the survival curve 5860. A steepened survival curve implies that the organism is maintained in a state of robust, high vitality (healthspan) for an extended duration, suffering virtually no decline until a sharp, precipitous drop-off occurs at the absolute biological limit 5860.
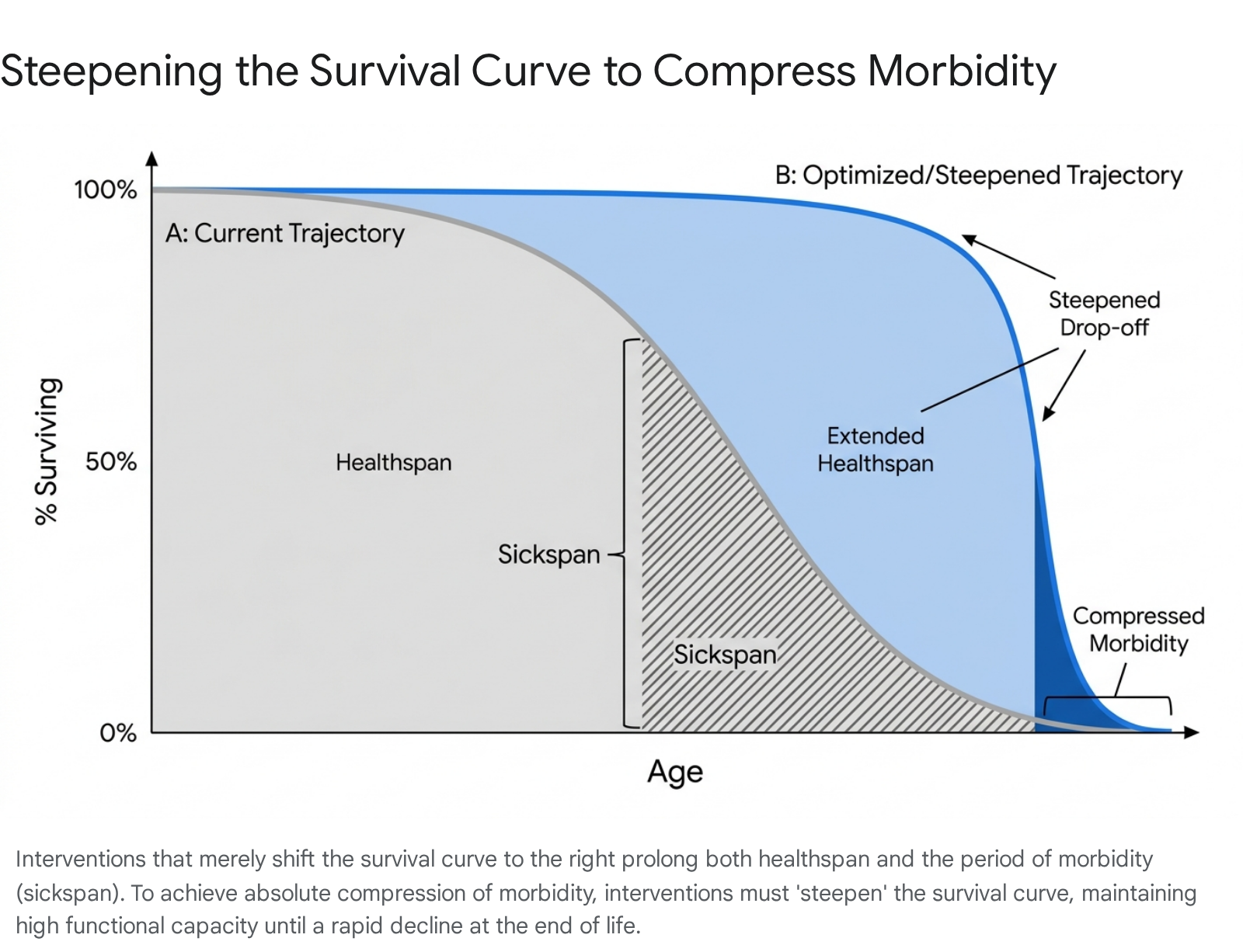
This rigorous mathematical reality underscores why future longevity interventions must focus relentlessly on preserving intrinsic functional capacity - building reserves through physical training and targeting root senescent decay - rather than relying on medical technologies that merely delay the final moment of mortality.
Conclusion
The vast corpus of global epidemiological data governing human longevity reveals a fundamental, systemic misalignment between the historical goals of modern medicine and the reality of demographic aging. While raw life expectancy has surged universally, the parallel expansion of chronic disease, multimorbidity, and functional decline indicates that merely living longer is a vastly insufficient objective. The widening gap between lifespan and healthspan generates profound economic liabilities, threatens to totally collapse international long-term care systems, and deeply compromises the quality of life for billions of individuals in advanced age.
Transitioning from a paradigm of lifespan extension to one of absolute healthspan optimization requires a systemic overhaul of both clinical targets and public health strategy. It necessitates moving aggressively away from the reactive medicalization of normal aging toward the proactive, preventive tenets of Medicine 3.0. This involves utilizing advanced functional metrics like VO2 max, grip strength, and rigorous multi-omic biomarkers to preserve intrinsic capacity decades before frailty sets in. Furthermore, the future success of geroscience relies entirely on identifying interventions - whether through intense, targeted physical conditioning to prepare for the marginal decade, or pharmacological agents like SGLT2 inhibitors and mTOR modulators - that do not merely stretch survival, but actively steepen the survival curve. By delaying the onset of chronic illness at a rate significantly faster than we delay death, society can achieve the ultimate goal of aging research: the compression of morbidity, ensuring that the final years of human life are characterized by vitality, dignity, and independence.