Comparison of intermittent fasting and caloric restriction mechanisms
Introduction
Dietary interventions designed to reduce body mass and improve cardiometabolic health have historically centered on continuous caloric restriction. Continuous caloric restriction involves a sustained reduction in daily energy intake - typically between ten and twenty-five percent below ad libitum consumption - without inducing malnutrition 12. More recently, intermittent fasting has emerged as a distinct nutritional paradigm. Intermittent fasting serves as a broad umbrella term encompassing several specific protocols, including alternate-day fasting, time-restricted eating, and periodic fasting regimens such as the 5:2 diet 23. While both intermittent fasting and continuous caloric restriction generally result in a net energy deficit over time, a growing body of research distinguishes the two approaches based on their molecular signaling cascades, the kinetics of their metabolic adaptations, and their differential effects on substrate utilization.
The core mechanistic debate within the current literature centers on whether the documented health benefits of intermittent fasting are simply the physiological result of an unintentional caloric deficit, or whether the specific temporal deprivation of nutrients triggers unique physiological adaptations that cannot be achieved through continuous restriction. Recent clinical trials and molecular analyses indicate that while the gross phenotypic outcomes of both diets, such as total weight loss, are often comparable in human subjects, the cellular mechanisms governing energy sensing, autophagic flux, and lean muscle mass preservation exhibit distinct kinetic signatures. These signatures are fundamentally dependent on whether nutrient deprivation is applied continuously or in cyclical pulses 245.
The Metabolic Switch and Substrate Partitioning
The primary physiological divergence between intermittent fasting and continuous caloric restriction lies in the induction and magnitude of the "metabolic switch." This switch represents the critical point of negative energy balance at which hepatic glycogen stores become sufficiently depleted to force the organism to transition from glucose oxidation to the mobilization of free fatty acids and the hepatic production of ketone bodies 1.
Hepatic Glycogen Depletion and Ketogenesis
Individuals maintaining a standard eating pattern of three or more meals per day rarely deplete their hepatic glycogen reserves. Consequently, their circulating ketone levels remain continuously low 15. Continuous caloric restriction reduces the overall daily influx of glucose, but because meals are distributed throughout the waking hours, hepatic glycogen is routinely replenished. This steady supply prevents a robust transition to ketogenesis unless the diet is specifically formulated to be severely carbohydrate-restricted, such as in a ketogenic diet 26.
In contrast, intermittent fasting imposes extended periods of nutrient abstinence that actively force the depletion of glycogen. Research demonstrates a clear physiological transition from glucose dependence to lipid metabolism and cellular repair across a continuous timeline. Hepatic glycogen stores are generally depleted between twelve and sixteen hours of fasting, which initiates the metabolic switch. However, achieving significant autophagic flux requires twenty-four to forty-eight hours of continuous fasting 78. As insulin levels drop significantly during this fasting window, the liver accelerates the conversion of mobilized fatty acids into ketones, primarily beta-hydroxybutyrate. These ketones do not merely serve as an alternative fuel source for the brain and skeletal muscle; they act as highly potent epigenetic signaling molecules that regulate the transcription of proteins involved in healthspan extension and cellular stress resistance 12.
Temporal Thresholds for Ketone Body Production
Research strongly differentiates intermittent fasting modalities based on their ability to consistently trigger this metabolic switch. Time-restricted eating protocols, such as the 16:8 method, provide a sufficient fasting window of sixteen to eighteen hours to initiate the early stages of metabolic switching and elevate morning ketone levels 5. Alternate-day fasting or extended periodic fasting, involving twenty-four to forty-eight hours of abstinence, pushes the organism deeper into nutritional ketosis, driving more profound shifts in lipid and cholesterol metabolism 1. Because chronic, continuous caloric restriction dampens but does not fully eliminate the insulin response throughout the day, the magnitude of the metabolic switch in continuous restriction is heavily blunted compared to pulsed fasting regimens 2.
Master Regulators of Cellular Energy: The AMPK and mTOR Axis
Both continuous caloric restriction and intermittent fasting exert their life-extending and health-promoting effects by modulating highly conserved cellular nutrient-sensing pathways. However, the kinetics of how these pathways are regulated - specifically whether they are subjected to sustained suppression or cyclical modulation - represents a fundamental mechanistic distinction between the two dietary strategies 24.
Adenosine Monophosphate-Activated Protein Kinase
Adenosine monophosphate-activated protein kinase (AMPK) operates as the master metabolic regulator and cellular energy sensor. AMPK exists as a heterotrimeric protein complex composed of a catalytic alpha subunit, a scaffolding beta subunit, and a regulatory gamma subunit. It is activated by cellular energy depletion, which is biochemically represented by an increased ratio of AMP or ADP to ATP 911. When activated by upstream kinases such as liver kinase B1 (LKB1) or calcium/calmodulin-dependent protein kinase kinase beta, AMPK phosphorylates target proteins to restore energy balance. It promotes catabolic processes that generate ATP, including fatty acid oxidation, mitochondrial biogenesis, and glucose uptake, while simultaneously shutting down energy-expensive anabolic processes like lipogenesis and protein synthesis 1110.
In skeletal muscle, the AMPK alpha 2 isoform plays a dominant role in regulating metabolic flexibility during exercise and fasting. Studies examining skeletal muscle from aged animal models undergoing continuous caloric restriction have demonstrated significant increases in the phosphorylation of the AMPK threonine 172 (Thr172) regulatory site, alongside increased phosphorylation of key downstream substrates such as acetyl-CoA carboxylase (ACC), which regulates lipid oxidation, and TBC1D1 and TBC1D4, which regulate glucose uptake 1111.
Mechanistic Target of Rapamycin
Operating in direct opposition to AMPK is the mechanistic target of rapamycin (mTOR), specifically the mTOR complex 1 (mTORC1). mTORC1 is a serine/threonine kinase that regulates cell growth, ribosome biogenesis, and protein synthesis. It is robustly activated by the presence of growth factors, insulin, and amino acids, particularly leucine 912. When activated, mTORC1 stimulates anabolic pathways via downstream targets such as p70 ribosomal S6 kinase and eukaryotic initiation factor 4E-binding protein, while actively inhibiting catabolic processes such as autophagy 912.
Pulsed Versus Chronic Kinase Modulation
Under continuous caloric restriction, mTORC1 is chronically inhibited, favoring catabolic processes that produce sufficient energy for basic survival, while AMPK is persistently activated. This chronic activation improves systemic insulin sensitivity and mitochondrial function, but researchers note it can manifest counterproductive effects in highly metabolic tissues. For instance, persistent AMPK activation and chronic mTOR suppression in skeletal muscle have been shown to reduce muscle hypertrophy in response to resistance training and limit gains in aerobic capacity by suppressing the signaling necessary for acute muscle repair and adaptation 4.
Intermittent fasting, conversely, embraces cyclical kinase activation. During the fasting window, AMPK is robustly activated, inhibiting mTOR and stimulating fatty acid oxidation, ketogenesis, and cellular repair 213. Upon refeeding, the rapid influx of nutrients suppresses AMPK and robustly stimulates mTOR, facilitating protein synthesis and tissue regeneration. This "pulsed" modulation maintains metabolic flexibility, preventing the cellular desensitization and lean mass degradation occasionally observed with the chronic mTOR suppression characteristic of severe, continuous caloric restriction 413.
Sirtuin Family Dynamics and NAD+ Biosynthesis
Sirtuins are a highly conserved family of nicotinamide adenine dinucleotide (NAD+)-dependent protein deacetylases and ADP-ribosyltransferases that function as metabolic sensors, translating dietary status into adaptive transcriptional outputs 1014. Both intermittent fasting and caloric restriction increase the cellular NAD+/NADH ratio, thereby activating sirtuins, but their distinct downstream targets govern different aspects of the systemic metabolic response 21014.
SIRT1: The Nuclear Metabolic Regulator
SIRT1 is localized primarily in the nucleus and acts as a primary mediator of calorie restriction. Once activated by elevated NAD+ levels resulting from slowed glycolysis, SIRT1 deacetylates and activates peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1-alpha). PGC-1-alpha serves as a master regulator of mitochondrial biogenesis 212. The mechanism by which AMPK impacts PGC-1-alpha relies heavily on SIRT1. Upon activation, AMPK directly phosphorylates PGC-1-alpha at specific threonine and serine residues, which structurally enables subsequent deacetylation and full activation by SIRT1 15.
Furthermore, SIRT1 deacetylates the FOXO family of transcription factors, driving their relocalization to the nucleus where they mediate stress resistance and longevity pathways 212. Fasting triggers a rapid increase in SIRT1 expression in the liver and skeletal muscle, which in turn upregulates AMPK via the deacetylation of LKB1, establishing a tightly regulated, mutually reinforcing feedback loop between the two nutrient sensors 1012.
SIRT3 and Mitochondrial Integrity
As the primary mitochondrial sirtuin, SIRT3 possesses robust deacetylase activity and its function is tightly linked to fasting and cellular energy deficits. SIRT3 deacetylates manganese superoxide dismutase, decreasing mitochondrial superoxide production and protecting the cell against oxidative stress 16. In addition to mitigating oxidative damage, SIRT3 promotes the catabolism of acetate and amino acids, deacetylating enzymes such as glutamate dehydrogenase to support the Krebs cycle during prolonged fasting 14. SIRT3 expression increases significantly during both fasting and caloric restriction, particularly in slow oxidative skeletal muscle, but is rapidly suppressed by high-fat ad libitum diets 17.
SIRT6 and Genomic Stability
Also localized within the nucleus, SIRT6 is crucial for regulating inflammation and maintaining genomic stability. Research indicates that SIRT1 activates the production of SIRT6, which subsequently deacetylates histones at the promoters of nuclear factor kappa B (NF-kB)-regulated genes. This targeted deacetylation directly suppresses chronic inflammatory pathways that contribute to numerous age-associated metabolic diseases 1418.
Human studies profiling sirtuin expression across diverse age groups reveal that while absolute protein levels of SIRT1, SIRT3, and SIRT6 naturally decline with age, interventions that mimic caloric restriction may rely on a phenomenon known as translational buffering. In extreme-age human cohorts, such as nonagenarians and centenarians, the protein-to-mRNA ratios for sirtuins often rise significantly, suggesting that cells upregulate the translational efficiency of these protective proteins to maintain critical functions despite age-related drops in transcription 18.
Table 1: Comparative Molecular Signaling in Dietary Restriction
| Molecular Target | Continuous Caloric Restriction | Intermittent Fasting | Primary Mechanism and Cellular Function |
|---|---|---|---|
| mTORC1 | Chronically suppressed 412. | Cyclical (suppressed during fast, activated during feed) 413. | Regulates ribosome biogenesis and protein synthesis. Pulsed activation prevents muscle degradation. |
| AMPK | Persistently elevated 419. | Robustly activated post 12-16 hours of fasting 720. | Senses AMP/ATP ratio. Directly phosphorylates ULK1 and PGC-1-alpha to drive catabolism. |
| SIRT1 | Upregulated continuously 210. | Spikes acutely during the fasting window 210. | NAD+-dependent deacetylase. Activates FOXO transcription factors and promotes mitochondrial biogenesis. |
| Hepatic Ketogenesis | Low; rarely crosses the metabolic switch threshold 12. | High; abundant production of beta-hydroxybutyrate 57. | Glycogen depletion forces the transition from glucose oxidation to systemic fatty acid utilization. |
Regulation of Autophagic Flux and Mitophagy
Autophagy is a highly conserved catabolic cellular housecleaning process through which damaged organelles, misfolded proteins, and intracellular pathogens are encapsulated and degraded by lysosomes. While both caloric restriction and intermittent fasting stimulate autophagy, the depth, systemic reach, and rapidity of autophagic flux differ fundamentally between the two interventions due to their differing impacts on the AMPK and mTORC1 regulatory axis 721.
Molecular Mechanisms of Autophagy Initiation
The initiation of macroautophagy and targeted mitophagy (the selective degradation of dysfunctional mitochondria) is directly controlled by the Unc-51 Like Autophagy Activating Kinase 1 (ULK1) complex. The biochemical regulation of ULK1 serves as a primary site of direct competition between AMPK and mTORC1 12.
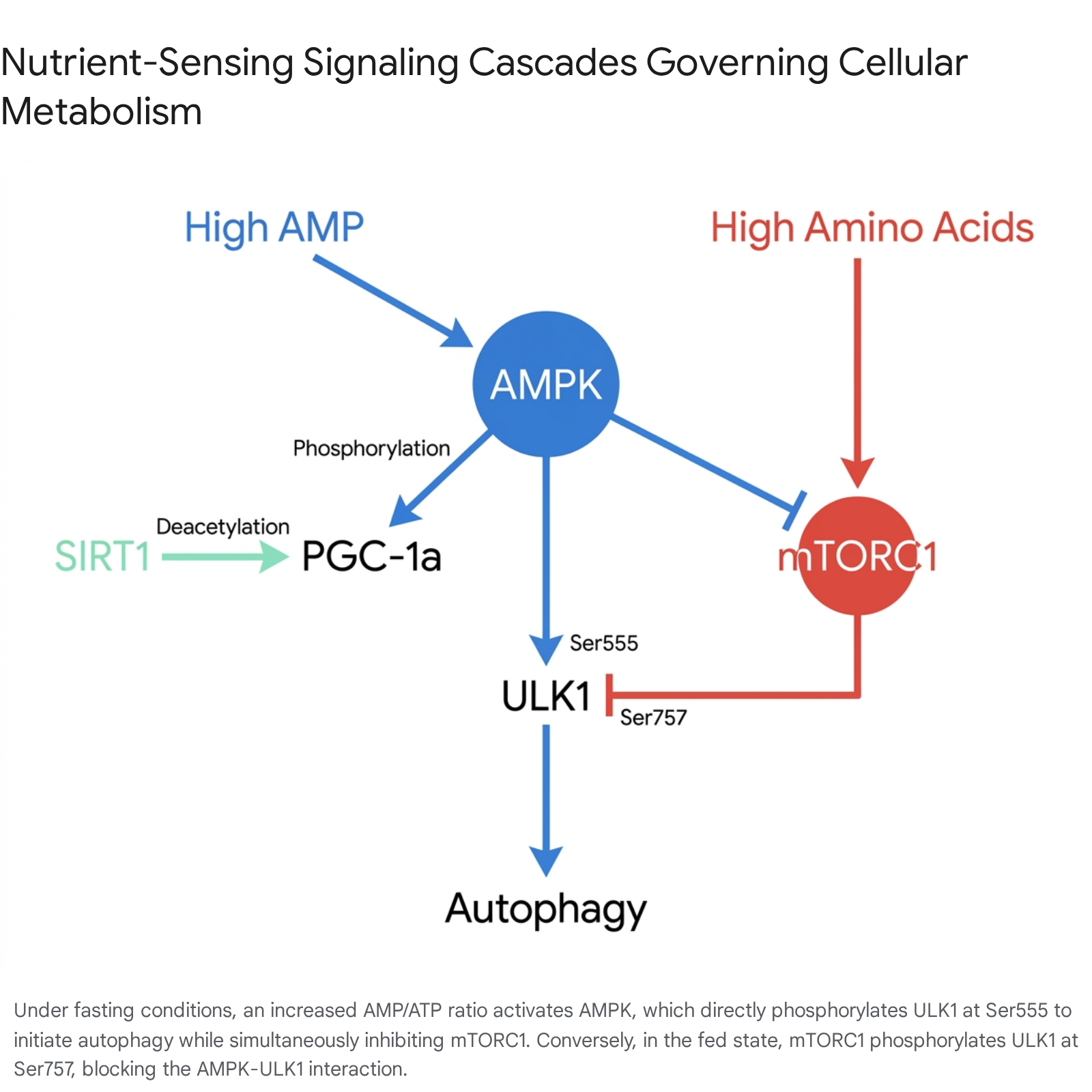
During the fed state, or under mild, continuous caloric restriction where exogenous nutrients are still regularly introduced into the system, mTORC1 remains at least partially active. Active mTORC1 phosphorylates ULK1 at the Serine 757 residue. This specific phosphorylation acts as an inhibitor, preventing ULK1 from interacting with AMPK, thereby keeping autophagy restrained to baseline housekeeping levels 12.
When an individual undergoes intermittent fasting and successfully crosses the metabolic switch threshold, the resulting profound energy deficit fully activates AMPK and achieves a near-complete suppression of mTORC1. Unrestrained by mTORC1, AMPK directly phosphorylates ULK1 at Serine 555. This phosphorylation robustly activates the ULK1 kinase, initiating the formation of autophagosomes via downstream targets such as Beclin-1 and modulating the clearance of cellular debris via Sequestosome 1 (SQSTM1/p62) 11122223. Genetic models demonstrate that the phosphorylation of ULK1 at Serine 555 is strictly required for the metabolic adaptations and improved mitochondrial health associated with severe caloric restriction and fasting 23.
Physiological Timelines of Human Autophagic Flux
Clinical research in humans provides a clear temporal framework for autophagy induction, illustrating precisely why intermittent fasting achieves a different cellular state than continuous caloric restriction 78:
- 0 to 12 Hours (Fed to Early Fasting): The body relies heavily on exogenous glucose and hepatic glycogen stores. While insulin levels begin to decline, mTORC1 remains active. Autophagy operates strictly at low, baseline levels.
- 12 to 16 Hours (The Initial Metabolic Shift): Glycogen stores are steadily depleted. As AMPK activity begins to rise in response to falling energy substrates, the mTORC1-mediated inhibition of ULK1 begins to lift. Early-stage autophagosome formation increases.
- 16 to 24 Hours (Significant Autophagic Activation): The organism completes the transition to fat burning and nutritional ketosis. AMPK phosphorylation peaks, triggering substantial cellular cleanup. Clinical studies suggest that time-restricted eating protocols, such as the 16:8 method, consistently capture the entry-level benefits of this specific phase 7.
- 24 to 48 Hours (Deep Autophagic Flux): Autophagic flux accelerates significantly, with biomarkers of cellular recycling increasing by up to three hundred percent compared to the baseline fed state. Human leukocyte models show that optimal, deep cellular repair requires this extended period of nutrient deprivation, a physiological state that remains entirely inaccessible through standard daily caloric restriction regimens 7821.
Body Composition and Lean Mass Preservation
A pervasive physiological challenge associated with continuous caloric restriction is the concomitant loss of lean body mass alongside the targeted reduction in adipose tissue. In traditional caloric restriction paradigms, approximately twenty-five percent of the total weight lost consists of fat-free mass. The degradation of skeletal muscle is particularly concerning in aging populations, as it impairs exercise capacity, lowers the basal metabolic rate, and is strongly associated with an increased risk of disability and mortality 12425.
Recent systematic reviews and network meta-analyses conducted in 2024 and 2025 indicate that intermittent fasting paradigms - particularly alternate-day fasting and early time-restricted eating - may preferentially target visceral fat mass while better preserving skeletal muscle tissue compared to continuous energy restriction 1326.
Hormonal Mediators: IGF-1 and Myostatin Dynamics
The differential impact on lean mass retention is largely mediated by fluctuations in Insulin-like Growth Factor 1 (IGF-1) and the cyclical nature of insulin secretion. Continuous caloric restriction generally suppresses circulating IGF-1, resulting in a persistent catabolic environment that slowly degrades muscle tissue over several months 127.
Conversely, specific intermittent fasting protocols, notably alternate-day fasting, have been shown to maintain or even transiently elevate IGF-1 levels during the refeeding or "feast" phases, actively mediating the maintenance of lean mass 1. Because intermittent fasting involves discrete, compressed feeding windows, the resultant acute spikes in insulin during ad libitum feeding periods are sufficient to stimulate muscle protein synthesis via the mTORC1 pathway. This establishes a metabolic rhythm where the body breaks down fat during the fasting window (driven by AMPK and lipolysis) and rebuilds lean tissue during the feeding window (driven by mTOR and IGF-1) 113.
Furthermore, the presence of ketone bodies during the fasting window provides a highly efficient alternative fuel source for skeletal muscle. The transcriptional regulator PPAR-alpha mediates this shift in muscle cell fuel preference from glucose to fatty acids and ketones, effectively sparing muscle amino acids from being catabolized and funneled into hepatic gluconeogenesis 1. When paired with resistance exercise, interventions combining caloric deficits with targeted anabolic stimuli consistently demonstrate the highest efficacy for reducing body fat while maintaining critical lean body mass 2428.
Clinical Efficacy and Metabolic Health Outcomes
While the molecular mechanisms clearly distinguish intermittent fasting from continuous caloric restriction at the cellular level, translating these biochemical differences into superior clinical outcomes in humans remains highly debated. A wealth of recent, rigorously designed randomized controlled trials provides a nuanced picture of their comparative efficacy in free-living populations.
Weight Loss and Fat Mass Reduction
When caloric intake is not strictly matched between study arms, both intermittent fasting and continuous restriction produce clinically meaningful weight loss. A comprehensive 2025 network meta-analysis of ninety-nine randomized controlled trials involving 6,582 participants found that alternate-day fasting was the only intermittent strategy to show statistically significant superiority over continuous caloric restriction in body weight reduction, yielding an additional 1.29 kilograms of weight loss 2629.
However, in rigorously controlled isocaloric trials - where total daily energy intake is perfectly matched between groups - the weight loss advantages of intermittent fasting largely disappear. The landmark TREATY trial, a twelve-month study published in the New England Journal of Medicine, demonstrated that time-restricted eating combined with caloric restriction did not produce significantly greater weight loss or body fat reduction than daily caloric restriction alone 5.
Similarly, the 2025 ChronoFast study, conducted by the German Institute of Human Nutrition, investigated isocaloric time-restricted eating in women with obesity. The trial found that an eight-hour eating window failed to provide measurable weight loss, cardiovascular benefits, or improvements in insulin sensitivity when overall calorie intake remained strictly unchanged. Interestingly, while metabolic measures were static, the timing of meals did act as a powerful cue for biological rhythms, shifting the body's internal clock by an average of forty minutes 30. These results suggest that the primary driver of weight loss in free-living time-restricted eating studies is often the incidental, unintentional reduction in overall calories, rather than the compressed eating window itself.
Glycemic Control and Insulin Resistance
Where intermittent fasting appears to show a more consistent, mechanistically distinct advantage over continuous restriction is in the realm of glycemic control. Even in studies where total weight loss is roughly equivalent, multiple recent meta-analyses indicate that fasting regimens drive superior improvements in insulin sensitivity.
Fasting strategies have been shown to reduce fasting insulin by significantly greater margins than continuous restriction and achieve steeper declines in the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR), the standard clinical metric for quantifying insulin resistance 35. In a twelve-month trial of adults presenting with obesity and baseline insulin resistance, alternate-day fasting lowered fasting insulin levels by fifty-two percent, compared to only fourteen percent for the continuous caloric restriction cohort 31. This clinical divergence suggests that the prolonged, uninterrupted periods of low insulin characteristic of intermittent fasting allow for essential pancreatic beta-cell rest and improved cellular receptor sensitivity, operating independently of the total energy deficit 532.
Table 2: Summary of Key Clinical Trials and Meta-Analyses (2024-2025)
| Study / Source | Intervention Design | Key Findings | Clinical Implications |
|---|---|---|---|
| BMJ Network Meta-Analysis (2025) 29 | 99 RCTs comparing IF (ADF, TRE, 5:2) to Continuous CR. | ADF was the only fasting strategy to yield statistically superior weight loss (-1.29 kg) compared to continuous CR. | TRE and continuous CR offer nearly identical weight loss benefits; ADF may offer a slight metabolic edge. |
| TREATY Trial (NEJM) 5 | 12-month isocaloric comparison of TRE + CR versus CR alone. | No significant difference in fat loss or weight reduction when total calories were matched. | Weight loss in TRE is driven almost entirely by the incidental reduction of energy intake. |
| ChronoFast Study (2025) 30 | Isocaloric 8-hour TRE crossover study in women with obesity. | No improvement in insulin sensitivity or blood lipids, but significant shifts in circadian rhythms. | Fasting windows alter biological clocks but do not inherently improve cardiovascular markers without an energy deficit. |
| Phase Obesity Trial 31 | 12-month comparison of ADF vs CR in insulin-resistant adults. | Equal weight loss, but ADF reduced fasting insulin by 52% compared to 14% for CR. | IF provides distinct mechanistic advantages for resolving insulin resistance and improving glucoregulatory factors. |
Genetic and Regional Phenotypic Variations
The metabolic responses to dietary interventions are not uniform; they are heavily influenced by genetic backgrounds and regional physiological phenotypes. Recent clinical trials examining South Asian and Sub-Saharan African populations highlight the necessity of tailoring fasting and restriction guidelines to specific metabolic profiles.
South Asian Populations and the Thin-Fat Phenotype
South Asian populations generally exhibit a distinct "thin-fat" phenotype. This profile is characterized by higher visceral adiposity, lower overall skeletal muscle mass, and a significantly earlier onset of systemic insulin resistance and Type 2 Diabetes Mellitus at lower Body Mass Indices compared to Caucasian populations 3334.
Because fat-free skeletal muscle mass is the primary physiological determinant of resting metabolic rate, South Asians generally possess a lower basal resting metabolic rate, which inherently alters their systemic response to caloric restriction 34. A 2025 prospective observational study evaluated a 16:8 time-restricted eating protocol over twelve weeks in Indian patients diagnosed with Type 2 Diabetes Mellitus. The intervention successfully lowered mean glycated hemoglobin (HbA1c) from 8.4% to 7.7%. Crucially, it allowed twenty percent of the participants to safely reduce their self-adjusted insulin doses without experiencing severe hypoglycemia - a unique advantage of intermittent fasting over continuous restriction in highly insulin-resistant cohorts 37. The heightened susceptibility of South Asians to central adiposity is closely linked to variations in adipokine production, notably higher leptin and lower adiponectin levels. Intermittent fasting interventions have demonstrated particular efficacy in reducing the atherogenic leptin-to-adiponectin ratio 3234.
African Populations and Community Interventions
Similarly, rigorous clinical trials examining intermittent fasting within Black and African communities have demonstrated high feasibility, cultural acceptance, and targeted metabolic improvements. A 2026 cluster-randomized controlled pilot study comparing intermittent fasting to continuous caloric restriction in African American church communities found that fasting led to statistically significant weight loss. More importantly, the fasting protocol naturally facilitated a significantly lower daily energy intake (a reduction of 414 kilocalories per day) compared to the continuous restriction cohort, alongside substantial reductions in total fat intake 35.
Culturally, intermittent fasting aligns seamlessly with various traditional and religious fasting practices prevalent in many African populations, which dramatically increases long-term adherence to the intervention 3640. Metabolically, alternate-day fasting in these specific groups has been correlated with substantial reductions in systolic blood pressure and low-density lipoprotein cholesterol. However, researchers caution that extended fasting without proper medical supervision can introduce dehydration and hypoglycemic risks for individuals presenting with advanced, unmanaged metabolic disease 4037.
Methodological Constraints and Cross-Species Translation
A critical factor frequently confounding the scientific consensus on intermittent fasting versus caloric restriction is the direct translation of molecular data from animal models to human physiology. The vast majority of spectacular mechanistic findings regarding lifespan extension, extreme autophagic flux, and massive sirtuin activation rely heavily on murine studies.
Metabolic Scaling and the Timeline Debunking
Mice and humans age at fundamentally different rates and possess vastly different mass-specific metabolic rates. An adult laboratory mouse possesses a basal metabolic rate per gram of body weight that is approximately seven times higher than that of a human 38. Consequently, a single twenty-four-hour fast in a mouse induces profound, potentially life-threatening physiological stress. This brief deprivation rapidly drops the animal's body temperature, drastically alters thyroid and insulin hormone levels, and forces the organism into deep survival mode. Metabolically, a twenty-four-hour fast in a mouse is roughly equivalent to a four-to-five-day continuous water fast in a human subject 38.
In murine studies, "short-term" fasting easily induces a twenty percent reduction in body weight and triggers dramatic, systemic autophagy. In stark contrast, twenty-four hours of fasting in a human produces only a minor fraction of that weight loss (typically less than two percent) and induces localized, rather than systemic, macroautophagy 3839.
Furthermore, a comprehensive 2021 study revealed that many of the longevity and metabolic benefits previously attributed purely to caloric restriction in mice were actually dependent on the fasting window. Because researchers typically fed restricted mice their daily food allotment all at once, the mice consumed it rapidly, inadvertently subjecting themselves to prolonged daily fasts. Mice that were calorically restricted but fed continuously throughout the day actually died younger than ad libitum control mice, emphasizing that the timing of food intake is functionally inseparable from the restriction itself in laboratory settings 40. In human trials, the inherent metabolic stability of the species blunts these extreme stress responses, clearly explaining why the clinical divergence between intermittent fasting and continuous caloric restriction is often significantly less dramatic than preclinical rodent models suggest 38.
Conclusion
Intermittent fasting and continuous caloric restriction utilize overlapping but mechanistically distinct molecular networks to improve metabolic health and alter body composition. While continuous caloric restriction relies on sustained, moderate energy deficits to chronically suppress mTOR and elevate AMPK, it largely fails to trigger the profound metabolic switch associated with prolonged hepatic glycogen depletion. Intermittent fasting, by imposing strict temporal boundaries on nutrient intake, capitalizes on the cyclical nature of human metabolism. It pulses AMPK and mTOR activation, driving deep autophagic flux via ULK1 phosphorylation during the fasting window, while permitting necessary anabolic repair and lean mass preservation during periods of refeeding.
Clinical evidence synthesized from rigorously controlled human trials indicates that while total weight loss between the two diets is largely comparable when overall energy intake is matched, intermittent fasting offers distinct mechanistic advantages. Fasting paradigms demonstrate superior efficacy in rapidly improving insulin sensitivity, reversing insulin resistance, and preserving fat-free skeletal muscle mass. As researchers continue to untangle these complex physiological responses across diverse genetic populations, it is evident that the temporal timing of nutrient intake exerts profound molecular effects entirely independent of the total calories consumed.