Clinical and social impacts of GLP-1 receptor agonists
Introduction: The Evolution of Incretin Therapies
The landscape of metabolic, neurobehavioral, and cardiovascular medicine has undergone a seismic paradigm shift driven by the clinical maturation of glucagon-like peptide-1 receptor agonists (GLP-1 RAs). Originally synthesized and approved over a decade ago exclusively for the glycemic management of type 2 diabetes mellitus, these agents have rapidly transcended their initial indications. As of 2026, the scientific and medical consensus recognizes GLP-1 RAs not merely as peripheral insulinotropic agents, but as profound modulators of systemic metabolic homeostasis, cardiovascular risk, renal preservation, and central nervous system (CNS) reward circuitry. This pharmacological class has evolved from a niche diabetes intervention into a foundational pillar of modern chronic disease management, touching nearly every facet of internal medicine.
This exhaustive report provides a granular, expert-level analysis of the contemporary GLP-1 ecosystem. By deconstructing the underlying neurobiological mechanisms of action, this analysis bridges the critical clinical gap between simple appetite suppression - often colloquially termed the reduction of "food noise" - and the complex attenuation of addictive behaviors and substance use disorders. Furthermore, the report rigorously evaluates the expanding clinical indications of these therapies, the physiological reality of the post-cessation rebound effect, the macroeconomic adaptations forced upon the global food and beverage industry, and the stark geopolitical divergences in drug pricing, health equity, and access. Finally, it contrasts the rapid hyper-medicalization of obesity facilitated by GLP-1s with the recent historical context of the fat acceptance and body positivity movements, highlighting the sociological friction generated by these highly effective pharmaceutical interventions.
Deconstructing the Pharmacopoeia: Active Ingredients Versus Branded Variants
A persistent source of public, media, and even clinical confusion surrounding modern weight-loss and diabetes pharmacotherapy is the frequent conflation of active pharmaceutical ingredients with their commercial brand names. The pharmacological landscape is not a monolith; it features distinct molecular entities, each engineered with unique receptor affinities, half-lives, and dosing schedules, which are then marketed under various trade names depending on the specific FDA-approved indication. Clarifying this taxonomy is essential for understanding the nuances of clinical efficacy and trial data.
Semaglutide represents the most globally recognized mono-agonist of the GLP-1 receptor. Developed by Novo Nordisk, the active ingredient semaglutide is marketed under three distinct brand names. As Ozempic, it is formulated as a weekly subcutaneous injection indicated primarily for type 2 diabetes and the reduction of major cardiovascular events 123. When formulated at a higher maximal dose (2.4 mg) for chronic weight management, cardiovascular risk reduction in patients with obesity, and recently for metabolic dysfunction-associated steatohepatitis (MASH), the identical molecule is marketed as Wegovy 24. Additionally, to overcome the limitations of subcutaneous delivery, Novo Nordisk engineered an oral formulation using an absorption enhancer (SNAC), which is marketed as Rybelsus for type 2 diabetes management 13.
Tirzepatide, developed by Eli Lilly, represents a pharmacological evolution as a first-in-class dual-agonist targeting both the GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptors. This dual mechanism drives superior weight loss outcomes compared to GLP-1 mono-agonists by synergistically modulating lipid metabolism and insulin sensitivity. Tirzepatide is marketed as Mounjaro for the treatment of type 2 diabetes and as Zepbound for chronic weight management and, most recently, obstructive sleep apnea 135.
Older generations of GLP-1 receptor agonists include liraglutide and dulaglutide. Liraglutide, an early daily-injected mono-agonist, is marketed as Victoza for diabetes and Saxenda for weight management 13. Dulaglutide is marketed as Trulicity, offering a weekly injection for diabetes and cardiovascular risk reduction, though it generally yields less profound weight loss than the newer molecules 23.
Looking toward the immediate future, the pharmaceutical pipeline is focused on maximizing efficacy and administration convenience. Orforglipron, currently in advanced Phase III trials by Eli Lilly, is a non-peptide, small-molecule GLP-1 receptor agonist designed for once-daily oral administration, potentially removing the barrier of complex manufacturing and injection fatigue 46. Concurrently, Eli Lilly is advancing retatrutide, an investigational "triple G" agonist targeting GLP-1, GIP, and glucagon receptors. The addition of glucagon receptor agonism is hypothesized to increase resting energy expenditure, breaking through the weight-loss ceiling observed with dual-agonists. In the Phase II TRIUMPH-4 trial, retatrutide demonstrated an unprecedented average body weight loss of 28.7% at 68 weeks - translating to roughly 71 pounds in the study cohort. Anticipated FDA submission for retatrutide is slated for late 2026, with potential approval and commercial availability by late 2027 or early 2028, pending the successful completion of the remaining Phase III trials 789.
To clarify this complex commercial and pharmacological matrix, the following table delineates the major agents available or under late-stage development as of 2026.
Table 1: Major GLP-1 and Multi-Agonist Medications
| Active Ingredient | Brand Name(s) | Manufacturer | Primary Mechanism of Action | FDA-Approved Indications (as of 2026) |
|---|---|---|---|---|
| Semaglutide | Ozempic | Novo Nordisk | GLP-1 Agonist (Injectable) | Type 2 Diabetes, Cardiovascular Risk Reduction, CKD 22 |
| Semaglutide | Wegovy | Novo Nordisk | GLP-1 Agonist (Injectable) | Chronic Weight Management, CV Risk Reduction, MASH 24 |
| Semaglutide | Rybelsus | Novo Nordisk | GLP-1 Agonist (Oral) | Type 2 Diabetes, Cardiovascular Risk Reduction 33 |
| Tirzepatide | Mounjaro | Eli Lilly | Dual GLP-1/GIP Agonist | Type 2 Diabetes 35 |
| Tirzepatide | Zepbound | Eli Lilly | Dual GLP-1/GIP Agonist | Chronic Weight Management, Obstructive Sleep Apnea (OSA) 512 |
| Liraglutide | Victoza | Novo Nordisk | GLP-1 Agonist (Injectable) | Type 2 Diabetes, Cardiovascular Risk Reduction 23 |
| Liraglutide | Saxenda | Novo Nordisk | GLP-1 Agonist (Injectable) | Chronic Weight Management 3 |
| Dulaglutide | Trulicity | Eli Lilly | GLP-1 Agonist (Injectable) | Type 2 Diabetes, Cardiovascular Risk Reduction 23 |
| Orforglipron | TBD (Investigational) | Eli Lilly | Small-Molecule GLP-1 Agonist | Pending (Phase III - target: Weight Management via oral pill) 46 |
| Retatrutide | TBD (Investigational) | Eli Lilly | Triple GLP-1/GIP/Glucagon | Pending (Phase III - target: Severe Obesity/T2D) 78 |
Neurological Mechanisms of Action: Bridging "Food Noise" and Addiction
The traditional clinical understanding of GLP-1 receptor agonists rested heavily on their peripheral metabolic and gastrointestinal effects. Biologically, natural GLP-1 is an incretin hormone secreted by the L-cells of the small intestine in response to nutrient ingestion. Exogenous GLP-1 RAs mimic this process to stimulate glucose-dependent insulin secretion from pancreatic beta cells, inhibit glucagon release from alpha cells, and significantly delay gastric emptying to increase postprandial satiety 45. However, while these peripheral mechanisms are vital for glycemic control, the defining clinical hallmark of next-generation incretin therapies is their profound action within the central nervous system (CNS), specifically targeting the neurobiological substrates that mediate reward, craving, and compulsion.
The subjective cessation of "food noise" - the intrusive, persistent cognitive preoccupation with consuming highly palatable, calorie-dense foods - is driven by GLP-1 receptors mapped densely across the brain's homeostatic and hedonic centers. Peripherally administered GLP-1 RAs are capable of crossing the blood-brain barrier, where they interact with receptors in the hypothalamus and the hindbrain, regions canonically responsible for assessing energy balance and generating primary satiety signals 6. However, contemporary neurobiological research has elucidated that these agents also actively and aggressively modulate the brain's reward circuitry, moving far beyond simple homeostatic appetite regulation 6.
Substance use disorders (SUDs) - whether involving alcohol, nicotine, or illicit opioids - and pathological overeating share a remarkably similar neurobiological etiology: the dysregulation of the mesolimbic dopamine pathway 717. When an individual consumes a highly palatable, hyper-processed food, uses tobacco, or ingests a narcotic, the ventral tegmental area (VTA) releases a massive surge of dopamine into the nucleus accumbens (NAc). This dopaminergic flood reinforces the behavior, coding it as highly salient and rewarding, and ultimately driving the compulsion to repeat the action despite negative consequences.
Extensive preclinical models and human functional magnetic resonance imaging (fMRI) studies indicate that GLP-1 RAs suppress this drug-induced and food-induced dopamine release 468. By binding to specific receptors in the CNS, semaglutide and tirzepatide effectively downregulate the salience of external rewarding stimuli. A landmark 2026 study published in Nature, funded by the National Institutes of Health, provided groundbreaking clarity on this mechanism. Investigations into newer, small-molecule oral GLP-1s, such as orforglipron and danuglipron, revealed targeted neural activation within the central amygdala, a deep brain region intimately associated with desire and pleasure-driven cravings 6. By activating these highly specific pathways in the central amygdala, GLP-1 therapies disrupt the compulsive loop of addiction, lowering the anticipatory dopamine release tied to reward-seeking behavior.
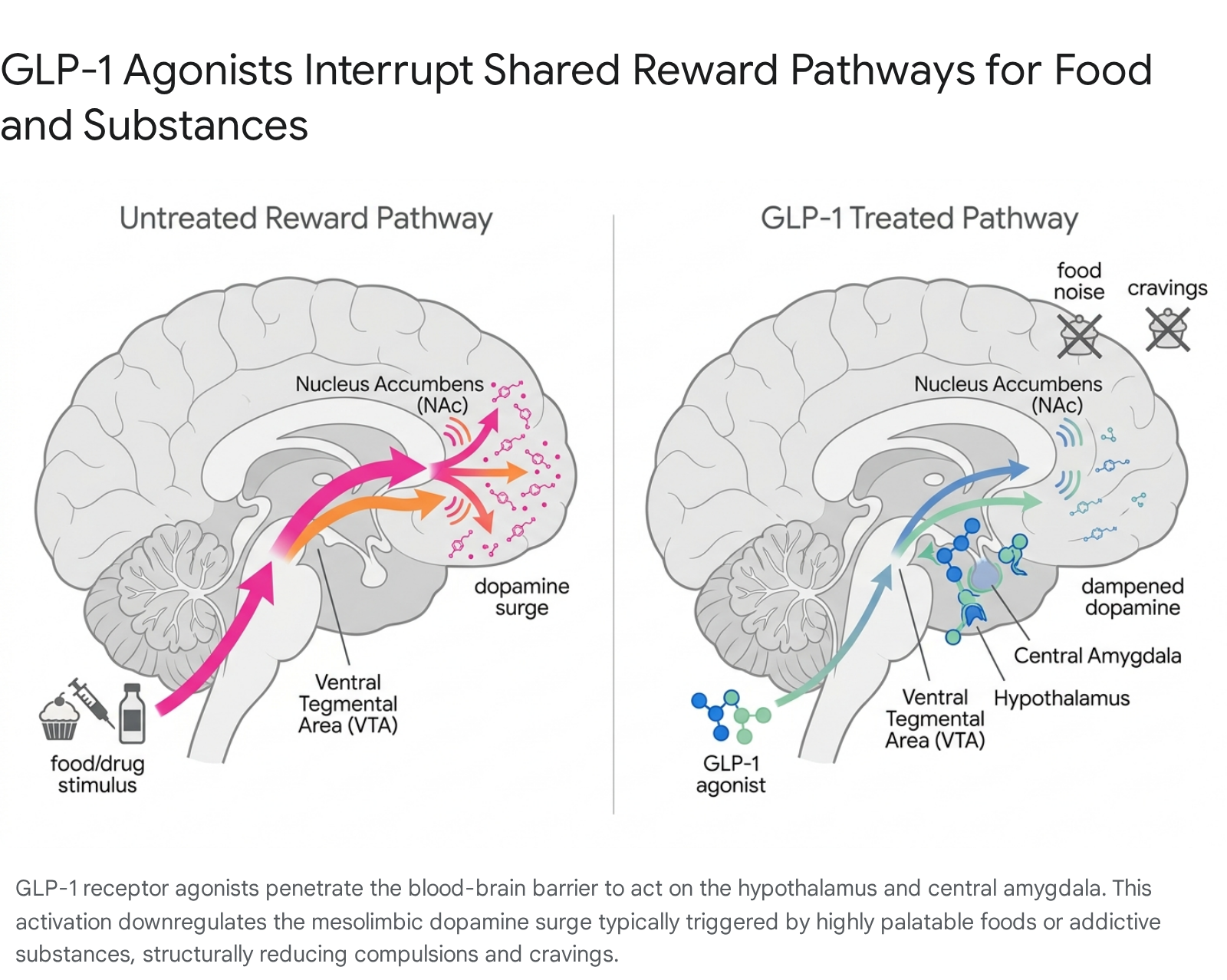
This neurobiological crossover elegantly explains the serendipitous clinical observations reported by thousands of patients worldwide: a medication originally developed to control blood glucose and shed adipose tissue simultaneously extinguishes the lifelong desire to smoke cigarettes, drink alcohol, or use illicit substances. The exact neural architecture that forces a patient to uncontrollably crave sugar is the identical architecture that drives a patient to crave fentanyl or alcohol; by dampening this circuit at the receptor level, GLP-1 RAs provide a unified pharmacological intervention for the disease of compulsion.
Clinical Translation: GLP-1s in Substance Use Disorders (SUD)
The translation of these preclinical neurobiological findings into rigorous human clinical trials has generated a wave of "cautious optimism" within the field of addiction psychiatry 9. Historically, the pharmacological armamentarium for treating substance use disorders has been severely limited, suffering from poor patient compliance, variable efficacy, and heavy social stigma. For instance, the FDA has approved only three medications for Alcohol Use Disorder (naltrexone, acamprosate, and disulfiram) over the past several decades, and these therapies remain vastly underutilized by the psychiatric and primary care communities 910. The repurposing of GLP-1 RAs, agents already integrated into mass-market primary care for diabetes and obesity, offers a highly accessible and potentially more efficacious intervention vector.
Alcohol Use Disorder (AUD)
Recent high-tier clinical trials provide compelling prospective evidence for the utility of incretins in combating alcohol addiction. A milestone randomized, double-blind, placebo-controlled trial published in The Lancet in May 2026 investigated the efficacy of semaglutide in 108 treatment-seeking adult patients with moderate-to-severe AUD and comorbid obesity. The trial took place at a mental health center in Denmark, combining pharmacological intervention with standardized cognitive behavioral therapy 91112.
At baseline, participants reported an average of 17 heavy drinking days over the preceding 30 days. After six months of intervention, participants receiving a once-weekly semaglutide injection experienced a profound 41.1% reduction in heavy drinking days, averaging a reduction of 12 days per month - this was 50% greater than the eight-day reduction observed in the placebo cohort 101113. Furthermore, total monthly alcohol consumption plummeted from an approximate baseline of 2,200 grams of alcohol to roughly 650 grams per month for those on semaglutide, vastly outperforming the placebo group's drop to 1,175 grams 1112.
Similarly, a 2025 Phase II study published in JAMA Psychiatry confirmed these therapeutic signals. Evaluating 48 patients with AUD, researchers found that low-dose semaglutide significantly reduced the amount of alcohol consumed during rigorous post-treatment laboratory self-administration tasks. The trial demonstrated medium-to-large statistical effect sizes for both the total grams of alcohol consumed ($\beta = -0.48; P = .01$) and the peak breath alcohol concentration achieved ($\beta = -0.46; P = .03$). Crucially, participants receiving semaglutide also experienced a statistically significant reduction in subjective weekly alcohol cravings ($\beta = -0.39; P = .01$), firmly establishing the drug's action on the psychological components of addiction 14.
Opioid, Nicotine, and Broad-Spectrum Addiction Mitigation
Beyond tightly controlled clinical trials, massive real-world observational datasets highlight the profound systemic benefits of GLP-1 adoption across broader addicted populations. A landmark observational study published in The BMJ in March 2026 monitored a cohort of 606,434 U.S. veterans diagnosed with type 2 diabetes over a three-year period. The sheer scale of this study allowed researchers to identify population-level psychiatric trends associated with incretin therapy.
For veterans with no prior history of SUDs, initiating a GLP-1 RA was linked to a 14% overall reduced risk of developing any new substance use disorder compared to those utilizing SGLT-2 inhibitors. When broken down by specific substance classes, the risk reductions were strikingly robust and broad-spectrum: researchers observed a 25% reduced risk for developing opioid use disorder, a 20% reduced risk for nicotine use disorder, a 20% reduced risk for cocaine use disorder, and an 18% reduced risk for alcohol use disorder 1315.
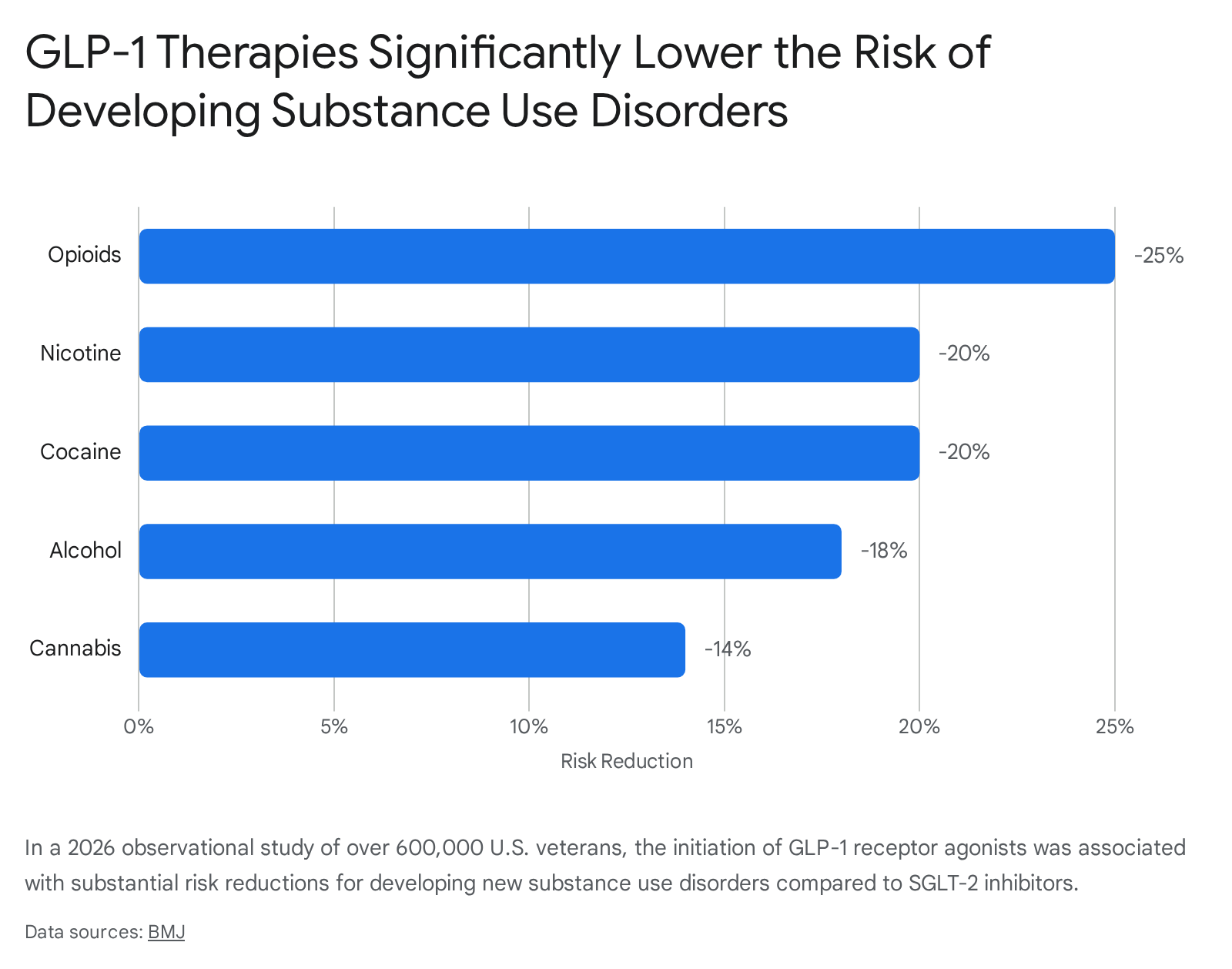
Perhaps even more critically for public health policy, among veterans who already carried a preexisting SUD diagnosis at the time of drug initiation, GLP-1 RAs correlated with massive risk reductions in severe, life-threatening clinical endpoints. Compared to standard care, GLP-1 users experienced a 50% reduction in all-cause mortality, a 39% reduction in drug overdoses, a 31% reduction in emergency department visits, and a 25% reduction in suicidal ideation or attempts 1526. These statistics suggest a profound stabilization of the psychiatric, behavioral, and compulsive comorbidities associated with severe addiction.
Recognizing this immense therapeutic potential, a multitude of randomized controlled trials have been rapidly initiated to formalize these indications. In the realm of nicotine addiction, studies such as NCT03712098 are investigating the effects of liraglutide on smoking abstinence and the mitigation of post-cessation weight gain (a primary driver of smoking relapse), while NCT07059377 is testing semaglutide as an adjunct to nicotine replacement therapy in diabetic patients 7816. For opioid use disorder (OUD), trials like NCT06548490 are assessing whether weekly semaglutide increases the probability that outpatient individuals enrolled in buprenorphine or methadone maintenance programs will refrain from using illicit and nonprescribed opioids over a 19-week period 1718.
The table below synthesizes the current landscape of high-impact clinical data regarding GLP-1 utility in SUDs.
Table 2: Ongoing Clinical Trials and Observational Data for GLP-1s in Substance Use Disorders
| SUD Category | Target Medication | Study Type & Status | Key Outcome Measures / Findings | Source Identifier |
|---|---|---|---|---|
| Alcohol (AUD) | Semaglutide | RCT (Lancet, 2026) | 41.1% reduction in heavy drinking days; Total alcohol consumption fell from 2200g to 650g/month. | 1011 |
| Alcohol (AUD) | Semaglutide | Phase II (JAMA Psych, 2025) | Reduced self-administration ($\beta = -0.48$); lowered peak breath alcohol concentration. | 14 |
| Nicotine/Tobacco | Liraglutide | Phase II Pilot (NCT03712098) | Investigating smoking abstinence and mitigation of post-cessation weight gain. | 7 |
| Nicotine/Tobacco | Semaglutide | Phase IV (NCT07059377) | Testing semaglutide as an adjunct to nicotine replacement therapy in diabetic patients. Est. Completion: 2027. | 8 |
| Opioids (OUD) | Semaglutide | Phase II (NCT06548490) | Assessing likelihood of participants remaining abstinent from illicit opioids while on buprenorphine/methadone. | 17 |
| Broad SUDs | GLP-1 RAs (Class) | Observational (BMJ, 2026) | 600,000+ patient cohort. -25% risk for opioids, -20% nicotine, -20% cocaine. 39% reduction in overdoses for existing SUDs. | 1526 |
Expanding Indications: Beyond Metabolism into Systemic Preservation
While the psychiatric benefits of GLP-1 RAs represent the vanguard of neurobehavioral research, the therapeutic scope of these agents continues to aggressively widen across traditional physiological disciplines, fundamentally altering treatment algorithms in nephrology, cardiology, and pulmonology. To adequately conceptualize this broad utility, the medical community has coalesced around the framework of "Cardiovascular-Kidney-Metabolic" (CKM) syndrome. This framing acknowledges the deeply intertwined pathology of obesity, hypertension, renal decline, and cardiovascular disease, positioning GLP-1s not as targeted symptom relievers, but as systemic disease modifiers at the center of CKM management 219.
Chronic Kidney Disease (CKD)
A historic shift in nephrology occurred in January 2025 when the FDA officially approved semaglutide (Ozempic) to reduce the risk of kidney disease worsening, end-stage kidney failure, and cardiovascular death in adults suffering from type 2 diabetes and chronic kidney disease (CKD) 220. This regulatory milestone was predicated entirely on the results of the massive Phase 3b FLOW trial, a multinational, double-blind study spanning 28 countries. The FLOW trial was halted early by an independent monitoring committee because the data demonstrated overwhelming efficacy that made continuing the placebo arm unethical 221.
The finalized data revealed that patients receiving a 1.0 mg weekly dose of semaglutide slowed the overall progression of their kidney disease by an astonishing 24%, while simultaneously reducing their risk of major cardiovascular events by 18% compared to the placebo cohort 23. Obesity profoundly contributes to CKD through renal hyperfiltration, barotrauma, adipose-driven systemic inflammation, and severe insulin resistance. By modulating these upstream drivers, semaglutide acts as a potent nephroprotective agent. For nephrologists, this approval represents a paradigm shift away from relying solely on SGLT-2 inhibitors, moving treatment guidelines toward a synergistic, dual-therapy approach combining "flozins" with GLP-1 RAs to maximize renal preservation 1921.
Obstructive Sleep Apnea (OSA)
Further disrupting classical treatment pathways, Eli Lilly's tirzepatide (Zepbound) achieved an unprecedented FDA approval in late 2024, becoming the very first pharmacological treatment indicated for moderate-to-severe obstructive sleep apnea in adults with obesity 1233. Historically, OSA management was entirely mechanical, relying on Continuous Positive Airway Pressure (CPAP) machines, oral appliance therapy, or invasive surgical interventions to prevent the airway collapse that defines the syndrome 1222.
Tirzepatide's approval was driven by the SURMOUNT-OSA phase III clinical trials, which evaluated the dual-agonist in patients both utilizing and not utilizing concurrent CPAP therapy. The results were revelatory: tirzepatide administration drove a profound 63% reduction in the apnea-hypopnea index (AHI). In real clinical terms, this equated to approximately 25 fewer breathing interruption events per hour of sleep, compared to merely 5 fewer events in the placebo group 45. Remarkably, between 42% and 50% of patients treated with tirzepatide achieved total OSA remission or transitioned to a state of mild, non-symptomatic disease by week 52 of the trial. This pharmacological success challenges the absolute necessity of mechanical CPAP therapy for obesity-driven sleep apnea, transforming OSA from a mechanically managed anatomical defect into a biologically modifiable metabolic syndrome 433.
Metabolic Dysfunction-Associated Steatohepatitis (MASH)
The liver has also emerged as a primary beneficiary of incretin therapy. In August 2025, Wegovy (semaglutide 2.4 mg) secured accelerated FDA approval for the treatment of noncirrhotic metabolic dysfunction-associated steatohepatitis (MASH) in adults with moderate-to-advanced liver fibrosis (stage F2 to F3). This approval, based on the Phase 3 ESSENCE trial, positions Wegovy as only the second drug globally approved for MASH, and the sole GLP-1 agent bearing the indication. At the week 72 interim analysis of the ESSENCE trial, 62.9% of semaglutide-treated patients achieved complete clinical resolution of steatohepatitis without any worsening of fibrosis, nearly doubling the 34.3% resolution rate observed in the placebo group 24.
The Rebound Effect: The Physiological Realities of Discontinuation
Despite the unparalleled, multi-system efficacy of GLP-1 receptor agonists, the scientific literature is unequivocal on one fundamental point: they are not curative. Obesity, type 2 diabetes, and the spectrum of CKM morbidities are chronic, progressive, and highly relapsing diseases. Discontinuation of incretin therapy reliably initiates a severe physiological rebound, exposing critical gaps in long-term disease maintenance strategies and testing the durability of modern healthcare budgets.
When GLP-1 receptor agonism ceases, the exogenous pharmacological suppression of appetite and the artificial slowing of gastric emptying are rapidly reversed. However, the body's deeply entrenched metabolic adaptations to the drug-induced weight loss remain active and hostile. During periods of rapid weight loss, mammalian biology initiates an evolutionary survival response aimed at preventing starvation. This response is characterized by a drastic reduction in resting energy expenditure, a surge in the secretion of orexigenic (hunger-promoting) hormones such as ghrelin from the stomach, and a precipitous drop in anorexigenic (satiety) hormones like leptin from adipose tissue and peptide YY (PYY) from the gut 352324. When the pharmacological "brake" of the GLP-1 drug is abruptly removed, these powerful hormonal currents operate unchecked, promoting intense hyperphagia (extreme hunger) and aggressive energy conservation, which ultimately drives rapid weight regain.
The clinical magnitude of this rebound is substantial. A comprehensive 2025 meta-analysis investigating the metabolic and cardiovascular deterioration following GLP-1 discontinuation meticulously quantified this trajectory. In patients treated for obesity, cessation of therapy led to a clinically significant mean body weight regain of 5.63 kg, alongside marked, rapid deteriorations in waist circumference, overall body mass index, and systolic blood pressure 25. Glycemic parameters also violently reversed course, with HbA1c rising by 0.25% in the obesity cohort and a troubling 0.65% in the type 2 diabetes cohort 25. Subgroup analyses within this data revealed that discontinuing semaglutide resulted in significantly more severe rebound effects than discontinuing the older liraglutide molecule, with semaglutide patients regaining an average of 8.21 kg compared to liraglutide's 4.29 kg 25.
Furthermore, a rigorous 2026 exponential recovery regression model tracking 3,236 participants characterized the exact mathematical trajectory of this weight rebound. The regression model estimated that weight regain inevitably plateaus at 75.6% of the total weight lost while on the treatment. The biological recovery rate constant corresponded to a half-life of exactly 23.0 weeks, indicating that one year after cessation of GLP-1 therapy, only an estimated 40.2% of the on-treatment weight loss remains preserved 39. These data - corroborated by extension studies from the STEP 1, STEP 4, and SURMOUNT-4 trials - unequivocally define GLP-1 RAs as chronic, lifelong therapies 232426. This physiological reality heavily compounds the ongoing socioeconomic challenges surrounding long-term drug costs, supply chain stability, and the reluctance of insurance providers to fund lifelong maintenance dosing.
Macroeconomic Ripples: The Food and Beverage Industry's Pivot
The pervasive, mass-market adoption of GLP-1s - with approximately 12% of U.S. adults reporting having utilized the medication class for weight loss or diabetes 8 - has triggered a defensive, strategic, and highly capitalized realignment within the global food and beverage industry. As these medications systemically curb overall caloric intake and fundamentally alter the neurological nature of consumer cravings, multi-national conglomerates are facing a structural economic shift. The industry is being forced away from the high-volume, impulse-driven consumption of ultra-processed foods that dominated the late 20th century, moving toward an era of highly intentional, discerning eating 27.
Food manufacturers are aggressively adapting their product lines to survive in what market analysts have dubbed the "GLP-1 economy." Designing products for the mainstream market now increasingly requires achieving "GLP-1 compatibility." This new product development framework prioritizes extreme portion flexibility, high nutrient density per bite, and digestive comfort (as GLP-1s often cause gastrointestinal distress with heavy, greasy meals) 27. Crucially, because incretin therapies inherently cause a loss of lean muscle mass alongside adipose tissue loss, protein preservation has become the paramount dietary goal for medicated patients. Consequently, consumer packaged goods giants are reformulating vast swathes of their portfolios to be aggressively protein-forward 2742.
A premier macroeconomic example of this pivot is Nestlé, the world's largest food and beverage company. Confronted with internal and industry data demonstrating a substantial shift in consumer behavior toward nutrition and portion control, Nestlé launched the Vital Pursuit brand in 2024. This line was explicitly marketed and formulated for individuals utilizing weight management medications, featuring high-protein, high-fiber, portion-controlled frozen meals 42. By early 2026, the financial success of this defensive strategy became evident in their fiscal reports. Nestlé's CEO, Philipp Navratil, explicitly noted to investors that the Vital Pursuit line had reached a much broader audience than originally anticipated. The brand's success helped drive positive real internal growth (RIG) of 0.8% across its frozen food sectors in 2025, accelerating to 1.4% in the second half of the year, effectively stabilizing the company's market share amidst broader industry headwinds and volume collapses in traditional snack categories 422829.
The future of the food sector in high-GLP-1-adoption markets relies entirely on extracting "higher value per unit" through functional nutrition, rather than relying on the sheer caloric volume of past decades 30.
Health Equity, Access, and Geopolitical Pricing Divergence
The structural biological reliance on GLP-1 RAs as chronic, lifelong therapies exacerbates profound existing disparities in global healthcare access. The pricing, distribution, and reimbursement models for these agents vary drastically depending on the geopolitical environment, revealing stark contrasts between free-market direct-to-consumer healthcare models and centralized public health systems.
The United States: MFN Pricing and the "TrumpRx" Intervention
In the United States, the historic list prices of GLP-1s - frequently exceeding $1,000 to $1,350 per month - created a massive systemic barrier to access. This extreme pricing structure heavily skewed utilization toward affluent demographics capable of paying out-of-pocket, or those privileged with premium, top-tier employer-sponsored health insurance. This paradigm, fueled by aggressive direct-to-consumer (DTC) pharmaceutical advertising, resulted in explosive, impulsive patient demand that routinely crashed global supply chains.
However, the U.S. regulatory and pricing landscape shifted dramatically and forcefully in November 2025, when the Trump Administration brokered a landmark pharmaceutical pricing agreement with the primary manufacturers, Eli Lilly and Novo Nordisk 314748. Executing the administration's "Most Favored Nation" (MFN) executive order - which mandates that U.S. prescription drug prices formally align with the lowest prices paid by other developed, wealthy nations - the administration slashed the direct-to-consumer costs of these medications by up to 74% 3147.
Scheduled to launch through a new federal direct-purchasing platform named "TrumpRx" in 2026, the out-of-pocket monthly costs for the injectable semaglutide agents Ozempic and Wegovy were forcefully capped at $350. Concurrently, Eli Lilly's Zepbound was reduced to an average of $346 per month 314832. Notably, the agreement preemptively addressed the future of the market, declaring that introductory doses of any future FDA-approved oral GLP-1 drugs (such as the impending orforglipron tablet) will be capped at just $150 per month through the platform 3150.
Simultaneously, the administration achieved a massive policy victory by expanding Medicare Part D coverage to include anti-obesity medications for the very first time. Under the negotiated agreement, Medicare and state Medicaid programs will procure these agents at a fixed, deeply discounted price of $245 per month, with senior beneficiaries liable for a strictly capped $50 monthly out-of-pocket co-pay 3148. In exchange for these unprecedented, market-altering price concessions, the pharmaceutical manufacturers were granted highly lucrative three-year tariff exemptions on imported branded drugs, legally contingent upon their continued capital investments in domestic U.S. manufacturing infrastructure 313250. While this federal intervention radically democratizes patient access, it introduces massive logistical and financial complexities for employer-sponsored health plans and Pharmacy Benefit Managers (PBMs), whose entire business models and rebate structures are deeply intertwined with historically inflated list prices 47.
Europe and the UK: Centralized Triage and Fragmented Reimbursement
Conversely, European markets utilize highly centralized, cost-efficacy frameworks to regulate GLP-1 adoption. Rather than allowing DTC advertising to drive impulsive consumer demand, European health ministries actively buffer their public health budgets against sudden utilization spikes through strict clinical triage and rigid prescribing pathways 30.
In the United Kingdom, the National Institute for Health and Care Excellence (NICE) updated its clinical guidelines in early 2026 to massively expand, yet carefully target, GLP-1 eligibility. The updated guidelines formally recommend GLP-1 RAs and tirzepatide as standard triple-therapy options for highly specific, high-risk cohorts: Type 2 diabetics diagnosed under the age of 40, those with established cardiovascular disease caused by blocked arteries, and those living with clinical obesity. This policy shift expanded eligibility to an estimated 810,000 new patients across the UK 3334. To creatively fund this massive expansion without bankrupting the system, the NHS strategically leveraged the recent patent expiration of older diabetes drugs. The systemic transition to generic dapagliflozin (an SGLT-2 inhibitor) is projected to save the NHS £560 million between 2025 and 2027, creating the exact fiscal space required to subsidize the expensive incretin therapies for those who need them most 3334.
In mainland Europe, however, the reimbursement landscape remains highly fragmented, rigid, and deeply conservative. While the World Health Organization (WHO) formally recognized obesity as a chronic, relapsing disease in December 2025 and recommended GLP-1s for its long-term treatment, individual national policies significantly trail behind these global mandates 35. In Germany, statutory public health insurance (GKV) explicitly and legally classifies weight-loss medications as "lifestyle drugs," categorically excluding them from public reimbursement. This forces German patients relying on GLP-1s purely for weight management into a strict out-of-pocket model 5436. Similarly, in France, access to Wegovy is largely restricted to private prescribers and out-of-pocket payment, with public coverage strictly gated behind severe, multi-morbid clinical requirements 5436. Consequently, European adoption will continue to grow methodically along strict medical prescribing pathways, fundamentally avoiding the rapid, consumer-driven hyper-adoption and subsequent supply shocks observed in the United States 30.
Sociological Crossroads: GLP-1s vs. the Fat Acceptance Movement
The sudden, meteoric ascendancy of highly effective GLP-1 RAs has triggered complex, often painful sociological friction, directly colliding with the core tenets of the fat acceptance and body positivity movements. Over the past decade, these vital sociological movements fought aggressively to decouple human body size from moral value, advocating that fatness is a natural, acceptable variation of human biology rather than an individual moral failure requiring eradication 2637.
The introduction and mass popularization of highly efficacious, medicalized weight-loss injections has inadvertently re-pathologized larger bodies in the public consciousness, reinforcing systemic anti-fat biases under the unassailable guise of "accessible medicine." A poignant 2025 study published in the International Journal of Behavioral Medicine, corroborated by a 2026 analysis, highlighted a novel and insidious layer of stigma facing modern GLP-1 users. In randomized psychological experiments, participants consistently judged individuals who lost weight using a GLP-1 RA significantly more harshly than those who lost weight through traditional lifestyle modifications (diet and exercise) alone 3839.
Utilizing a pharmaceutical intervention was widely perceived by the public as taking an undisciplined "shortcut." This reaction reveals deeply entrenched societal beliefs regarding weight controllability: the public largely believes that weight loss must fundamentally involve suffering, immense willpower, and self-deprivation to be deemed morally valid and worthy of respect 3840. Even more startlingly, the research found that GLP-1 users were occasionally viewed less positively regarding their social desirability and moral character than individuals with obesity who made no attempt to lose weight at all 3940. The stigma of the "easy way out" proved heavier than the stigma of remaining fat.
Furthermore, as these drugs become cheaper and universally ubiquitous - particularly following the 2025 U.S. Medicare inclusions and the TrumpRx price cuts - clinical bioethicists warn of a looming, dangerous era of "coerced weight loss." In a society that intrinsically stigmatizes fatness, the sudden availability of a highly effective "cure" shifts the entirety of the burden of treatment onto the patient. Fat individuals who decline a GLP-1 prescription - whether due to valid concerns about severe gastrointestinal side effects, the loss of lean muscle mass, or simply a desire to accept their current body size - risk being perceived by their healthcare providers as inherently irrational, non-compliant, or careless regarding their own health 37. This dynamic threatens to make thinness an unspoken, coerced prerequisite for receiving equitable, unbiased medical care across all disciplines, forcing patients to medicate not for their metabolic health, but to survive a biased medical system.
Conclusion
The era of GLP-1 receptor agonists represents one of the most profound and rapid pharmacological revolutions of the 21st century. By successfully targeting the deep neurological intersection of metabolic homeostasis and mesolimbic reward circuitry, these agents have redefined the clinical management of obesity, type 2 diabetes, cardiovascular disease, chronic kidney disease, and, increasingly, the devastating cycle of substance use disorders.
The clinical efficacy of these molecules is undeniably historic. From the near-eradication of obstructive sleep apnea to striking, life-saving reductions in alcohol, opioid, and nicotine consumption, incretin therapies offer a unified, multi-system approach to chronic disease management that was previously unimaginable. However, the harsh physical reality of the discontinuation rebound necessitates that these medications be viewed strictly as lifelong interventions, not as temporary cures.
As the global macroeconomic landscape adapts - evidenced by the massive food industry's pivot toward functional nutrition and the aggressive, geopolitical restructuring of drug pricing via Most Favored Nation policies - the broader societal implications remain incredibly complex. The ultimate historical legacy of GLP-1 receptor agonists will be determined not solely by their ability to reduce HbA1c or lower body mass indices, but by how global healthcare systems and societies manage the ethical, sociological, and immense financial realities of sustaining millions of human beings on chronic neurometabolic therapy for the remainder of their lives.