Clinical Mechanisms of the Placebo Effect
Conceptual Framework and Definitions
The placebo effect is a distinct psychobiological phenomenon encompassing the physiological and psychological changes that occur when an individual receives an inert treatment but harbors expectations of therapeutic benefit 12. While historically dismissed as a nuisance variable in clinical trials, the placebo effect is now understood as a fundamental component of the human healing response, mediated by specific neural networks and neurotransmitter systems 134.
In clinical research, a critical distinction exists between the "placebo response" and the "placebo effect." The placebo response is a broad umbrella term describing all clinical outcomes observed within the control arm of a clinical trial. This response includes the genuine psychobiological placebo effect, but also incorporates extraneous variables such as the natural history of the disease, spontaneous remission, statistical regression to the mean, and the Hawthorne effect - where patient behavior alters simply due to being observed 1355. Conversely, the genuine placebo effect isolates the specific psychobiological mechanisms triggered by the therapeutic context, the patient-provider relationship, and the patient's internal expectations 135.
The reciprocal phenomenon is the nocebo effect, which describes the worsening of symptoms or the manifestation of adverse side effects triggered by negative expectations 126. Nocebo effects frequently emerge when patients anticipate harm, experience anxiety regarding a medical intervention, or are exposed to negatively framed medical information 78. Both the placebo and nocebo effects demonstrate that the context surrounding a treatment - including verbal suggestions, environmental cues, and prior learning - can independently alter physiological function 237.
Neurobiology of Placebo Responses
The physiological basis of the placebo effect has been extensively mapped, particularly within the domains of nociception (pain perception) and movement disorders. Advanced functional neuroimaging, including functional magnetic resonance imaging (fMRI) and positron emission tomography (PET), alongside pharmacological blockade experiments, has isolated the specific neurotransmitter networks and anatomical brain regions responsible for translating subjective expectation into somatic experience 34910.
Pain Modulation and the Opioidergic System
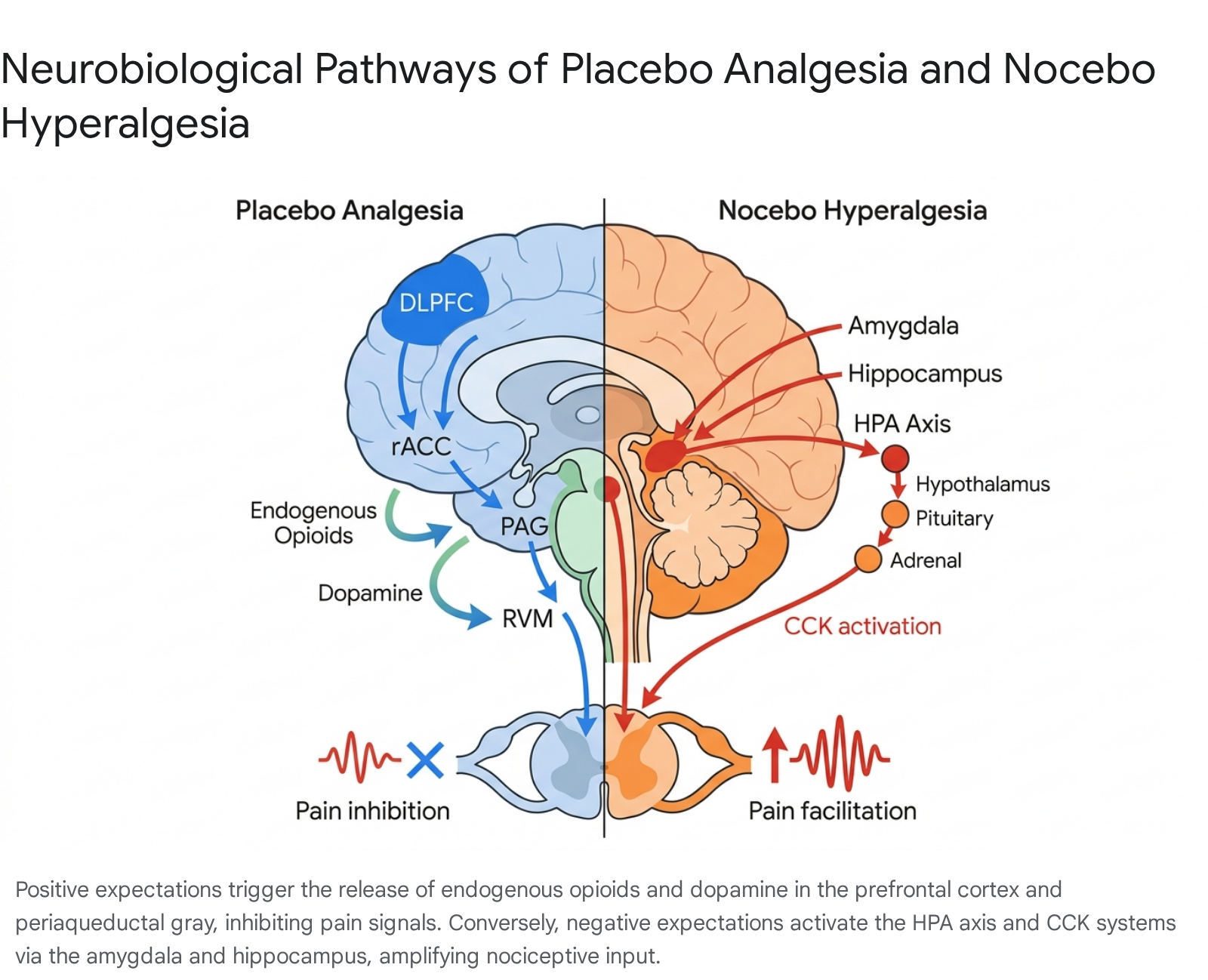
Placebo analgesia is primarily mediated by the descending pain-modulating circuit, a network that relies heavily on the deployment of endogenous opioids. Upon the expectation of pain relief, robust hemodynamic activation is observed in the dorsolateral prefrontal cortex (DLPFC), the rostral anterior cingulate cortex (rACC), and the periaqueductal gray (PAG) 41011. This cortical and midbrain activation initiates a downward cascade to the rostral ventromedial medulla (RVM) and the spinal cord, which actively inhibits ascending pain signals in the mid and posterior cingulate cortex, insula, and thalamus 4911.
The specific involvement of the endogenous opioid system is confirmed by pharmacological blockade studies. When patients are administered naloxone - a mu-opioid receptor antagonist - prior to a placebo intervention, the placebo analgesic effect is completely abolished 12. Furthermore, PET imaging reveals that the administration of a placebo activates the exact same cortical and brainstem regions as the administration of the synthetic opioid remifentanil, indicating a shared neural architecture between pharmacological and psychological analgesia 1112.
Dopaminergic Pathways and Movement Disorders
While the opioidergic system dominates nociceptive modulation, the dopaminergic system mediates placebo responses in motor control and reward expectation. In patients with Parkinson's disease, the administration of a placebo triggers significant dopamine release in the ventral (nucleus accumbens) and dorsal striatum 12. In some cases, this placebo-induced extracellular dopamine concentration increases by over 200%, a magnitude comparable to the neurochemical response observed following the administration of amphetamines in healthy individuals 12.
Intraoperative single-neuron recordings provide further objective evidence of this phenomenon. During the surgical implantation of deep brain stimulation (DBS) electrodes in Parkinson's patients, researchers observed that administering a placebo decreased the firing rates of neurons in the subthalamic nucleus and the substantia nigra pars reticulata, while simultaneously increasing firing rates in the ventral anterior and anterior ventral lateral thalamus 12. This neuroelectric shift correlates directly with a measurable, objective reduction in clinical muscle rigidity 12.
Alternative Neurochemical Pathways
Recent neurobiological profiling indicates that placebo effects are not monolithic; they engage diverse biochemical networks depending on the specific clinical context. Endocannabinoid systems, utilizing CB1 receptors, modulate certain types of placebo analgesia that remain functional even when endogenous opioids are blocked 12. Furthermore, lipidic pathways involving arachidonic acid, prostaglandins, and thromboxane have been implicated in the placebo modulation of specific conditions, such as hypoxia-induced headaches 12.
Neurobiology and Mechanisms of the Nocebo Effect
The nocebo effect operates through neurobiological mechanisms that are functionally inverse to those of the placebo effect. Driven fundamentally by the anticipation of harm, fear, and state anxiety, the nocebo effect actively amplifies sensory transmission and exacerbates symptomatology 41013.
Cholecystokinin and the Hypothalamic-Pituitary-Adrenal Axis
Nocebo hyperalgesia is primarily mediated by the pro-nociceptive cholecystokinin (CCK) system 41213. CCK functions as an endogenous anti-opioid, counteracting the body's natural pain-relieving mechanisms and facilitating the transmission of pain signals from the periphery to the brain 13. When an individual expects pain to worsen, neuroimaging demonstrates heightened activation in the anterior cingulate cortex, insula, amygdala, and hippocampus 91014.
This anticipation of an adverse event simultaneously triggers a systemic stress response via the hypothalamic-pituitary-adrenal (HPA) axis, resulting in elevated cortisol levels and broad sympathetic nervous system arousal 1013. The definitive role of the CCK pathway is validated by pharmacological research showing that proglumide - a non-specific CCK-1/2 receptor antagonist - completely blocks nocebo hyperalgesia 1112. Unlike placebo analgesia, nocebo hyperalgesia is unaffected by the administration of naloxone, confirming that it operates independently of the endogenous opioid system 11.
Psychological Drivers: State Anxiety and Expectancy
The induction of the nocebo effect relies heavily on immediate cognitive and affective states rather than immutable personality traits. A 2023 systematic review and meta-analysis by Rooney et al., which analyzed 59 independent studies comprising over 3,000 participants, quantified the impact of these variables 815. The analysis found that nocebo manipulations reliably generate a moderate overall effect size across health outcomes (Hedges' g = 0.522) 816.
The primary drivers of this effect are situationally induced negative expectancy (g = 0.837) and acute state anxiety (g = 0.312) 81517. Interestingly, the meta-analysis found no significant correlation between the magnitude of a nocebo response and dispositional or "trait" anxiety (r = 0.021) 81517. This indicates that patients are not inherently "nocebo responders" based on a baseline anxious personality; rather, the nocebo effect is highly malleable and heavily dependent on the specific clinical context, the immediate threat perception, and the precise framing of information provided by healthcare professionals 817.
Conditioning, Social Learning, and Extinction
While explicit verbal instruction (e.g., warning a patient about a side effect) effectively induces nocebo responses, social learning and observation are equally potent. Meta-analytical data demonstrates that observing another individual experience an adverse reaction generates a socially-induced nocebo effect with a medium-to-large magnitude (g = 0.74), an effect size comparable to direct classical conditioning 18.
Furthermore, evidence suggests that the nocebo effect is biologically more powerful and more resistant to extinction than the placebo effect. A 2025 within-subject experimental study by Kunkel et al. directly compared the magnitude and persistence of placebo and nocebo effects on experimental pain. On the initial day of induction, nocebo effects were vastly stronger (Cohen's d = 1.46) than placebo effects (d = 0.38) 1920. Upon re-testing one week later without further conditioning, both effects persisted, but the nocebo effect maintained a consistently higher magnitude 1920. Neuroscientists attribute this asymmetry to an evolutionarily advantageous "better safe than sorry" biological strategy, where the central nervous system prioritizes, amplifies, and durably encodes aversive, threat-related stimuli to ensure survival 192021.
| Neurobiological Domain | Placebo Effect (Analgesia/Relief) | Nocebo Effect (Hyperalgesia/Adverse) |
|---|---|---|
| Primary Psychological Driver | Positive Expectancy, Conditioning | Negative Expectancy, State Anxiety |
| Key Neurotransmitters | Endogenous Opioids, Dopamine, Endocannabinoids | Cholecystokinin (CCK), Cortisol |
| Core Brain Regions Activated | DLPFC, rACC, PAG, Nucleus Accumbens | Amygdala, Hippocampus, Insula, HPA Axis |
| Pain Pathway Modulation | Descending Inhibition (Suppression) | Descending Facilitation (Amplification) |
| Pharmacological Blockade | Naloxone (Mu-opioid antagonist) | Proglumide (CCK-1/2 antagonist) |
Genetic Mediators and the Placebome
The pronounced variability in placebo responsiveness among individuals - where some experience profound relief from inert treatments while others experience none - has driven genomic research. This research has culminated in the concept of the "placebome," a network of genetic biomarkers that predispose patients to stronger or weaker placebo and nocebo responses 2322. Because the placebo effect relies on the synthesis, signaling, and degradation of specific neurotransmitters, genetic variations governing these neurochemical pathways directly influence clinical outcomes 2322.
Catechol-O-Methyltransferase (COMT)
In 2012, researchers identified the catechol-O-methyltransferase (COMT) gene as the first definitive placebo biomarker 2323. COMT codes for an enzyme responsible for metabolizing and degrading dopamine in the prefrontal cortex. Common genetic variations, or single nucleotide polymorphisms (SNPs) such as rs4680, dictate the efficiency of this enzyme 2224.
Individuals possessing the met/met genotype produce a less active form of the COMT enzyme, leading to slower dopamine degradation and consequently higher baseline levels of dopamine in the prefrontal cortex 222425. Clinical trial data demonstrates that individuals with the met/met genotype mount significantly stronger placebo responses compared to those with the val/val genotype (which codes for a highly efficient enzyme and lower baseline dopamine) 2224.
Monoamine Oxidase A and Other Pathways
Polymorphisms in the monoamine oxidase A (MAO-A) gene also heavily influence placebo susceptibility. MAO-A is another critical enzyme involved in the regulation of monoamines, including dopamine and norepinephrine 25. In studies involving patients diagnosed with major depressive disorder (MDD), researchers observed that individuals possessing genotypes coding for the highest MAO-A enzyme activity exhibited significantly lower placebo responses 25.
The expanding placebome framework now includes evidence of genetic variations in the endogenous opioid pathways, the endocannabinoid system (such as the FAAH gene), and the serotonin system 232224.
Implications for Clinical Trial Design
The discovery of the placebome holds profound implications for the design and interpretation of randomized controlled trials (RCTs). The foundational assumption of the RCT is that the placebo control arm accurately establishes a baseline against which an active drug can be measured. However, if a trial inadvertently enrolls a disproportionate number of high-placebo responders (e.g., COMT met/met genotypes), the exceptionally high response rate in the control arm may artificially obscure the genuine efficacy of the active drug 232223.
Furthermore, researchers highlight the risk of placebo-drug interactions. Because active pharmacological agents and placebos often utilize the exact same neurotransmitter pathways, they can interfere with one another 2323. For example, the antidepressant paroxetine is metabolized by an intermediate that interacts with the COMT pathway. Consequently, a patient's COMT genotype simultaneously alters both their baseline placebo response and their specific pharmacological response to paroxetine 24. To untangle these variables, researchers increasingly advocate for the inclusion of a "no-treatment" control arm in future clinical trials, allowing for the precise isolation of true drug effects from contextually driven placebo responses 232223.
Efficacy Across Clinical Outcomes
The potency of the placebo effect is strictly bounded by the nature of the target pathology. Research systematically demonstrates a severe divergence in placebo efficacy between conditions governed by subjective, central nervous system-regulated symptoms and those characterized by objective, organic disease markers 12.
Psychiatric and Subjective Disorders
Conditions relying primarily on patient self-reporting - such as depression, generalized anxiety disorder, chronic pain, and irritable bowel syndrome - are highly susceptible to placebo interventions 12. A comprehensive 2024 systematic review and meta-analysis published in JAMA Psychiatry analyzed 90 high-quality RCTs to quantify placebo responses across nine distinct psychiatric diagnoses 262728.
The analysis revealed that placebo treatments generate substantial, clinically meaningful symptom improvement across the psychiatric spectrum, though the magnitude varies widely based on the underlying pathology 2628. The strongest placebo effect sizes (measured as standardized pre-post effect size, dav) were observed in major depressive disorder (dav = 1.40) and generalized anxiety disorder (dav = 1.23) 2628. Mid-range effects were noted in panic disorder (dav = 1.09) and PTSD (dav 0.68 - 0.92) 2628. The weakest placebo effects were documented in conditions with rigid neurological or rigid behavioral pathologies, such as schizophrenia (dav = 0.59) and obsessive-compulsive disorder (dav = 0.65) 2628.
The nature of the condition also dictates patient retention in placebo arms. The same dataset revealed that dropout rates for placebo-treated patients in schizophrenia trials reached 41%, primarily due to a severe lack of clinical effect 27. Conversely, in trials for OCD, the primary cause of placebo dropout was the occurrence of nocebo-driven adverse events, highlighting the different psychobiological profiles of these patient populations 27.
| Psychiatric Disorder | Placebo Effect Size (dav / SMD) |
|---|---|
| Major Depressive Disorder (MDD) | 1.40 |
| Generalized Anxiety Disorder (GAD) | 1.23 |
| Panic Disorder | 1.09 |
| Post-Traumatic Stress Disorder (PTSD) | 0.68 - 0.92 |
| Obsessive-Compulsive Disorder (OCD) | 0.65 |
| Schizophrenia | 0.59 |
| Data derived from 2024 JAMA Psychiatry meta-analysis of 90 high-quality RCTs 2628. |
Placebo Limits in Oncology and Organic Diseases
While placebos can drastically alter subjective experience, they largely fail to alter fundamental disease pathologies or objective biomarkers 1. This limitation is starkly illustrated in the field of oncology. Placebos are highly effective in mitigating the severe, subjective side effects of cancer and antineoplastic treatments, producing significant improvements in cancer-related fatigue, subjective pain, and treatment-induced nausea 129.
However, exhaustive reviews of placebo-controlled oncology trials confirm that placebos do not halt organic disease progression 13033. An analysis of trials evaluating objective tumor response (measured by rigid structural shrinkage using WHO criteria) found that tumor response occurred in only 10 out of 375 placebo-treated patients (2.7%) 30. Similarly, positive responses in objective serum markers occurred in only 1.7% of placebo patients 30. These nominal percentages are statistically indistinguishable from baseline rates of spontaneous fluctuation 30. The data solidifies the consensus that while the placebo effect profoundly modulates the central nervous system's perception of symptoms, it cannot destroy pathogens, shrink tumors, or reverse organic structural damage 13033.
Open-Label Placebos and Non-Deceptive Administration
Historically, the clinical utility of the placebo effect was severely restricted by the ethical requirement of deception. Because modern biomedical ethics and informed consent protocols prohibit physicians from lying to patients about the nature of their prescriptions, classical placebo administration is generally barred from standard care 343531. To circumvent this ethical barrier, researchers developed the "open-label placebo" (OLP) paradigm. In OLP trials, patients are administered an inert substance but are explicitly informed that it contains no active medication; the treatment rationale relies on explaining that the body can undergo positive mind-body conditioning simply by engaging in the ritual of medicine 32333934.
Efficacy of Open-Label Placebos
The core hypothesis of OLP research is that deception is not a fundamental prerequisite for eliciting a psychobiological placebo response 3435. Recent large-scale meta-analyses confirm this hypothesis, demonstrating that OLPs can generate statistically significant clinical benefits 3936.
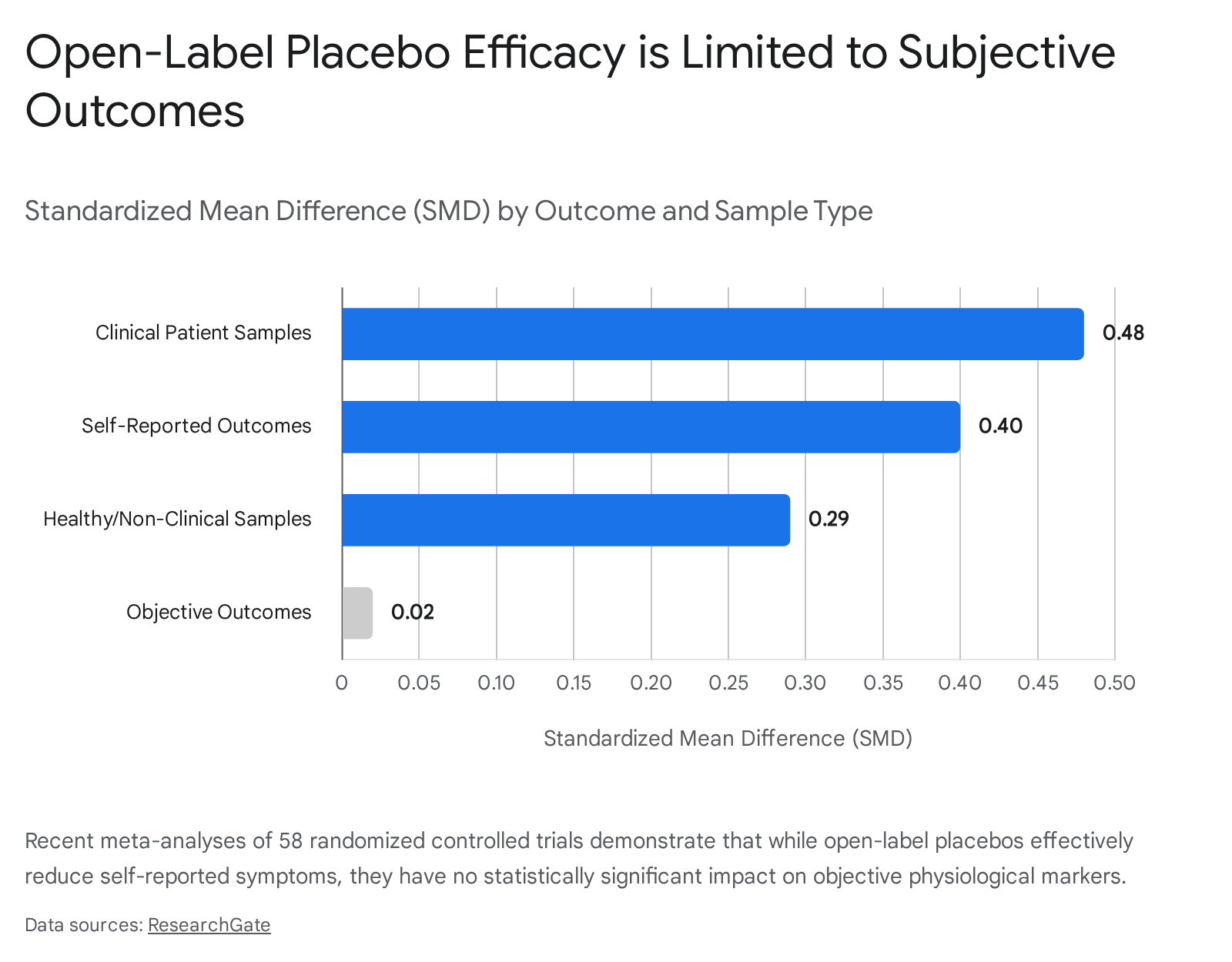
A comprehensive 2024 meta-analysis by Fendel et al., which updated previous findings by examining 58 RCTs comprising 4,569 participants, found a small but highly significant positive effect of OLPs across various health conditions (overall SMD = 0.36) 36. The analysis provides crucial nuance regarding where OLPs are most effective.
The data reveals a stark divergence between subjective and objective metrics: OLPs demonstrate a meaningful, sustained effect on self-reported, subjective outcomes (SMD = 0.40), but fail to produce any statistically significant change in objective, measurable physiological outcomes (SMD = 0.02) 3637. Furthermore, OLPs prove significantly more effective in clinical patient samples seeking treatment for actual pathology (SMD = 0.48) than in experimental settings utilizing non-clinical, healthy volunteers (SMD = 0.29) 36.
Comparison Between Open-Label and Double-Blind Placebos
A persistent question in placebo research is the "deception penalty" - whether full transparency inherently diminishes the magnitude of the placebo effect compared to traditional, deceptive administration. In a landmark nested RCT focusing on patients with irritable bowel syndrome (IBS), researchers directly compared the efficacy of an OLP arm to a traditional double-blind placebo (DBP) arm 38.
The study demonstrated that, for IBS symptom improvement, there was no statistically significant difference in efficacy between the fully transparent OLP group and the deceptive DBP group (p = 0.485) 38. Qualitative interviews nested within the trial revealed nuanced psychological differences in how patients processed the treatment. While both cohorts shared feelings of hope, uncertainty, and curiosity, patients in the OLP group were noticeably more hesitant to attribute their symptom relief directly to the physical pill itself, recognizing instead the therapeutic value of the broader clinical context and their relationship with the providers 38.
Cultural and Socioeconomic Determinants
The placebo effect does not operate independently of external reality; it is a culturally embedded construct heavily dictated by societal norms, media narratives, aesthetic preferences, and the macroeconomic context of healthcare delivery 3946.
Cross-Cultural Variations in Treatment Expectancy
The symbolic meaning that specific patient populations ascribe to illness and medical rituals heavily modulates the strength of the placebo effect. Anthropological analyses of international double-blind clinical trials reveal vast geographical disparities in baseline placebo response rates 3940. For example, in global trials evaluating treatments for gastric ulcers, German participants historically generated an exceptionally high average placebo healing rate of 59% 39. In stark contrast, Dutch and Danish participants exhibited a 22% placebo healing rate, while patients in Brazil demonstrated a minimal placebo response of just 7% 39.
Cultural conditioning also alters the specific efficacy of physical aesthetics. The color, shape, and invasiveness of a placebo fundamentally alter its psychobiological impact based on regional associations. White capsules are frequently associated with analgesia by Caucasian populations in the United States, whereas the same white capsules are perceived as stimulants by certain African American cohorts 41. Furthermore, highly invasive placebos, such as sham surgeries or sham acupuncture, consistently generate stronger placebo responses than oral pills. The increased procedural "drama" and heightened ritual of invasive interventions signal greater therapeutic potency, leveraging the patient's cultural reverence for advanced medical intervention 4642.
Cultural belief systems can also drive profound nocebo effects, ultimately impacting mortality. A landmark 1993 study by Phillips et al. analyzed mortality rates among Chinese Americans who adhered to traditional Chinese medical astrology, which dictates that a person's fate and organ health are intrinsically tied to their birth year 39. The researchers found that individuals who developed diseases associated with their astrological birth phase (e.g., patients born in "lung years" who contracted respiratory diseases) died an average of 4.5 to 7 years earlier than a comparable cohort of European Americans with the same diseases, demonstrating the deadly physiological impact of deeply internalized cultural fatalism 39.
Economic Variables and Global Trial Recruitment
In contemporary clinical trials, the macroeconomic status of the host country has emerged as a major predictor of placebo response rates. Systematic reviews published in 2025 and 2026 analyzed global recruitment data across dozens of trials for rheumatoid arthritis, psoriatic arthritis, and plaque psoriasis 4344. The data revealed a robust negative association between a country's per-capita Gross National Income (GNI) and its baseline placebo response rates 4344.
As pharmaceutical companies expand trial recruitment into less affluent countries across the Global South, the placebo response rates within those specific cohorts inflate significantly. In psoriatic arthritis trials, researchers calculated a 5.7% decrease in placebo response rates for every $10,000 increase in a country's per-capita GNI 44. Experts attribute this variance to the stark contrast in baseline healthcare quality. For patients residing in lower-income regions with severely limited access to modern biological therapies, simply being enrolled in a high-quality, closely monitored clinical trial represents a massive elevation in their standard of care 4344. This sudden influx of medical attention generates intense positive expectancy, which, when combined with statistical regression to the mean, produces dramatically elevated placebo outcomes that complicate global efficacy assessments 4344.
| Sociocultural Factor | Impact on Placebo / Nocebo Outcomes |
|---|---|
| Geographic Variation | Ulcer placebo healing rates vary drastically (e.g., Germany 59% vs. Brazil 7%) 39. |
| Aesthetic Preferences | Pill color induces divergent expected effects (e.g., white pills viewed as analgesics vs. stimulants based on demographic) 41. |
| Intervention Invasiveness | Sham surgeries/injections yield stronger placebo responses than oral pills due to heightened medical "ritual" 4642. |
| Macroeconomic Context (GNI) | Lower-income countries exhibit significantly higher placebo response rates in global trials due to perceived upgrades in care 4344. |
| Cultural Fatalism | Deeply held cultural beliefs regarding illness inevitability can accelerate mortality (severe nocebo effect) 39. |
Ethics and Clinical Implementation
Despite the documented neurobiological efficacy of the placebo effect in treating subjective symptoms, integrating placebos into routine clinical care remains fraught with ethical controversies and systemic logistical barriers.
The Deception Debate and Paternalism
Surveys of general practitioners (GPs) across the United States and Europe reveal a glaring disconnect between ethical guidelines and real-world clinical behavior. Despite explicit American Medical Association (AMA) and General Medical Council (GMC) guidelines prohibiting deceptive placebo use, research indicates that between 77% and 97% of primary care physicians admit to regularly prescribing "impure" placebos 313545. Impure placebos are off-label active interventions - such as prescribing vitamins for unverified fatigue or utilizing low-dose antibiotics for presumed viral infections - administered purely for their psychological, placebo-generating value 354546. This practice is ethically hazardous, as it exposes patients to genuine pharmacological side effects (nocebo or otherwise) while undermining the foundational trust of the doctor-patient relationship 3545.
While Open-Label Placebos (OLPs) were designed to eradicate this deception, some bioethicists argue they merely reframe it. Critics, such as Tresker and Louhiala (2026), contend that OLP scripts remain subtly misleading because they conflate the inert pill itself with the contextual healing generated by the clinical encounter 4748. They argue that telling a patient an inert pill "can help" through mind-body mechanisms is a conceptual sleight of hand; if the pill is inert, it cannot possess therapeutic properties, and attributing the benefit to the pill rather than the clinical ritual borders on misdirection 4748.
The Worthwhile Wager Framework
Conversely, proponents of OLP integration advocate for the "worthwhile wager" framework. Ethicists Hardman and Miller (2025) argue that for chronic, functional conditions lacking clear biomedical solutions (such as IBS or non-specific lower back pain), prescribing an OLP is ethically superior to the paternalistic prescribing of off-label active medications 3349.
In this framework, the clinician presents the OLP transparently, explaining that the body can automatically respond to the medical ritual itself. The patient is not required to genuinely believe the placebo will cure them; rather, they are instructed to "act as if" it will, engaging in the daily ritual of pill-taking to stimulate non-conscious expectancy and conditioning 33. Because OLPs carry zero risk of pharmacological toxicity and zero risk of drug-drug interactions, the "wager" offers potential symptomatic upside with absolute minimization of iatrogenic harm 33.
Medical Billing and Regulatory Friction
Beyond philosophical debates, the transition of OLPs from the laboratory to the primary care clinic faces severe regulatory and financial barriers, primarily regarding medical billing infrastructure.
Currently, billing procedures for clinical research are strictly delineated by entities like the Centers for Medicare & Medicaid Services (CMS). Clinical trial costs are divided into "research costs" (absorbed by trial sponsors) and "routine patient care costs" (billed to insurance). To ethically bill insurance during a clinical trial, practitioners must utilize highly specific CPT modifiers (e.g., modifier Q1 or Q0) and secondary ICD-10 diagnosis codes (e.g., Z00.6 for "examination of a participant in a clinical trial") alongside the trial's official National Clinical Trial (NCT) identifier 575059.
However, no such infrastructure exists for standard clinical care. Modern insurance frameworks require a "medically necessary" procedure mapped to an organic diagnosis 5951. A physician cannot currently bill an insurance provider for prescribing a transparently inert "sugar pill," nor can they easily code for a "placebo administration visit" outside of an approved experimental protocol. Until medical coding schema adapt to recognize the standalone therapeutic value of clinical context and psychological conditioning, the widespread adoption of open-label placebos in general practice will remain stalled by systemic regulatory friction 575951.