Clinical evidence for dietary interventions in mental illness
Introduction
Mental disorders, including major depressive disorder, anxiety, bipolar disorder, and schizophrenia, constitute a leading cause of global disability and carry substantial economic and societal costs. For decades, psychiatric treatment has predominantly relied upon a pharmacocentric model combined with psychotherapy. However, treatment-resistant populations, high relapse rates, and the metabolic side effects of psychotropic medications have exposed the limitations of relying exclusively on these interventions 123. In response, nutritional psychiatry has emerged as a dedicated scientific discipline investigating the impact of diet, specific nutrients, and overall dietary patterns on brain function and mental health 34.
Observational epidemiology consistently demonstrates that high-quality dietary patterns are inversely associated with the risk of common mental disorders, whereas Western diets rich in ultra-processed foods are correlated with higher psychiatric morbidity 156. Moving beyond observational correlations, recent randomized controlled trials suggest that dietary modification can serve as a viable clinical intervention. Research indicates that the brain relies on continuous nutrient provision to maintain structural integrity, regulate neurotransmitter synthesis, and manage oxidative stress 2. Consequently, nutrition is increasingly recognized as a central, modifiable determinant of mental health rather than a peripheral lifestyle factor.
Biological Mechanisms of Diet and Brain Function
The theoretical foundation of nutritional psychiatry rests on several intersecting biological pathways that mediate the relationship between gastrointestinal inputs and psychological well-being. These pathways function simultaneously, connecting dietary composition to neural architecture and emotional regulation.
The Microbiome Gut Brain Axis
The gut-brain axis constitutes a bidirectional communication network linking the enteric nervous system to the central nervous system 78. The gut microbiota - trillions of microorganisms residing in the gastrointestinal tract - plays an integral role in this network. Communication between the gut and the brain occurs via multiple distinct routes, including neural, immune, and endocrine pathways 7910.
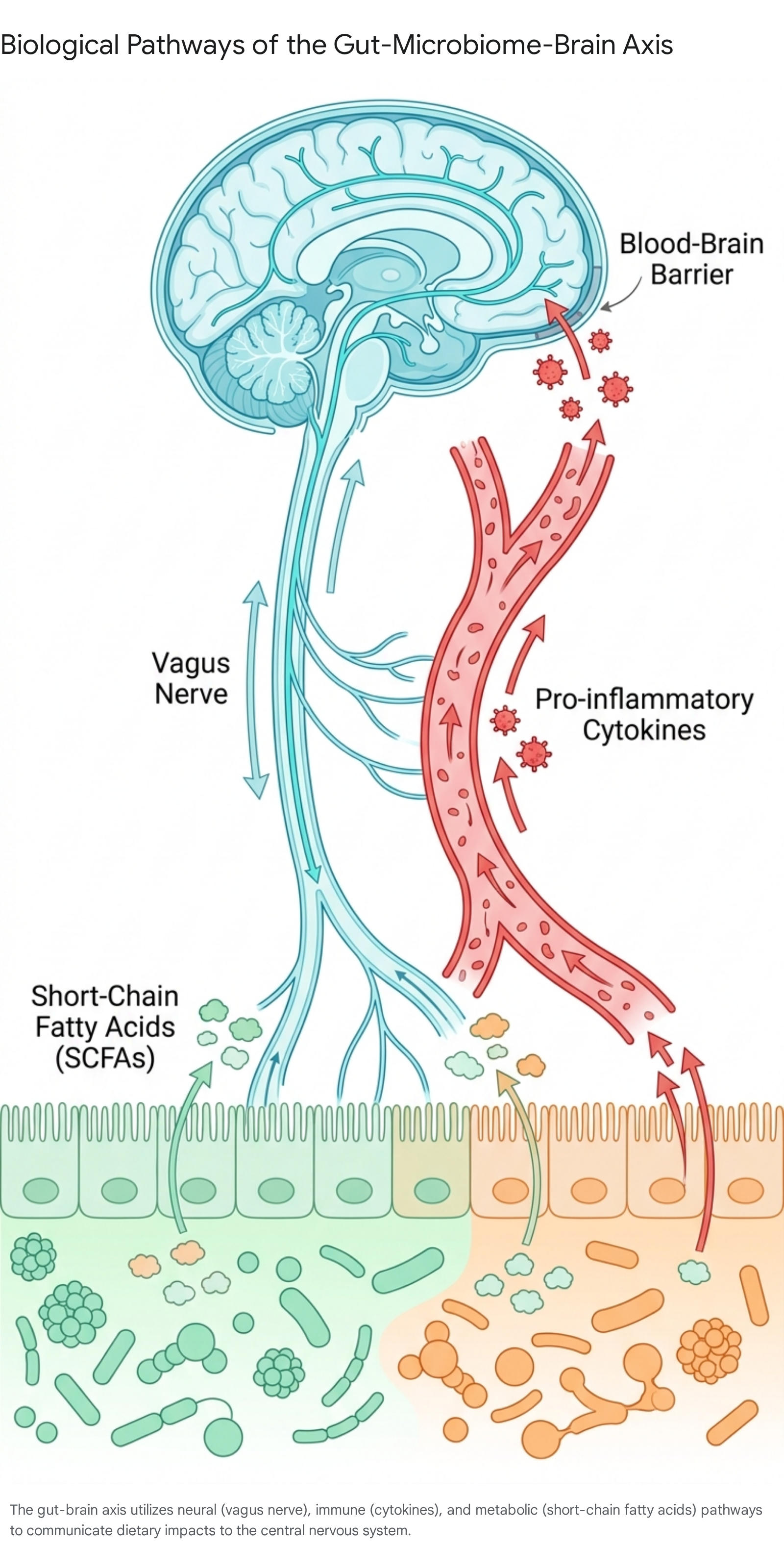
Commensal bacteria ferment dietary fiber and prebiotics to produce short-chain fatty acids, primarily butyrate, propionate, and acetate. Short-chain fatty acids strengthen the intestinal barrier, modulate systemic immunity, and cross the blood-brain barrier to influence neuroplasticity and the production of brain-derived neurotrophic factor 8911. Furthermore, the gut microbiome is directly involved in the synthesis and regulation of critical neurotransmitters. Approximately 90% of the human body's serotonin is produced in the gastrointestinal tract, and microbial balance directly affects the availability of serotonin, dopamine, and gamma-aminobutyric acid 4811.
Neural and Endocrine Signaling Pathways
The vagus nerve acts as a direct physical conduit between the gut and the brain, facilitating rapid, bidirectional communication. Preclinical studies indicate that the introduction of specific gut bacteria can activate neuronal regions via vagal afferent pathways, independently influencing anxiety-like behaviors without requiring a systemic immune response 78. Specific probiotic strains, including Lactobacillus and Bifidobacterium, influence the body's stress response system by modulating the hypothalamic-pituitary-adrenal axis, thereby affecting cortisol levels and overall stress resilience 7811.
Systemic Inflammation and Immune Regulation
Dietary patterns heavily influence systemic inflammation, which is increasingly recognized as a core mechanism in the pathophysiology of mood disorders, schizophrenia, and neurodevelopmental conditions 4912. Dysbiosis, driven by Western diets high in ultra-processed foods and low in dietary fiber, can compromise the integrity of the intestinal lining. This results in increased intestinal permeability, allowing pro-inflammatory molecules to enter systemic circulation 11.
Once in the bloodstream, pro-inflammatory cytokines such as interleukin-6 and tumor necrosis factor-alpha can traverse the blood-brain barrier, inducing neuroinflammation. Neuroinflammation alters neurotransmitter metabolism, impairs neuroplasticity, and exacerbates psychiatric symptoms 4911. Conversely, high-quality dietary patterns promote the proliferation of beneficial bacteria, such as Faecalibacterium prausnitzii, which stimulate the production of the anti-inflammatory cytokine interleukin-10, mitigating systemic inflammatory responses 9.
Oxidative Stress and Neuroprotection
The brain's high oxygen consumption renders it exceptionally vulnerable to oxidative stress - an imbalance between the production of reactive oxygen species and the body's antioxidant defenses 11. Chronic oxidative stress leads to neuronal damage affecting DNA, lipids, and proteins, which accelerates neurodegenerative processes and worsens psychiatric symptoms 11.
Nutrient-dense diets supply essential exogenous antioxidants, including vitamins C and E, zinc, magnesium, and polyphenols. These compounds scavenge reactive oxygen and nitrogen species, protecting the brain from oxidative injury 1113. Furthermore, dietary antioxidants modulate redox-sensitive signaling pathways and transcription factors, such as NF-κB, which are intimately involved in cellular stress responses and synaptic plasticity 11.
Clinical Evidence in Major Depressive Disorder
Major depressive disorder is the most extensively researched condition within nutritional psychiatry. Epidemiological and interventional data converge to demonstrate that dietary quality significantly impacts the onset, severity, and remission of depressive symptoms.
Epidemiological Associations
Longitudinal and cross-sectional studies consistently link Western dietary patterns - characterized by high intakes of ultra-processed foods, refined sugars, saturated fats, and processed meats - with a substantially increased risk of depression 51415. A study investigating ultra-processed food consumption found a 22% increased risk of depression and a 48% increased risk of anxiety among individuals with the highest intake levels 5. Similarly, the ALIMENTAL study, an international cross-sectional survey of over 15,000 adults, demonstrated that high consumption of ultra-processed foods significantly increased the odds of depression across both sexes 15.
Conversely, diets rich in whole foods, such as vegetables, fruits, legumes, nuts, seeds, and fish, act as a protective factor. Adherence to these high-quality dietary patterns is associated with an approximately 30% decreased risk of developing depression 1617.
Randomized Controlled Trials
The translation from observational epidemiology to clinical intervention was catalyzed by the "Supporting the Modification of lifestyle In Lowered Emotional States" trial. Conducted over 12 weeks, this was the first randomized controlled trial to test a whole-diet intervention for clinical depression 18. Sixty-seven adults with moderate to severe depression were randomized to either a dietary intervention or a social support control group 1819.
The dietary intervention utilized a modified Mediterranean diet, guided by a clinical dietitian, which emphasized whole grains, vegetables, fruits, legumes, low-fat dairy, and olive oil, while strictly limiting refined cereals, fried foods, fast foods, and sugary drinks 1920. The control condition consisted of a "befriending" protocol matched for visit frequency and length, explicitly avoiding psychotherapy techniques or nutritional advice to isolate the dietary variable 1819.
The outcomes of the trial demonstrated a significant reduction in depressive symptoms for the dietary cohort. The diet group achieved a 7.1-point greater reduction on the Montgomery - Åsberg Depression Rating Scale compared to the control group. Furthermore, 32.3% of the dietary group achieved full remission (defined as a score below 10), compared to only 8.0% in the control group.
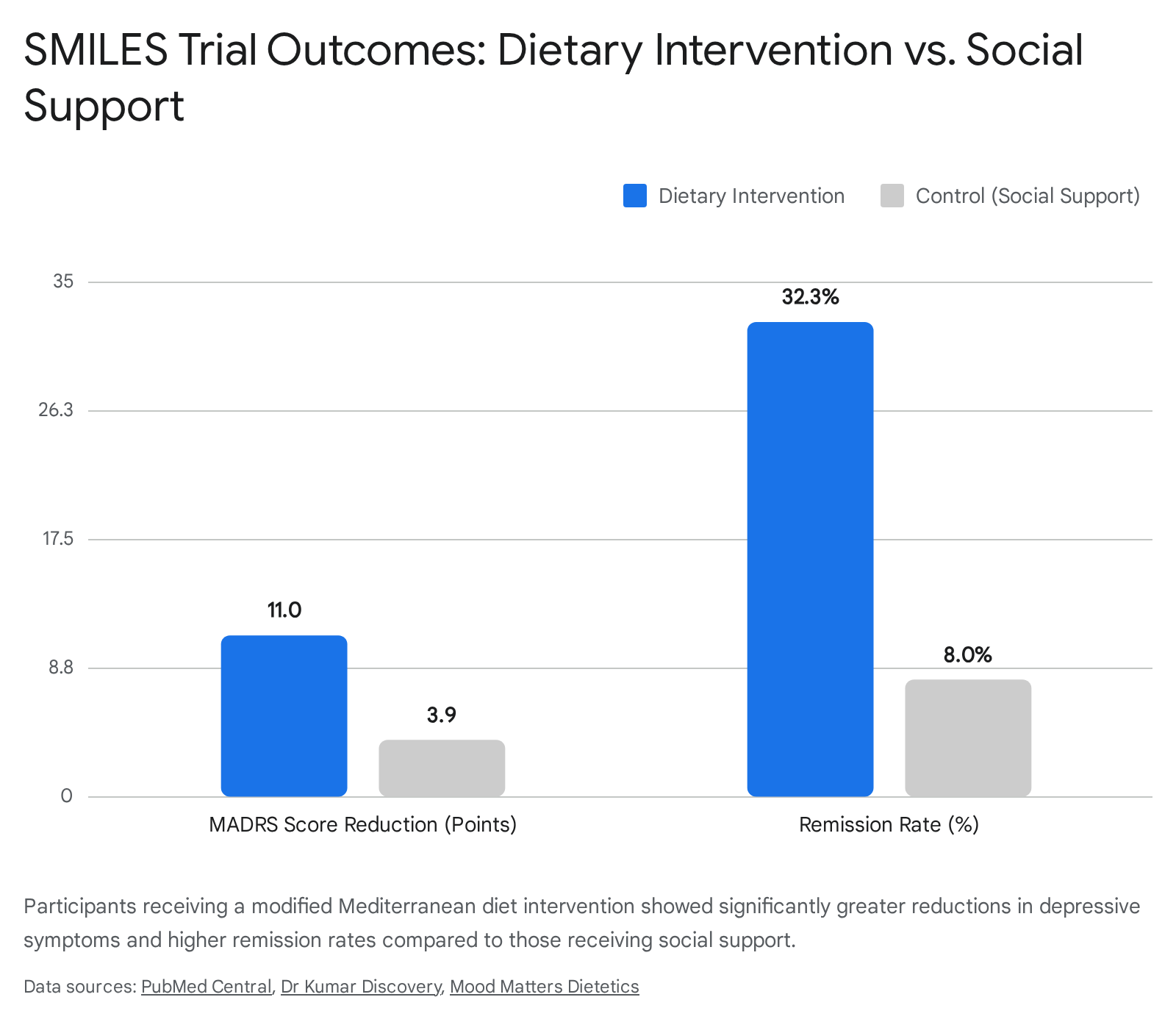
This resulted in a robust number needed to treat of 4.1, indicating that for every four patients treated with the dietary intervention, one achieved remission 181920.
Interpreting the Evidence Base
Despite these promising results, early trials face methodological scrutiny. Critics note that subjective outcome measures are highly susceptible to expectancy bias, particularly in lifestyle trials where blinding the intervention is virtually impossible 21. The exceptionally large effect size (Cohen's d = 1.16) in the trial exceeds those typically seen in established cognitive behavioral therapy or pharmacotherapy trials 21. Furthermore, the control condition suffered a dropout rate four times higher than the dietary intervention, suggesting that the social support protocol may not have been perceived as an equally credible or attractive treatment by participants 21.
Nevertheless, subsequent trials and umbrella reviews continually corroborate the utility of high-quality diets as an adjunctive treatment for major depressive disorder 21722. Clinical guidance currently positions dietary modification as a critical component of comprehensive psychiatric care, highly effective when utilized alongside established pharmacological and psychological treatments rather than as a standalone cure 223.
Dietary Interventions in Bipolar Disorder and Schizophrenia
Bipolar disorder and schizophrenia pose profound treatment challenges, with traditional antipsychotics and mood stabilizers frequently inducing severe metabolic side effects. Patients often suffer from treatment-emergent weight gain, dyslipidemia, impaired glucose tolerance, and an elevated risk of cardiovascular disease, leading to a drastically reduced life expectancy 242526.
Metabolic Comorbidities and Mental Illness
The paradigm of "metabolic psychiatry" has gained significant traction, positing that serious mental illnesses share foundational pathologies of brain energy metabolism, mitochondrial dysfunction, and redox dysregulation 242526. By conceptualizing severe psychiatric disorders partially as metabolic dysfunctions of the brain, researchers have begun applying dietary interventions historically used for physical metabolic conditions to psychiatric populations.
Medical Ketogenic Diet Interventions
The most notable dietary intervention in this domain is the medical ketogenic diet - a high-fat, moderate-protein, and very low-carbohydrate regimen that forces the brain to utilize ketone bodies for energy rather than glucose 2425. A pioneering pilot trial investigated a 4-month ketogenic intervention in adults with bipolar disorder or schizophrenia who concurrently suffered from metabolic abnormalities or psychotropic-induced weight gain 272829.
The dual outcomes of the trial were substantial across both physical and psychiatric domains. Metabolically, 100% of participants who met the criteria for metabolic syndrome at baseline achieved remission by the study's conclusion 2829. Adherent individuals experienced an average 12% reduction in body mass index, a 13% reduction in waist circumference, a 36% reduction in visceral adipose tissue, and a 27% decrease in insulin resistance 28.
Psychiatrically, participants with schizophrenia exhibited a 32% reduction in Brief Psychiatric Rating Scale scores 28. The overall cohort demonstrated an average 31% improvement in Clinical Global Impression severity, with 75% of participants achieving a recovered state on clinical mood monitoring scales 2829. Pilot data from the University of Edinburgh further indicates that ketogenic therapy for bipolar disorder yields improvements in mood stability, energy, and anxiety 30. While these remain preliminary pilot trials with small sample sizes, they provide compelling proof-of-concept that targeting systemic metabolism via nutritional ketosis can yield simultaneous metabolic and psychiatric benefits, prompting the initiation of larger, randomized controlled trials 242526.
Nutritional Approaches to Attention-Deficit Hyperactivity Disorder
In the context of attention-deficit hyperactivity disorder, the role of nutrition has transitioned from an alternative therapy to an evidence-based adjunctive consideration 31. Research indicates a dual approach is most effective: mitigating dietary risk factors through exclusion and correcting existing micronutrient deficiencies.
Micronutrient Deficiencies and Supplementation
Multiple meta-analyses confirm that children and adults with attention-deficit hyperactivity disorder exhibit higher rates of specific nutrient deficiencies, notably in omega-3 fatty acids, zinc, iron, and magnesium. These micronutrients are essential cofactors for neurotransmitter synthesis and dopamine regulation 123132.
Clinical evaluations demonstrate that low serum ferritin (indicative of poor iron status) correlates with greater symptom severity. Furthermore, children with lower iron levels often require higher doses of stimulant medication to achieve the same clinical efficacy as those with adequate iron status 3233. While broad-spectrum supplementation strategies yield mixed results, targeted supplementation for patients with confirmed deficiencies has shown adjunctive benefits in improving focus, emotional dysregulation, and hyperactivity 123334.
Dietary Exclusion and Inflammatory Triggers
Diets high in ultra-processed foods, refined sugars, and saturated fats - often termed a "Western" dietary pattern - are consistently linked to more severe attention-deficit hyperactivity disorder symptoms 3132. Furthermore, rigorous clinical trials provide evidence that specific dietary exclusions can lead to measurable symptom reduction. Carefully conducted elimination diets, which remove artificial food colorings and preservatives, have demonstrated significant reductions in hyperactive behaviors in sensitive subsets of children 3132. Current clinical consensus suggests that while pharmacotherapy remains the primary treatment, optimizing nutrient intake and minimizing highly processed foods offer a valuable complementary strategy to manage baseline symptomatology 3233.
Impact of Diet on Anxiety Disorders
The evidence linking diet to anxiety disorders exists, but it is currently less robust than the literature surrounding depression. Observational studies demonstrate a moderate correlation between high-quality diets and a decreased risk of anxiety, while linking ultra-processed food consumption to increased anxiety risk 51635.
Observational Evidence and Limitations
Despite strong epidemiological correlations, clinical trial outcomes utilizing whole-diet interventions for primary anxiety disorders remain inconclusive. Meta-analyses indicate that while dietary interventions reliably improve depressive symptoms, their effect on anxiety symptoms is less consistent and frequently fails to reach statistical significance 1636.
Some reviews suggest that anxiety symptoms may be more responsive to specific micronutrient supplementation, such as magnesium or B vitamins, particularly in cases of established clinical deficiency, rather than broad dietary pattern modifications alone 123537. Probiotic interventions targeting the gut-brain axis have also failed to produce statistically significant improvements in primary anxiety symptoms in multiple trials 4. Thus, the clinical application of diet specifically for the treatment of generalized anxiety requires further large-scale, highly powered randomized trials to establish causality and treatment efficacy 36.
Analysis of Specific Dietary Patterns
Nutritional psychiatry has increasingly pivoted away from reductionist approaches that isolate single nutrients, recognizing that nutrients operate synergistically within the complex matrix of whole foods 21738. Consequently, contemporary research emphasizes the evaluation of comprehensive, culturally relevant dietary patterns.
The Mediterranean and MIND Diets
The Mediterranean diet is characterized by a high intake of vegetables, fruits, legumes, nuts, whole grains, extra virgin olive oil, and fish, with moderate consumption of dairy and minimal intake of red meat and refined sugars 11938. It currently holds the strongest evidence base in nutritional psychiatry 615. Meta-analyses of clinical trials conclude that adherence to a Mediterranean pattern reduces depressive symptoms by 32% to 45% 1739. The benefits are largely attributed to the diet's high content of anti-inflammatory polyphenols, antioxidants, and omega-3 polyunsaturated fatty acids, which collectively reduce neuroinflammation and promote gut microbiome diversity 173840.
The MIND diet combines elements of the Mediterranean and DASH (Dietary Approaches to Stop Hypertension) diets, specifically emphasizing neuroprotective foods like leafy greens and berries 1741. Originally designed to combat cognitive decline in older populations, emerging cross-sectional evidence indicates that high adherence to the MIND diet is significantly associated with lower risks of psychological distress and depression across broader demographics 174142.
The Traditional Japanese Washoku Diet
While Western nutritional models dominate the clinical literature, culturally specific dietary patterns in other global regions show significant psychoprotective effects. The traditional Japanese diet, known as Washoku, has been extensively evaluated in occupational cohorts.
A comprehensive longitudinal study evaluating over 12,000 Japanese workers found that strict adherence to the Washoku pattern - rich in fish, rice, miso soup, seaweed, soy products, mushrooms, and green tea - resulted in a 17% to 20% reduction in depressive symptoms 434445. These results remained robust even after adjusting for non-dietary lifestyle factors such as smoking, alcohol intake, obesity, and working hours 4445. Researchers attribute these psychoprotective effects to multiple factors: the high folate content of seaweed and soy aids in serotonin and dopamine synthesis; fermented foods support a healthy gut microbiome; and the characteristic umami flavor profile stimulates the parasympathetic nervous system, promoting mental stability and relaxation 434446.
Comparison of Evidence-Based Dietary Patterns
| Dietary Pattern | Core Nutritional Components | Primary Psychiatric Applications | Proposed Biological Mechanisms |
|---|---|---|---|
| Mediterranean Diet | Extra virgin olive oil, fish, legumes, nuts, high vegetable and fruit intake. | Major Depressive Disorder, General Cognitive Health. | High polyphenol and omega-3 content lowers systemic inflammation; supports gut microbiome diversity 1740. |
| MIND Diet | Leafy green vegetables, berries, whole grains, nuts, olive oil, fish. | Cognitive Decline, Depressive Symptoms. | High antioxidant profile neutralizes oxidative stress; specific phytonutrients from berries protect neuroplasticity 1741. |
| Washoku (Japanese) | Soy products, seaweed, fermented foods (miso), fish, green tea. | Depression, Occupational Stress, Anxiety. | Folate supports neurotransmitter synthesis; fermented foods balance the gut-brain axis; umami promotes parasympathetic relaxation 434446. |
| Medical Ketogenic | High fat (70-80%), moderate protein, very low carbohydrate. | Bipolar Disorder, Schizophrenia, Treatment-Resistant Depression. | Shifts brain energy metabolism from glucose to ketones; mitigates mitochondrial dysfunction and neuroinflammation 242526. |
The Superfood Fallacy and Reductionism
As consumer interest in nutrition has expanded, the commercial wellness industry has aggressively marketed "superfoods" - a term defined entirely by commercial utility and marketing strategy rather than scientific taxonomy 384748. The superfood narrative incorrectly suggests that specific, often exotic, individual ingredients hold medicinal properties capable of independently curing illness or offsetting the systemic damage of an otherwise poor diet 3849.
Psychiatric and nutritional researchers actively critique this framework. Studies examining consumer psychology demonstrate that belief in superfoods is frequently driven by intuitive thinking, exotic marketing narratives, and subjective social norms rather than analytical health literacy or scientific consensus 4748. The clinical reality is that isolated nutrients rarely produce dramatic psychiatric outcomes outside the context of treating a diagnosed clinical deficiency 2.
For instance, while broad-spectrum omega-3 supplements have proven beneficial as an adjunct for depression and attention-deficit hyperactivity disorder, extracting single compounds without modifying the broader inflammatory environment (such as a diet highly reliant on ultra-processed foods) blunts their efficacy 173233. The field of nutritional psychiatry is explicitly moving away from "shotgun" nutraceutical formulations. Instead, the focus has shifted toward integrating holistic dietary modifications within a personalized medicine framework, prioritizing whole-food synergy, cultural accessibility, and sustainable dietary adherence over the search for single nutritional "magic bullets" 22338.
Methodological Challenges in Nutritional Psychiatry
Despite a rapidly growing body of promising data, nutritional psychiatry faces substantial methodological hurdles that complicate the translation of findings into universal clinical guidelines. Umbrella reviews of randomized controlled trials frequently classify the overall quality of available evidence as low to moderate due to the inherent logistical limitations in conducting dietary research 62252.
Trial Design and Blinding Limitations
Unlike pharmacological trials utilizing visually identical placebo pills, it is practically impossible to successfully double-blind a whole-food dietary intervention. Participants are inherently aware of the dietary changes they are making. This lack of blinding introduces significant expectancy bias, where the psychological anticipation of a benefit from a "healthy diet" inflates the reported improvement in subjective mood questionnaires 621. Furthermore, designing an appropriate control group presents a persistent challenge. Control conditions, such as unstructured social support, often suffer from higher dropout rates if participants do not perceive the control intervention as an equally credible or attractive treatment 21.
Adherence and Baseline Confounding
Long-term compliance with restrictive dietary interventions ranges historically from 30% to 50% outside highly controlled clinical environments, making it difficult to assess the true long-term efficacy of the diets 53. Additionally, many trials fail to adequately screen for baseline dietary status. If an intervention group already consumes a relatively high-quality diet prior to enrollment, a "ceiling effect" severely limits any observable psychiatric improvements 653.
Nutrition research is also heavily confounded by innumerable lifestyle variables. Individuals who adhere to healthy diets frequently engage in more physical activity, report better sleep hygiene, and possess higher socioeconomic status, making causal isolation between the diet itself and the psychiatric outcome statistically difficult 44452. Finally, psychiatric neuroplasticity and microbiome remodeling require extensive time, yet many dietary trials are limited to 8 to 12 weeks, failing to capture critical long-term relapse prevention data 46.
Translation to Clinical Practice and Guidelines
Historically, traditional psychiatry viewed nutrition as peripheral to treatment, focusing primarily on neuropharmacology. However, converging evidence has prompted authoritative bodies to integrate dietary recommendations directly into clinical practice guidelines.
International Society for Nutritional Psychiatry Research Guidelines
To address systemic methodological deficits and standardize the field, the International Society for Nutritional Psychiatry Research published comprehensive clinical trial guidelines in 2024. These guidelines mandate multidisciplinary research teams that explicitly include registered dietitians to ensure intervention fidelity 650.
The guidelines emphasize the necessity of tracking objective biological markers - such as inflammatory cytokines and microbiome sequencing - alongside subjective symptom reporting to definitively establish mechanisms of action 26. Crucially, the guidelines recommend the utilization of weight-neutral dietary interventions for depression. This ensures that the observed psychiatric benefits are attributed directly to the nutritional composition of the diet rather than being a secondary psychological benefit of weight loss 6.
Integration into Established Psychiatric Frameworks
Clinical practice guidelines across multiple major organizations now formally recognize lifestyle and dietary interventions as a foundational step in psychiatric management. The Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for mood disorders emphasize diet as an important early-stage component in the treatment of major depressive disorder 16. Additionally, organizations such as the American Psychiatric Association and the Veterans Affairs/Department of Defense continually update clinical guidelines to reflect holistic, integrative models of care that encompass substance use, depression, and schizophrenia management 555152.
Translation into real-world community settings requires a multidisciplinary approach. Psychiatrists are increasingly encouraged to conduct basic dietary assessments during intake, collaborating with specialized dietitians to formulate culturally appropriate, financially accessible interventions 62346. Importantly, the current consensus frames dietary interventions as a powerful adjunctive therapy. While high-quality diets provide significant, evidence-based support for neurobiological health, they are not a standalone replacement for acute psychiatric care, crisis intervention, or necessary pharmacotherapy in cases of severe mental illness 223.
Conclusion
Converging evidence from epidemiological cohorts, mechanistic biological studies, and randomized controlled trials confirms that diet is a critical, modifiable determinant of mental health. The gut-microbiome-brain axis, systemic inflammation, and oxidative stress provide clear physiological pathways through which gastrointestinal inputs actively influence neural architecture, neurotransmitter synthesis, and emotional regulation.
While the commercial promotion of isolated "superfoods" lacks clinical validity, sustained adherence to nutrient-dense, whole-food dietary patterns - such as the Mediterranean, MIND, and traditional Japanese diets - yields significant and measurable reductions in depressive symptoms and occupational stress. Furthermore, highly specialized interventions like the medical ketogenic diet show profound promise in addressing both the debilitating metabolic comorbidities and the primary psychiatric symptoms of treatment-resistant bipolar disorder and schizophrenia.
As the discipline of nutritional psychiatry matures, overcoming methodological limitations via standardized, well-powered, and long-term clinical trials will be paramount. Ultimately, nutritional psychiatry does not seek to replace traditional psychiatric modalities. Rather, it aims to expand the therapeutic arsenal, firmly establishing the nutritional composition of the food we consume as a fundamental pillar of psychiatric prevention and comprehensive clinical treatment.