Clinical evidence for anti-aging interventions
Mechanisms of Biological Aging and Geroscience
The biological process of aging constitutes the primary, systemic risk factor for the development of chronic, life-threatening pathologies, including cardiovascular disorders, neurodegeneration, metabolic dysfunction, and various malignancies 123. Geroscience, an interdisciplinary field of biogerontology and clinical medicine, operates on the foundational paradigm that aging is driven by a highly conserved set of molecular and cellular mechanisms 1245. These established hallmarks of aging include genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication 145.
Historically, clinical research and regulatory frameworks have adhered to a disease-centric model, addressing individual pathologies reactively as they become clinically apparent. However, treating solitary diseases in geriatric populations often fails to increase overall healthspan - the period of life spent free from chronic disease and functional decline - due to the competing risks of secondary age-related conditions 67. Geroscience proposes a shift toward prophylactic, systemic interventions designed to modulate the fundamental biology of aging before overt tissue pathology manifests 136. Advancing this paradigm requires rigorous clinical evidence. While numerous compounds have demonstrated profound lifespan-extending properties in invertebrate and murine models, translating these findings to human populations presents substantial methodological, regulatory, and physiological challenges 7810.
Regulatory Classifications and Clinical Endpoints
A significant barrier to the execution of human clinical trials for longevity therapeutics has been the historical absence of a regulatory classification for aging as a treatable disease indication 910. Without recognized clinical endpoints, pharmaceutical developers have struggled to design trials capable of securing authorization from major regulatory bodies such as the United States Food and Drug Administration (FDA) and the European Medicines Agency (EMA) 91011.
Recent developments within the World Health Organization (WHO) International Classification of Diseases (ICD-11) framework have partially addressed this impediment. Following proposals from longevity research organizations, the ICD-11 implementation included an extension code for "Ageing-related" (XT9T) within the "Aetiology" and "Causality" sections, formally acknowledging aging as a causal factor for pathological conditions 91412. Additionally, the ICD-11 code MG2A, previously defined by the vague term "Old age," has been formally reclassified as "Aging-associated decline in intrinsic capacity" 13. These diagnostic codes provide a foundational architecture for researchers to measure interventions directly targeting aging mechanisms, utilizing quality-adjusted life years and healthy-years equivalents as trial endpoints rather than the incidence of a single disease 913.
Regulatory bodies are also adapting to these paradigm shifts. The EMA's regulatory science strategy to 2025 emphasizes the integration of real-world data, 'omics' methods, and precision medicine biomarkers into the clinical evaluation pipeline, indicating a willingness to move beyond traditional disease models if supported by robust surrogate endpoints 14151617. Similarly, major clinical trials, such as the $38 million VITAL-H study, are currently working to establish "intrinsic capacity" - a composite measure of physical and mental functionality - as a regulatory-grade endpoint acceptable to the FDA 3.
Maturation of Epigenetic and Multi-Omic Biomarkers
The transition from animal lifespan studies to human trials necessitates the use of validated surrogate biomarkers. Because human longitudinal lifespan studies are prohibitively expensive and require decades to complete, researchers rely on molecular and physiological biomarkers to assess the short-term efficacy of geroprotective interventions 1118.
Epigenetic clocks, which quantify biological age by analyzing DNA methylation patterns at specific CpG sites across the genome, have emerged as the leading diagnostic tools 1819. Early iterations of these clocks successfully predicted all-cause mortality better than chronological age, but were often insensitive to short-term lifestyle interventions 1823. Advanced multi-omic clocks (such as OMICmAge) and systems-level biomarkers now allow researchers to deconvolve biological aging across specific organ systems, providing granular data on how interventions affect the cardiovascular, hepatic, renal, and immune systems independently 182420. As aging is not uniform across all tissues, these organ-specific clocks are crucial for evaluating the targeted efficacy of emerging gerotherapeutics 20.
Lifestyle Interventions and Systemic Homeostasis
Behavioral and dietary modifications remain the most extensively validated methods for extending human healthspan and mitigating the risks of age-related disease 2122. In the absence of definitively proven, safe pharmacological monotherapies for maximum human lifespan extension, lifestyle interventions serve as the baseline standard against which all emerging medical therapies are measured 2123.
Caloric Restriction and Nutrient Sensing
Caloric restriction - defined as a reduction in total daily energy intake without malnutrition - is the most robust and highly conserved intervention for extending lifespan across multiple model organisms, including yeast, nematodes, fruit flies, rodents, and non-human primates 22324. In human populations, sustained caloric restriction elicits profound metabolic adaptations, including enhanced stress resistance, reduced systemic inflammation, increased insulin sensitivity, and optimized mitochondrial quality control 224.
The physiological benefits of caloric restriction are directly mediated by the modulation of highly conserved nutrient-sensing pathways. A reduction in systemic energy availability downregulates the insulin/insulin-like growth factor 1 (IGF-1) signaling cascade and inhibits the mechanistic target of rapamycin (mTOR), a central regulator of cellular growth, proliferation, and protein synthesis 24. Simultaneously, this state of energetic stress activates AMP-activated protein kinase (AMPK) and sirtuins, which collectively upregulate autophagic clearance, DNA repair mechanisms, and mitochondrial biogenesis 224. Autophagy, the cellular process of degrading and recycling damaged organelles and misfolded proteins, is considered an indispensable mechanism for lifespan extension and the prevention of neurodegenerative and oncological pathologies 230.
Physical Activity and Sarcopenia
Regular physical activity operates synergistically with dietary interventions to preserve structural and functional healthspan 21. Exercise induces complex, pleiotropic cellular adaptations, such as the activation of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), which enhances mitochondrial calcium handling, regulates apoptosis, and mitigates endoplasmic reticulum stress in skeletal muscle 21.
Currently, structured resistance and endurance training are the only available interventions proven to mitigate and reverse sarcopenia, the age-related decline in skeletal muscle mass and functional strength 21. Sarcopenia is a primary physiological driver of frailty, loss of independence, and all-cause mortality in older adults. While various pharmacological agents attempt to mimic the molecular signatures of exercise, none have successfully replicated the broad, systemic benefits of mechanical loading and cardiovascular exertion 21.
The Complexity of Pharmacological Mimetics
The pursuit of "exercise mimetics" or "caloric restriction mimetics" (CRMs) aims to capture the biochemical benefits of lifestyle interventions using targeted pharmacological agents 25. The conceptual mapping of these interventions is straightforward: specific compounds are utilized to engage specific biological targets, thereby mimicking the systemic benefits of caloric restriction or physical activity. For instance, rapamycin is deployed to inhibit the mTOR pathway, metformin is utilized to activate AMPK, NAD+ precursors are administered to stimulate sirtuin activity, and senolytics are used to achieve the selective clearance of senescent cells 22430.
However, the human physiological response to lifestyle interventions is highly complex and integrated. Clinical data indicate that pharmacological monotherapies can occasionally interfere with natural physiological adaptations. In multiple clinical trials, the administration of metformin and resveratrol was shown to blunt the beneficial metabolic, hypertrophic, and mitochondrial adaptations to endurance and resistance exercise in human subjects 21. This interference underscores the profound difficulty of using isolated molecular compounds to replicate the systemic homeostasis achieved through behavioral and physical interventions 21.
Preclinical Validation: The Interventions Testing Program
Because longitudinal human lifespan studies require decades to complete, the longevity field relies heavily on rigorous mammalian models to identify viable pharmacological candidates for human translation 2526. Established by the US National Institute on Aging (NIA) in 2004, the Interventions Testing Program (ITP) represents the definitive gold standard for preclinical longevity research 52526.
The ITP circumvents the reproducibility issues historically common in aging research by utilizing a highly controlled methodology. Compounds are tested in genetically heterogeneous mice - ensuring genetic diversity that better mimics human populations - across three independent geographical research sites simultaneously (The Jackson Laboratory, University of Michigan, and UT Health San Antonio) 25262728. Over two decades, the ITP has evaluated over 50 putative geroprotectors, dismissing many popular, commercially available compounds while validating a select few 102627.
As of late 2025, the program has identified 15 individual agents capable of significantly extending median lifespan. Acarbose proved highly effective, particularly in males (yielding up to a 22% extension), while 17α-estradiol (a non-feminizing estrogen derivative) demonstrated strong lifespan efficacy exclusively in male subjects 272829. Other validated compounds include canagliflozin (an SGLT2 inhibitor), astaxanthin (up to 12% extension), glycine, and epicatechin (approximately 5% extension in males) 262730.
The Shift Toward Combination Therapies
A major paradigm shift emerging from recent ITP data is the evaluation of multi-pathway polypharmacy 1025. Recognizing that aging is driven by multiple, distinct biological mechanisms, researchers hypothesized that combining compounds with different modes of action could yield additive or synergistic biological benefits 1025.
This approach was profoundly validated when the ITP tested the combination of rapamycin (targeting the mTOR pathway) and acarbose (targeting postprandial glucose metabolism and the gut microbiota). The rationale was that the insulin-sensitizing action of acarbose could compensate for the potentially deleterious insulin-desensitizing effects occasionally induced by rapamycin 1029. The combined administration resulted in the largest lifespan extensions ever recorded by the program: a 28% increase in median lifespan for female mice and a remarkable 34% increase for male mice 29.
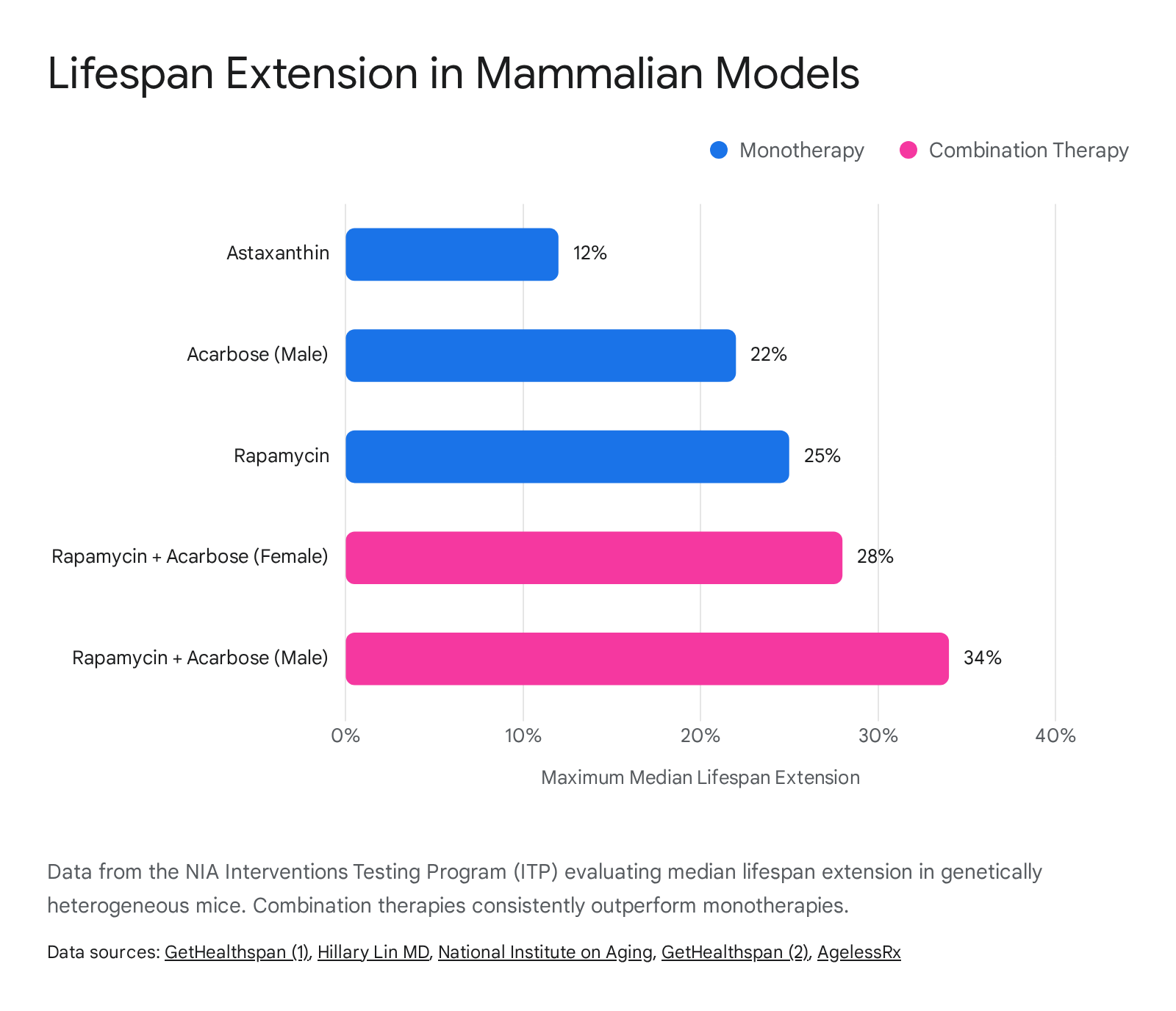
The success of this specific combination reinforces the emerging clinical consensus that human longevity interventions will likely mirror modern oncology and infectious disease protocols, utilizing customized combination therapies to target multiple hallmarks of aging simultaneously 2225. Large-scale human clinical trials, such as the VITAL-H study, are already adopting this philosophy by evaluating the combinatorial effects of FDA-approved drugs on intrinsic capacity in older adults 3.
| Compound / Intervention | Primary Biological Target | Maximum Reported Lifespan Extension (ITP Murine Data) | Key Observations |
|---|---|---|---|
| Rapamycin | mTORC1 Inhibition | ~25% | Consistent across sexes; highly reproducible across independent labs. |
| Acarbose | Alpha-glucosidase (Carbohydrate absorption) | 22% (Males), 5% (Females) | Significant sex dimorphism; modulates gut microbiota. |
| Rapamycin + Acarbose | mTOR + Metabolic / Microbiota | 34% (Males), 28% (Females) | Highest lifespan extension recorded by the ITP; synergistic effect. |
| 17α-estradiol | Non-feminizing estrogen signaling | ~19% (Males only) | Highly sex-specific; no lifespan benefit observed in females. |
| Astaxanthin | Oxidative stress / Antioxidant | ~12% | Naturally occurring compound; stable dietary inclusion. |
Pharmacological Interventions: mTOR and Metabolic Modulators
The field of longevity medicine has identified several existing, FDA-approved pharmaceutical compounds that modulate the mechanisms of aging 13. These medications, originally developed for specific disease indications such as immunosuppression, glycemic control, or obesity management, are currently under extensive investigation for their off-label geroprotective properties 73138.
mTOR Inhibitors: Rapamycin and Everolimus
Rapamycin (sirolimus) is a macrolide compound originally isolated from the soil bacterium Streptomyces hygroscopicus discovered on Easter Island (Rapa Nui) in 1975 73132. It is currently FDA-approved for the prevention of organ transplant rejection and as an anti-tumorigenic agent for specific rare diseases 313240. Rapamycin functions as a highly specific inhibitor of the mechanistic target of rapamycin (mTOR), a central cellular pathway that becomes chronically overactive with advancing age 72431. By dampening mTOR activity, rapamycin mimics a state of nutrient scarcity, thereby upregulating cellular cleanup processes like autophagy and mitophagy, reducing chronic inflammation, and preserving DNA integrity 73040.
In murine models, rapamycin is the most robust and highly replicated pharmacological agent for lifespan extension, reliably increasing median survival even when administration begins late in life (the equivalent of a 60-year-old human) 73031. However, translating these findings to humans requires a fundamental alteration in dosing strategy. In transplant medicine, rapamycin is dosed continuously on a daily basis to maintain steady trough levels. This continuous dosing suppresses immune function and can induce severe metabolic side effects, including hyperlipidemia, insulin resistance, and impaired wound healing 313334. These adverse effects are mechanistically linked to the unintended chronic inhibition of mTOR complex 2 (mTORC2), which occurs alongside the intended inhibition of mTOR complex 1 (mTORC1) 34.
For longevity applications, clinical practitioners utilize an off-label, low-dose, intermittent protocol - typically 5 to 6 milligrams administered once weekly 31. This pulsed dosing allows mTOR activity to rebound between administrations, which appears to maximize autophagic clearance while avoiding severe immunosuppression and preserving mTORC2 function 314035.
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial, published in 2025, provided critical human safety and efficacy data. In this 48-week, randomized, double-blind, placebo-controlled trial, healthy adults received either 5 mg weekly, 10 mg weekly, or a placebo 73136. While the trial did not meet its primary endpoint of significant visceral fat reduction, secondary analyses revealed compelling sex-specific benefits 3136. Women receiving 10 mg weekly exhibited a statistically significant 5% increase in lean tissue mass and a reduction in self-reported pain scores, while the 5 mg cohort reported improvements in general health and emotional well-being 731. Crucially, the trial confirmed that intermittent, low-dose rapamycin was well-tolerated over a full year without an increase in moderate to severe adverse events compared to placebo 73136.
Further evidence for mTOR inhibition in humans stems from studies utilizing everolimus, a rapamycin analog (rapalog). Clinical trials evaluating low-dose, intermittent everolimus in older adults demonstrated a 20% improvement in immunological response to the influenza vaccine and a reduction in respiratory infections 314037. Additional trials, such as the VIBRANT study at Columbia University, are currently investigating whether weekly rapamycin administration can slow ovarian aging in women 31. Despite these physiological improvements, human data have yet to establish that rapamycin directly extends maximum human lifespan, necessitating careful patient monitoring and lipid tracking during off-label use 73234.
Metabolic Modulators: Metformin
Metformin is a synthetic biguanide and the globally established first-line oral therapeutic for type 2 diabetes 333847. Metformin primarily acts by inhibiting mitochondrial complex I in the respiratory chain, which subsequently alters the cellular AMP-to-ATP ratio and activates AMPK 333847. This biochemical sequence mimics cellular energy deprivation, improving insulin sensitivity, reducing hepatic gluconeogenesis, and dampening systemic oxidative stress 243338.
Historically, large-scale observational data - such as analyses from the UK Prospective Diabetes Study (UKPDS) - suggested that diabetic patients taking metformin exhibited lower rates of all-cause mortality, cardiovascular events, and certain cancers compared to non-diabetic controls, sparking immense scientific interest in metformin as a "geroprotector" 384739. The landmark Targeting Aging with Metformin (TAME) trial was subsequently designed to test if the drug could delay the onset of a composite of age-related diseases (heart disease, cancer, dementia) in over 3,000 non-diabetic older adults, serving as a critical regulatory test case for the FDA 47394041.
However, recent rigorous analyses and controlled animal trials have cast significant doubt on metformin's efficacy as a primary longevity therapeutic in metabolically healthy individuals. Large meta-analyses of vertebrate lifespan studies show that while dietary restriction and rapamycin consistently extend lifespan, metformin frequently fails to produce statistically significant extensions, with its effects being highly dependent on dosage, sex, and age at the onset of treatment 33385142. Furthermore, studies in genetically diverse C. elegans demonstrated that metformin response is highly genotype-dependent, extending lifespan in some strains while actually decreasing survival in others 41.
Human trials evaluating metformin in healthy, non-diabetic populations have yielded mixed results regarding aging biomarkers, with evidence indicating that the drug may interfere with mitochondrial adaptations and impair the hypertrophic and cardiovascular benefits of exercise 2140. The clinical consensus in 2025 and 2026 is that metformin provides profound healthspan benefits by delaying metabolic dysfunction in at-risk or diabetic populations, but it may not fundamentally alter the underlying biology of aging in those without pre-existing metabolic disease 384043.
Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists
Initially developed for glycemic control and weight management, GLP-1 receptor agonists (e.g., semaglutide) and dual GLP-1/GIP agonists (e.g., tirzepatide) have rapidly emerged as potent gerotherapeutics 42238. Beyond their established capacity to induce 10% to 15% reductions in body weight - primarily through the depletion of metabolically harmful visceral adipose tissue - these agents exhibit profound pleiotropic effects that intersect directly with the core hallmarks of aging, including deregulated nutrient sensing and chronic inflammation 2238.
Human randomized controlled trials published throughout 2024 and 2025 demonstrated systemic cardiovascular, renal, and hepatic protection extending far beyond simple caloric deficit 2438. Landmark cardiovascular trials (SELECT, SUSTAIN-6, LEADER) established 20% to 26% reductions in major adverse cardiovascular events, while a massive 2025 Lancet meta-analysis involving over 85,000 participants confirmed a 16% reduction in the risk of kidney failure and a 22% slowing of renal filtration decline 38. In the liver, the ESSENCE Phase 3 trial demonstrated that 62.9% of patients on semaglutide achieved resolution of steatohepatitis without worsening fibrosis 38.
At the molecular level, emerging evidence indicates that GLP-1 therapies actively modulate biological aging. A highly significant 2025 double-blind, placebo-controlled clinical trial evaluated the epigenetic effects of semaglutide over 32 weeks in human adults 24. After adjusting for weight loss and baseline inflammatory parameters, semaglutide significantly decelerated epigenetic aging across multiple models, including PhenoAge (-4.9 years), GrimAge V2 (-2.3 years), and the DunedinPACE clock (indicating a ~9% slower pace of aging) 24. Furthermore, eleven distinct organ-system clocks showed concordant deceleration, particularly within the inflammatory, brain, and cardiac domains 24. Preclinical models and early human data further suggest neuroprotective potential, with GLP-1 signaling requiring a distinct central nervous system route to reduce neuroinflammation, clear amyloid pathology, and coordinate systemic multi-omic reversal of aging 43844.
Cellular Energy Modulators and NAD+ Precursors
Nicotinamide adenine dinucleotide (NAD+) is an essential coenzyme present in all living cells, serving as a critical mediator of cellular respiration, mitochondrial energy production, and DNA repair 2355. Crucially, NAD+ acts as the requisite substrate for sirtuins, a highly conserved family of protein deacetylases closely associated with longevity, metabolic regulation, and genomic stability 523. The interaction between NAD+ and sirtuins is fundamentally interdependent; sirtuin activity declines commensurately with the natural, age-related depletion of cellular NAD+ pools 52355.
Direct oral supplementation of NAD+ is pharmacokinetically inefficient due to poor cellular permeability and rapid degradation in the gastrointestinal tract. Consequently, researchers and clinical practitioners have focused on administering molecular precursors - primarily nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR) - which utilize specific enzymatic salvage pathways to rapidly elevate intracellular NAD+ concentrations 23304355.
Clinical Evidence for Nicotinamide Mononucleotide (NMN)
Recent human clinical trials have provided substantial safety and efficacy data for oral NMN administration, moving the compound beyond theoretical animal models into practical human application. In a landmark study conducted by Keio University School of Medicine in Japan, researchers conducted the first rigorous safety evaluations of both single-dose and long-term oral NMN in healthy adult males 564546. The trials confirmed that doses up to 500 mg were rapidly absorbed, safely metabolized, and resulted in dose-dependent elevations of systemic NAD+ levels without inducing significant adverse physiological, cardiovascular, or ophthalmological effects 564546.
Subsequent Phase II randomized controlled trials have expanded upon these initial safety findings to identify metabolic and functional benefits. A seminal 10-week study published in Science involving prediabetic postmenopausal women demonstrated that 250 mg/day of NMN significantly improved skeletal muscle insulin sensitivity and upregulated mTOR and Akt signaling networks 55. Further multi-center clinical trials evaluating dosages ranging from 300 mg to 900 mg over 30 to 60 days reported statistically significant improvements in physical endurance, measured via six-minute walking distances, alongside measurable improvements in patient-reported outcomes and circulating NAD+ metabolites 4347. Additional 12-week trials utilizing 125 mg twice daily showed significant reductions in total LDL and non-HDL cholesterol, diastolic blood pressure, and modest attenuation of postprandial hyperinsulinemia 4647.
Volatile Regulatory Status of NMN Supplements
Despite the accumulating clinical safety data, the regulatory environment governing NMN has been highly volatile, reflecting the ongoing tension between pharmaceutical drug development and the dietary supplement industry. In November 2022, the FDA invoked the "drug preclusion" clause of the Dietary Supplement Health and Education Act (DSHEA), effectively banning the sale of NMN as a dietary supplement because it was concurrently undergoing clinical investigation as an Investigational New Drug (IND) by pharmaceutical entities 604862.
Following sustained legal challenges, federal lawsuits, and citizen petitions led by the Natural Products Association (NPA), the FDA officially reversed this decision in late September 2025 604849. The agency concluded that there was sufficient historical evidence demonstrating that NMN had been lawfully marketed as a dietary supplement in the United States prior to the authorization of the initial IND, thereby exempting it from the drug preclusion clause 6249.
As of early 2026, NMN is legally permitted for sale as a dietary supplement in the United States, provided manufacturers submit the requisite New Dietary Ingredient Notifications (NDIN) to ensure manufacturing safety 4950. Conversely, the European Union maintains significantly stricter constraints, requiring extensive and costly Novel Food authorization before NMN can be legally marketed - a process that remains ongoing and currently prevents lawful retail sale in the region 50.
Senotherapeutics and Cellular Senescence
Cellular senescence is a state of irreversible cell cycle arrest triggered by severe DNA damage, telomere shortening, mitochondrial dysfunction, or oncogenic stress 12351. While initially serving as a protective mechanism against malignant transformation and aiding in embryonic development and wound healing, senescent cells accumulate exponentially with advancing age 151. These cells resist natural apoptosis and secrete a highly toxic mixture of pro-inflammatory cytokines, chemokines, and matrix metalloproteinases - collectively termed the senescence-associated secretory phenotype (SASP) 192352. The persistent SASP drives chronic, low-grade systemic inflammation (often referred to as "inflammaging") and degrades surrounding tissue architecture, contributing heavily to osteoarthritis, pulmonary fibrosis, and neurodegeneration 52.
Senolytics are a highly specific class of pharmacological agents engineered to selectively induce apoptosis in these dysfunctional senescent cells while leaving healthy, proliferating cells unharmed 15253.
Clinical Trials of Dasatinib, Quercetin, and Fisetin
The most extensively studied senolytic intervention in humans is the combination of Dasatinib (a targeted tyrosine kinase inhibitor approved for leukemia) and Quercetin (a naturally occurring plant flavonoid), commonly referred to as the D+Q protocol 52354. In human pilot studies targeting idiopathic pulmonary fibrosis (IPF) - a classic senescence-driven disease - a short, intermittent burst of oral D+Q (administered over three weeks) significantly improved physical function, including walking speed, six-minute walk distance, and chair-rise ability 23.
Fisetin, another naturally occurring flavonoid found abundantly in strawberries and apples, has demonstrated potent senolytic properties in preclinical models 2352. In aged mice, fisetin effectively cleared senescent cells across multiple tissues, improved physical homeostasis, and extended median lifespan by approximately 10% 52. Early human trials administering short, high-dose bursts of fisetin (e.g., 20 mg/kg/day for two consecutive days) to older adults with osteoarthritis showed reductions in systemic inflammatory markers and improvements in self-reported physical function without serious adverse events 5255. Ongoing clinical trials, such as those registered by the American Society of Clinical Oncology (ASCO), are currently evaluating fisetin's ability to alleviate persistent frailty and functional decline in breast cancer survivors, a population known to experience a rapid, chemotherapy-induced increase in senescent cell burden 55.
Nuanced Clinical Outcomes and Biomarker Challenges
Despite immense preclinical promise, human clinical trials for senolytics have revealed the profound complexities of biological aging. A 2024 Phase II trial conducted by the Mayo Clinic evaluated the D+Q combination in healthy older women with osteoporosis 51. The treatment was administered intermittently (three consecutive days every 28 days for 20 weeks). While the overall treatment group did not show statistically significant differences in the reduction of bone resorption, a granular subgroup analysis revealed that women with a demonstrably high baseline burden of senescent cells responded positively, exhibiting measurable improvements in bone mineral density 51. This highlights a critical limitation in current gerotherapeutics: therapies like senolytics may only benefit individuals harboring a pathological threshold of senescent cells, underscoring the urgent need for precision medicine and companion diagnostics to identify ideal candidates before treatment 51.
Furthermore, longitudinal studies measuring the effect of senolytics on epigenetic age have yielded paradoxical results. A 2024 clinical study evaluating the D+Q protocol (and subsequently D+Q combined with Fisetin) over six months found that the treatment actually induced a short-term acceleration in first-generation epigenetic clocks and mitotic clocks derived from peripheral blood immune cells, alongside a decrease in telomere length 2353. Second and third-generation epigenetic clocks showed no significant changes 2353. Researchers hypothesize that this epigenetic acceleration does not indicate accelerated biological aging, but rather reflects the rapid proliferation, turnover, and reconstitution of immune cell populations following the targeted clearance of senescent immune cells 23. These findings mandate the development of more robust, tissue-specific biomarkers that are resistant to transient immune fluctuations to accurately gauge the physiological impact of senolytic therapies 2353.
| Clinical Trial / Study | Investigated Compound | Target Population | Primary Outcomes / Observations |
|---|---|---|---|
| PEARL Trial (2025) | Rapamycin (5mg & 10mg weekly) | Healthy adults (50-85) | Safe over 48 weeks; 5% increase in lean mass & reduced pain (women on 10mg); improved general well-being (5mg). |
| TAME Trial (Upcoming) | Metformin | Non-diabetic adults (65-79) | Evaluating composite delay of age-related diseases (CVD, cancer, dementia); sets precedent for FDA aging endpoints. |
| Keio University Phase II | NMN (up to 500mg) | Healthy adults, prediabetics | Safe, dose-dependent NAD+ increase; modest attenuation of postprandial hyperinsulinemia; improved endurance. |
| Epigenetic GLP-1 Trial | Semaglutide | Adults with lipohypertrophy | Decelerated PhenoAge by 4.9 years; slowed DunedinPACE clock by ~9%; reduced inflammation markers. |
| Mayo Clinic Phase II | Dasatinib + Quercetin (D+Q) | Older women with osteoporosis | Improved bone mineral density strictly in the subgroup with a high baseline senescent cell burden. |
Synthesis of Clinical Efficacy
The scientific evaluation of anti-aging interventions is currently undergoing a rapid and unprecedented transition from basic biological research into rigorous human clinical application 13. While no single pharmacological agent has been definitively proven to extend maximum human lifespan in controlled trials, a compelling body of clinical evidence supports the capacity of several interventions to delay age-related pathology and extend functional healthspan 3237.
Lifestyle modifications - specifically caloric restriction, intermittent fasting, and regular physical activity - remain the most effective, pleiotropic, and universally safe methods for modulating nutrient-sensing pathways, clearing cellular waste, and maintaining physiological resilience 2124.
Among pharmacological candidates, rapamycin exhibits the strongest preclinical data and promising early human safety profiles when dosed intermittently, successfully balancing autophagic clearance with immunological safety 731. Concurrently, metabolic modulators, particularly GLP-1 receptor agonists, have demonstrated an unprecedented ability to preserve organ function and decelerate biological aging clocks in human subjects, positioning them as leading prophylactic agents 2438. NAD+ precursors like NMN show excellent safety and measurable metabolic benefits in phase II trials, further supported by clear regulatory pathways for consumer access in the United States 5662. Finally, senolytics offer a highly targeted approach for clearing toxic cellular burdens, though precision companion diagnostics will be strictly required to optimize their deployment and avoid unnecessary treatment in healthy populations 51.
As international regulatory bodies such as the EMA and FDA begin to integrate epigenetic biomarkers, real-world data, and composite endpoints of "intrinsic capacity" into their approval frameworks, the next decade of geroscience will likely witness the approval of the first generation of multi-pathway, prophylactic gerotherapeutics designed to compress morbidity and fundamentally alter the trajectory of human aging 111425.