Clinical Efficacy and Safety of Intermittent Fasting 2024-2026
The paradigm of metabolic health, obesity management, and human longevity has undergone a profound transformation over the past decade. Historically, continuous energy restriction - the daily, persistent reduction of caloric intake - has served as the undisputed cornerstone of dietary intervention for obesity and cardiometabolic diseases. However, the chronic behavioral fatigue, metabolic adaptation, and long-term adherence failure associated with continuous energy restriction have driven the scientific and medical communities to explore alternative temporal dietary patterns. Intermittent fasting, which involves cycling between defined periods of voluntary energy restriction and ad libitum eating, has subsequently emerged as one of the most dominant and vigorously debated areas of clinical investigation in contemporary nutritional science 122.
Driven by pioneering foundational work from researchers such as Mark Mattson, Satchin Panda, Valter Longo, and Krista Varady, the understanding of intermittent fasting has rapidly evolved from early animal-model enthusiasm to highly rigorous, large-scale human clinical trials 453. Between 2024 and 2026, the scientific literature expanded exponentially. This period saw the publication of massive network meta-analyses, multi-year randomized controlled trials, and vigorous academic debates regarding long-term cardiovascular safety. This report provides an exhaustive, quantitative synthesis of the current evidence surrounding intermittent fasting, addressing comparative efficacy, underlying cellular mechanisms, epidemiological safety controversies, demographic-specific applications, and contemporary public health guidelines.
1. Taxonomic Classification of Intermittent Fasting Protocols
Before evaluating the quantitative efficacy and physiological impact of intermittent fasting, it is necessary to establish a precise taxonomy of the regimens currently utilized in clinical settings. The physiological responses to fasting, as well as the behavioral sustainability of the diets, are highly protocol-dependent 1. The literature generally recognizes four distinct categories of fasting based on the temporal distribution of energy intake.
The first category is alternate-day fasting, which involves alternating between days of ad libitum energy intake and days of severe caloric restriction. On the fasting days, individuals typically consume either zero calories or a highly restricted intake of approximately 25% of their baseline energy needs, which usually equates to roughly 500 kilocalories 4. This approach introduces a profound, acute bioenergetic challenge every 48 hours, forcing rapid metabolic adaptations 2.
The second major category is time-restricted eating, which operates on an entirely different mechanistic principle. Rather than imposing explicit caloric targets, time-restricted eating confines daily caloric intake to a specific window - commonly ranging from four to ten hours - while fasting for the remaining hours of the diurnal cycle. The most ubiquitous variant is the 16:8 method, comprising a 16-hour fasting window and an eight-hour eating window 445. This approach relies heavily on circadian biology, aligning nutrient intake with periods of peak metabolic efficiency 6.
The third category encompasses periodic fasting, widely popularized as the 5:2 or 4:3 diets. These regimens allow individuals to maintain unrestricted, ad libitum dietary habits for a set number of days per week (e.g., five days) while severely restricting calories - typically imposing a 75% to 80% deficit - on two or three non-consecutive days 478. This protocol aims to capture the metabolic benefits of severe fasting while offering greater social and behavioral flexibility than alternate-day fasting 9.
The final distinct category is the fasting-mimicking diet. Developed primarily to simulate the deep cellular effects of prolonged water fasting safely, this is a multi-day, commercially formulated, plant-based protocol. It is typically executed over five consecutive days once per month. The fasting-mimicking diet is precisely calibrated in its macronutrient profile to remain low in proteins and carbohydrates while being high in specific fats, effectively tricking the body's nutrient-sensing pathways into a fasted state while still providing essential micronutrients and mitigating the severe adverse effects of total starvation 410.
2. Quantitative Efficacy: Intermittent Fasting versus Continuous Energy Restriction
A primary, long-standing debate in nutritional epidemiology has been whether intermittent fasting possesses unique metabolic and thermodynamic advantages, or if it simply serves as a behavioral vehicle for establishing a standard caloric deficit. For years, the literature was saturated with underpowered, short-term trials that yielded conflicting results. The release of highly powered, long-term randomized controlled trials and comprehensive network meta-analyses in 2025 and 2026 has provided unprecedented clarity on this fundamental issue.
2.1 The 2025 BMJ Network Meta-Analysis
In 2025, a landmark network meta-analysis published in The BMJ aggregated data from 99 randomized clinical trials involving 6,582 adults, making it the most comprehensive synthesis of fasting data to date 111512. The participant pool had a median age of 45 years, an average body mass index of 31.3 $\text{kg/m}^2$, and nearly 90% presented with existing health conditions such as type 2 diabetes or metabolic syndrome 1112. This advanced network analysis allowed researchers to directly and indirectly compare various fasting modalities against continuous energy restriction and unrestricted ad libitum diets across numerous cardiometabolic parameters.
The primary findings definitively established that all forms of intermittent fasting and continuous energy restriction yielded statistically significant body weight reductions when compared to unrestricted diets 1314. However, when comparing intermittent fasting directly to traditional continuous energy restriction, the differences were remarkably nuanced. Alternate-day fasting was the singular fasting modality that demonstrated a statistically significant advantage over continuous energy restriction for weight loss, yielding an additional mean weight reduction of $-1.29$ kg (95% CI, $-1.99$ to $-0.59$ kg) 1512. Alternate-day fasting also produced trivial to moderate additional improvements in overall body mass index, lipid parameters, and systolic blood pressure relative to standard continuous restriction 14. Conversely, popular protocols such as time-restricted eating and whole-day fasting (e.g., the 5:2 diet) did not significantly outperform continuous energy restriction in long-term weight reduction, demonstrating comparable efficacy 15.
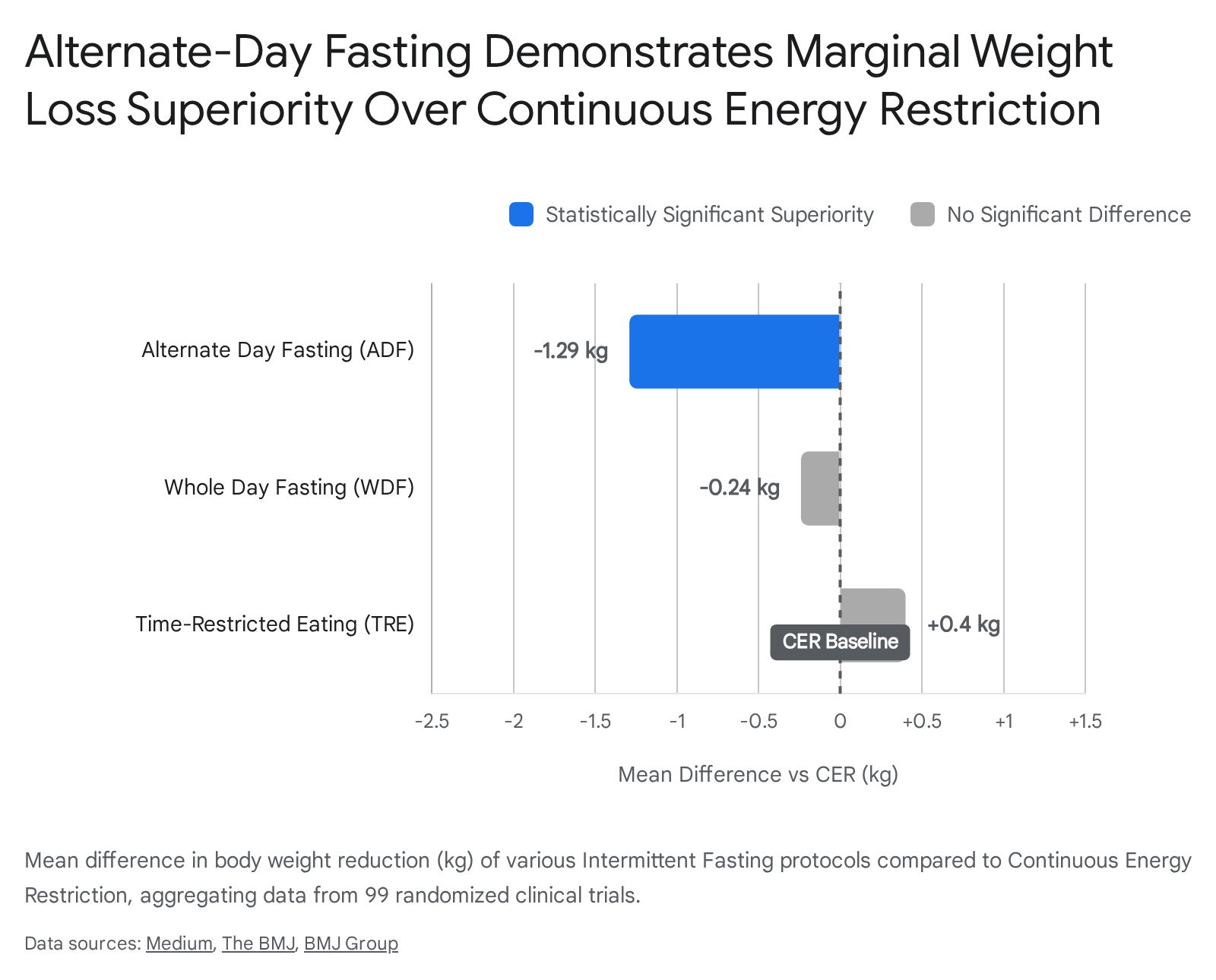
The researchers also noted that the slight weight loss advantage of alternate-day fasting over continuous energy restriction tended to diminish in trials extending beyond 24 weeks, suggesting that long-term metabolic adaptation or waning behavioral adherence eventually equalizes the outcomes 1513.
2.2 The 2026 Cochrane Database Systematic Review
Further contextualizing the overall clinical impact of fasting, a highly rigorous 2026 Cochrane Database Systematic Review evaluated 22 randomized controlled trials encompassing 1,995 overweight or obese participants. This review specifically focused on comparing various intermittent fasting approaches against standard, regular dietary advice or waitlist control groups 1516. The Cochrane authors determined that intermittent fasting resulted in a mean difference in weight loss of only $-0.33$ kg (95% CI, $-0.92$ to $0.26$ kg) when compared to regular dietary advice 1718.
Overall, participants engaged in intermittent fasting typically lost approximately 3% of their baseline body weight. The researchers noted that evidence suggesting fasting could reliably help patients achieve a 5% reduction in body weight - a threshold widely considered clinically meaningful for reducing cardiometabolic risk - was of very low certainty 1819. Consequently, the lead authors of the review cautioned that while intermittent fasting is a viable option for some individuals, the data does not justify the extraordinary enthusiasm it receives in popular media, as it performs comparably to conventional dietary counseling without demonstrating clear superiority in long-term weight reduction or quality-of-life improvements 1820.
2.3 The 4:3 Intermittent Fasting Trial (Annals of Internal Medicine, 2025)
To address the persistent lack of long-term, head-to-head data comparing specific periodic fasting regimens with daily caloric restriction under tightly controlled conditions, a watershed 12-month randomized clinical trial was published in the Annals of Internal Medicine in 2025. Catenacci and colleagues evaluated 165 overweight or obese adults (mean body mass index 34.1 $\text{kg/m}^2$, 73.9% female) assigned to either a 4:3 intermittent fasting group or a daily caloric restriction group 821. Crucially, the study design prescribed both cohorts an identical aggregate weekly energy deficit of 34.3% and provided both groups with identical high-intensity behavioral support and exercise prescriptions (300 minutes per week) 822.
The results demonstrated a modest but statistically significant divergence in favor of the 4:3 intermittent fasting protocol. At the 12-month mark, the 4:3 fasting group lost an average of 7.7 kg ($-7.6\%$ of baseline body weight), whereas the daily caloric restriction group lost 4.8 kg ($-5.0\%$ of baseline body weight) 821.
| Clinical Outcome (12 Months) | 4:3 Intermittent Fasting (n=84) | Daily Caloric Restriction (n=81) | Statistical Significance |
|---|---|---|---|
| Absolute Weight Loss | $-7.7$ kg | $-4.8$ kg | $P = 0.040$ |
| Percentage of Baseline Body Weight | $-7.6\%$ | $-5.0\%$ | Significant |
| Participants Achieving $\ge 5\%$ Weight Loss | 58% | 47% | Clinically Meaningful |
| Participants Achieving $\ge 10\%$ Weight Loss | 38% | 16% | Superiority Indicated |
Table 1: Anthropometric outcomes from the 12-month 4:3 Intermittent Fasting Trial compared to Daily Caloric Restriction 82122.
Beyond the pure anthropometric data, secondary behavioral analyses of this trial highlighted the psychological mechanisms driving the differential outcomes. Participants assigned to the 4:3 fasting cohort exhibited significant reductions in binge eating and uncontrolled eating behaviors compared to the daily restriction group. The researchers hypothesized that the inherent flexibility of eating without strict caloric limits for four days out of the week fostered higher long-term adherence and a less rigid, more adaptive psychological relationship with food 9. This flexibility ultimately allowed the 4:3 cohort to spontaneously reduce their overall caloric intake more effectively than those tasked with calculating daily deficits 921.
3. Cardiometabolic Risk Factors and Biomarkers
While weight loss is the primary driver of cardiometabolic improvement, extensive research indicates that intermittent fasting exerts independent, direct effects on metabolic disease markers. Even in scenarios where absolute weight loss is comparable between fasting and continuous restriction cohorts, differences frequently emerge in specific biochemical indices 615.
Systematic reviews demonstrate that fasting interventions significantly enhance insulin sensitivity. For instance, a 12-month study evaluating individuals with existing insulin resistance found that alternate-day fasting resulted in massive reductions in fasting insulin levels and the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) - decreasing by 52% and 53%, respectively - compared to reductions of only 14% and 17% in a continuous calorie restriction group, despite both groups achieving comparable overall weight loss 23. This suggests that the extended periods of low circulating glucose uniquely repair pancreatic $\beta$-cell function and peripheral tissue insulin receptivity.
Lipid metabolism also adapts to prolonged fasting. Short-term and mid-term fasting protocols have consistently been shown to lower total cholesterol, low-density lipoprotein cholesterol (LDL-C), and triglycerides 46. Some studies report reductions in total cholesterol ranging from 31.8 to 37 mg/dL, and triglyceride reductions between 25 and 38 mg/dL following alternate-day fasting protocols 4. Furthermore, fasting elicits notable hemodynamic improvements, with multiple meta-analyses documenting reductions in systolic blood pressure ranging from 2.14 mmHg to 9.5 mmHg, often outperforming the blood pressure reductions seen in continuous energy restriction groups 424.
4. Chrononutrition: The Role of Circadian Alignment
The thermodynamic accounting of caloric intake fails to explain the entirety of the body's response to food. The integration of chronobiology into fasting research - a field termed chrononutrition - has revealed that the precise temporal placement of meals within the 24-hour day is highly relevant to metabolic endpoints, largely independent of total caloric volume.
A distinct bifurcation in metabolic efficacy exists between early time-restricted eating (eTRE; typically involving an eating window from early morning to mid-afternoon, such as 08:00 to 16:00) and late time-restricted eating (lTRE; involving eating from noon into the evening, such as 12:00 to 20:00). Network meta-analyses and highly controlled crossover trials demonstrate that early time-restricted eating aligns caloric intake with the body's natural diurnal peaks of insulin sensitivity and metabolic efficiency 2325. The human circadian system dictates that glucose tolerance and pancreatic insulin secretion are optimal in the morning and progressively decline toward the evening. Consequently, early time-restricted eating yields significantly greater improvements in fasting blood glucose, HOMA-IR, and postprandial insulin levels compared to late time-restricted eating 425.
Further supporting this, an observational study from the Barcelona Institute for Global Health tracked over 7,000 adults over five years. The data revealed that participants who routinely consumed breakfast early in the day and extended their overnight fasting window by finishing dinner early exhibited a significantly lower body mass index over time compared to those with delayed eating patterns 26.
However, the intrinsic benefits of the shortened eating window are heavily dependent on an actual reduction in calories. The ChronoFast trial, published in 2026 in Science Translational Medicine, examined 31 women with overweight or obesity undergoing strictly isocaloric early time-restricted eating. In this rigorously controlled environment where the 8-hour window was matched calorie-for-calorie against a standard eating pattern, the researchers observed no clinically meaningful changes in insulin sensitivity, blood sugar, or cardiovascular markers 27. The shortened window did, however, significantly shift the body's internal circadian clock and alter sleep architecture 27. This trial reinforced the concept that while chrononutrition optimizes the physiological processing of food, the primary metabolic benefits of time-restricted eating observed in the general population are largely driven by the unintended, spontaneous reduction of total calories caused by limiting the time available to eat 27.
5. Cellular and Molecular Mechanisms of Fasting
The translation of intermittent fasting from an epidemiological observation to a targeted medical intervention relies upon the deep elucidation of its subcellular mechanics. The "Cyclic Metabolic Switching" theory, formalized in the scientific literature by neuroscientist Mark Mattson in 2025, posits that the true health benefits of intermittent fasting are not derived from a continuous, monotonic state of caloric deficit. Rather, the benefits arise precisely from the rhythmic oscillation between states of targeted metabolic stress during fasting, followed by periods of robust cellular recovery and growth during feeding 2.
5.1 The Bioenergetic Challenge and the Metabolic Switch
When exogenous nutrient supply ceases for an extended duration - typically surpassing 10 to 14 hours - hepatic glycogen stores are progressively depleted 5. This bioenergetic challenge fundamentally shifts the body's systemic respiratory quotient. To maintain energy homeostasis, adipose tissue begins hydrolyzing stored triglycerides into free fatty acids. These fatty acids are transported to the liver, where they undergo accelerated beta-oxidation and are converted into ketone bodies, predominantly $\beta$-hydroxybutyrate 528.
This transition - widely referred to as the metabolic switch - induces profound downstream neuroendocrine and biochemical responses. The sharp reduction in circulating systemic glucose and amino acids down-regulates the insulin and insulin-like growth factor 1 (IGF-1) signaling pathways 5. Concurrently, the depletion of intracellular ATP alters the cellular adenylate energy charge, increasing the $\text{AMP}:\text{ATP}$ ratio 5. This shift acts as a primary trigger, directly activating AMP-activated protein kinase (AMPK), which functions as the cell's master energy-sensing regulator 5. Furthermore, the fasting state elevates cellular $\text{NAD}^+$ levels, serving as a critical cofactor for sirtuins (SIRT1), a family of epigenetic modifiers involved in DNA repair and metabolic regulation 5.
5.2 Autophagy, mTOR Inhibition, and Cellular Recycling
The activation of AMPK, combined with the repression of the IGF-1 axis, leads to the direct inhibition of the mammalian target of rapamycin (mTOR) signaling pathway 15. mTOR is a central kinase complex that acts as the primary driver of cellular anabolism, protein synthesis, and cellular proliferation. Its sustained inhibition during the fasting window forces the cell to halt energy-expensive growth processes and instead initiate macroautophagy, commonly referred to simply as autophagy 529. Autophagy is a highly conserved, fundamental cytoprotective mechanism wherein cells systematically package damaged organelles, misfolded proteins, and excess lipid droplets into autophagosomes, delivering them to lysosomes for degradation and molecular recycling 2930.
Historically, while the induction of autophagy by fasting has been extensively documented in murine models and cell cultures, direct evidence in humans has been methodologically challenging to capture. However, groundbreaking translational research published in 2025 provided clear evidence of this phenomenon in human subjects. Exploratory analyses documented significant up-regulation of specific autophagy-related genes - including LAMP2, LC3B, and ATG5 - in human peripheral blood mononuclear cells following periods of intermittent fasting 2335. Furthermore, an intensive six-month clinical trial of intermittent time-restricted eating demonstrated a tangible increase in physiological autophagic flux within these cells compared to standard care control cohorts 3035. This provided robust, peer-reviewed evidence that temporal nutrient restriction can actively modulate autophagy in humans, offering a direct molecular mechanism for how fasting delays the onset of age-related cellular dysfunction and mitigates biological aging 2930.
6. The Cardiovascular Risk Controversy: Analyzing the 2024 AHA/NHANES Data
As intermittent fasting surged in popularity, its long-term safety profile came under intense public and scientific scrutiny. One of the most fiercely debated developments in the literature occurred in March 2024, when a large-scale epidemiological study generated global media headlines by questioning the cardiovascular safety of time-restricted eating.
6.1 The Zhong Study Findings
Presented at the American Heart Association's (AHA) Epidemiology and Prevention Scientific Sessions, a study led by Dr. Victor Wenze Zhong analyzed dietary data from over 20,000 adults enrolled in the United States National Health and Nutrition Examination Survey (NHANES) between 2003 and 2018 313233. Cross-referencing participant data with mortality records from the Centers for Disease Control and Prevention's National Death Index, the researchers reported a startling finding: individuals who restricted their daily eating to an 8-hour window (the equivalent of a 16:8 time-restricted eating diet) exhibited a highly significant 91% increased risk of dying from cardiovascular disease (Hazard Ratio 1.91; 95% CI 1.20-3.03) over a median follow-up of eight years, compared to those who consumed their meals over a standard 12-to-16-hour window 3132.
The elevated risk was particularly pronounced in vulnerable subpopulations; for patients with pre-existing cardiovascular disease, an eating window of 8 to 10 hours was linked to a 66% higher risk of death from heart disease or stroke 32. The study also noted that time-restricted eating did not confer any overall longevity benefits or reduce the risk of all-cause mortality 32. Paradoxically, the data indicated that among cancer patients, consuming food over a much wider window extending beyond 16 hours per day was associated with a lower risk of cancer-related mortality 3233.
6.2 Methodological Critiques and the Scientific Consensus
The widespread dissemination of these alarming statistics prompted an immediate, rigorous, and highly critical response from the broader cardiometabolic research community. Retrospectively evaluating the discourse from the vantage point of 2026, the scientific consensus largely views the 91% hazard ratio not as a causal biological reality, but as a severe artifact of observational confounding and inherent epidemiological limitations 344041.
Cardiologists and nutrition experts identified several profound methodological flaws that fundamentally undermined the AHA abstract's conclusions: 1. Extreme Dietary Recall Limitations: The classification of participants' eating windows was predicated on merely two 24-hour dietary recall questionnaires completed during their first year of enrollment. Extrapolating a two-day, self-reported snapshot to represent an individual's rigid, habitual eating pattern over a median eight-year follow-up period introduces an extraordinary degree of measurement error and recall bias 323440. 2. Reverse Causality and Health-Seeking Behaviors: The NHANES dataset is purely observational and cannot identify why participants were eating in a restricted window. Experts noted that individuals compressing their meals into less than eight hours likely included shift workers, individuals experiencing high psychological stress, or, crucially, those with underlying, undiagnosed pathologies. Severe illnesses, gastrointestinal distress, or cancer-induced anorexia naturally suppress appetite and compress eating windows. Therefore, the restricted eating pattern was highly likely a symptom of existing poor health leading to premature mortality, rather than the primary cause of it 3341. 3. Absence of Diet Quality Assessment: The study failed to account for the nutrient density or macronutrient composition of the diets. Cardiovascular outcomes could easily be driven by the consumption of hyper-caloric, ultra-processed foods within that 8-hour window, rather than the duration of the fasting period itself 324041. 4. Statistical Noise and Data Dredging: Detailed reviews of the actual poster presentation revealed that the authors conducted at least 36 separate subgroup analyses. Finding an isolated spike in cardiovascular mortality in one specific subgroup without a corresponding, logical increase in all-cause mortality strongly suggests the presence of random statistical noise - a common outcome when dredging massive, multi-variable datasets 34.
While the Zhong study serves as a stark reminder of the limitations of retrospective epidemiological datasets, separate, highly controlled prospective clinical trials provide a more nuanced picture of risk. The WONDERFUL trial, for example, noted that intermittent fasting can transiently elevate short-term clinical mortality risk scores (such as the Intermountain Mortality Risk Score) due to the acute physiological stress it places on the body, while simultaneously acting as an adaptive trigger that drastically decreases 10-year chronic disease risk formulas through profound long-term improvements in insulin sensitivity and lipid profiles 35. The overriding clinical consensus maintains that properly implemented intermittent fasting does not inherently drive cardiovascular pathology 3343.
7. Body Composition and Sarcopenia Mitigation
A highly valid clinical concern accompanying any energy restriction protocol is the concomitant loss of lean body mass. When the body is placed in a prolonged caloric deficit, it inevitably catabolizes both adipose tissue and skeletal muscle to meet its energetic demands. The loss of fat-free mass is deleterious, as it directly decreases the basal metabolic rate, impairs functional mobility, and predisposes individuals to frailty and rapid weight regain 10.
Meta-analyses evaluating intermittent fasting, particularly extended protocols or alternate-day regimens, confirm that without targeted behavioral and nutritional interventions, a significant proportion of the weight lost consists of fat-free mass 636. Consequently, clinical guidelines established by 2026 strongly emphasize two vital, non-negotiable adjuncts to any fasting regimen designed for sustainable health:
First, practitioners emphasize "protein pacing." Concentrating an entire day's worth of protein intake into a highly compressed eating window or a single massive meal impairs optimal muscle protein synthesis. Skeletal muscle anabolism is best stimulated by evenly distributed doses of essential amino acids throughout the feeding period. Nutritional evidence indicates that distributing approximately 30 grams of high-quality protein evenly across three distinct meals within the feeding window significantly minimizes muscle catabolism and preserves lean mass much more effectively than haphazard eating 4345.
Second, the integration of resistance training is considered paramount. A 2025 systematic review and meta-analysis from the University of Mississippi, which analyzed 15 studies over the past decade, confirmed that combining time-restricted eating (specifically an 8-hour window) with regular resistance training not only preserves lean muscle mass but significantly accelerates the reduction of visceral fat compared to fasting in isolation 46. Fasting should be viewed as an adjunct to physical conditioning, not a replacement for it.
8. Neuroprotection, Cognitive Function, and Aging
The translation of cyclic metabolic switching to neurobiology has yielded highly promising clinical findings regarding cognitive health, directly challenging longstanding myths about fasting and mental acuity.
Patients frequently express anxiety that abstaining from food will impair acute cognitive performance, inducing lethargy, lack of focus, or "brain fog" during the workday. A comprehensive meta-analysis of 63 global studies encompassing over 3,400 individuals, published in the Psychological Bulletin in 2025, decisively refuted this notion. The data demonstrated that short-term fasting (up to 24 hours) neither enhances nor impairs acute mental performance in healthy adults 28. The human brain rapidly adapts to systemic glycogen depletion by up-regulating the utilization of astrocyte-derived ketone bodies (such as $\beta$-hydroxybutyrate), ensuring a stable, highly efficient, and continuous supply of ATP to neurons even in the absence of exogenous glucose 28.
Conversely, the chronic implementation of intermittent fasting demonstrates measurable neuroprotective effects that accumulate over time. In a clinical trial sponsored by the National Institute on Aging (NIA) involving 40 insulin-resistant seniors (mean age 63), participants assigned to a 5:2 intermittent fasting protocol exhibited significantly reduced insulin resistance within both the body and the brain. Furthermore, the fasting cohort demonstrated superior improvements in executive function - the higher-order cognitive domain responsible for planning, strategizing, and problem-solving - compared to a control group following a standard healthy living diet 37. Both dietary interventions successfully reduced biological markers of brain aging on MRI scans, validating the underlying thesis that reversing systemic metabolic syndrome and insulin resistance inherently protects the cerebral microvasculature and delays cognitive decline 37.
9. Geriatric Considerations: The Limits of Fasting in Older Adults
While intermittent fasting offers potent neuro-metabolic benefits, older populations represent a highly vulnerable demographic that requires strict protocol titration. The physiological resilience necessary to endure profound cyclic metabolic switching wanes significantly with advancing age, making aggressive fasting regimens potentially hazardous for the elderly.
A robust 2026 network meta-analysis published in the journal Nutrients evaluated 31 studies focusing exclusively on the safety and efficacy of fasting in adults over the age of 60 3839. The analysis identified a distinct therapeutic window for fasting safety in geriatrics. Moderate approaches, specifically time-restricted eating using a 16:8 protocol and Islamic Sunnah Fasting (ISF), emerged as the safest and most effective regimens. These moderate protocols yielded clinically meaningful reductions in body weight (averaging $-1.92$ kg to $-2.36$ kg), lowered blood pressure, and improved overall metabolic markers without inducing dangerous losses of lean muscle mass 3940.
However, extreme fasting regimens proved highly deleterious in this age group. The researchers found that compressing the eating window to less than 10 hours per day, or extending the fasting period beyond 12.4 to 14 hours, correlated directly with severe adverse outcomes. Elderly individuals subjected to these extreme windows exhibited reduced cognitive function scores and a staggering 58% increased risk of cardiovascular mortality 383940. This critical data firmly establishes that moderation is not optional for geriatric patients; highly restricted eating windows pose an unacceptable risk of malnutrition, sarcopenia, and cardiovascular stress in older demographics 3840.
10. Institutional Integration and Public Health Guidelines (2025 - 2026)
As the clinical data surrounding intermittent fasting has matured and moved beyond transient dietary trends, leading global health institutions have begun formally addressing its application within national nutritional frameworks.
10.1 The USDA 2025 - 2030 Dietary Guidelines for Americans
Released in January 2026, the Dietary Guidelines for Americans, 2025-2030 represents a profound paradigm shift in federal nutrition policy, described by officials as a fundamental "reset" aimed at combating the chronic disease epidemic 4152. While the guidelines do not explicitly endorse specific temporal fasting windows like 16:8 or 5:2, their foundational philosophy aligns perfectly with the biochemical prerequisites necessary for safe, effective fasting 4142.
Moving away from strict caloric mathematics and complex dietary architecture, the core message of the 2025 - 2030 guidelines is remarkably simple: "eat real food" 4143. The guidelines instruct a dramatic reduction in the consumption of highly processed foods laden with refined carbohydrates, artificial additives, and low-calorie non-nutritive sweeteners 4152. Crucially, the new guidelines prioritize the consumption of high-quality protein at every meal, updating the standard recommendations to a bodyweight-based target of 1.2 to 1.6 grams of protein per kilogram of body weight per day 5242.
Furthermore, the guidelines remove historical, restrictive caps on full-fat dairy and meat consumption, advising only that saturated fat remains below 10% of total daily calories 4244. This systemic shift toward nutrient density, high protein, and healthy fats indirectly supports the physiological requirements of intermittent fasting; a diet rich in these macronutrients drastically increases satiety, stabilizes postprandial glycemic excursions, and facilitates successful adherence to prolonged fasting windows without the debilitating hypoglycemic crashes associated with carbohydrate-dominant, highly processed diets 5242. The visual representation of these guidelines also abandoned the traditional "MyPlate" in favor of an inverted food pyramid, heavily emphasizing protein and whole foods at its broad apex 5244.
10.2 The NHS UK Framework and Pharmacological Contraindications
The United Kingdom's National Health Service (NHS) has adopted a highly pragmatic, evidence-based approach to the clinical deployment of intermittent fasting. Current 2026 NHS guidance officially acknowledges that intermittent fasting (specifically the 5:2 and 16:8 protocols) produces clinically meaningful weight loss - typically 3% to 8% of body weight over 8 to 24 weeks - that is entirely comparable to standard continuous caloric restriction 5645. The NHS positions intermittent fasting not as a metabolic magic bullet, but as a highly viable behavioral alternative for patients who suffer from chronic restriction fatigue under traditional daily dieting regimens.
However, the NHS emphasizes stringent risk stratification and highlights profound pharmacological interactions. Intermittent fasting is explicitly contraindicated for pregnant women, pediatric populations, and individuals with a history of eating disorders, as the strict rules surrounding feeding windows can trigger or exacerbate restrictive psychological patterning and binge/purge cycles 5645.
Furthermore, the integration of fasting with modern pharmacological management of type 2 diabetes requires extreme caution. The NHS mandates that diabetic patients utilizing insulin, insulin secretagogues (such as sulfonylureas), or Sodium-Glucose Cotransporter-2 (SGLT2) inhibitors must undergo rigorous physician-managed medication titration prior to initiating any fasting protocol. Fasting while taking SGLT2 inhibitors or exogenous insulin exponentially increases the risk of severe hypoglycemia and euglycemic diabetic ketoacidosis, a potentially fatal metabolic complication 5658.
| Patient Profile / Demographic | Intermittent Fasting Suitability | Primary Clinical Concerns and Contraindications |
|---|---|---|
| Healthy Adults (BMI > 25) | Highly Suitable | Ensuring adequate protein intake; managing transient hunger and hydration 4356. |
| Type 2 Diabetes (Diet-controlled) | Suitable (with monitoring) | Excellent potential to reduce HOMA-IR and fasting glucose; requires glycemic monitoring 4345. |
| Type 2 Diabetes (on SGLT2i / Insulin) | Contraindicated without supervision | High risk of euglycemic ketoacidosis or severe hypoglycemia; requires medication titration 56. |
| Geriatric Populations (>60 years) | Use with Extreme Caution | Risk of sarcopenia and frailty; eating windows must remain $\ge 10$ hours to prevent cardiovascular stress 39. |
| History of Eating Disorders | Strictly Contraindicated | High risk of exacerbating binge/purge cycles and restrictive psychological behaviors 2856. |
| Pregnancy and Lactation | Strictly Contraindicated | Potential impairment of fetal development and maternal nutritional inadequacy 56. |
Table 2: Clinical Risk Stratification and Suitability for Intermittent Fasting based on 2026 Public Health Frameworks.
11. Conclusion
The rigorous scientific evaluation of intermittent fasting between 2024 and 2026 has successfully stripped away the intense hyperbole and social media speculation surrounding the practice, leaving behind a scientifically validated, behaviorally potent dietary tool. The culmination of massive network meta-analyses and long-term randomized controlled trials overwhelmingly indicates that, from a pure thermodynamic and weight-loss perspective, time-restricted eating and continuous energy restriction are functionally equivalent 1512. Alternate-day fasting provides only a marginal statistical advantage over standard dieting, and the long-term sustainability of any protocol remains the primary determinant of success 91512.
However, the physiological impact of intermittent fasting cannot be reduced entirely to the simple mathematics of a caloric deficit. The temporal restriction of nutrients induces profound, independent biological adaptations that continuous grazing does not - specifically, the entrainment of peripheral circadian rhythms, the up-regulation of macroautophagy, and the reduction of systemic insulin resistance via the activation of AMPK and the mechanism of cyclic metabolic switching 2232729.
The alarming cardiovascular mortality risks highlighted in observational epidemiological studies (such as the 2024 NHANES analysis) serve as a stark reminder of the dangers of extreme restriction devoid of nutrient density, though they suffer from severe methodological flaws and do not negate the robust safety profile demonstrated in dozens of highly controlled clinical trials 14313240. Moving forward, clinical success dictates that fasting protocols must be highly individualized. Intermittent fasting yields optimal, life-extending cardiometabolic benefits only when combined with high-quality protein pacing to prevent sarcopenia, regular resistance training to preserve functional mobility, and the consumption of nutrient-dense, unprocessed whole foods as emphasized by modern dietary guidelines 43454641.