Circulating Blood Factors and Plasma Exchange in Aging
Systemic Regulation of Biological Aging
The systemic regulation of biological aging is governed by a highly dynamic and complex milieu of circulating factors within the blood plasma. Historically, the aging process was considered an inevitable, cell-autonomous deterioration of biological function. However, seminal heterochronic parabiosis experiments - where the circulatory systems of young and old murine models were surgically fused - demonstrated that systemic factors actively dictate tissue-specific aging and regenerative capacity. Exposure to a youthful systemic environment was shown to restore regenerative potential in aged tissues, while exposure to an aged systemic environment accelerated functional decline in young tissues 123.
Early interpretations of these parabiosis models operated on the hypothesis that the aging process was primarily driven by the progressive depletion of youthful "activator" or "rejuvenating" factors. Consequently, longevity research focused heavily on identifying and isolating specific youth-associated proteins, such as specific growth factors and hormones, to be administered as exogenous therapeutics 145.
Recent advancements in geroscience, however, have precipitated a paradigm shift. Novel experimental designs utilizing neutral blood exchange (NBE) - which dilutes the plasma of older animals using a saline and 5% albumin solution without introducing any young blood components - have demonstrated multi-tissue rejuvenation equivalent to, or occasionally exceeding, that of heterochronic parabiosis 366. Old plasma dilution alone improves skeletal muscle repair, reduces liver adiposity and fibrosis, attenuates neuroinflammation, and increases hippocampal neurogenesis 3667. These findings indicate that the fundamental driver of systemic aging is not solely the absence of youthful factors, but rather the accumulation of circulating pro-aging inhibitors that actively suppress stem cell function, impair cellular communication, and sustain chronic inflammation 37.
The identification of these specific circulating activators and inhibitors, alongside the clinical application of therapeutic plasma exchange (TPE) in humans, forms the basis of current translational longevity medicine.
Youth-Associated Activators
The search for circulating proteins that actively promote cellular plasticity and tissue regeneration has identified several candidates that decline with chronological age. While the systemic dilution of inhibitors is highly effective, the presence of specific molecular activators remains a critical component of biological resilience and homeostatic maintenance.
Tissue Inhibitor of Metalloproteinases 2
Tissue inhibitor of metalloproteinases 2 (TIMP2) has emerged as a well-documented youth-associated blood-borne factor. TIMP2 is widely expressed throughout the body, is highly enriched in human umbilical cord plasma as well as young murine plasma, and reliably crosses the blood-brain barrier from the systemic circulation into the central nervous system 8910. Systemic levels of TIMP2 rapidly decline from early development into adulthood and old age 11.
Physiologically, the TIMP family primarily regulates the extracellular matrix (ECM) by inhibiting matrix metalloproteinases (MMPs), which are peptidases responsible for ECM degradation. Specifically, TIMP2 forms a trimolecular complex with MMP14 and proMMP2, acting as a crucial regulator of ECM remodeling, angiogenesis, and tissue architecture 1213.
In the context of central nervous system aging, systemic administration of TIMP2 in aged murine models has been shown to revitalize adult hippocampal neurogenesis, enhance synaptic plasticity, and improve hippocampus-dependent memory performance 81014. Furthermore, TIMP2 acts directly on microglia - the resident innate immune cells of the brain. The deletion of TIMP2 exacerbates age-associated microglial phenotypes, leading to transcriptomic shifts toward cell activation, increased microgliosis, and diminished phagocytic clearance of myelin and cellular debris. Conversely, treating aged models with TIMP2 reduces the proportion of pro-inflammatory microglia and enhances physiological phagocytosis, positioning the protein as a key modulator of age-related microglial dysfunction 1116.
Translational evidence for TIMP2 in human cognitive aging is expanding rapidly. Proteomic polygenic risk score (protPRS) analyses indicate that a genetic predisposition for higher lifelong plasma TIMP2 levels is significantly associated with improved global cognition and episodic memory in older adults, even among those at risk for Alzheimer's disease 1516. Cross-sectional and longitudinal human cohorts demonstrate that higher plasma TIMP2 correlates with larger brain volumes and that longitudinal decreases in the protein associate with steeper trajectories of cognitive decline 814.
Behavioral Modulation of TIMP2
TIMP2 levels are not strictly predetermined by genetics or age; they are demonstrably responsive to neuroprotective lifestyle behaviors. Physical activity - specifically objectively measured average daily step counts - has been positively correlated with circulating TIMP2 levels in cross-sectional human studies 81014.
Longitudinal changes in multi-domain lifestyle factors positively associate with changes in TIMP2 levels over time. In controlled murine models, exposure to an enriched, plasticity-inducing environment for three weeks elevates plasma TIMP2 levels. While wildtype mice exposed to enrichment exhibit elevated adult hippocampal neurogenesis, this neurogenic effect is entirely lost in models where TIMP2 has been genetically deleted 814. This establishes TIMP2 not merely as a biomarker, but as an essential mechanistic pathway through which lifestyle behaviors confer neuroprotection and cognitive resilience 81720.
Age-Associated Inhibitors and Pro-Aging Factors
The accumulation of circulating inhibitors drives the progressive deterioration of homeostatic repair mechanisms. These molecules disrupt cellular signaling and actively suppress the regenerative capacity of resident stem cell populations.
Senescence-Associated Secretory Phenotype
Cellular senescence is a primary hallmark of biological aging characterized by irreversible cell-cycle arrest in response to genomic instability, oxidative stress, oncogene activation, or telomere attrition. While senescent cells cease to divide, they remain metabolically highly active and secrete a complex, deleterious array of pro-inflammatory cytokines, chemokines, matrix metalloproteinases, and growth factors. This specific secretory profile is collectively termed the Senescence-Associated Secretory Phenotype (SASP) 181920.
The SASP is a fundamental driver of "inflammaging" - the chronic, sterile, low-grade systemic inflammation pervasive in older populations. Canonical SASP factors include Interleukin-6 (IL-6), Tumor Necrosis Factor-alpha (TNF-α), Interleukin-1 alpha and beta (IL-1α, IL-1β), and Interleukin-8 (CXCL-8) 181921. As these factors accumulate in systemic circulation, they exert paracrine and endocrine effects that impair neighboring healthy cells, inhibit progenitor cell proliferation, and disrupt the extracellular matrix 1820.
In the tumor microenvironment, senescent macrophages and cancer-associated fibroblasts continuously release SASP factors. The localized accumulation of IL-6 and IL-8 activates downstream signaling cascades, including the STAT3 and NF-κB pathways, which promote angiogenesis, tumor cell survival, and epithelial-to-mesenchymal transition (EMT), facilitating metastasis and immune evasion 1920. Systemically, elevated levels of highly cited SASP markers such as IL-6 and TNF-α correlate strongly with biological age acceleration, cardiovascular disease, and all-cause mortality 1825. Furthermore, systematic reviews of clinical trials indicate that standard physical exercise interventions have inconsistent or negligible impacts on circulating SASP-related cytokines like IL-6 and TNF-α, suggesting that more direct physiological interventions may be required to clear these established inflammatory mediators 2125.
Chemokines and Inflammatory Mediators
Beyond canonical SASP proteins, specific chemokines accumulate in aged plasma and directly impair tissue function. Eotaxin-1 (CCL11) is a chemokine traditionally associated with eosinophil recruitment in allergic responses. However, heterochronic parabiosis and plasma transfer studies identified CCL11 as a potent pro-aging systemic factor. Circulating levels of CCL11 increase progressively with age in both murine models and humans 922.
Systemic administration of CCL11 to young mice impairs adult neurogenesis and negatively impacts hippocampal-dependent learning and memory 9. In human observational cohorts, simultaneous increases in circulating CCL11 and other pro-aging chemokines like CCL2 in patients with Alzheimer's disease predict accelerated cognitive decline and inferior visual and verbal memory scores 16. The active removal or dilution of these circulating inhibitors serves as the primary theoretical mechanism for therapeutic apheresis interventions.
The Growth Differentiation Factor 11 Controversy
Few circulating factors have been subject to as much scientific scrutiny and debate as Growth Differentiation Factor 11 (GDF11). A member of the transforming growth factor-beta (TGF-β) superfamily, GDF11 shares 89% sequence homology with GDF8 (myostatin), a well-characterized inhibitor of skeletal muscle growth 12328.
In 2013 and 2014, foundational publications utilizing heterochronic parabiosis models reported that systemic GDF11 levels declined with age. Exogenous administration of recombinant GDF11 was claimed to reverse age-related cardiac hypertrophy, improve skeletal muscle structure and strength, and enhance neurogenesis and brain revascularization 2232425. These early studies categorized GDF11 as a premier systemic rejuvenating factor capable of mimicking the physiological benefits of young blood 525.
Subsequent rigorous investigations by independent laboratories and pharmaceutical groups failed to reproduce these geronic benefits and yielded diametrically opposed conclusions. Methodological evaluations revealed that the antibodies and assays utilized in the initial 2013 - 2014 studies exhibited high cross-reactivity with myostatin (GDF8), rendering the assertion that GDF11 specifically declined with age biologically unsubstantiated 423. High-resolution RNA-sequencing and GDF11-specific immunoassays subsequently demonstrated that GDF11 expression actually increases as a function of age in skeletal muscle and systemic circulation 426.
Mechanistic analyses clarify why elevated GDF11 acts as a pro-aging factor rather than a rejuvenator. GDF11 initiates cellular signaling by binding to activin type II receptors (ActRIIA/B) and recruiting type I receptors (ALK4/5/7). This receptor complex activation induces the phosphorylation of canonical SMAD2/3 transcription factors 2526. In skeletal muscle, the activation of the SMAD2/3 pathway by GDF11 promotes the transcription of E3 ubiquitin ligases, specifically Atrogin-1 and MuRF1. These ligases are the central mediators of ubiquitin-proteasome-dependent muscle proteolysis 26. Consequently, exposure to exogenous GDF11 inhibits satellite cell expansion, suppresses myoblast differentiation, and activates a pro-atrophic catabolic program, closely mimicking the deleterious effects of myostatin 282526.
Current clinical consensus aligns with these later findings. In human cohorts, circulating GDF11 concentrations are significantly higher in older adults compared to younger demographics and are independently associated with the clinical diagnosis of sarcopenia, physical frailty, and accelerated functional decline 26.
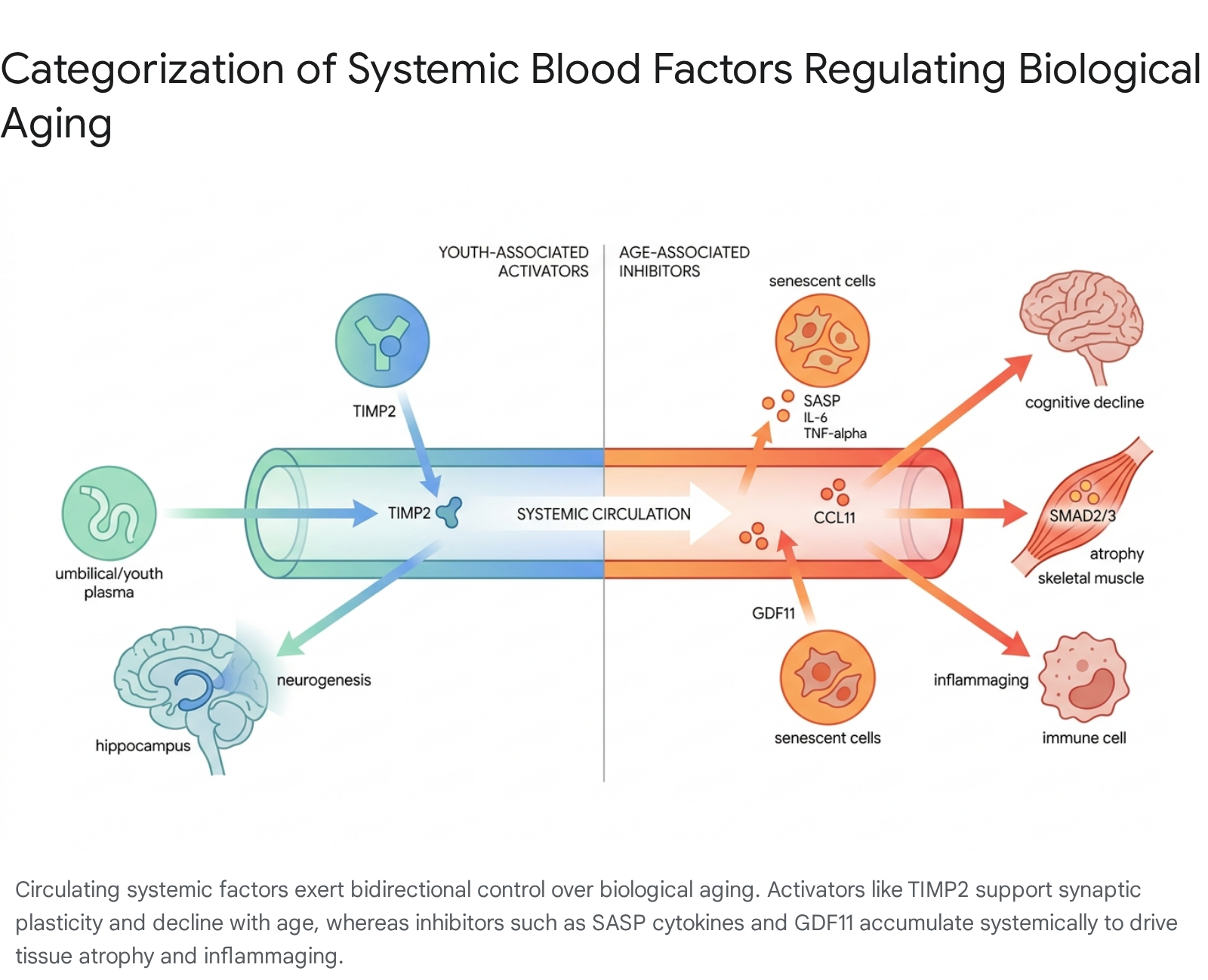
Summary of Systemic Blood Factors in Aging
| Factor Classification | Specific Molecule | Expression Trajectory in Aging | Physiological Effect | Clinical/Translational Impact |
|---|---|---|---|---|
| Activator / Rejuvenator | TIMP2 (Tissue Inhibitor of Metalloproteinases 2) | Declines | Regulates ECM; revitalizes adult hippocampal neurogenesis and synaptic plasticity. | Higher lifelong genetic proxy levels correlate with better episodic memory and global cognition 81516. |
| Inhibitor / Pro-Aging | GDF11 (Growth Differentiation Factor 11) | Increases | Activates SMAD2/3 signaling; upregulates ubiquitin ligases (Atrogin-1, MuRF1). | Induces skeletal muscle atrophy; elevated levels are strongly associated with clinical sarcopenia 2526. |
| Inhibitor / Pro-Aging | SASP (e.g., IL-6, TNF-α, IL-8) | Increases | Promotes persistent localized and systemic inflammation; induces cellular senescence in neighboring cells. | Drives "inflammaging," impedes stem cell repair, and facilitates tumor progression and immune evasion 181920. |
| Inhibitor / Pro-Aging | CCL11 (Eotaxin-1) | Increases | Impairs neurogenesis and neurovascular function. | Elevated systemic levels are predictive of rapid cognitive decline and memory impairment in neurodegenerative states 91622. |
Therapeutic Plasma Exchange as an Anti-Aging Intervention
Given that the systemic accumulation of inhibitors drives the aging phenotype, interventions capable of clearing these deleterious factors offer significant geronic utility. Therapeutic Plasma Exchange (TPE) is a well-established, FDA-approved extracorporeal blood purification technique utilized for over a century to manage severe autoimmune, neurological, and metabolic conditions, including Guillain-Barré syndrome, myasthenia gravis, and thrombotic microangiopathy 27282930.
Mechanisms of Plasma Dilution
During TPE, blood is continuously drawn from the patient via venous access and passed through an apheresis machine, which centrifugally or filtration-separates the plasma from cellular components (erythrocytes, leukocytes, and thrombocytes). The native, aged plasma - containing concentrated SASP factors, inflammatory cytokines, circulating autoantibodies, and pathogenic extracellular vesicles - is discarded. The patient's cellular components are then reconstituted with a sterile replacement fluid and returned to the systemic circulation 303132.
The replacement fluid utilized in TPE typically consists of a physiological saline solution mixed with 5% human serum albumin to maintain oncotic pressure and circulatory stability 632. In advanced longevity protocols, some regimens co-administer Intravenous Immunoglobulin (IVIG) to replace native immunoglobulins discarded during the procedure and to confer secondary immunomodulatory benefits 313233.
The anti-aging efficacy of TPE relies on a systemic biological "reset." The process does not simply remove toxic factors momentarily; it causes a massive, acute dilution of autoregulatory proteins that otherwise engage in sustained, deleterious crosstalk with multiple cellular signaling pathways. By diluting these factors, TPE breaks pathological feedback loops, leading to long-lasting changes in gene expression and the restoration of normal cellular communication pathways 66. Clinical multi-omic studies demonstrate that TPE actively restores key signaling pathways - including JAK-STAT, MAPK, TGF-β, NF-κB, and Toll-like receptors - toward youthful parameters. This systemic shift effectively reawakens dormant regenerative mechanisms and stem cell proliferation previously suppressed by the aged systemic milieu 37.
Albumin Redox State and Oxidative Stress
A critical variable in the TPE protocol is the utilization of human serum albumin as the primary replacement fluid. Albumin is the most abundant circulating protein in mammalian plasma, functioning not only to maintain fluid balance and transport ligands but also acting as the body's primary extracellular redox buffer 323435.
The redox buffering capacity of albumin is dictated by a single, free, and highly reactive cysteine residue at position 34 (Cys34). Depending on oxidative conditions, albumin exists in three distinct states: 1. Human Mercaptalbumin (HMA): The reduced, free thiol form. HMA acts as a potent free radical scavenger. In healthy, youthful populations, HMA constitutes 70 - 80% of circulating albumin 293436. 2. Human Nonmercaptalbumin-1 (HNA-1): The reversibly oxidized state, forming a mixed disulfide (often with cysteine or homocysteine). HNA-1 comprises 20 - 30% of total albumin 2934. 3. Human Nonmercaptalbumin-2 (HNA-2): The irreversibly oxidized form (sulfinic or sulfonic acid), reflecting severe, accumulated oxidative damage. HNA-2 normally constitutes 2 - 5% of circulating albumin 293436.
In aging and chronic inflammatory diseases (e.g., renal failure, advanced cirrhosis), the ratio shifts heavily toward the oxidized fractions (HNA-1 and HNA-2). Oxidized albumin ceases to function as a systemic protectant and can per se aggravate pathological conditions by failing to neutralize reactive oxygen and nitrogen species 343536.
Interestingly, while the macro-level dilution of aging factors during TPE induces deep systemic rejuvenation, the immediate infusion of commercial, pharmaceutical-grade albumin replacement fluids introduces a complex redox dynamic. Clinical studies tracking the albumin redox state (ARS) of patients undergoing repeated TPE sessions have demonstrated that commercial albumin solutions contain disproportionately high levels of oxidized fractions. Consequently, over a course of five TPE procedures, patient levels of irreversibly oxidized HNA-2 can increase significantly (from ~2.8% to 13.6%) and remain elevated for weeks post-procedure 293738.
Despite this acute alteration in the albumin redox profile, the overarching multi-omic rejuvenation achieved by TPE - via the bulk clearance of highly inflammatory SASP and pro-aging regulators - appears to overwhelmingly supersede the transient oxidative stress induced by the replacement fluid itself. The data implies that the therapeutic threshold for biological age reduction relies heavily on clearing targeted signaling inhibitors rather than simply restoring a chemically perfect systemic redox balance 66.
| Albumin Redox State | Molecular Characteristics | Abundance in Healthy Plasma | Implications in Aging and TPE |
|---|---|---|---|
| Human Mercaptalbumin (HMA) | Reduced, free thiol at Cys34. | 70 - 80% | Functions as primary extracellular antioxidant. Declines with age and chronic disease 2936. |
| Human Nonmercaptalbumin-1 (HNA-1) | Reversibly oxidized (mixed disulfide). | 20 - 30% | Represents moderate oxidative stress. Elevated in commercial albumin replacement fluids 2938. |
| Human Nonmercaptalbumin-2 (HNA-2) | Irreversibly oxidized (sulfinic/sulfonic acid). | 2 - 5% | Represents severe oxidative damage. Can spike from ~2.8% to 13.6% following a course of TPE 29. |
Clinical Evidence: The 2025 Multi-Omic Trials
The translational validity of TPE as an anti-aging modality was firmly established in 2025 through a landmark randomized, single-blind, placebo-controlled clinical trial published in the peer-reviewed journal Aging Cell. Conducted by Circulate Health in collaboration with the Buck Institute for Research on Aging, the study measured the rejuvenative potential of TPE in 44 healthy adults over the age of 50 3133394041.
To quantify the physiological impact, researchers employed advanced multi-omics profiling. This entailed measuring systemic changes across the epigenome, proteome, metabolome, and glycome, alongside assessing longitudinal shifts in immune cell composition 3740. Participants were randomized into several distinct treatment regimens: * Biweekly TPE + IVIG: Two TPE treatments per week for one week each month, repeated for three months, accompanied by intravenous immunoglobulin therapy. * Biweekly TPE (Alone): The same intensive schedule without the addition of IVIG. * Monthly TPE: One TPE session per month for six months. * Sham (Placebo): Simulated apheresis procedures mimicking the intervention schedules to maintain objectivity 3140.
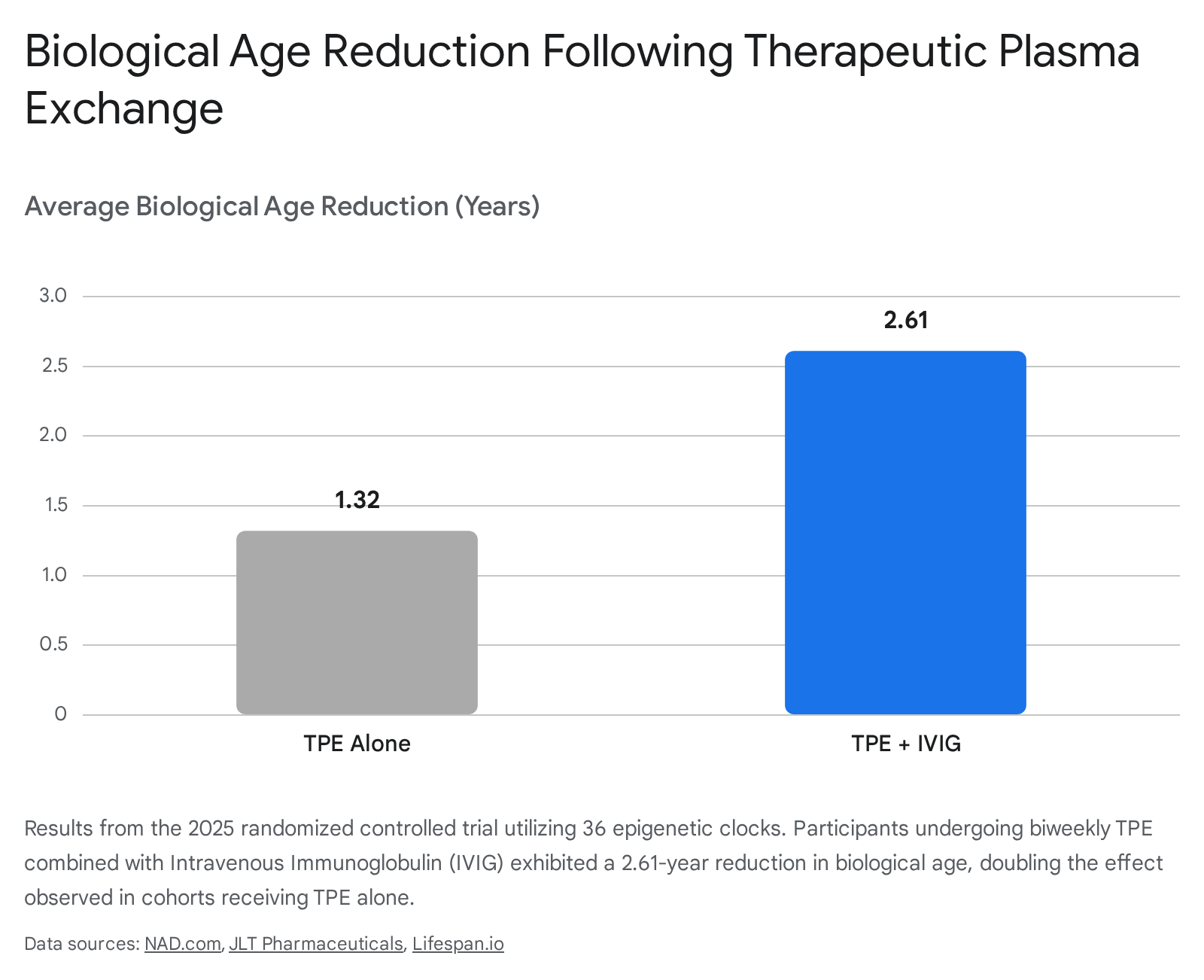
The results demonstrated a highly significant, coordinated molecular rejuvenation. Biological age was calculated utilizing an aggregation of 36 distinct epigenetic clocks - molecular algorithms that measure DNA methylation patterns (such as GrimAge, Horvath, and Hannum clocks) to accurately predict biological aging, mortality risk, and disease onset 313342.
Participants receiving the intensive biweekly TPE + IVIG protocol exhibited a profound average biological age reduction of 2.61 years compared to the placebo group. Those receiving TPE alone demonstrated a robust, though smaller, average reduction of 1.32 years 31333941.
The procedural safety profile was exceptional, with only two minor adverse events documented across all treatment arms (representing 0.45% of total procedures), and none necessitating study discontinuation due to the TPE process itself 33340.
Epigenetic and Immunological Rejuvenation
The multi-omic analysis of the 2025 Buck Institute trial provided granular insights into the exact physiological pathways reset by plasma exchange.
The most pronounced transformations occurred within the immune system, successfully reversing established hallmarks of immunosenescence. The TPE + IVIG protocol stimulated a marked proliferation in naïve CD4+ and CD8+ T cells - vital components for immune resilience that typically plummet with advanced age. Concurrently, the treatment induced a sharp decline in highly inflammatory monocyte populations and natural killer (NK) cells, shifting the immune topography back to a youthful state 3342.
Proteomic and metabolomic shifts corroborated the epigenetic data. TPE directly modulated proteins associated with chronic inflammation and cellular senescence, while generating a surge in complement system proteins critical for enhanced immune defense 3339. Predictive biomarker analysis indicated that baseline health status heavily influenced patient response; individuals entering the trial with markers indicating suboptimal metabolic health - such as higher circulating glucose, elevated bilirubin, higher liver enzymes, or lower naïve monocyte counts - achieved the steepest relative reductions in biological age compared to individuals in excellent baseline health 31333941.
Clinical Paradigms: Therapeutic Plasma Exchange Versus Young Plasma Exchange
As longevity medicine transitions from academic research to clinical practice, a clear delineation must be drawn between evidence-based apheresis modalities and highly commercialized, scientifically unproven interventions.
Young Plasma Exchange (YPE) - frequently marketed as "young blood transfusion" - involves the direct intravenous infusion of blood plasma harvested from young donors (typically aged 18 - 25) into older recipients. Inspired by the initial interpretations of the murine parabiosis experiments, YPE operates on the assumption that introducing exogenous youth factors is sufficient to override the aged recipient's systemic environment 484344.
Translating YPE from murine models to human clinical trials has yielded highly inconclusive results. A prominent 2019 randomized trial conducted at Stanford University investigating weekly infusions of young donor plasma in older adults with Alzheimer's disease failed to produce any statistically significant improvements in cognitive function or memory 48. Follow-up studies using fractionated young plasma products similarly failed to demonstrate robust, statistically powered anti-aging effects across validated epigenetic clocks, with leading researchers like Dr. Steve Horvath noting the minimal changes (e.g., a 0.82-year reduction on GrimAge) as clinically unremarkable 4844. Furthermore, the introduction of exogenous foreign plasma carries elevated risks of severe allergic reactions, transfusion-related acute lung injury (TRALI), and infection, prompting explicit warnings from the U.S. Food and Drug Administration (FDA) against clinics offering YPE for anti-aging purposes outside of regulated clinical trials 48.
In contrast, TPE replaces native plasma with a sterile, synthetic 5% albumin solution, relying entirely on the dilution of endogenous inhibitors rather than the introduction of foreign donor plasma. TPE is an established medical procedure with a 50-year safety record and has recently demonstrated verifiable, multi-omic biological age reversal in controlled human settings 34851.
| Feature | Therapeutic Plasma Exchange (TPE) | Young Plasma Exchange (YPE) |
|---|---|---|
| Primary Mechanism | Dilution and removal of accumulated pro-aging inhibitors and SASP factors. | Addition of exogenous youthful activators and circulating proteins from donors. |
| Replacement Fluid | Sterile, physiological saline combined with 5% human serum albumin. | Native plasma harvested from young human donors (ages 18 - 25). |
| Biological Age Efficacy | Proven: Averages up to 2.61 years of age reversal measured across 36 epigenetic clocks 3133. | Unproven: Mixed or negligible changes in human epigenetic age 4844. |
| Safety and Regulation | FDA-cleared procedure; decades of safety data across autoimmune and neurological indications 33943. | High risk of transfusion reactions; FDA has issued explicit warnings against off-label longevity use 48. |
| Translational Viability | Highly viable; standardizes cellular reset without donor dependency. | Low viability; relies on limited donor pools and introduces immunological variables. |
Future Directions in Systemic Rejuvenation
The revelation that systemic biological aging is largely driven by accumulating inhibitors rather than solely a deficiency of activators alters the trajectory of longevity research. Therapeutic Plasma Exchange stands as the most immediate, clinically validated intervention for systemic rejuvenation. By physically filtering out SASP factors, inflammatory cytokines, and pathogenic signaling molecules, TPE allows the endogenous cellular machinery to revert to a youthful state of repair and homeostasis.
Future optimization of this therapy will require investigating long-term treatment cadences to prevent the compensatory mechanisms that begin to dampen the anti-aging effects after continuous, repeated sessions 4142. Additionally, refining the commercial albumin solutions to minimize acute shifts in the Cys-34 redox state (HNA-2 accumulation) could potentially amplify the regenerative outcomes 2938. Ultimately, targeting specific components of the systemic milieu - whether by directly supplementing proven activators like TIMP2 or aggressively diluting inhibitors via TPE - represents the frontier of actionable interventions to reverse human biological age.