Chronic stress and immune system alteration
Introduction to Psychoneuroimmunology and Stress Transduction
Psychoneuroimmunology (PNI) represents an interdisciplinary synthesis of neuroscience, endocrinology, and immunology, fundamentally altering the biomedical understanding of human health. Historically, the immune system was conceptualized as a highly autonomous network of cells and organs functioning independently to identify and eliminate foreign pathogens 123. Contemporary PNI research has systematically dismantled this paradigm, demonstrating that psychological states, emotional regulation, and cognitive appraisals are intricately linked to physiological host defense mechanisms 134. The central premise of PNI is that the central nervous system (CNS) continuously monitors environmental threats and psychological stressors, translating these subjective experiences into objective neurochemical signals that modulate immune cell proliferation, cytokine secretion, and inflammatory baseline states 12.
The physiological consequences of stress are highly dependent on the chronicity and intensity of the stressor 45. Acute, short-term stress - defined as lasting from minutes to hours - typically acts as an immunoprotective and highly adaptive evolutionary mechanism. In the face of an immediate physical threat, acute stress enhances innate and adaptive immune responses by stimulating the rapid trafficking, maturation, and functional capacity of dendritic cells, neutrophils, macrophages, and lymphocytes 5. This transient mobilization prepares the organism for potential injury, accelerating wound healing and augmenting the initial response to infectious agents or vaccinations 5.
However, modern psychological stress is rarely transient. Chronic stress - arising from prolonged caregiving, occupational burnout, socioeconomic disadvantage, or social isolation - imposes an unrelenting allostatic load on the organism 346. This sustained neuroendocrine activation shifts the immune system from a state of acute readiness to one of chronic dysregulation. Prolonged exposure to stress hormones induces a state of systemic, low-grade inflammation while simultaneously suppressing protective cellular immunity 56. Over time, this dysregulation accelerates immunological aging, exacerbates pathological auto-immune responses, and provides a fertile pathophysiological foundation for numerous stress-associated morbidities, including cardiovascular disease, metabolic syndrome, and severe psychiatric conditions such as major depressive disorder 3478.
Cellular and Molecular Mechanisms of Stress Transduction
The biological transduction of psychological stress into immunological alteration is governed primarily by two major efferent pathways: the hypothalamic-pituitary-adrenal (HPA) axis and the sympathetic nervous system (SNS) 7910. The coordinated activation of these systems dictates the body's immediate and sustained physiological reactions to perceived threats.
The Hypothalamic-Pituitary-Adrenal Axis and Sympathetic Activation
Upon the cognitive appraisal of a stressor, the paraventricular nucleus of the hypothalamus secretes corticotropin-releasing hormone (CRH) 47. This hormone travels to the anterior pituitary gland, stimulating the release of adrenocorticotropic hormone (ACTH) into the systemic circulation. ACTH subsequently binds to receptors in the adrenal cortex, triggering the synthesis and secretion of glucocorticoids, predominantly cortisol in humans 478. Simultaneously, the SNS is activated, stimulating the adrenal medulla and peripheral sympathetic nerve terminals to release catecholamines, including epinephrine and norepinephrine 47.
The magnitude and consequence of the physiological stress response reflect both the systemic concentrations of these hormones and the localized sensitivity of target tissues. A comprehensive 2022 analysis utilizing single-cell RNA sequencing and human proteome data mapped the expression of stress hormone receptors across 55 different human organ systems 11. The findings revealed that glucocorticoid receptors (GR) are ubiquitously expressed across the body, but immune tissues and circulating immune cells express the highest absolute levels of GR RNA and protein 11. In contrast, alpha- and beta-adrenergic receptors (AR-α1B, AR-α2B, AR-β2, AR-β3) exhibit more specialized and localized expression patterns 11.
Because mounting a sustained immune and neuroendocrine response requires immense cellular energy, chronic stress places a profound metabolic burden on the organism. The 2022 mapping study confirmed this by identifying a strong positive association between the density of glucocorticoid and adrenergic receptors and the expression of key mitochondrial pathways, underscoring the severe energetic cost associated with prolonged stress transduction 11.
Circadian Regulation and Immune Cell Redistribution
The immune system does not operate at a static level; it is heavily influenced by circadian rhythms, which are directly orchestrated by the diurnal secretion patterns of cortisol and catecholamines 910. Under healthy homeostatic conditions, human T-cell counts fluctuate significantly, generally exhibiting a pronounced dip during the daytime and peaking at night 9.
Research has demonstrated that specific subpopulations of T-cells are differentially regulated by these neuroendocrine rhythms. Naive CD4+ and CD8+ T cells exhibit a distinct daytime nadir that negatively correlates with circulating cortisol levels. Elevated daytime cortisol upregulates the expression of the chemokine receptor CXCR4 on naive and central memory T cells, directing their redistribution out of peripheral blood and into the bone marrow 9. Conversely, terminally differentiated effector CD8+ T cells peak during the daytime, positively correlating with systemic epinephrine rhythms. Catecholamine signaling via β-adrenoceptors upregulates fractalkine receptors (CX3CR1) on these effector cells, promoting their rapid mobilization from the marginal pool into the active circulation 9.
Consequently, chronic stress fundamentally disrupts this precise circadian choreography. When chronic psychological stress blunts the diurnal cortisol slope or sustains high levels of catecholamines throughout the night, the organism maintains a persistent state of immunological hyper-vigilance 910. This circadian disruption favors immediate, generic effector defense mechanisms but severely diminishes the immune system's capacity to initiate highly specific adaptive immune responses to novel pathogens 9.
Stress-Induced Shifts in T-Cell Differentiation
CD4+ T helper (Th) cells play an indispensable role in coordinating diverse immune responses 1312. Following activation by antigen-presenting cells, naive CD4+ T cells differentiate into functionally distinct effector subsets - most notably Th1, Th2, Th17, and regulatory T cells (Treg) - depending precisely on the specific cytokine milieu and neuroendocrine signals present during their activation 121314.
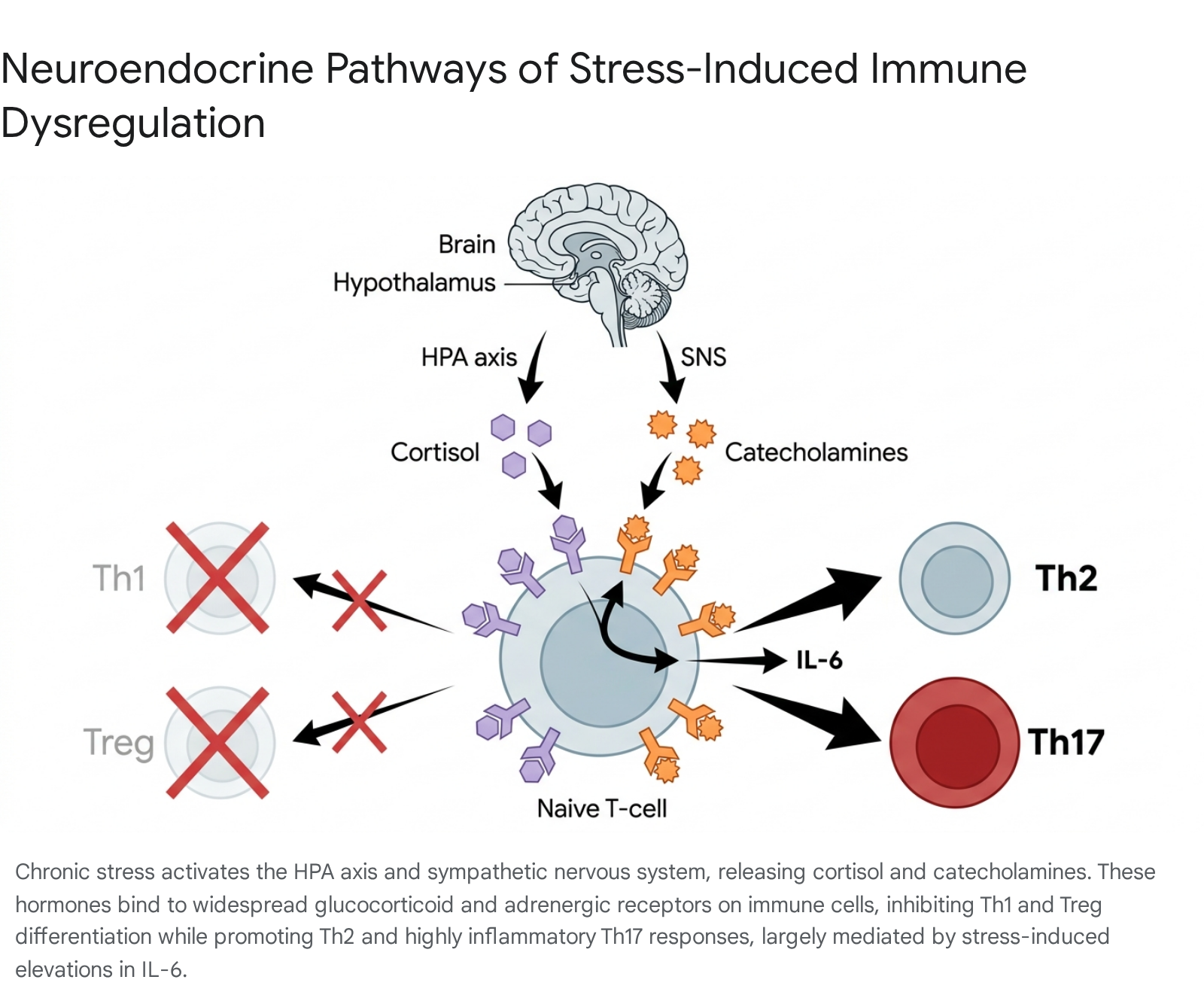
The Th1 and Th2 Paradigm
Th1 cells are essential for mediating cellular immunity against intracellular pathogens, such as viruses and certain bacteria, primarily by secreting pro-inflammatory cytokines like interferon-gamma (IFN-γ) and interleukin-2 (IL-2) 1313. In contrast, Th2 cells mediate humoral (antibody-driven) immunity against extracellular pathogens, allergens, and parasites, secreting cytokines such as IL-4, IL-5, and IL-13 1313.
Chronic stress profoundly alters the delicate balance between these two subsets, generally inducing a suppression of Th1 cellular immunity while preserving or actively enhancing Th2 humoral immunity 515. Elevated concentrations of catecholamines typically inhibit the production of Th1 cytokines (IFN-γ, TNF-α) through direct receptor interactions on the T-cell surface, while exerting less suppressive or even inductive effects on Th2 cytokines 515. This stress-induced "Th2 shift" increases susceptibility to viral infections while potentially exacerbating allergic and asthmatic responses 51314.
Interestingly, this catecholaminergic control can be pathologically impaired in pre-existing autoimmune conditions. Studies comparing healthy donors to patients with rheumatoid arthritis (RA) demonstrate that RA patients exhibit a significantly reduced inhibitory effect of catecholamines on IFN-γ production 15. In these individuals, the typical stress-induced shift toward a Th2 profile is abrogated due to defects in CD8+ T-cell regulatory mechanisms, allowing chronic stress to continuously fuel unmitigated Th1-driven joint inflammation 15.
Th17 and Regulatory T Cell Balance
Beyond the Th1/Th2 paradigm, PNI research has increasingly focused on the critical balance between Th17 cells and Treg cells. Th17 cells, which secrete IL-17A, IL-17F, and IL-22, are powerful drivers of neutrophil recruitment and tissue inflammation, playing a central role in autoimmune pathologies 131416. Conversely, Treg cells (defined by the transcription factor Foxp3) suppress aggressive immune responses, preventing autoimmunity and maintaining systemic immune homeostasis 1216.
Th17 and Treg cells share a common naive CD4+ precursor and both strictly require transforming growth factor-beta (TGF-β) for their initial differentiation 1214. The presence of interleukin-6 (IL-6) acts as the critical biological switch determining the ultimate cellular fate 1314. When chronic stress triggers the systemic elevation of IL-6, the cytokine synergizes with TGF-β to entirely abort the differentiation of immunosuppressive Treg cells 1314. Instead, it drives the overexpression of the transcription factor RORγt, pushing the naive T-cells to differentiate into highly pathogenic, pro-inflammatory Th17 cells 1216.
This pathological differentiation is further compounded by intracellular metabolic pathways. Chronic stress frequently elevates the mechanistic target of rapamycin (mTOR) signaling pathway, which controls lymphocyte proliferation. Hyperactive mTOR signaling directly promotes Th17 differentiation while inhibiting the stability and suppressive phenotype of Treg cells, cementing a chronic inflammatory state that exacerbates autoimmune rheumatic diseases and neuroinflammation 16.
Immunological Profiles of Chronic Stress Modalities
Psychological stress is a heterogeneous construct. Distinct chronotypes of chronic stress - ranging from caregiver burden and occupational burnout to profound socioeconomic strain - generate overlapping but distinctly measurable immunological signatures 191718.
Caregiver Burnout and Immunological Aging
Informal caregiving, specifically caring for family members with progressive conditions such as dementia or developmental disabilities like autism, represents one of the most thoroughly investigated models of chronic stress in PNI 191719. Caregiving imposes relentless emotional, physical, and financial demands that consistently disrupt physiological homeostasis, resulting in measurable acceleration of immunological aging 1719.
In highly stressed caregiving cohorts, cellular immunity exhibits profound structural alterations. Compared to age-matched, non-caregiving controls, caregivers display significantly higher percentages of CD4+ and CD8+ effector memory T cells, alongside markedly lower percentages of central memory T cells and naive CD8+ T cells 17. This restructuring creates a highly elevated Effector-to-Naive (E:N) cellular ratio, indicative of a depleted naive T-cell reservoir and a hyper-differentiated effector state 17. Caregivers also present with significantly elevated populations of KLRG1+ T cells 19. Because KLRG1 is a primary inhibitory receptor associated with terminal cellular senescence, its widespread expression functionally explains the impaired neutrophil phagocytosis, poor wound healing, and blunted vaccination responses traditionally documented in chronically stressed populations 1920.
Furthermore, caregiving stress generates unremitting, low-grade systemic inflammation. Longitudinal community studies evaluating older adults have demonstrated that the average rate of increase in circulating IL-6 over a six-year period is approximately four times steeper in spousal dementia caregivers compared to non-caregivers 2122. Crucially, this inflammatory momentum becomes biologically embedded; the steep annual escalation of IL-6 among former caregivers remains statistically indistinguishable from current caregivers even several years following the death of the impaired spouse 2123.
Caregiving Intensity Thresholds
The deterioration of psychological and physiological well-being in caregivers is not linear; it is characterized by specific "tipping points" related to caregiving intensity. Large-scale longitudinal analyses utilizing fixed-effects estimators have quantified the exact thresholds at which informal caregiving becomes definitively toxic to psychological health 2425.
When providing care to a spouse, there is no safe threshold; all levels of informal care intensity are associated with diminished well-being 2425. However, when caring for an elderly parent, the tipping point occurs at a mere 5 hours of care per week 2425. Conversely, when caring for a child with a disability or long-term illness, the threshold extends to 50 hours per week 2425. According to the 2025 Caregiver Intensity Index (CII), roughly 24% of informal caregivers in the United States operate "in the red" - a state of extremely high intensity associated with severe mental health deterioration, sleep disruption, and financial insecurity 2630. However, systemic support serves as a massive buffer; when caregivers report adequate structural, social, or workplace support, the percentage of individuals falling into this high-risk red zone drops to 14%, representing a 71% reduction in crisis risk 30.
Occupational Burnout and Socioeconomic Strain
Occupational stress and systemic socioeconomic status (SES) vulnerabilities generate potent biological embedding, acting as primary health determinants with effect sizes rivaling traditional physical risk factors 2728. The economic and physiological toll is immense; work-related stress is associated with billions in annual healthcare costs due to downstream metabolic and inflammatory disorders 27.
Chronic occupational strain - particularly jobs characterized by high psychological demands, low control, and working hours exceeding 55 hours weekly - increases the relative risk of developing depressive symptoms by up to 14%, a risk that is further amplified in individuals occupying lower socioeconomic positions 27. Physiologically, occupational burnout induces a state of glucocorticoid resistance. Overwhelmed by chronic cortisol exposure, immune cells downregulate their GR expression, rendering them impervious to cortisol's standard anti-inflammatory signaling 27. This results in chronically elevated baseline levels of pro-inflammatory cytokines such as IL-6, TNF-α, and C-reactive protein (CRP) 27.
Paradoxically, while baseline inflammation is dangerously elevated, the innate immune system's capacity to respond to novel, acute challenges becomes exhausted. Clinical studies subjecting physicians suffering from severe occupational burnout to the Trier Social Stress Test demonstrated that burned-out individuals exhibited an attenuated and blunted IL-6 reactivity response to the acute stressor compared to healthy matched controls 29. This diminished reactivity signals a deeply dysregulated innate immune response, leaving the individual highly vulnerable to cardiovascular events and opportunistic infections 29.
Lower SES acts as an independent, chronic psychosocial stressor that fundamentally alters immune trajectories from a young age 2830. Adolescents living in impoverished neighborhoods exhibit altered immune profiles, most notably expressing higher levels of stimulated Th2 (IL-5) and Th1 (IFN-γ) cytokines alongside chronically lowered morning cortisol 35. This physiological profile suggests that chronic financial strain, combined with a perceived lack of environmental control, reprograms neuroendocrine-immune setpoints during crucial developmental windows 2835. Furthermore, utilizing advanced "Elo-rating" systems to objectively rank SES, researchers have found that lower SES correlates directly with a higher lifetime pathogen burden, such as significantly increased cytomegalovirus (CMV) seropositivity, which itself drives lifelong systemic inflammation 3031.
Comparative Table of Chronic Stress Immunological Signatures
| Stressor Profile | Primary Neuroendocrine & Immune Signatures | Key Clinical / Cellular Findings |
|---|---|---|
| Caregiver Burden | Steeper IL-6 trajectory; Elevated E:N T-cell ratio. | 4x faster age-related increase in IL-6; High CD4+/CD8+ effector memory cells; Increased KLRG1+ senescence markers 171921. |
| Occupational Burnout | Glucocorticoid resistance; Attenuated acute IL-6 reactivity. | High baseline TNF-α and CRP; Blunted innate immune response to novel acute stressors; 2.5x higher depression risk 2729. |
| Low Socioeconomic Status | Altered Th1/Th2 stimulated profiles; Lowered morning cortisol. | Elevated stimulated IL-5 and IFN-γ; Higher lifetime pathogen burden (e.g., CMV seropositivity) driving baseline inflammation 28303531. |
| Social Isolation / Loneliness | Elevated suPAR (Isolation); Elevated IL-6 / reduced IGF-1 (Loneliness). | Isolation drives broad acute-phase reactants (CRP, Fibrinogen, WBC); suPAR reflects multi-decade biological embedding of isolation 323334. |
Social Disconnection and Systemic Inflammation
The psychological distress derived from social disconnection is increasingly recognized as a profound physiological threat. However, rigorous PNI research necessitates distinguishing between objective social isolation (living alone, lacking a quantifiable social network) and subjective loneliness (the perceived, emotional discrepancy between desired and actual social connections), as they operate through divergent immunological pathways 1033.
Divergent Pathways of Isolation and Loneliness
Longitudinal analyses utilizing data from the English Longitudinal Study of Ageing (ELSA) provide compelling evidence that objective social isolation triggers widespread, acute-phase systemic inflammation. Individuals who live alone or lack social engagement consistently present with elevated levels of CRP, fibrinogen, and elevated white blood cell (WBC) counts 3233. Crucially, these associations remain highly statistically significant even after rigorous adjustments for time-invariant factors (genetics, gender, baseline social class) and time-varying confounders (income fluctuations, physical health changes, and clinical depression) 3233.
Subjective loneliness, while intrinsically linked to isolation, produces a distinct biological signature. Chronic loneliness is primarily associated with the dysregulation of anti-inflammatory feedback loops rather than broad acute-phase activation 3233. Lonely individuals exhibit significantly reduced levels of insulin-like growth factor-1 (IGF-1), an anti-inflammatory peptide, alongside selective, chronic elevations in IL-6, independent of their objective living status 1032.
The Role of suPAR in Biological Embedding
Traditional inflammatory markers like CRP and IL-6 are highly volatile, frequently fluctuating in response to acute minor infections or circadian changes. To better capture the long-term biological embedding of social stress, researchers have recently identified soluble urokinase plasminogen activator receptor (suPAR) as a superior, highly stable biomarker for chronic inflammation 34.
Multi-cohort investigations encompassing over 8,400 participants tracked the inflammatory consequences of social isolation from early childhood into mid-adulthood 34. The findings demonstrated that individuals experiencing objective social isolation possessed suPAR levels roughly 24% higher than non-isolated individuals 34. Strikingly, while acute markers like CRP and IL-6 normalized after rigorous statistical adjustments for lifestyle factors (smoking, BMI), suPAR remained significantly elevated 34. This indicates that social isolation triggers an insidious, deep-rooted dysregulation of immune function that becomes biologically embedded over decades, physically confirming why long-term social disconnection carries mortality risks comparable to smoking and clinical obesity 34.
Neuroinflammation and Psychiatric Pathologies
Historically, clinical psychiatry conceptualized major depressive disorder (MDD) almost exclusively as a neurochemical imbalance confined strictly within the central nervous system. Modern PNI research has fundamentally shattered this localized paradigm, demonstrating that peripheral, systemic chronic inflammation penetrates the blood-brain barrier, initiating neuroinflammation that radically alters neurotransmitter synthesis, synaptic plasticity, and mood-regulating neurocircuitry 835.
Microglial Activation and the NLRP3 Inflammasome
Chronic peripheral stress disrupts CNS homeostasis primarily by activating microglia and astrocytes, the brain's resident innate immune cells 8363738. A major technological breakthrough in 2025 utilized advanced single-cell RNA sequencing to map gene expression across thousands of cells from post-mortem brain tissue of depressed patients 39. The study confirmed that clinical depression induces measurable, physical transcriptomic changes at the single-cell level, specifically identifying profound pathological alterations in prefrontal cortex microglia and excitatory neurons involved in stress regulation 3839.
The primary molecular engine driving this neuroinflammatory cascade is the NLRP3 inflammasome, a multiprotein intracellular complex 836. Chronic psychological stress increases the circulation of danger-associated molecular patterns (DAMPs) and extracellular ATP in the brain 836. These molecules bind to P2X7 receptors on microglial cell surfaces, triggering a cascade that oligomerizes the NLRP3 sensor, the ASC adaptor protein, and pro-caspase-1 836. The subsequent conversion to active caspase-1 allows the complex to cleave pro-IL-1β and pro-IL-18 into their mature, highly inflammatory forms 836.
The sustained secretion of IL-1β within stress-sensitive brain regions, such as the hippocampus and prefrontal cortex, actively promotes excitotoxicity, impairs synaptic plasticity, and drives the behavioral symptoms of depression 837. Notably, rodent models exploring post-stroke depression (PSD) have demonstrated that subjects exhibiting specific "depression resilience" possess a unique ability to suppress this pathway. In stroke-resilient mice, microglial reactivity is significantly reduced, accompanied by the active inhibition of NLRP3 and NF-κB signaling, suggesting that inflammasome blockade is a fundamental mechanism of neurological resilience 37.
The Inflammatory Subtype of Major Depressive Disorder
Driven by these neurobiological discoveries, pioneers in immunopsychiatry have accumulated compelling evidence supporting the formal recognition of an "inflammatory subtype" of major depression 354041. It is currently estimated that this biologically distinct subtype accounts for approximately 25% to 30% of all patients diagnosed with MDD 40.
The inflammatory subtype is clinically defined by consistently elevated peripheral inflammatory markers - most commonly utilizing a clinical cutoff of C-reactive protein (CRP) > 2.0 mg/L or > 3.0 mg/L - alongside elevated concentrations of TNF and IL-6 in both blood and cerebrospinal fluid 404142. Patients presenting with this inflammatory profile exhibit a distinct clinical phenotype characterized by a severe neurovegetative symptom cluster often termed "sickness behavior." This presentation is dominated by profound anhedonia, severe psychomotor slowing, chronic fatigue, and notable disruptions in sleep and appetite architecture 354041.
Crucially, patients harboring the inflammatory subtype of depression respond poorly to standard psychiatric interventions 404143. Traditional antidepressants, such as selective serotonin reuptake inhibitors (SSRIs), are frequently ineffective because they fail to target the underlying, upstream immune dysregulation driving the neurotransmitter depletion 4041. However, these patients demonstrate superior response rates to alternative therapies, including ketamine administration, electroconvulsive therapy (ECT), and emerging pharmacological anti-inflammatory protocols 4041.
Efficacy of Anti-Inflammatory Clinical Interventions
The recognition of inflammatory depression has spurred intense clinical investigation into repurposing immunomodulatory drugs for psychiatric use. Previous early-stage trials evaluating anti-inflammatory treatments (such as infliximab, minocycline, and tocilizumab) for MDD yielded highly mixed and often disappointing results 434445. PNI researchers hypothesized that these trials failed because they recruited highly heterogeneous depressive populations without screening for baseline systemic inflammation 4345. Administering an anti-inflammatory agent to a depressed patient without underlying inflammation provides no target for the drug's mechanism of action.
To address this, a comprehensive, pre-registered 2025 meta-analysis evaluated data exclusively from randomized controlled trials (RCTs) that strictly utilized an established clinical cutoff for baseline elevated inflammation (CRP ≥ 2 mg/L) prior to treatment 434647. The results were revelatory: in patients with confirmed inflammatory profiles, anti-inflammatory treatments significantly reduced specific neurovegetative symptoms compared to placebos. The interventions produced a moderate clinical effect size in reducing anhedonia (Hedges' g = 0.40) and overall depressive symptom severity (Hedges' g = 0.35) 434647.
However, it is vital to express calibrated uncertainty regarding the current clinical ceiling of these treatments. While targeted symptom reduction was significant, the overall treatment response rates and total clinical remission rates did not achieve statistical significance compared to placebo controls 434647. This indicates that while anti-inflammatory therapies are safe and effective at alleviating the severe neurovegetative burden (anhedonia, fatigue) of inflammatory depression, they may need to be deployed as adjuncts rather than monotherapies to achieve full clinical remission 434446.
| Clinical Metric | Effect Size (Hedges' g) | 95% Confidence Interval | Clinical Significance |
|---|---|---|---|
| Anhedonia Reduction | 0.40 | 0.08 to 0.71 | Statistically significant moderate reduction in the inability to feel pleasure 434647. |
| Depressive Symptom Severity | 0.35 | 0.05 to 0.64 | Statistically significant moderate reduction in overall symptom burden 434647. |
| Total Remission Rates | N/A (Relative Risk = 1.18) | 0.71 to 1.95 (RR) | Not statistically significant compared to placebo (confidence interval crosses 1.0) 434647. |
Global Perspectives: Stress and Immunity in Non-WEIRD Populations
A historical and methodological limitation of psychoneuroimmunology has been its overwhelming reliance on study cohorts drawn from Western, Educated, Industrialized, Rich, and Democratic (WEIRD) populations. Recent global initiatives are rapidly expanding the field, demonstrating that diverse genetic ancestries, regional infectious disease burdens, and cultural expressions of distress profoundly modulate stress immunology 304849.
Biomarker Trajectories in Latin American Cohorts
Populations with admixed ancestries exhibit distinct physiological baseline immune functions and unique responses to environmental stressors. Massive longitudinal biobanks, such as the Mexico City Prospective Study (MCPS) and the OriGen initiative, have begun tracking metabolic and psychoneuroimmunological data across hundreds of thousands of Latin American participants 485051.
Within these vast cohorts, higher scores on the Perceived Stress Scale (PSS) show robust, independent correlations with elevated CRP, IL-6, and TNF-α, persisting even after stringent adjustments for major confounding variables like BMI, sex, and baseline clinical depression 50. Furthermore, specific ethnic variations modulate how this inflammation translates into physical decline. Studies evaluating physical performance measures (such as gait speed and balance) have found that Mexican American cohorts exhibit a significantly more direct and severe inverse relationship with IL-6 and TNF-α levels compared to age-matched Non-Hispanic White cohorts 52. This suggests that the functional, physical consequences of stress-induced inflammation may manifest earlier or more aggressively in certain demographic groups 52.
Oncology data similarly reveals complex intersections between ethnicity, chronic stress, and the localized immune environment. Analyses of Triple-Negative Breast Cancer (TNBC) tissue samples demonstrate that African American women frequently present with significantly higher quantities of tumor-infiltrating lymphocytes (sTILs) and broader inflammatory gene expression than clinically matched non-African American women 49. Paradoxically, despite this demonstrably robust immune infiltration, African American women experience significantly higher recurrence rates and worse overall survival 49. This indicates that chronic socioeconomic stressors and systemic health disparities may rapidly exhaust the local immune response, ultimately inducing highly immunosuppressive regulatory phenotypes within the tumor microenvironment that permit cancer progression despite initial immune recognition 49.
Sub-Saharan Africa and Cultural Manifestations of Trauma
In Sub-Saharan Africa, regional hardships related to economic instability, shifting agricultural practices, and infectious disease density impose a unique chronic stress burden. To quantify this, researchers utilize hair cortisol analysis - a powerful biomarker that captures cumulative HPA axis activity over months, circumventing the daily volatility of blood or saliva tests 53. Studies conducted in Kenyan settlement communities adjacent to large floriculture farms revealed that hair cortisol concentrations averaged an astonishing 639 ng/g 53. This is more than double the concentration found in standard Caucasian reference groups (299 ng/g), providing objective biological confirmation that the severe socio-economic vulnerabilities prevalent in the region generate profound, chronic physiological stress 53.
Furthermore, the psychological appraisal and cultural interpretation of stress fundamentally alter clinical manifestations, challenging the global applicability of Western psychiatric diagnostics like the DSM-5. In many Sub-Saharan contexts, severe psychological trauma is not primarily conceptualized as an internalized psychiatric failure. Instead, distress is frequently expressed through severe somatic ailments - such as unexplainable rheumatism, prolonged fevers, or rapid muscular atrophy - or attributed to external spiritual causes, curses, and witchcraft 54. If allopathic physicians fail to identify a biological pathogen for these stress-induced somatic symptoms, patients frequently turn to traditional healers or religious interventions 54. This highlights a vital consideration for the future of global PNI: the cultural and psychological framing of a stressor - whether internalized as a mental disorder or externalized as a spiritual attack - directly dictates the coping mechanisms deployed, which in turn modulates the specific neuroendocrine cascades and resultant immunological profiles 5455.
Methodological Challenges and Future Directions
Despite robust and highly replicated findings, the field of psychoneuroimmunology continues to grapple with significant methodological challenges. The primary debates center on the high temporal variability of immune biomarkers and the profound difficulty of isolating direct psychobiological stress pathways from secondary behavioral coping mechanisms.
Isolating Psychological Stress from Health Behaviors
A persistent and critical debate within modern PNI is whether chronic stress directly alters immunity via internal neuroendocrine pathways (the direct biological effect), or whether psychological stress merely drives individuals toward maladaptive health behaviors - such as poor diet, sleep deprivation, smoking, alcohol consumption, and sedentary lifestyles - that subsequently trigger systemic inflammation (the indirect behavioral effect) 42856.
Extensive population-level data indicates that both pathways are highly active, but behavioral mediators play a larger role than previously estimated in early PNI models. For instance, in a massive analysis utilizing data from 293,864 participants in the UK Biobank, researchers found that individuals experiencing high levels of social isolation faced a 39% greater relative risk of developing incident chronic obstructive pulmonary disease (COPD) compared to non-isolated peers 57. However, rigorous cause-mediation analyses revealed that current smoking status, unhealthy dietary patterns, elevated BMI, and physical inactivity collectively accounted for between 32.8% and 46.1% of the association between isolation/loneliness and the onset of the disease 57. Similarly, in evaluating mental health in post-pandemic cohorts, poor sleep duration and diminished physical activity were found to mediate up to 14.7% of the positive physiological correlation between clinical depression and clinical obesity 6358.
To adequately control for these profound behavioral confounds, rigorous contemporary PNI studies employ advanced fixed-effects modeling and exhaustive time-varying covariates. Importantly, even when statistical models stringently adjust for baseline BMI, age, race, pharmaceutical interventions (e.g., statins, antidepressants), smoking status, and preexisting health conditions, the independent, direct effect of chronic psychological stress on biomarkers like IL-6, suPAR, and T-cell effector ratios remains highly statistically significant 1732344158. This conclusively validates the presence and potency of direct neuro-immune signaling pathways, proving that stress damages the immune system independently of lifestyle degradation 3334.
Advancements in Multi-Omics and Tissue-Agnostic Biomarkers
A major limitation of early PNI research was the reliance on single-protein, cross-sectional blood assays - such as taking an isolated measurement of CRP or IL-6 at a single point in time. The immune system is incredibly dynamic; cytokine half-lives are short, and systemic levels fluctuate rapidly based on minor acute infections, fasting states, and time of day 959.
To overcome the noise inherent in single-protein measurements, leading PNI scientists are forcefully advocating for the widespread integration of exploratory, hypothesis-generating multi-omics research 59. By transitioning away from isolated ELISA tests and leveraging cross-sectional whole-exome sequencing, comprehensive RNA sequencing, and broad proteomics, researchers can identify stable, network-level genomic changes rather than transient cytokine spikes 5960.
The identification of pan-cancer, tissue-agnostic biomarkers is a primary example of this technological evolution. By combining massive datasets measuring tumor mutational burden with localized T-cell infiltration scores and systemic transcriptomic profiles, researchers are moving toward a future where immunotherapies and psychiatric interventions can be personalized. Ultimately, treatments will be tailored based not only on the biological architecture of a disease but on the distinct neuro-immunological stress resilience profile of the individual patient 496061.