Cellular Senescence Mechanisms and Senotherapeutics
The Epistemological Shift: Deconstructing the "Zombie Cell" Misconception
The advent of modern geroscience has fundamentally shifted the biomedical understanding of aging, transitioning from a model of inevitable thermodynamic decay to one of programmed, manipulable biological signaling. At the core of this paradigm is cellular senescence, a state of stable, essentially irreversible cell-cycle arrest induced by a multitude of endogenous and exogenous stressors, including telomere attrition, oxidative stress, oncogene activation, and epigenetic dysregulation 123. In popular scientific discourse, and occasionally in reductionist clinical literature, senescent cells are frequently - and erroneously - characterized as "zombie cells." This pervasive moniker implies a state of metabolic death, biological dormancy, or passive existence. In biological reality, the exact opposite is true. The "zombie cell" conceptualization fails completely to capture the intense, hyper-metabolic nature of senescence 4.
Senescent cells are highly active, demanding engines of biosynthesis. While they have permanently exited the cell cycle and no longer divide, they undergo significant morphological hypertrophy, expand their lysosomal and mitochondrial networks, and drastically upregulate the transcription and translation of a vast array of bioactive molecules 45. Rather than decaying passively, these cells consume substantial cellular energy to synthesize and excrete the Senescence-Associated Secretory Phenotype (SASP) - a potent, complex cocktail of pro-inflammatory cytokines, chemokines, matrix metalloproteinases (MMPs), and growth factors 46. By remaining metabolically hyperactive and continuously engaging the mechanistic target of rapamycin (mTOR) pathway, senescent cells act as pathological signaling hubs 6. They exert profound paracrine and endocrine effects that degrade surrounding tissue architecture, exhaust stem cell niches, and drive the systemic, low-grade chronic inflammation known as "inflammaging" 478. Thus, targeting cellular senescence therapeutically is not a matter of clearing "dead" cellular debris, but rather identifying and dismantling active, rogue biological factories that are actively driving age-related tissue dysfunction across the organism's lifespan.
Comparative Analysis of Fundamental Cellular States
To fully comprehend the pathology of cellular senescence, it is absolutely necessary to structurally and metabolically delineate it from other fundamental cellular states. The assumption that an arrested cell is a senescent cell overlooks the highly specific biological programming that differentiates senescence from quiescence or apoptosis. Table 1 provides a definitive structural comparison of senescent cells against apoptotic, quiescent, and healthy proliferating cells, highlighting the unique biochemical, morphological, and secretory signatures of the senescent phenotype.
Table 1: Biological and Metabolic Characteristics of Primary Cellular States
| Characteristic | Healthy Proliferating Cell | Quiescent Cell ($G_0$ Phase) | Apoptotic Cell | Senescent Cell |
|---|---|---|---|---|
| Cell Cycle Status | Active progression through $G_1$, S, $G_2$, and M phases | Reversibly arrested in the $G_0$ resting phase | Terminal, programmed cell death execution | Irreversibly arrested, typically at the $G_1$ or $G_2/M$ boundary |
| Metabolic Activity | High (focused on DNA replication and cytokinesis) | Low (focused on basal cellular maintenance) | Rapidly declining to absolute zero | Exceptionally High (hyper-metabolic, continuous SASP synthesis) |
| Morphological Presentation | Normal, tissue-specific size and architecture | Small, condensed, with minimal cytoplasm | Shrinking, membrane blebbing, nuclear fragmentation | Enlarged, flattened, highly vacuolized, hypertrophic |
| Secretory Profile | Homeostatic, highly context-dependent | Minimal to zero external signaling | "Find-me/Eat-me" signals (e.g., extracellular ATP, fractalkine) | SASP (highly pro-inflammatory, tissue-remodeling, profibrotic) |
| Lysosomal Mass | Normal physiological baseline | Normal physiological baseline | Degraded, permeabilized, or ruptured | Massively expanded (exhibiting high $SA-\beta-gal$ activity) |
| Resistance to Cell Death | Normal (regulated by standard survival signals) | Normal baseline resistance | Actively undergoing organized destruction | Highly resistant (massive upregulation of SCAP survival networks) |
| DNA Damage Status | Transient (repaired efficiently during the cell cycle) | Minimal | Purposeful, organized internucleosomal DNA fragmentation | Persistent (unresolved $\gamma H2AX$ foci, telomeric dysfunction) |
The Mechanistic Pathways of Senescence Initiation and Maintenance
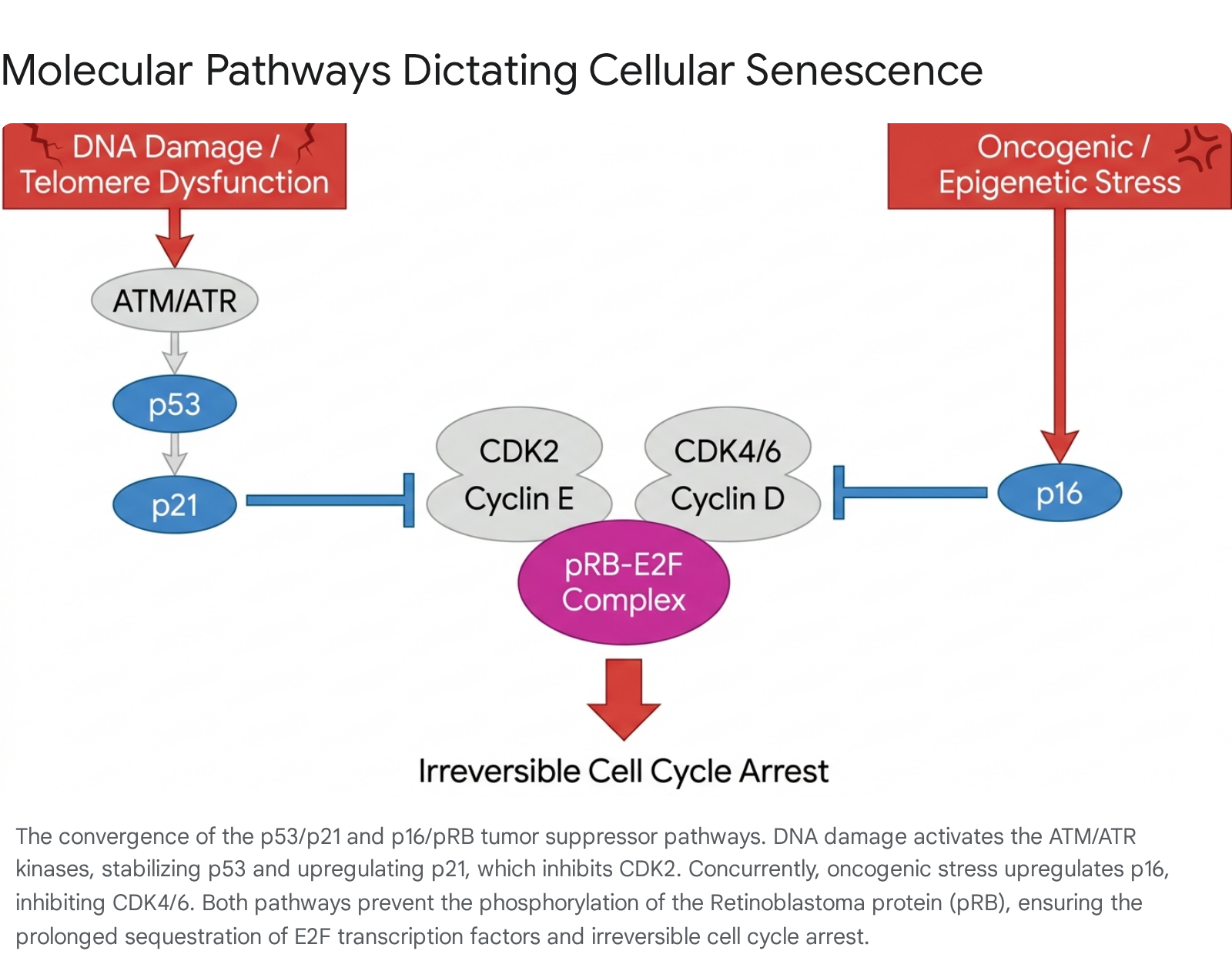
The induction, establishment, and maintenance of cellular senescence are governed by two primary, overlapping tumor-suppressor networks: the $p53/p21^{CIP1/WAF1}$ pathway and the $p16^{INK4a}/pRB$ pathway. These pathways act as biological circuit breakers, decisively halting cellular proliferation when a cell experiences critical genomic, oncogenic, or metabolic stress 910. Understanding the structural breakdown of these pathways is vital for recognizing how senescence is initiated versus how it is chronically sustained.
The DNA Damage Response and the $p53/p21$ Axis
The $p53/p21$ axis is primarily engaged in response to severe genotoxic stress, such as telomere shortening, double-strand DNA breaks, ionizing radiation, or severe oxidative DNA damage. When structural DNA damage occurs, sensor kinases, specifically Ataxia Telangiectasia Mutated (ATM) and Ataxia Telangiectasia and Rad3-related (ATR), are immediately recruited to the sites of DNA damage, often marked by the phosphorylation of histone H2AX ($\gamma H2AX$) 11112. These primary kinases initiate a downstream phosphorylation cascade that stabilizes and activates the master transcription factor $p53$.
Once activated, $p53$ upregulates the transcription of the $CDKN1A$ gene, which encodes the potent cyclin-dependent kinase (CDK) inhibitor $p21^{CIP1/WAF1}$ 912. The $p21$ protein is highly promiscuous; it binds to and inhibits multiple CDK complexes, most notably the CDK2/Cyclin E complex. Under normal, healthy proliferative conditions, CDK2 phosphorylates the Retinoblastoma protein ($pRB$), inactivating it to allow cell cycle progression. However, in the presence of high intracellular $p21$, $pRB$ remains in a hypophosphorylated, highly active state. Hypophosphorylated $pRB$ tightly binds and sequesters the E2F family of transcription factors, preventing the transcription of critical genes required for the transition from the $G_1$ phase to the S phase of the cell cycle, thereby enforcing immediate cell cycle arrest 910. It has been extensively reported that $p21$ serves as the primary marker for the initiation of cellular senescence 91314.
Oncogene-Induced Senescence and the $p16^{INK4a}/pRB$ Axis
While the $p53/p21$ pathway is the acute responder that initiates senescence following DNA damage, the $p16^{INK4a}$ pathway is the mechanism crucial for its irreversible maintenance, particularly in response to oncogenic stress (Oncogene-Induced Senescence, OIS) or cumulative epigenetic de-repression during chronological aging. The $CDKN2A$ genetic locus encodes $p16^{INK4a}$, which serves as a highly specific inhibitor of CDK4 and CDK6 910.
When $p16^{INK4a}$ levels rise due to stress signaling or epigenetic alterations, the protein competitively binds to CDK4 and CDK6, displacing Cyclin D. This competitive inhibition prevents the CDK4/6-Cyclin D complex from performing the initial, necessary phosphorylations of $pRB$. Consequently, $pRB$ remains completely unphosphorylated and firmly bound to E2F. The continuous, unyielding expression of $p16^{INK4a}$ establishes a rigid, durable state of growth arrest that is overwhelmingly resistant to mitogenic stimulation or growth factor signaling. Over time, senescence initially induced by $p21$ often transitions to a $p16^{INK4a}$-dominated state, a biological handoff that fundamentally cements the permanent nature of the senescence phenotype, making the cell essentially blind to proliferative cues 913.
The Biomarker Conundrum: ICSA Consensus and Multi-Marker Panels
Historically, geroscience research and early clinical evaluations have heavily relied on the expression of $p16^{INK4a}$ as the definitive, solitary proxy for quantifying cellular senescence. However, this over-reliance has revealed critical methodological limitations that actively complicate both basic tissue research and the establishment of reliable clinical trial endpoints 915.
The Severe Limitations of Relying Solely on $p16^{INK4a}$
The fundamental flaw in utilizing $p16^{INK4a}$ as a standalone diagnostic marker is that its expression is not strictly exclusive to the state of cellular senescence. Emerging transcriptomic and proteomic evidence decisively demonstrates that $p16^{INK4a}$ can be highly expressed in entirely non-senescent physiological states. For instance, during the physiological activation and polarization of specific immune cells - most notably macrophages - $p16^{INK4a}$ is dramatically upregulated independently of any senescence induction or cell cycle arrest 917. Consequently, relying on $p16^{INK4a}$ alone in highly inflammatory tissue microenvironments, such as atherosclerotic plaques or obese visceral adipose tissue, will inevitably yield massive false positives, mistaking active immune infiltration for senescent cell burden.
Conversely, relying solely on $p16^{INK4a}$ can also produce false negatives. In certain types of stress-induced premature senescence, particularly where the $p53/p21$ pathway is the dominant, enduring driver, $p16^{INK4a}$ expression may remain relatively low despite irreversible cell cycle arrest. Similarly, $p21^{CIP1/WAF1}$ expression carries its own diagnostic vulnerabilities; it is actively modulated by the circadian clock and transient DNA damage responses, meaning its temporary elevation does not guarantee a permanent, stable senescent state 9. Furthermore, genetic testing complexities arise regarding transcript variants; clinical trials must differentiate between $p16$ variant 5 expression in T-cells versus combined variant 1+5 expression to accurately predict skeletal and metabolic responses to senolytic therapies 16.
The 2024/2025 ICSA Consensus Guidelines
To permanently resolve these analytical bottlenecks and establish standardized rigor, the International Cell Senescence Association (ICSA) updated its consensus guidelines, explicitly and forcefully advocating against single-marker reliance 35131517. The 2024/2025 ICSA consensus mandates a highly rigorous, multi-marker screening workflow to accurately define senescence both in vitro and in vivo. The ICSA algorithm recommends a required triad of overlapping phenotypic and molecular markers:
- Cell-Cycle Arrest Markers: The verification of durable, irreversible arrest requires the combined evaluation of $p16^{INK4a}$ (indicating senescence sustenance) and $p21^{CIP1/WAF1}$ (indicating senescence initiation), explicitly coupled with an absence of proliferative markers such as Ki-67 or lamin B1 degradation 1314.
- Lysosomal Mass and Alterations ($SA-\beta-gal$): The application of Senescence-Associated $\beta$-Galactosidase ($SA-\beta-gal$) staining remains a core recommendation. $SA-\beta-gal$ measures the enzymatic activity of lysosomal $\beta$-D-galactosidase, which becomes vastly overexpressed within the hyper-expanded, dysfunctional lysosomal compartments of senescent cells. Crucially, this activity is exclusively detectable at a suboptimal, acidic pH of 6.0 91314. However, researchers must account for false positives in cell types with naturally high lysosomal mass, such as osteoclasts and mature neurons 9.
- Lipofuscin Accumulation (The Archival Solution): Lipofuscin is a highly cross-linked, undegradable biochemical aggregate of oxidized proteins, lipids, and trace metals that inexorably accumulates in the aging lysosome 131819. The ICSA now heavily prioritizes lipofuscin detection due to recent methodological breakthroughs that solve historical tissue analysis problems.
Advanced Detection: Sudan Black B, SenTraGor, and In Vivo Probes
A significant diagnostic advancement aligning closely with the ICSA recommendations is the refined use of Sudan Black B (SBB) and its advanced, biotinylated, lipophilic chemical analog, SenTraGor (also designated as GL13 or GLF16). While $SA-\beta-gal$ has served as the traditional "gold standard" for decades, it suffers from a fatal methodological flaw in clinical pathology: the enzymatic assay requires fresh or snap-frozen tissue and is entirely inapplicable to archival material 19.
SenTraGor specifically and robustly reacts with lipofuscin granules. This allows for highly sensitive, antibody-enhanced detection of senescent cells in practically any biological material, including decades-old formalin-fixed, paraffin-embedded (FFPE) clinical tissue biopsies 31920. This chemical analog bridges the gap between basic geroscience and retrospective human epidemiological studies. Furthermore, to translate longitudinal senescence tracking to human clinical trials without requiring repeated invasive biopsies, researchers are validating non-invasive in vivo molecular probes. Prominent examples include NBGal, a highly specific $\beta$-galactosidase-activatable near-infrared (NIR) senoprobe, and advanced PET imaging agents like $[^{18}F]$-PyGal, which allow for real-time, non-invasive spatial mapping of the senescent burden in living mammalian organisms 917.
The Senescence-Associated Secretory Phenotype (SASP) and Tissue Damage
If irreversible cell cycle arrest is the defining internal feature of senescence, the Senescence-Associated Secretory Phenotype (SASP) is its defining external weapon. The SASP fundamentally transforms a locally arrested cell from a benign bystander into a systemic driver of profound tissue pathology 82321.
The continuous synthesis and secretion of the SASP is largely governed by a network of stress-responsive transcription factors, including NF-$\kappa$B, C/EBP$\beta$, and p38 MAPK 211. Additionally, the cGAS-STING pathway plays a critical role; it detects cytosolic DNA fragments leaking from the degraded, unstable nuclei of senescent cells, triggering robust type I interferon responses and supercharging the inflammatory secretome 211. The SASP is intrinsically highly heterogenous; its exact biochemical composition is dictated by the cell type of origin and the exact nature of the senescence-inducing stress (e.g., Oncogene-induced senescence yields a different profile than Mitochondrial dysfunction-associated senescence, MiDAS, which notably lacks IL-1-dependent inflammation) 421.
Despite this heterogeneity, a conserved, pathological "core" of the SASP exists, which inflicts massive collateral damage on the structural and functional tissue microenvironment. Table 2 categorizes the main components of this SASP core and details their specific mechanisms of tissue destruction.
Table 2: Core Components of the SASP and Mechanisms of Tissue Damage
| SASP Category | Primary Components | Cellular Mechanism of Action / Target | Specific Tissue Pathology & Disease Exacerbation |
|---|---|---|---|
| Pro-inflammatory Cytokines | Interleukin-6 (IL-6), IL-$1\alpha$, IL-$1\beta$, TNF-$\alpha$ | Sustains chronic systemic inflammation (inflammaging); recruits innate immune cells; induces secondary paracrine senescence in adjacent healthy cells. | Cardiovascular & Systemic: Promotes profound endothelial dysfunction, atherosclerotic plaque instability, and the progression of cardiac hypertrophy. Drives systemic metabolic syndrome 4212223. |
| Chemokines | Interleukin-8 (IL-8 / CXCL8), MCP-1 (CCL2), CXCL1 | Acts as potent chemoattractants; chronically anchors macrophages and T-cells to the senescent microenvironment, preventing resolution. | Metabolic Organs (Liver/Adipose): Drives relentless macrophage infiltration into visceral adipose tissue and the liver, exacerbating severe insulin resistance, NAFLD, and progressive NASH 4417. |
| Matrix Metalloproteinases (MMPs) | MMP-1, MMP-2, MMP-3, MMP-13 | Hyper-degrades extracellular matrix (ECM) proteins including collagen and elastin; structurally disrupts tissue architecture and integrity. | Musculoskeletal System: Drives Intervertebral Disc Degeneration (IVDD) by actively degrading the annulus fibrosus and nucleus pulposus matrices; promotes end-stage osteoarthritis via articular cartilage degradation 241021. |
| Growth & Angiogenic Factors | VEGF, TGF-$\beta$, Amphiregulin (AREG), IGFBPs | Alters local stem cell regenerative niches; drives abnormal, leaky vascularization; induces epithelial-mesenchymal transition (EMT). | Oncology & Fibrosis: TGF-$\beta$ drives massive profibrotic tissue scarring (e.g., idiopathic pulmonary fibrosis). VEGF and EMT drivers promote tumor angiogenesis, invasiveness, and metastasis in late-life cancers 44102124. |
SASP-mediated tissue damage is compounding and recursive. For instance, in Intervertebral Disc Degeneration (IVDD), senescent cells residing in the nucleus pulposus secrete exceptionally high levels of IL-1, MMP-1, and MMP-13, which relentlessly degrade the surrounding proteoglycan matrix and collagen II networks 102023. This architectural degradation alters the mechanical load distribution of the spine, inducing further biomechanical stress, which in turn generates a new wave of senescent cells in a vicious, self-amplifying degenerative cycle 102023. Similarly, in the aging liver, SASP factors alter the delicate metabolic zonation, driving neighboring healthy hepatocytes toward steatosis and accelerating the pathological transition from simple fatty liver to severe, irreversible cirrhosis 72526. Furthermore, the SASP exhibits a dangerous temporal biphasic nature; initially, it may be immunosuppressive and profibrotic (characterized by TGF-$\beta$ secretion), but over time it rigidly progresses to become overwhelmingly pro-inflammatory and fibrolytic (characterized by extreme IL-6 and IL-8 expression), throwing tissue homeostasis into absolute chaos 21.
The Biological Danger of Eradicating All Senescent Cells
Given the catastrophic, systemic effects of the accumulated SASP, the intuitive clinical response is to seek the total, unmitigated eradication of all senescent cells from the human body. However, such a simplistic approach is profoundly biologically dangerous. Cellular senescence is a highly conserved, pleiotropic biological program that evolved primarily as a protective, homeostatic mechanism. Transient, acutely induced senescence is absolutely vital for human survival and tissue maintenance 2427.
Broad, unselective, and continuous eradication of senescent cells carries severe clinical risks, as it strips the body of essential physiological functions. Senescent cells play indispensable, beneficial roles in several critical biological arenas:
- Wound Healing and Tissue Repair: Following acute physical injury, transient senescent cells - particularly senescent endothelial cells and fibroblasts - rapidly appear at the wound site. These cells secrete highly specific, beneficial SASP components, most notably Platelet-Derived Growth Factor AA (PDGF-AA). This specific signaling induces optimal myofibroblast differentiation, initiates vascularization, and accelerates the precise architectural remodeling required for rapid tissue healing 8.
- Acute Tumor Suppression: Senescence serves as an immediate, uncompromising biological brake on malignant transformation. If a cell sustains severe oncogenic mutations (such as the hyperactivation of the RAS oncogene), entering immediate senescence prevents it from continuously dividing and forming a full-blown malignancy 2428.
- Limitation of Acute Fibrosis: Paradoxically, while chronic, long-term SASP promotes systemic fibrosis, acute senescence plays the opposite role. For example, during acute liver damage, hepatic stellate cells enter senescence, which actively limits liver fibrosis by halting their own proliferation and preventing the overproduction and massive deposition of ECM proteins into the fibrotic scar 7.
- Embryonic Development: Senescence is a rigidly programmed, non-pathological necessity during mammalian embryogenesis. It is required for the proper anatomical remodeling, shaping, and regression of transient embryonic structures, such as the mesonephros, the endolymphatic sac, and the neural tube 7.
As noted by leading researchers in the field, "simply clearing away everything could have negative long-term consequences," including severely compromised wound healing and unchecked tissue regeneration leading to hyperplasia 29. Therefore, the clinical goal of advanced senotherapeutics is not total systemic eradication. Rather, the objective is the restoration of homeostatic balance - specifically utilizing targeted interventions to clear the chronically accumulated, pathogenic subsets of senescent cells that the aging immune system has failed to remove, while meticulously preserving the transient senescent cells required for acute injury response and fundamental tissue maintenance.
Therapeutic Modalities: Senolytics vs. Senomorphics
To precisely address the pathological burden of accumulated senescent cells, the pharmaceutical and biotechnology industries have developed two distinct, sophisticated classes of gerotherapeutics: Senolytics and Senomorphics (also referred to as senostatics) 273330.
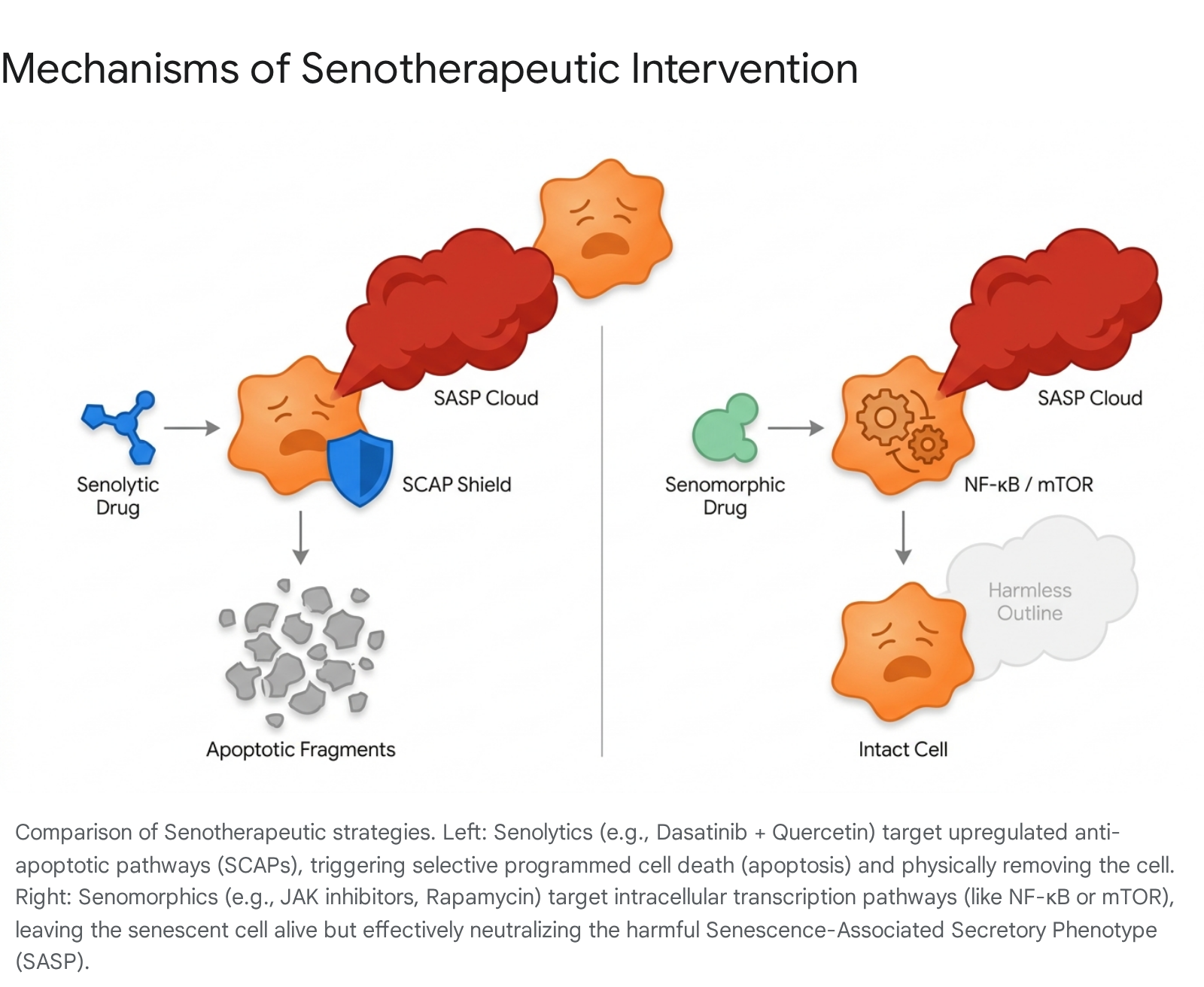
Senolytics are small molecules, peptides, or biological agents rationally designed to selectively induce apoptosis (programmed cell death) exclusively in senescent cells, while sparing adjacent healthy tissue. Because senescent cells are continuously subjected to their own highly toxic, pro-apoptotic SASP, they only survive by massively upregulating Senescent Cell Anti-Apoptotic Pathways (SCAPs) 173132. These SCAP networks depend heavily on survival proteins such as the BCL-2/BCL-xL family, the PI3K/AKT signaling cascade, and specific ephrins and Src family tyrosine kinases 173132. Senolytics ruthlessly exploit this singular metabolic vulnerability. By transiently inhibiting these specific SCAPs, senolytics remove the biological "shield" of the senescent cell. Unable to withstand its own internal toxic environment, the senescent cell quickly triggers self-destruction, physically removing itself from the tissue 1630. Prominent examples include Dasatinib (a broad-spectrum tyrosine kinase inhibitor), Navitoclax (a targeted BCL-2/BCL-xL inhibitor), and highly active natural flavonoids such as Quercetin and Fisetin. Because senolytics physically remove the offending cells, they can be administered in intermittent "hit-and-run" dosing regimens, minimizing prolonged systemic drug exposure 3733.
Senomorphics, on the other hand, do not kill the senescent cell. Instead, they modulate the cell's pathological behavior by neutralizing or drastically altering the SASP. Senomorphics target the deep intracellular signaling pathways - such as mTOR, p38 MAPK, or the JAK/STAT inflammatory cascade - that are responsible for transcribing, translating, and assembling the inflammatory secretome 273934. By effectively dampening the SASP, senomorphics alleviate chronic inflammation and halt tissue degradation without inducing cellular ablation. While senomorphics are often considered potentially safer due to the avoidance of mass cellular die-off and the preservation of the cell's structural presence, they present a significant clinical challenge: they strictly require continuous, long-term administration to keep the SASP suppressed 373334. If a senomorphic is withdrawn, the surviving senescent cell will immediately resume pumping out its toxic secretome.
Prioritizing 2023+ Global Human Clinical Trials
The transition of senotherapeutics from tightly controlled murine models to rigorous human clinical trials represents a monumental leap for translational longevity medicine. Between 2023 and 2026, the landscape of clinical trials has rapidly matured, expanding far beyond initial Phase 1 safety studies to aggressively target specific, severe age-related pathologies across global research hubs in the United States, Europe, and Asia.
The United States: Advancing Multimorbidity and Metabolic Indications
In the US, the oral combination of Dasatinib and Quercetin (D+Q) remains the absolute vanguard of clinical senolytic testing. Building on early, foundational Phase 1 successes in Idiopathic Pulmonary Fibrosis (IPF) and Diabetic Kidney Disease (DKD) - where researchers demonstrated the first direct, peer-reviewed evidence of a reduced senescent cell burden in human adipose and skin tissues post-treatment - the field is now targeting severe neurodegeneration 171631. The SToMP-AD (Senolytic Therapy to Modulate Progression of Alzheimer's Disease) phase 2 trial, spearheaded by leading researchers at the Mayo Clinic, is actively evaluating the central nervous system penetration of D+Q. This trial aims to decisively determine if clearing senescence in specific neuroglial cell populations can halt or modulate the progression of early Alzheimer's Disease 1631. Additionally, at St. Jude Children's Research Hospital, an innovative trial is utilizing D+Q to treat childhood cancer survivors, a population that exhibits accelerated, premature biological aging and frailty due to the severe genotoxic stress of early-life chemotherapy and radiotherapy 2935.
Simultaneously, the private US biotechnology sector is heavily capitalizing on second-generation, highly selective senotherapeutics. Unity Biotechnology recently announced comprehensive 36-week results from their Phase 2b ASPIRE trial (May 2025). Their novel Bcl-xL inhibitor, UBX1325, achieved statistical non-inferiority against standard-of-care anti-VEGF therapies (aflibercept) in patients suffering from Diabetic Macular Edema (DME). Crucially, the data demonstrated a highly durable treatment effect that dramatically reduces the need for frequent, invasive intraocular injections 42. In parallel, BioAge Labs secured a massive $170 million in Series D financing in early 2024 to accelerate the Phase 2 clinical development of senomorphic candidates specifically targeting metabolic aging, insulin resistance, and age-related muscle preservation 4243. Furthermore, researchers at Tulane University recently published compelling data in Nature Aging on a novel candidate, 753b. This drug exhibits profound senolytic clearance in advanced models of metabolic dysfunction-associated steatotic liver disease (MASLD), successfully slowing disease progression and halting the fatal development of hepatocellular carcinoma 26.
Europe: Organ Rejuvenation, Fibrosis Reversal, and PLA2 Targeting
European clinical trials are pioneering the highly innovative application of senolytics in whole-organ preservation and the reversal of chronic fibrotic diseases. At the Charité - Universitätsmedizin in Berlin, researchers are exploring the revolutionary concept of ex vivo organ rejuvenation for transplantation 3637. Older donor organs typically exhibit exceptionally high senescent cell burdens, which provoke massive, destructive immune responses and drive graft rejection upon transplantation. Preclinical and ex vivo human studies at Charité demonstrate that perfusing older cardiac allografts with D+Q during machine perfusion effectively clears senescent cells, significantly reduces cell-free mitochondrial DNA (a massive driver of age-associated inflammation), and dramatically improves the survival and functional metrics of older donor organs 36.
In the Netherlands, the highly anticipated TRUTH Study (NCT05506488, running from July 2023 to February 2026) is evaluating D+Q for the direct treatment of fibrotic Non-Alcoholic Fatty Liver Disease (NAFLD) 38. Given that NAFLD affects up to 30% of the Western population and is heavily driven by SASP-induced hepatic scarring, this Phase II interventional trial aims to prove definitively that short-term senolytic therapy can successfully reverse hepatic fibrosis in humans aged 18-65 by clearing pathogenic, senescent hepatocyte populations, allowing the liver to functionally regenerate 3538.
In Austria, Vienna-based biotech Rockfish Bio is advancing a highly targeted senolytic approach, moving completely away from the broad-spectrum, off-target toxicity risks associated with early kinase inhibitors 3948. They identified a unique metabolic vulnerability: senescent cells actively resist apoptosis by altering the enzymatic conversion of phospholipase A2 (PLA2) 49. By developing novel small molecules (such as their lead candidate RFB01016) that selectively target this specific PLA2 metabolic pathway, Rockfish Bio has achieved an unparalleled therapeutic index of up to 700 in preclinical models. Treatment completely restored neuromuscular function and massively extended median survival times by 40% in naturally aged mice. Supported by major investments, they are currently executing IND-enabling studies, preparing for highly specific first-in-human clinical trials slated for late 2025 394849.
Asia: Seno-Antigen Vaccines, CAR-T Therapy, and Epigenetic Reprogramming
Asia is aggressively pushing the absolute boundaries of longevity medicine, moving beyond small molecules and into the realm of advanced immunotherapeutics, vaccines, and multimodal cellular reprogramming. At Juntendo University in Japan, a landmark discovery via deep transcriptome analysis identified Glycoprotein Nonmetastatic Melanoma Protein B (GPNMB) as a transmembrane "seno-antigen" - a specific surface marker highly and exclusively expressed on the most malignant, pro-inflammatory senescent vascular endothelial cells, particularly within atherosclerotic plaques 324041. Instead of utilizing traditional small molecule drugs, the Juntendo team developed a revolutionary senolytic "aging vaccine" targeting GPNMB 2940. In advanced murine models (including high-fat diet models and progeria models), the vaccine successfully trained the immune system to hunt and selectively destroy GPNMB-expressing cells via antibody-dependent cellular cytotoxicity (ADCC). This precision elimination led to dramatic extensions in lifespan, improvements in glucose tolerance, and severe reductions in metabolic dysfunction 293240. The Juntendo team is currently advancing this toward human clinical application, seeking to utilize an mRNA-vaccine platform - similar in delivery to successful COVID-19 vaccines - allowing for highly precise, immune-mediated senolysis that completely spares beneficial, physiological senescent cells 2941. Parallel immunotherapy research highlighted in Nature Aging (2025) demonstrates that uPAR-positive senescent cells can be safely eliminated via a single administration of engineered CAR-T cells, reversing liver fibrosis and metabolic dysfunction with unparalleled long-term memory 42.
In Singapore, the PROMETHEUS trial, originating from the National University of Singapore's Academy for Healthy Longevity and intimately tied to the XPRIZE Healthspan competition, represents the zenith of personalized, multimodal senotherapeutics 5354. Scheduled to yield critical results by April 2025, this trial explicitly abandons the outdated "one-size-fits-all" pharmacological approach. Instead, it categorizes patients aged 50 to 80 utilizing individual "gerotypes" - profiling subjects based on deep multi-omics, epigenetic clocks, and precise immune biomarkers (e.g., CD4/CD8 ratios) 5455. The trial tests the profound efficacy of autologous "Prometheus Cells" - a patient's own dermal fibroblasts that have been epigenetically reprogrammed using bioactive molecules to a highly functional, stem-like state. These cells are designed to modulate the immune system, clear local senescence, and secrete trophic factors like BDNF and PDGF to promote massive tissue repair 5543. Crucially, this advanced cellular therapy is dynamically stacked with personalized lifestyle interventions (diet, sleep optimization) and highly targeted supplementation (e.g., NAD+ boosters like NMN, Urolithin A, and senolytics). Operating under the "SEMI" model (Senolytics, Epigenetics, Metabolism, and Immunology), the PROMETHEUS trial serves as the first rigorously controlled human clinical trial testing the additive and synergistic effects of a comprehensive, multimodal longevity protocol designed to fundamentally reverse biological age 5355.
Conclusion
The biomedical campaign against cellular senescence has conclusively transitioned from the theoretical realm of basic geroscience to tangible, aggressive clinical reality. Senescent cells are definitively not inert, dying "zombie" entities, but rather hyper-metabolic drivers of massive structural tissue destruction via the continuous secretion of the SASP. As highlighted by the updated, rigorous 2024/2025 ICSA guidelines, understanding and quantifying the nuanced biology of senescence requires advanced, multi-marker profiling. The field has rightfully moved away from solitary, error-prone reliance on $p16^{INK4a}$ toward comprehensive diagnostic panels that include $p21$, $SA-\beta-gal$, and highly specific lipofuscin analogs like SenTraGor, allowing for the analysis of archival clinical tissues.
Crucially, the next frontier of senotherapeutics recognizes the profound biological danger of total senescent cell eradication. Because acute senescence is necessary for wound healing, embryogenesis, and tumor suppression, the future of treatment relies entirely on precision medicine. The breathtaking progression of clinical trials globally - from the Mayo Clinic's D+Q interventions in Alzheimer's disease to Rockfish Bio's PLA2-targeted molecules, and Juntendo University's breakthrough GPNMB immune-vaccines - demonstrates a concerted, worldwide effort to selectively target only pathological senescence. As these advanced clinical trials yield their comprehensive results through 2025 and 2026, the medical community stands on the precipice of a revolutionary era where therapeutically targeting the fundamental hallmarks of aging may successfully compress human morbidity and structurally reverse age-related chronic disease across the global population.