Cardiovascular and neuroprotective GLP-1 agonist longevity evidence
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have transitioned from targeted glycemic control agents for type 2 diabetes mellitus to foundational therapies for chronic weight management and metabolic syndrome. Recent large-scale cardiovascular, oncological, and neurological outcome trials suggest these agents initiate systemic physiological shifts that extend far beyond adipose tissue reduction. Emerging evidence indicates that GLP-1 RAs function as direct disease-modifying agents with the capacity to influence overall longevity and morbidity. Through mechanisms involving systemic inflammation reduction, mesolimbic reward pathway modulation, and direct cardioprotection, these molecules are redefining the pharmacological management of chronic metabolic disease. This exhaustive analysis evaluates the clinical data surrounding GLP-1 RAs, focusing on cardiovascular risk reduction, systemic inflammation, neuroprotection, oncological incidence, demographic trial diversity, and the emerging physiological risks associated with treatment cessation and sarcopenia.
Cardiovascular Outcomes and Mortality Reduction
The cardiovascular efficacy of GLP-1 RAs has been established through several pivotal phase 3 trials, shifting the clinical paradigm from isolated risk factor management to comprehensive cardiovascular disease secondary prevention. The accumulated data indicate that both subcutaneous and oral formulations significantly reduce the incidence of major adverse cardiovascular events (MACE) in highly vulnerable populations, demonstrating benefits irrespective of baseline diabetic status.
The SELECT Trial and Secondary Prevention
The Semaglutide Effects on Cardiovascular Outcomes in People with Overweight or Obesity (SELECT) trial serves as the primary benchmark for cardiovascular risk reduction in patients without diabetes. Enrolling 17,604 patients across 41 countries and more than 800 sites, the trial evaluated subcutaneous semaglutide (2.4 mg weekly) in adults aged 45 and older with a body mass index (BMI) of 27 or greater and established cardiovascular disease 12345. The inclusion criteria required a prior myocardial infarction, stroke, or symptomatic peripheral arterial disease, while explicitly excluding patients with a history of diabetes or a hemoglobin A1c (HbA1c) of 6.5% or higher 566.
Over a mean follow-up of 39.8 months, semaglutide reduced the primary composite outcome - death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke - by 20%, with events occurring in 6.5% of the semaglutide cohort versus 8.0% of the placebo group (Hazard Ratio [HR] 0.80; 95% Confidence Interval [CI] 0.72 - 0.90; p<0.001) 135. Secondary analyses of the SELECT trial demonstrated a 15% reduction in cardiovascular death (HR 0.85; 95% CI 0.71 - 1.01; p=0.07) and a 19% reduction in all-cause mortality (HR 0.81; 95% CI 0.71 - 0.93), reinforcing the longevity implications of the therapy 3578. The trial also documented a 17% reduction in total strokes and a 10% reduction in total myocardial infarctions 4.
Safety outcomes in the SELECT trial revealed that the overall proportion of patients with serious adverse events was lower in the semaglutide group (33.4%) compared to placebo (36.4%; p<0.001), primarily driven by a reduction in cardiac disorders 5910. However, adverse events leading to trial drug discontinuation were significantly higher in the semaglutide group (16.6% versus 8.2% for placebo; p<0.001), almost entirely attributable to gastrointestinal complications such as nausea, diarrhea, and vomiting (10.0% versus 2.0%) 159. The incidence of acute pancreatitis and acute kidney failure did not differ significantly between the groups 5.
Consequently, regulatory bodies have formally recognized these cardiovascular benefits. In March 2024, the U.S. Food and Drug Administration (FDA) expanded the label for semaglutide (Wegovy) to explicitly include the reduction of major adverse cardiovascular events in adults with established cardiovascular disease and overweight or obesity 71011121413. In July 2024, the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion. While the EMA concluded that cardiovascular risk reduction is inherent to the drug's existing weight management indication and did not grant a separate isolated cardiovascular indication, the product information was formally updated to reflect the 20% MACE reduction demonstrated in the SELECT trial, aiding in clinical decision-making and regional reimbursement negotiations 814.
The SOUL Trial and Oral Administration Efficacy
Parallel to the subcutaneous trials, the SOUL trial investigated the cardiovascular efficacy of oral semaglutide in patients with type 2 diabetes and established atherosclerotic cardiovascular disease (ASCVD), chronic kidney disease (CKD), or both 151617. Enrolling 9,650 participants across 444 sites in 33 countries, patients were randomized to receive oral semaglutide (escalated to a maximum dose of 14 mg) or placebo, superimposed on standard cardiovascular and glycemic care 1617.
Over a median follow-up of 49.5 months, the trial demonstrated a 14% reduction in the primary three-point MACE endpoint. Primary outcome events occurred in 12.0% of the oral semaglutide group (3.1 events per 100 person-years) compared to 13.8% of the placebo group (3.7 events per 100 person-years) (HR 0.86; 95% CI 0.77 - 0.96; p=0.006) 1617. The incidence of serious adverse events was balanced between the two cohorts (47.9% in the oral semaglutide group versus 50.3% in the placebo group), with gastrointestinal disorders maintaining a manageable profile (5.0% versus 4.4%, respectively) 17. Subgroup analyses indicated pronounced efficacy in Asian populations, with a reported 27% reduction in MACE among participants from that region 15.
The cardiovascular safety and efficacy of oral semaglutide was further supported by the OASIS 1 and OASIS 2 trials, which evaluated an escalated 50 mg oral dose for weight management. In OASIS 1, enrolling adults with overweight or obesity without diabetes, oral semaglutide 50 mg yielded a 15.1% reduction in baseline body weight at 68 weeks, compared to 2.4% with placebo (p<0.0001) 181920. A substantial 85% of participants on the active drug achieved at least a 5% weight reduction, compared to 26% on placebo 181920.
| Cardiovascular Outcome Trial | Primary Intervention | Target Population | MACE Risk Reduction (HR) | All-Cause Mortality Reduction (HR) | Median/Mean Follow-Up |
|---|---|---|---|---|---|
| SELECT | Semaglutide 2.4 mg (Subcutaneous) | Overweight/obese, established CVD, no T2DM | 20% (HR 0.80) | 19% (HR 0.81) | 39.8 months |
| SOUL | Semaglutide up to 14 mg (Oral) | T2DM, established ASCVD and/or CKD | 14% (HR 0.86) | Not explicitly powered/reported as primary | 49.5 months |
| Meta-Analysis (21 Trials) | Various GLP-1 RAs (Subcutaneous/Oral) | Broad populations across 99,599 patients | 13% (HR 0.87) | 12% (HR 0.88) | 2.4 years |
A comprehensive meta-analysis incorporating 21 randomized controlled trials and 99,599 patients across eight different GLP-1 RAs (including lixisenatide, liraglutide, exenatide, semaglutide, efpeglenatide, dulaglutide, albiglutide, and tirzepatide) confirmed high-certainty evidence for broad class efficacy. The pooled data demonstrated that GLP-1 RAs reduced all-cause mortality with a number needed to treat (NNT) of 121 (Incidence Rate Ratio [IRR]: 0.88; 95% CI: 0.84 - 0.92), cardiovascular death with an NNT of 170 (IRR: 0.87; 95% CI: 0.81 - 0.92), and overall MACE with an NNT of 66 (IRR: 0.87; 95% CI: 0.83 - 0.91) 24.
Heart Failure Efficacy and the STEP-HFpEF Trial
The cardiometabolic burden of obesity frequently manifests as heart failure with preserved ejection fraction (HFpEF), a highly prevalent condition historically resistant to standard pharmacological interventions. The 2023 STEP-HFpEF trial evaluated semaglutide 2.4 mg in obese patients with HFpEF, demonstrating marked improvements in both patient-reported symptomatology and hard clinical outcomes 25. Patients treated with semaglutide experienced a significant 16.6-point increase in the Kansas City Cardiomyopathy Questionnaire (KCCQ) clinical summary score and a 20-meter improvement in the 6-minute walk distance test 25. Crucially, the trial recorded a 13% reduction in body weight and a remarkable 79% reduction in heart failure hospitalizations (HR 0.79) over 52 weeks 25. These positive outcomes were mechanistically attributed to a combination of total weight loss, decreased epicardial fat, improved myocardial energy metabolism, and a reduction in systemic inflammation 25.
A prespecified secondary analysis of the SELECT trial corroborated these heart failure benefits across a broader demographic over a longer time horizon. Among the 4,286 patients (24.3% of the total cohort) with pre-existing heart failure at baseline, semaglutide significantly reduced the composite heart failure endpoint (cardiovascular death or hospitalization for heart failure) 32122. The therapeutic benefit was observed regardless of the underlying subtype, demonstrating efficacy in both heart failure with reduced ejection fraction (HFrEF) (HR 0.65 for MACE; HR 0.79 for composite HF) and HFpEF (HR 0.69 for MACE; HR 0.75 for composite HF) 522. The incidence of serious adverse events remained lower with semaglutide versus placebo, regardless of heart failure classification 22.
Weight-Independent Cardioprotection Mechanisms
A critical debate within metabolic literature concerns whether the cardiovascular benefits of GLP-1 RAs are strictly secondary to weight reduction, or if they involve weight-independent direct cardioprotective mechanisms. Post-hoc temporal analyses of the SELECT trial reveal that cardiovascular benefits manifest significantly prior to the achievement of clinically meaningful weight loss 2. Within the first three months of treatment, prior to the achievement of maximum weight loss, patients receiving semaglutide experienced a 37% reduced risk of MACE (HR 0.63; 95% CI 0.41 - 0.95). By six months, the risk of cardiovascular death was reduced by 50% (HR 0.50; 95% CI 0.26 - 0.93), the composite heart failure risk dropped by 59% (HR 0.41; 95% CI 0.24 - 0.67), and all-cause mortality fell by 40% (HR 0.60; 95% CI 0.36 - 1.01) 223.
Furthermore, statistical modeling indicates that the magnitude of cardiovascular benefit in the SELECT trial did not correlate linearly with the amount of weight lost by week 20, nor did it differ significantly across baseline BMI categories 24. Patients who were only modestly overweight achieved comparable cardiovascular protection to those with severe clinical obesity 24. Based on this divergence, researchers estimate that approximately two-thirds of the MACE reduction observed with semaglutide cannot be accounted for by adiposity reduction alone 24. These findings reframe GLP-1 RAs as direct disease-modifying agents acting upon vascular endothelium, plaque stabilization, and myocardial tissue, operating through physiological pathways parallel to, but distinct from, energy balance and lipolysis 2425.
Modulation of Systemic Inflammation
Obesity represents a state of chronic, low-grade systemic inflammation, driven by the active secretion of pro-inflammatory cytokines and adipokines from visceral adipose tissue 31. This inflammatory state accelerates endothelial dysfunction, insulin resistance, and atherogenesis, driving the progression of coronary heart disease and peripheral vascular disease 25. The presence of GLP-1 receptors on various immune cells, including macrophages and T cells, indicates that GLP-1 RAs directly modulate systemic inflammatory cascades independent of glycemic control 2531.
Biomarker Reduction and Endothelial Function
Clinical trials consistently report substantial reductions in highly sensitive C-reactive protein (hs-CRP), a primary biomarker of systemic and vascular inflammation 31. Data indicates a 40% to 60% decrease in hs-CRP among patients treated with GLP-1 RAs 31. In the semaglutide SUSTAIN and PIONEER trials, observed reductions in blood glucose and body weight explained only 20% to 60% of the total hs-CRP reductions 25. In the PIONEER 2 trial specifically, oral semaglutide reduced CRP by 30%, whereas the SGLT2 inhibitor empagliflozin had no significant effect on CRP despite achieving a comparable 4% body weight loss, highlighting a unique anti-inflammatory mechanism intrinsic to GLP-1 receptor agonism 25.
Proteomic analyses from the STEP clinical program further reveal semaglutide-induced alterations in immune regulatory pathways that occur independently of metabolic improvements 25. Beyond vascular inflammation, targeted observational studies report significant reductions in erythrocyte sedimentation rate (ESR), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-alpha). In patients with rheumatoid arthritis and concurrent obesity, GLP-1 RA therapy yielded significant reductions in disease activity scores, subjective pain scores (mean change -0.6 cm versus 1.3 cm for placebo), and joint inflammation, with pain reduction showing no direct correlation to the absolute volume of weight lost 3132. Meta-analyses across 40 randomized controlled trials confirm that GLP-1 RAs significantly reduce CRP levels compared to placebo (Standardized Mean Difference [SMD] -0.59) and other oral antidiabetic drugs (SMD -1.06), as well as significant reductions in TNF-alpha and IL-6 26.
| Inflammatory Marker | Physiological Target | Observed GLP-1 RA Effect | Clinical Implication |
|---|---|---|---|
| hs-CRP | Vascular inflammation | 40 - 60% decrease | Reduced cardiovascular atherogenesis and plaque instability |
| ESR | General systemic inflammation | 20 - 40% decrease | Improvements in systemic autoimmune conditions (e.g., Rheumatoid Arthritis) |
| IL-6 | Pro-inflammatory cytokine | Significant decrease | Reduced visceral fat-mediated metabolic inflammation |
| TNF-alpha | Inflammatory signaling | Significant decrease | Improved peripheral insulin sensitivity |
| Fibrinogen | Clotting / coagulation cascade | Modest decrease | Reduced thrombotic risk in cardiovascular disease |
Epidemiological Conflicting Evidence
Despite robust clinical trial data supporting anti-inflammatory efficacy, conflicting evidence exists within specific large-scale observational cohorts. An analysis utilizing the Epic Cosmos dataset, encompassing nearly 3,000 matched patient pairs prescribed semaglutide or liraglutide, found no statistically significant reduction in CRP or ESR levels following treatment initiation after applying rigorous adjustment modeling for the percentage of BMI reduction 27. While follow-up CRP levels broadly decreased for all groups, the statistical significance dissolved when controlling strictly for adiposity changes 27. This discrepancy highlights an ongoing epidemiological debate: while GLP-1 RAs definitively lower systemic inflammation, the exact degree to which this effect is mediated centrally through immune cell receptors versus peripherally through the mass reduction of cytokine-secreting visceral adipose tissue remains difficult to isolate in real-world clinical populations.
Neuroprotection and Cognitive Health
The physiological expression of GLP-1 receptors within the central nervous system has prompted extensive investigation into the neuroprotective and neuromodulatory capabilities of these agents. GLP-1 signaling in the brain appears to influence microglial activation, neuroinflammation, and cellular apoptosis, suggesting viable applications in neurodegenerative disease modification.
Parkinson's Disease Clinical Trial Outcomes
Preclinical murine models indicating that GLP-1 RAs can rescue dopaminergic neurons from neurotoxicity have translated into targeted clinical trials for Parkinson's disease. In April 2024, the New England Journal of Medicine published the results of a Phase 2 clinical trial evaluating the Type 2 diabetes drug lixisenatide in patients with early-stage Parkinson's disease 28. The trial met its primary endpoint, demonstrating that participants treated with lixisenatide maintained stable motor symptom scores (measured via the MDS-UPDRS part III) over the duration of the trial, whereas the placebo cohort exhibited standard, progressive motor decline 28. A subsequent follow-up assessment conducted two months post-treatment cessation revealed that the gap in motor progression between the active and placebo cohorts persisted, suggesting a durable, potentially disease-modifying neuroprotective effect rather than transient symptom masking 28.
However, the intervention was associated with notable tolerability challenges; more than half of the participants reported significant gastrointestinal issues including nausea and vomiting, frequently necessitating protocol-driven dose reductions 28. A previous Phase 2 trial for a similar agent, exenatide, yielded comparable possibilities for motor stabilization, prompting a large, multicenter Phase 3 clinical trial currently underway to definitively establish efficacy 28. Conversely, trials of other agents within the incretin class, such as NLY01 and PT320, have reported no meaningful neuroprotective effects 28. These divergent outcomes indicate that neurological efficacy may rely heavily on specific molecular structures, lipid solubility, blood-brain barrier penetrance profiles, and exact receptor binding affinities, rather than a universal class effect.
Addiction and Mesolimbic Reward Pathway Modulation
Beyond metabolic and neurodegenerative applications, GLP-1 RAs profoundly alter the mesolimbic reward system, a complex neural network traditionally associated with the reinforcement of addictive behaviors and hedonic drive. The convergence of metabolism and neuroscience represents one of the most significant emerging therapeutic frontiers for these molecules.
Mechanism of Dopaminergic Downregulation
GLP-1 medications target the hindbrain, specifically accessing circumventricular organs such as the area postrema, as well as the nucleus tractus solitarius (NTS) 293031. The NTS serves as a critical signaling relay, projecting GLP-1-secreting neurons directly to the ventral tegmental area (VTA) and the central amygdala via the mesolimbic pathway 29323334. The VTA relies on dopamine projections to the nucleus accumbens to govern reinforcement learning, the motivation to work for a reward, and the anticipatory pleasure of consumption 2942.
Recent neuroimaging and genetically engineered murine studies demonstrate that GLP-1 RAs actively lower dopamine activity in this reward circuit. Utilizing a Pavlovian sucrose paradigm, researchers found that while semaglutide did not alter VTA dopamine activity during the "reward-seeking" or cue phase, it significantly enhanced VTA signaling during the "reward-collection" or consummatory phase, effectively triggering an early, amplified sense of satiation 293235. By engaging the central amygdala, these drugs turn down the "reward noise" and internal urgency dial that drives compulsion, altering how the brain assigns value to highly rewarding, high-calorie foods 31333442.
Dissociation of Emesis and Reward Circuits
A central scientific debate regarding this mechanism revolves around whether the reduction in intake is a secondary consequence of subclinical nausea or a primary modulation of desire. Early hypotheses suggested that GLP-1 RAs suppressed feeding largely by triggering the area postrema - the brain's emetic center - causing aversion 3031. However, novel research demonstrates that the satiety and reward-dampening effects are neurologically dissociable from the emetic effects 3031.
Targeting the area postrema with GLP-1 RAs produces both weight loss and nausea, but targeting the distinct NTS-to-amygdala pathway successfully reduces food motivation without triggering gastric discomfort 303133.
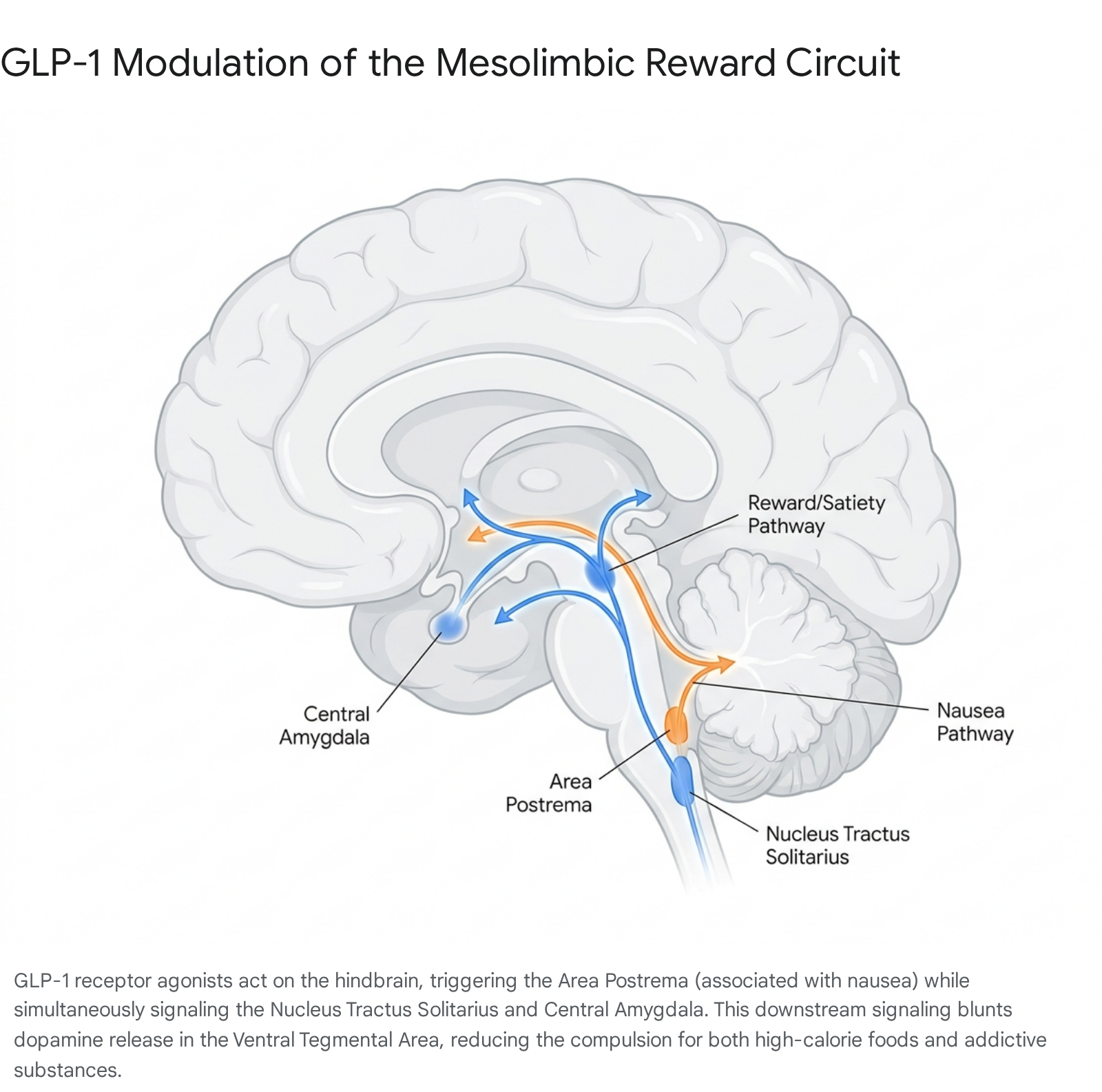
This anatomical separation opens the therapeutic possibility for developing next-generation, highly tailored weight-loss and anti-addiction molecules designed to preferentially activate the mesolimbic reward circuit while bypassing the emetic centers, potentially resolving the high rates of gastrointestinal discontinuation seen with current generation peptides 3033. Furthermore, evidence indicates that co-agonism with Glucose-Dependent Insulinotropic Polypeptide (GIP), as seen in tirzepatide, actively blunts the nauseating effects of GLP-1R agonism via central GABAergic neurons, resulting in improved clinical tolerability at higher doses 30.
Clinical Observations in Substance Use Disorders
The reduction in dopaminergic impulsivity extends beyond food consumption, impacting broad substance use disorders (SUD). A 2024 large-scale observational study involving over 600,000 U.S. veterans diagnosed with type 2 diabetes and concurrent opioid use disorder (OUD) or alcohol use disorder (AUD) found that patients prescribed GLP-1 RAs experienced significantly lower rates of substance-related harm 3637.
Specifically, GLP-1 RA use was associated with a 40% lower rate of opioid overdose and a 50% lower rate of alcohol intoxication compared to baseline non-user controls 36. Functional magnetic resonance imaging (fMRI) studies assessing brain responses to reward-related cues tentatively suggest that acute GLP-1 RA administration reduces brain reactivity in appetite and reward regions, blunting the craving that pulls individuals toward their addiction 3746. While clinical data regarding non-food emotional stimuli remains limited and heterogeneous, the cross-domain efficacy against multiple addictive substances suggests GLP-1 RAs target a common biological pathway underlying compulsion, serving as a "reward muffler" that prevents the nervous system from firing in extremes 423746.
Oncological Incidence and Disease Modification
Obesity is a recognized pathophysiological driver for at least 13 distinct malignancies, largely mediated through chronic low-grade inflammation, hyperinsulinemia, and elevated circulating sex hormones 3839. Given their profound capability to resolve these underlying physiological aberrations, GLP-1 RAs have been heavily scrutinized for both their long-term oncological safety and their potential chemo-preventative properties.
Reductions in Obesity-Associated Malignancies
A comprehensive 2025 cohort study published in JAMA Oncology, utilizing real-world target trial emulation data from over 86,000 adults, evaluated cancer incidence among patients with obesity prescribed GLP-1 RAs compared to propensity score-matched nonusers 3839. The analysis revealed a statistically significant decrease in overall cancer risk among GLP-1 users, yielding an aggregate hazard ratio of 0.83 (Incidence rate: 13.6 per 1,000 person-years for GLP-1 users versus 16.4 for nonusers) 3839.
When stratified by specific tumor types, GLP-1 RAs were associated with marked and statistically significant risk reductions in several hormone- and adiposity-driven cancers. Ovarian cancer incidence was reduced by nearly half (HR 0.53; p=0.04), meningioma incidence was lowered (HR 0.69; p=0.05), and endometrial cancer risk decreased significantly (HR 0.75; p=0.05) 3839. Bariatric surgery comparisons further validate these findings; research published in 2024 indicates that while bariatric surgery leads to a 22% lower risk of developing cancer, GLP-1 RA treatment was associated with a 39% lower risk of developing obesity-related cancers compared to no treatment, alongside lower all-cause mortality rates 40.
Colorectal Cancer Primary and Secondary Prevention
Further real-world data specifically positions GLP-1 RAs as a powerful strategy for colorectal cancer (CRC) prevention. A large-scale analysis examining over 1.6 million patients with type 2 diabetes compared GLP-1 RAs against standard therapies. In head-to-head comparisons for primary CRC prevention, patients receiving GLP-1 RAs were 36% less likely to develop CRC overall (HR 0.643; 95% CI 0.531 - 0.778) and up to 42% less likely in specific high-risk subgroups 41. When compared specifically against insulin therapy, GLP-1 RA use was associated with a 46% reduction in CRC risk (HR 0.56; 95% CI 0.44 - 0.72) 41.
The benefits appear to extend into secondary prevention and prognostic improvement. Among a cohort of patients with established colon cancer across the University of California Health system, GLP-1 RA usage was associated with 62% lower odds of 5-year mortality (OR 0.38; 95% CI 0.21 - 0.64), a survival benefit that persisted even after strict adjustment for age, BMI, and disease severity 41. Preclinical studies suggest these effects may be mediated directly at the cellular level rather than entirely dependent on whole-body weight loss. For instance, specific GLP-1 receptors have been identified directly on meningioma cells, and compounds like exenatide have been shown to directly inhibit the proliferation and invasion of ovarian cancer cells in vitro, suggesting a primary anti-tumor effect within responsive tissues 38.
Long-Term Safety Signals and Malignant Neoplasms
Despite these highly encouraging signals, researchers and oncologists urge caution in interpreting GLP-1 RAs as definitive chemopreventive agents. The observational nature of current large-scale cohort studies renders them vulnerable to unmeasured confounding variables, such as parallel changes in lifestyle, physical activity, or socioeconomic access to preventative care, which typically correlate with the prescription of high-cost novel therapeutics 3842.
Furthermore, while the JAMA Oncology study demonstrated broad risk reduction across 12 of 13 obesity-associated cancers, it identified a borderline, non-significant trend toward an increased risk of kidney cancer among GLP-1 users (HR 1.38; 95% CI 0.99 - 1.93; p=0.04) 383943. Early regulatory concerns regarding an increased risk of pancreatic and medullary thyroid cancers, which initially led to heavy regulatory scrutiny of incretin-based therapies, appear largely unsupported by recent high-quality randomized controlled trials (RCTs). A comprehensive meta-analysis of 93 RCTs confirmed no significant increase in the incidence of malignant neoplasms, including pancreatic cancer (HR 0.78; 95% CI 0.61 - 0.95), among patients receiving GLP-1 RAs compared to placebo 404142. Data from the 17,000-patient SELECT trial further corroborates this safety profile, reporting nearly identical rates of fatal malignant neoplasms between the semaglutide group (4.8%) and the placebo group (4.7%; p=0.92) over a median follow-up of nearly 40 months 5942. The prevailing clinical consensus indicates that while apparent reductions in gastrointestinal, liver, and colorectal cancers are compelling and hypothesis-generating, the primary conclusion to be drawn from current RCT data is the stringent oncological safety, rather than explicitly proven pharmacological prevention, of the drug class 42.
Treatment Cessation and Sarcopenia Risks
As the prescription rates for GLP-1 RAs scale globally - reaching millions of active users - poor long-term adherence has emerged as a primary clinical challenge. Driven by high out-of-pocket costs, supply shortages, and chronic gastrointestinal tolerability issues, data indicates that between 46% and 65% of users discontinue treatment within 12 months 14454. The physiological and metabolic consequences of treatment cessation represent a critical, newly recognized risk factor affecting patient longevity.
Cardiovascular Reversal and Metabolic Whiplash
Recent evidence demonstrates that the cardioprotective benefits and metabolic stability provided by GLP-1 RAs are highly contingent upon continuous, uninterrupted administration. A landmark 2026 study published in BMJ Medicine, tracking over 333,000 U.S. veterans treated through the Veterans Health Administration, evaluated the cardiovascular risk trajectories of patients who discontinued GLP-1 therapy compared to those who maintained it 54454647.
The findings quantify a profound physiological rebound termed "metabolic whiplash." Patients who maintained continuous GLP-1 RA treatment for three years achieved an 18% reduction in the risk of major cardiovascular events (heart attack, stroke, or death) compared to control groups on standard therapies 544546. However, upon cessation, the accumulated cardiovascular benefits degraded rapidly. Within six months of stopping the medication, patient cardiovascular risk rose by 4% relative to continuous users 45. After one year of cessation, the risk increased by 14%, and by two years, cardiovascular risk escalated by 22%, effectively erasing all protective benefits gained during the active treatment window 54454647.
Furthermore, patients who took the medication for less than 18 months prior to cessation exhibited cardiovascular outcomes completely indistinguishable from those who had never taken the drug, suggesting that cardioprotection is not a permanent structural change but rather a state that requires long-term, sustained receptor agonism 5446. The rapid return of baseline inflammation, hypertension, and hyperlipidemia outpaces the visible regain of body weight, exposing the cardiovascular system to sudden stress 47.
Sarcopenic Obesity in Older Adults
The rapid, profound weight loss induced by GLP-1 RAs is not exclusively lipolytic; a substantial and concerning proportion of the total weight lost - ranging from 15% to 40% depending on the cohort - consists of lean skeletal muscle mass 4448. This dynamic introduces the severe risk of accelerated sarcopenia, which is particularly hazardous for older adults who already experience natural age-related muscle degeneration and frailty 4344.
The primary physiological hazard materializes during the post-cessation phase. When GLP-1 RAs are discontinued, patients almost universally experience rapid weight regain 445446. However, metabolic composition studies indicate that this rapid regain is disproportionately composed of adipose tissue rather than the recovery of the previously lost lean muscle mass 44. Repeated cycles of treatment initiation, weight loss, cessation, and rapid regain (weight cycling) can systematically deplete a patient's skeletal muscle reserves while simultaneously expanding visceral fat depots. This process precipitates a state of "sarcopenic obesity" - a condition characterized by excessive adiposity masking dangerously low muscle mass 434448. Sarcopenic obesity is heavily associated with severe mobility restriction, increased fall risk, frailty, insulin resistance, and heightened all-cause mortality, effectively negating the overarching longevity benefits of the initial weight loss intervention 434448. Clinical consensus now mandates that GLP-1 RA therapy, particularly in older populations, must be paired with aggressive dietary protein optimization and frequent, structured resistance training to preserve lean mass during the active catabolic weight-loss phase 4648.
Demographic Diversity in Clinical Evidence
The epidemiological generalizability of GLP-1 RA longevity data relies heavily upon the demographic diversity of the foundational clinical trials. The Semaglutide Treatment Effect in People with Obesity (STEP) clinical program, which secured global regulatory approvals for weight management, has faced necessary scientific scrutiny regarding participant representation and the applicability of its findings to global minority populations.
Efficacy Across Racial and Ethnic Subgroups
Post-hoc analyses of the major STEP trials (specifically STEP 1, 2, and 3) were conducted to determine if the efficacy and safety profile of semaglutide 2.4 mg varied across different racial and ethnic demographics 49505152. The pooled data confirmed that the drug's performance is biologically consistent across all assessed subgroups. Reductions in body weight, waist circumference, and systolic blood pressure showed no statistically significant interaction with patient race (p ≥ 0.07) or ethnicity (p ≥ 0.40) 49505152.
For instance, the estimated treatment difference (ETD) in percent body weight reduction from baseline for semaglutide versus placebo ranged tightly between 9.3% and 12.5% across all subgroups 51. Specifically, the ETD was -9.7 percentage points for Asian participants, -11.6 for Black participants, and -12.5 for White participants 52. Ethnic comparisons showed an ETD of -10.9 percentage points for Hispanic or Latino patients and -12.1 for non-Hispanic patients 52. Adverse events and tolerability profiles were similarly consistent, indicating that the biological mechanism of GLP-1 RAs operates uniformly regardless of genetic background 4950.
| Demographic Subgroup | Mean Percent Weight Change (vs Placebo) | Efficacy Interaction p-value | Trial Data Source |
|---|---|---|---|
| White | -12.5 percentage points | p = 0.07 (overall race) | STEP 1, 2, 3 Pooled |
| Black | -11.6 percentage points | p = 0.07 (overall race) | STEP 1, 2, 3 Pooled |
| Asian | -9.7 percentage points | p = 0.07 (overall race) | STEP 1, 2, 3 Pooled |
| Hispanic/Latino | -10.9 percentage points | p = 0.40 (overall ethnicity) | STEP 1, 2, 3 Pooled |
| Non-Hispanic | -12.1 percentage points | p = 0.40 (overall ethnicity) | STEP 1, 2, 3 Pooled |
East Asian Cohort Specificity and Risk Thresholds
Recognizing that different populations experience metabolic complications at varying BMI thresholds, specialized trials were designed. The STEP 6 and STEP 7 trials, alongside the OASIS 2 trial, explicitly targeted East Asian populations in Japan, South Korea, China, and Brazil 63535466. These trials utilized lower BMI inclusion thresholds (e.g., BMI ≥ 27.0 with comorbidities, as standard in East Asian guidelines) to reflect the heightened risk of type 2 diabetes and visceral adiposity in these genetic cohorts at lower absolute weights 5354.
In the STEP 7 trial, semaglutide 2.4 mg yielded a mean body weight reduction of -12.1% compared to -3.6% for placebo over 44 weeks among predominantly East Asian participants 6355. The OASIS 2 trial, evaluating oral semaglutide 50 mg in East Asian populations, demonstrated a -14.3% reduction in body weight compared to -1.3% for placebo 54. Furthermore, the SOUL trial's subgroup analysis of Asian populations demonstrated an exceptional 27% reduction in MACE, outperforming the 14% global average, highlighting the profound cardiovascular benefit of incretin therapy in this specific demographic 1516.
Structural Representation Gaps
Despite uniform biological efficacy when diverse populations are included, severe structural gaps in trial representation persist across the broader GLP-1 literature. A comprehensive meta-analysis of 27 randomized controlled trials involving GLP-1 medicines for obesity highlighted that overall trial cohorts remain predominantly White/Caucasian (79%) 5056. There is significant and consistent underrepresentation of Black (9%), Asian (13%), Hispanic (22%), and Indigenous (2%) populations relative to both national demographic baselines and the actual global distribution of obesity 56.
For example, representation gaps reached an alarming -58% relative to the general population of Brazil, and -68% compared to South Africa 56. Furthermore, 84.2% of all trial sites were located in high-income countries, standing in sharp contrast to the reality that the global prevalence of obesity and its metabolic complications predominantly affects low- and middle-income nations 56. The lack of representation from diverse cohorts limits the epidemiological precision of these longevity findings, particularly because baseline metabolic risk, diabetes onset thresholds, and cardiovascular morbidity rates differ vastly across genetic and socioeconomic backgrounds. To ensure equitable global health outcomes, future longevity and cardiovascular outcome trials must aggressively prioritize the inclusion of underrepresented global populations 495156.