Biological sex differences in aging and longevity
Introduction to the Sex Gap in Aging
The demographic reality that females consistently outlive males is one of the most robust, universally observed phenomena in human biology and gerontology. Across diverse populations, historical epochs, and varying levels of industrialization, the survival advantage of women remains a demographic constant. Historically, epidemiological consensus heavily favored behavioral and sociodemographic explanations for this discrepancy. The traditional narrative posited that the male longevity deficit was predominantly a consequence of lifestyle choices, including higher rates of tobacco and alcohol consumption, greater engagement in risky behaviors, and increased exposure to occupational hazards.
However, contemporary research has fundamentally reshaped this understanding. A landmark 2026 analysis published in the Journal of the American Medical Association (JAMA) evaluated a national cohort spanning two decades and decisively concluded that the sex-based mortality gap endures even after rigorously accounting for a comprehensive spectrum of behavioral, clinical, and sociodemographic factors 12. The study, which adjusted for variables such as smoking, alcohol use, chronic conditions, access to healthcare, education, income, and occupational exposures, demonstrated that males maintain a 63% greater risk of all-cause mortality compared to females 13. This heightened risk translates to a mortality rate of 1,268 per 100,000 person-years for males, compared to 1,117 for females 1. The persistence of this gap over a period when male health behaviors generally improved firmly redirects the scientific focus toward intrinsic biological, genetic, and evolutionary drivers of aging 12.
While women enjoy an undisputed survival advantage, they concurrently bear a disproportionate burden of morbidity. Females spend a significantly greater proportion of their extended lifespans in states of poor health, experiencing higher rates of clinical frailty, systemic disability, and non-fatal chronic diseases. This profound biological dichotomy is known as the morbidity-mortality paradox. This exhaustive report synthesizes contemporary demographic data from the 2024 United Nations World Population Prospects (WPP) and the World Health Organization (WHO), evaluates the latest 2023 and 2024 epigenetic clock research, analyzes prevailing evolutionary frameworks, and examines cutting-edge therapeutic interventions that reveal the distinctly sex-dimorphic nature of the aging process.
Global Demographics and Geographic Disparities
The global landscape of aging is undergoing a profound transition, heavily influenced by shifting fertility rates, advancements in medical technology, and the lingering impacts of global health emergencies. Understanding the sex differences in aging requires situating the biological data within the broader macroeconomic and demographic context.
The Trajectory of Global Life Expectancy and the COVID-19 Disruption
According to the 2024 Revision of the United Nations World Population Prospects, global life expectancy at birth reached 73.3 years in 2024, an increase of 8.4 years since 1995, with projections indicating an average longevity of 77.4 years by 2054 34. This overarching trend of increased longevity, however, was severely disrupted by the COVID-19 pandemic, which precipitated a massive contraction in both lifespan and healthspan.
Prior to the pandemic, global life expectancy had increased by more than six years between 2000 and 2019, climbing from 66.8 years to 73.1 years 5. Concurrently, Healthy Life Expectancy (HALE) - a metric defining the average number of years a person can expect to live in "full health" free from disabling disease - increased by 9%, from 58.1 to 63.5 years 567. Crucially, the 5.3-year increase in HALE did not keep pace with the 6.4-year increase in overall life expectancy, indicating an expansion of morbidity in later life 58.
The pandemic effectively wiped out a decade of progress. By 2021, global life expectancy dropped by 1.8 years to 71.4 years, and global HALE retreated to 61.9 years, returning populations to 2012 levels 589. The impact was notably sex-dimorphic; while male life expectancy dropped relatively evenly across 2020 and 2021, female life expectancy experienced a much steeper drop in the second year of the pandemic 12. Males ultimately experienced 45% more health loss from COVID-19 than females, cementing the virus as a primary driver of the male mortality disadvantage during this period 1011.
Demographic Groupings and Survival Ratios
The gap between male and female longevity varies across geographic regions, largely correlating with a nation's position within the demographic transition. The UN categorizes the global population into distinct groups based on population growth trajectories, which correspond closely with life expectancy outcomes:
The "Growing Group" consists of nations whose populations are projected to continue expanding through 2054, largely concentrated in Sub-Saharan Africa and parts of South Asia 3. Nigeria, located within a region holding the lowest life expectancy globally (averaging 62.3 years in 2024), exemplifies early-stage demographic transition where infectious diseases and maternal mortality remain significant challenges 315. India, another key member of this group, demonstrates a rapidly changing demographic profile where female life expectancy (approximately 72 years) outpaces male life expectancy (approximately 69 years), despite women facing intense burdens of morbidity throughout their extended lives 31512.
The "Peaking Group" includes 48 countries projected to reach their maximum population size between 2025 and 2054, including Brazil and Vietnam 3. In 2024, the average life expectancy for this group reached 76.5 years, with women outliving men by more than 2.8 years 3. These middle-income nations exhibit a dual burden of disease, simultaneously managing lingering infectious diseases while experiencing rapid increases in non-communicable, age-related conditions like cardiovascular disease and stroke 151217.
The "Already Peaked Group" comprises 63 countries, including Japan, the United States, and much of Western Europe, where population sizes peaked prior to 2024 34. The average life expectancy in this highly industrialized cohort was 78.8 years in 2024 3. The sex gap here is pronounced; women average 82.0 years of life compared to 75.6 years for men 3. Japan represents the apex of global longevity, boasting a female life expectancy exceeding 88 years and a male life expectancy near 82 years, supported by the highest global male HALE at 74.1 years 181913. Conversely, the United States presents a unique crisis within the developed world, experiencing a marked slowdown in life expectancy gains even prior to COVID-19, driven by stagnant progress against cardiovascular diseases, rising obesity, and substance abuse - all of which disproportionately depress male life expectancy 1221.
Comparative Healthspan vs. Lifespan Analysis
To illustrate the morbidity-mortality paradox mathematically, it is necessary to compare total Life Expectancy (LE) against Healthy Life Expectancy (HALE). The difference between these two figures represents the absolute number of years lived in poor health or disability.
| Country | UN Demographic Classification | Male LE (Years) | Female LE (Years) | Male HALE (Years) | Female HALE (Years) | Expected Years in Poor Health (M / F) |
|---|---|---|---|---|---|---|
| Japan | Already Peaked | 82.0 | 88.0 | 74.1 | 76.4 | 7.9 / 11.6 |
| USA | Already Peaked | 76.5 | 81.4 | 66.1 | 68.2 | 10.4 / 13.2 |
| Brazil | Peaking (2025-2054) | 72.0 | 79.0 | 65.4 | 67.5 | 6.6 / 11.5 |
| Vietnam | Peaking (2025-2054) | 71.0 | 77.0 | 65.3 | 67.0 | 5.7 / 10.0 |
| India | Growing | 69.0 | 72.0 | 63.0 | 64.5 | 6.0 / 7.5 |
| Nigeria | Growing | 55.0 | 58.0 | 54.0 | 56.0 | 1.0 / 2.0 |
The data confirms a universal trend: while females possess longer total lifespans, they also endure a systematically higher number of years living with disease or disability.
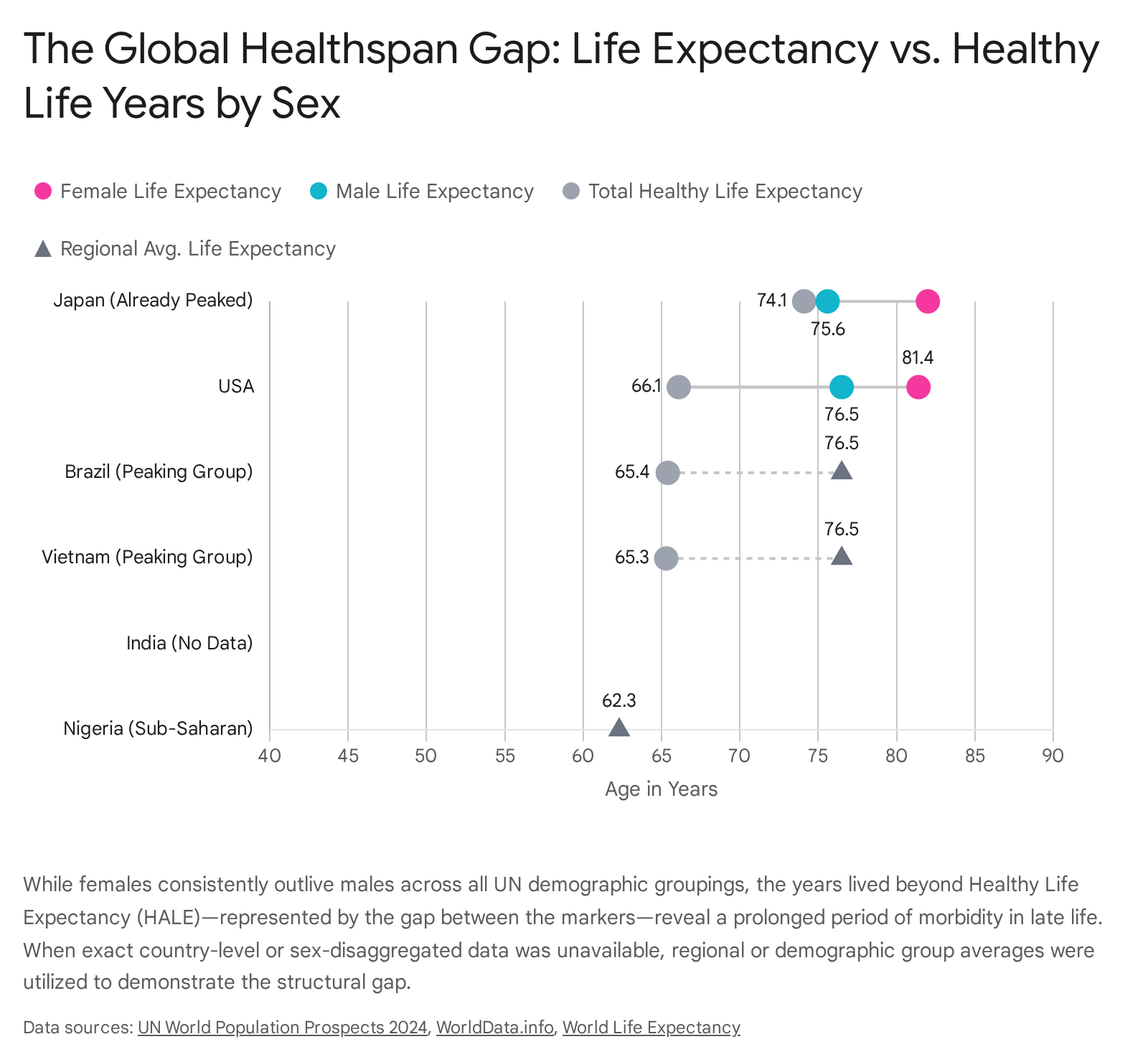
In advanced economies like the United States and Japan, women spend upwards of 11 to 13 years in suboptimal health, whereas men experience significantly compressed periods of late-life morbidity before succumbing to mortality.
The Morbidity-Mortality Paradox: Epidemiological Realities
The morbidity-mortality paradox represents a fundamental divergence in how human bodies deteriorate over time. From early adolescence through late middle age, females consistently report worse self-rated health and undergo more hospitalization episodes than men 2214. Yet, at almost every age interval, male mortality rates exceed female mortality rates, a trend that persists well into the 90-to-94-year age bracket 15.
Global Burden of Disease Disparities
A comprehensive systematic analysis of the Global Burden of Disease (GBD) Study 2021 provides the exact quantification of this health loss. Analyzing the top 20 causes of disease burden from 1990 to 2021 across seven world regions highlights that global disability-adjusted life years (DALYs) are strictly partitioned along sex lines 11.
Females bear an overwhelming burden of morbidity-driven conditions - non-fatal diseases that cause decades of chronic pain and disability. The largest absolute differences in DALYs disadvantaging females stem from musculoskeletal disorders, notably low back pain, which accounted for 478.5 more DALYs per 100,000 individuals among females than males globally in 2021 1011. Depressive disorders (348.3 more DALYs per 100,000) and headache disorders (332.9 more DALYs per 100,000) similarly afflict female populations at vastly higher rates 101116. Furthermore, because women survive longer, they are disproportionately subjected to late-life neurodegenerative declines; mortality and disability from Alzheimer's disease and other dementias among females nearly tripled between 2000 and 2021 1617. The female health penalty is largely defined by the gradual, non-lethal erosion of musculoskeletal and mental health 10.
Males, conversely, are heavily disadvantaged by mortality-driven conditions. The pathophysiology of the male body is highly susceptible to acute, fatal, and systemic failures. Ischemic heart disease presents the second-largest absolute sex difference globally, generating 1,611.8 more DALYs per 100,000 among males 1011. Men suffer 45% more health loss from heart disease than women, a gap that peaks drastically in Central and Eastern Europe where the male burden is 49% higher 10. Beyond cardiovascular vulnerability, males generate significantly higher DALYs from road injuries, stroke, chronic kidney disease, lung cancer, and infectious agents like COVID-19, which caused 1,767.8 more DALYs per 100,000 in men 101116.
Frailty Indices and Physiological Resilience
The application of deficit accumulation Frailty Indices (FI) further illuminates the mechanics of the paradox. A frailty index assesses physiological dysregulation by counting a broad spectrum of minor and major health deficits. Extensive meta-analyses incorporating tens of thousands of community-dwelling older adults uniformly conclude that females exhibit higher mean FI scores than males at all ages 152718.
The critical revelation of frailty modeling is the sex-specific response to accumulated damage. While women accumulate deficits faster and maintain a higher baseline of systemic frailty, men possess a radically lower threshold for lethal failure 1529.
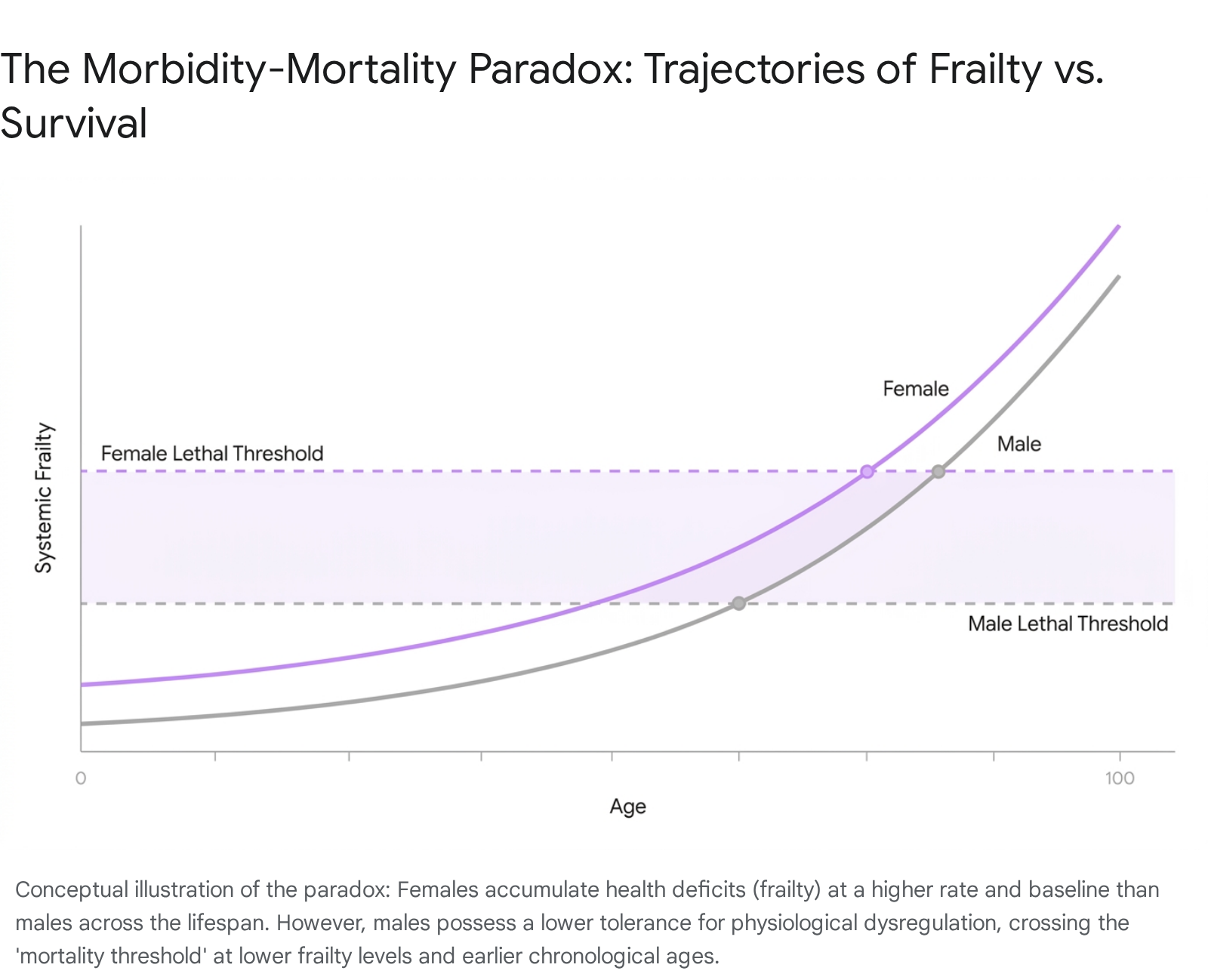
In clinical terms, a male with an FI score of 0.35 faces an exponentially higher near-term mortality risk than a female with the identical score 15. This phenomenon indicates that female physiology harbors an intrinsic resilience - an ability to tolerate high loads of systemic damage, immune dysregulation, and multimorbidity without progressing to organismal death 18. Preclinical research demonstrates that this female resilience extends beyond humans; across multiple strains of laboratory mice, females reliably generate higher frailty scores while outliving their male counterparts, indicating a deeply conserved mammalian trait 2718.
Molecular and Biological Clocks: 2023-2024 Epigenetic Insights
To decouple the effects of chronological time from true physiological decline, geroscience has increasingly relied on biological age estimators, chief among them being epigenetic clocks. Epigenetic clocks quantify age-related biological changes by measuring DNA methylation (DNAm) levels at specific cytosine-guanine (CpG) sites across the genome 31. As an organism ages, the epigenome typically undergoes a global loss of methylation (hypomethylation) in intergenic regions and targeted hypermethylation at specific promoter regions, leading to chromosomal instability and altered gene expression 19.
The ASPREE Cohort and Advanced Clocks
First-generation clocks (such as HorvathAge and HannumAge) were calibrated strictly against chronological age. However, second and third-generation clocks developed in recent years (including PhenoAge, GrimAge, GrimAge2, and DunedinPACE) are calibrated against clinical biomarkers, mortality risk, and the pace of physiological decline, offering a much more precise proxy for true biological aging 22021.
A highly significant 2024 study published in Geroscience utilized the ASPREE cohort - comprising 560 relatively healthy older adults aged 70 and above - to investigate sex differences using these advanced clocks 20. The study generated a system-wide Frailty Index using 67 health measures and estimated age acceleration (AA) across multiple epigenetic models. The findings definitively proved the molecular basis of the morbidity-mortality paradox. Despite females possessing a significantly higher Frailty Index than males, they consistently demonstrated dramatically reduced epigenetic age acceleration 20. Biologically, at the cellular level, the men were aging faster.
The study further identified profound sex differences in how epigenetic aging correlates with specific clinical conditions. In females, biological age acceleration (measured by GrimAA and Grim2AA) was most strongly associated with conditions like obesity and depression, linking directly to the high-morbidity, low-mortality DALY data 20. In males, however, age acceleration measured by GrimAA and DunedinPACE was inextricably linked to lethal, systemic failures, including diabetes, severe hypertension, and chronic kidney disease 20.
Conserved Epigenetic Mechanisms in Mammalian Populations
The sex divergence in epigenetic aging is not merely an artifact of modern human lifestyles. In a 2024 investigation of Mediterranean "Blue Zones" (Sardinia and Ikaria), regions celebrated for extreme longevity where men are as likely as women to reach centenarian status, epigenetic profiling revealed that male residents still exhibit greater age acceleration than their female counterparts . Even when behavioral and environmental factors allow men to match female chronological survival, their underlying biological clocks tick at a faster, more precarious rate .
Furthermore, 2023 research extended epigenetic clock methodology to wild, free-ranging mammalian populations. Developing bespoke clocks for species like the white-tailed deer, black bear, and mountain goat using over 30,000 CpG sites, researchers confirmed that methylation-based aging is a deeply conserved molecular mechanism 31. Across varying mammalian life histories, the universal DNA methylation clock demonstrates that the fundamental machinery of aging - and the sex-specific disparities in how that machinery degrades - operates independently of the human sociodemographic experience 3119. Studies on rhesus macaques utilizing high-throughput sequencing of liver and hippocampus tissues mirror human findings, confirming that age-associated CpG sites overwhelmingly undergo hypomethylation with advancing age in both sexes, but the rate and specific locus targeting remain strongly sex-associated 19.
Endocrine Dynamics and Ovarian Aging
The biochemical environment in which the epigenome operates is heavily regulated by sex steroid hormones. The protective effects of estrogen during the female reproductive years are well-documented. Estrogens exert highly favorable effects on cardiovascular hemodynamics, lipid profiles, and endothelial function, buffering women against ischemic heart disease and related metabolic syndromes during early and middle adulthood 1522.
However, the eventual exhaustion of the ovarian reserve fundamentally alters the trajectory of female aging. The age at natural menopause (ANM) serves as a critical inflection point in the female healthspan. The menopausal transition triggers a precipitous decline in circulating estrogens, precipitating a cascade of systemic physiological dysregulations. This hormonal withdrawal is associated with severe vasomotor symptoms, rapid bone mineral density deterioration, and an exponentially elevated risk for cardiovascular disease, insulin resistance, and neurodegeneration 22.
Recent genome-wide association studies (GWAS) and Mendelian randomization analyses highlight a strong genetic basis for ANM, identifying over 200 genetic loci - many intimately linked to DNA damage response genes (e.g., ETAA1, PALB2) - that dictate the rate of ovarian aging 22. The punctuated nature of female reproductive senescence creates a distinct biphasic aging profile. While men experience a gradual, linear decline in androgens resulting in steady somatic deterioration, women maintain an artificially protected biological state until menopause, after which they suffer an accelerated accumulation of chronic morbidities 2236. Consequently, ovarian aging acts as a principal catalyst for the morbidity phase of the morbidity-mortality paradox, guaranteeing that the extended years of female life are disproportionately burdened by chronic disease 22.
Evolutionary Frameworks of Sex-Dimorphic Aging
To comprehend why the genomes and epigenomes of males and females execute distinct aging programs, one must evaluate the biological imperatives that shaped them. Evolution does not prioritize post-reproductive longevity; it optimizes organisms for successful reproduction and the propagation of genetic material. Because males and females utilize vastly divergent strategies to achieve reproductive success, natural selection has engineered different somatic architectures for each sex.
The Disposable Soma Theory and Terminal Investment
The "Disposable Soma" theory provides the foundational logic for sex-dimorphic aging. The theory posits that every organism possesses a finite energy budget that must be strategically partitioned between somatic maintenance (cellular repair, antioxidant defense, immune function) and reproductive effort 36. Because evolution favors reproduction, the body (the soma) is biologically "disposable" once genetic transmission is complete.
In mammals, the energetic costs of reproduction are highly asymmetrical. Females must invest massive, sustained biological resources into gestation, lactation, and the extended rearing of altricial young. To ensure the survival of her offspring, evolutionary pressure mandates that the female somatic machinery be highly durable, requiring intense investment in DNA repair pathways, robust immune responses, and cellular maintenance over a long, protected reproductive window 36.
Conversely, in mammalian mating systems characterized by mild to severe polygyny, male reproductive success hinges primarily on intra-sexual competition - securing access to mates through physical dominance, rapid growth, and the maintenance of high androgen levels 36. This evolutionary strategy demands a massive early-life investment in physical performance and sexually selected traits at the direct expense of long-term somatic maintenance 36. From a Darwinian perspective, investing cellular energy into late-life DNA repair is wasted on a male whose reproductive success relies entirely on out-competing rivals in his youth. Consequently, male biology is intrinsically programmed for faster senescence, driving the accelerated epigenetic clocks and lower mortality thresholds observed in human demographic data 152036.
Genomic Architectures: The Unguarded X Hypothesis
At the structural level of the genome, the "Unguarded X" hypothesis offers a mechanistic explanation for heightened male vulnerability. In humans and most mammals, females are the homogametic sex (XX), possessing two X chromosomes, while males are the heterogametic sex (XY). The male Y chromosome is highly degenerate, containing very few functional genes 3738.
This genetic asymmetry leaves the male X chromosome "unguarded." If a male inherits a recessive, deleterious mutation on his single X chromosome, it is entirely phenotypically expressed because he lacks a healthy backup copy to mask the defect 3739. Females, possessing two X chromosomes, benefit from cellular mosaicism and redundancy, which effectively buffers their physiology against the continuous accumulation of minor, life-shortening X-linked genetic errors 38. A massive 2024 meta-analysis encompassing 229 species across 99 families verified that the homogametic sex (XX in mammals, ZZ in birds) consistently outlives the heterogametic sex, confirming that asymmetrical chromosome inheritance is a fundamental driver of sexually dimorphic lifespan across the tree of life 3840.
Mitonuclear Interactions and the "Mother's Curse"
The third evolutionary pillar explaining the sex gap centers on mitochondrial inheritance. Mitochondria, the organelles responsible for cellular energy production, maintain their own distinct circular genome (mtDNA). Crucially, mtDNA is inherited in a strictly matrilineal fashion; fathers do not pass their mitochondria to their offspring 3740.
Because natural selection operates only on genes passed to the next generation, selection pressure is entirely "blind" to mtDNA mutations that harm only males. If a mitochondrial mutation severely impairs male lifespan or fertility, but remains neutral or mildly beneficial to females, it will freely accumulate and spread through the population because its negative effects on males do not impede its transmission through daughters 3940.
This phenomenon, termed the "Mother's Curse," essentially dictates that mitochondrial function is evolutionarily optimized exclusively for female physiology 39. Because mitochondria are the primary generators of reactive oxygen species (ROS) and cellular oxidative stress, sub-optimal mitochondrial efficiency in males leads to a systemic accumulation of free radical damage, particularly within high-energy tissues like the brain and myocardium 40. Studies on Tigriopus californicus, a species completely lacking sex chromosomes, demonstrate that complex mitonuclear incompatibilities alone are sufficient to drive male-biased mortality, proving that the Mother's Curse independently accelerates male biological aging in the absence of an Unguarded X 40.
Vanguard Therapeutics: Sex-Dimorphic Responses to Rapamycin
As biogerontology pivots from observing aging to actively designing therapeutics to delay it, the integration of sex-specific biology is paramount. The mechanistic target of rapamycin (mTOR) signaling pathway serves as a master regulator of cellular metabolism, growth, and nutrient sensing. Pharmacological inhibition of this pathway using the drug rapamycin is currently the most robust, reproducible intervention known to extend lifespan and delay age-related diseases across multiple animal models 3623. However, recent data highlights that rapamycin's mechanism of action is profoundly sex-dimorphic.
A groundbreaking 2024 transcriptomic analysis mapped the effects of rapamycin on kidney podocytes - highly specialized, post-mitotic cells critical for renal filtration that rely heavily on precise mTOR signaling 23. Under normal, untreated physiological conditions, male and female podocytes exhibit entirely distinct transcriptomic signatures, clustering far apart in genetic principal component analyses 23.
Following systemic mTOR inhibition with rapamycin, female podocyte transcriptomes demonstrated extreme stability, exhibiting significant differential expression (DE) in only two genes 23. The male transcriptome, however, underwent a massive restructuring. Rapamycin induced significant differential expression in 119 genes within male cells, predominantly affecting biological processes related to mitochondrial energy machinery, endocytosis, and anabolic protein synthesis 23.
Crucially, rapamycin treatment effectively abolished the natural sex-specific clustering. It achieved this not by altering females, but by violently shifting the male transcriptomic profile to align with the female baseline. Approximately 87% of the genes significantly upregulated by rapamycin in males were classified as intrinsically "female-biased" genes under normal conditions, while 66% of the downregulated genes were intrinsically "male-biased" 23.
This transcriptomic reprogramming was regulated by 83 identified sexually dimorphic transcriptional regulators, including Kdm5a, Rb1, Xbp1, and Ppargc1α 23. Interaction analyses identified specific transcription factors like Mrtfb, Cnbp, and Stat6 that were radically upregulated in males but downregulated in females in response to the drug 23. Furthermore, the intervention altered the expression of estrogen (ESR1, ESR2) and androgen receptors, and forced higher phosphorylation of AMPK (a critical metabolic guardian) specifically in males, demonstrating a stronger male dependence on intact mTOR signaling to maintain homeostasis 23.
These 2024 findings confirm a paradigm-shifting reality for geroscience: male and female cells utilize fundamentally distinct metabolic strategies to navigate stress and aging. Interventions like rapamycin appear to extend male lifespan precisely by overriding the resource-intensive, evolutionarily programmed male metabolic signaling (dictated by the Disposable Soma theory) and forcing the male cellular machinery into a more resilient, "female-like" state of maintenance and repair 3623.
Conclusion
The divergence in human aging is an intricate biological tapestry woven from evolutionary imperatives, genomic architectures, and endocrine shifts. As confirmed by the 2026 JAMA analysis and extensive demographic data from the UN and WHO, the sex survival gap is not an artifact of modern behavior, but a fundamental biological reality 13. Women consistently outlive men globally, yet they are subjected to a profound morbidity-mortality paradox, enduring extended decades of physical frailty, musculoskeletal pain, and neurological decline while remaining remarkably resilient against the acute, fatal system failures that claim male lives prematurely 101118.
The underlying mechanisms are deeply conserved across the mammalian class. While men suffer from the evolutionary legacy of the Unguarded X chromosome and the Mother's Curse - driving accelerated epigenetic clocks and oxidative mitochondrial decay - women are protected by genetic redundancy and estrogenic shielding until the punctuated disruption of menopause 20223839.
The future of aging research and clinical intervention must fully embrace this biological dimorphism. The transcriptomic response to rapamycin proves that therapeutics cannot be applied uniformly across the sexes; extending healthspan requires recognizing that male and female cells require distinctly different metabolic manipulations to achieve longevity 23. Ultimately, understanding the sex differences in aging is not merely an epidemiological exercise, but the essential key to unlocking precision geroscience and improving both the length and quality of human life.