Biological and Psychological Mechanisms of Weight Regain
The long-term maintenance of lost weight remains the preeminent challenge in the clinical management of obesity. While behavioral, pharmacological, and surgical interventions consistently produce initial reductions in body mass, the overarching trajectory of diet-induced weight loss is characterized by an early phase of rapid reduction, a plateau occurring between six and twelve months, and a prolonged phase of progressive weight regain 123. Meta-analyses of long-term weight loss studies reveal that more than half of lost weight is typically regained within two years, and by five years, over 80% of the lost weight is recovered by the majority of individuals 45.
This near-ubiquitous phenomenon of weight recidivism is not an indicator of personal failure, moral failing, or a lack of psychological willpower, but rather the result of a coordinated, highly evolved physiological and psychological response to perceived energy deprivation 166. Biological, behavioral, and environmental factors conspire to resist weight loss, driving the body toward its baseline adiposity through profound metabolic adaptations, sustained endocrine alterations, and the onset of behavioral fatigue 16. Understanding the exact mechanisms behind this failure requires examining the theoretical models of body weight regulation, the specific neuroendocrine responses to caloric restriction, the psychological burdens of long-term behavioral maintenance, and the longitudinal data governing modern obesity treatments.
Theoretical Models of Body Weight Regulation
To understand why weight loss reverses, researchers rely on theoretical models that attempt to explain the homeostatic mechanisms governing human adiposity. The close correspondence between prolonged energy intake and expenditure, coupled with the body's apparent defense of fat stores during caloric restriction, indicates the presence of a biological regulatory system 7810. However, the exact nature of this regulation remains a subject of ongoing scientific debate, leading to several distinct frameworks that attempt to map the interplay of genetics and environment.
The Set-Point and Settling Point Models
Historically, the set-point model has dominated the discussion of body weight regulation. Rooted in genetics, molecular biology, and physiology, this model suggests the existence of an active feedback mechanism linking stored energy in adipose tissue to food intake and energy expenditure via a specific, centrally encoded reference point in the brain 7811. When body fat drops below this set-point, the body initiates compensatory mechanisms to decrease energy expenditure and increase appetite, effectively driving the individual back to their starting weight 910.
While the set-point model effectively explains the biological resistance to weight loss and the phenomenon of post-diet hyperphagia, it struggles to account for the impact of social and environmental influences 78. Most importantly, the set-point model fails to explain the modern obesity epidemic, which has seen a widespread and profound increase in body weight across populations since the 1980s 7811. If the body tightly defends a specific target weight, the vast upward shift in population body mass indices contradicts the existence of a rigid, universal biological set-point 812.
An alternative framework, the settling point model (often associated with control theory models), proposes that body weight is not actively defended at a specific target but rather stabilizes at a dynamic equilibrium 7109. In this model, weight is the result of passive feedback between the size of body stores and environmental factors such as food availability and physical activity 1011. While the settling point model accommodates the reality of the obesogenic environment and explains population-level weight gain, it is criticized for inadequately explaining the fierce, genetically driven biological adaptations that occur when individuals attempt to restrict calories, which appear far more active than passive 7813.
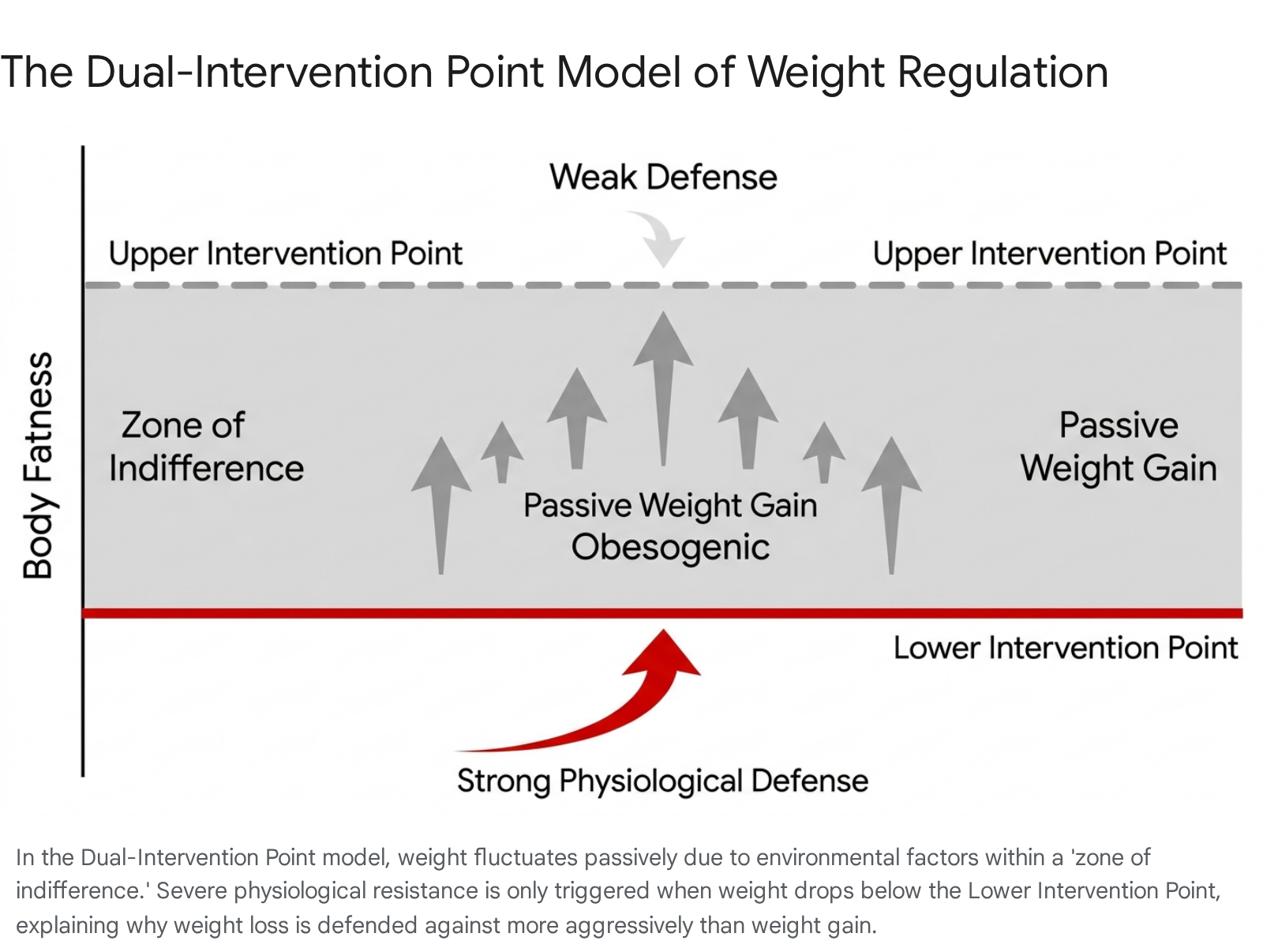
The Dual-Intervention Point Model
To reconcile the limitations of both the set-point and settling point frameworks, contemporary researchers have increasingly adopted the Dual-Intervention Point (DIP) model 91415. The DIP model theorizes that rather than a single fixed set-point, humans possess two boundaries: a lower intervention point and an upper intervention point 914.
Below the lower intervention point, active physiological feedback mechanisms aggressively kick in to prevent starvation 912. These mechanisms include massive reductions in total daily energy expenditure and spikes in extreme hunger, mimicking states of low energy availability 1215. Above the upper intervention point, physiological mechanisms theoretically engage to prevent excessive fat accumulation. Evolutionarily, this upper boundary would have been dictated by the risk of predation, as excessive body mass would impair an organism's ability to flee 1415.
Crucially, the space between these two boundaries is termed the zone of indifference 91415. Within this zone, physiological regulation of body weight is weak or absent, and body weight fluctuates almost entirely based on environmental factors, social cues, and passive energy balance 79. Because the risk of predation has been functionally eliminated in modern human societies, the evolutionary pressure to maintain a strict upper intervention point is exceptionally weak 1415. The "drifty gene hypothesis" suggests that random genetic drift has led to significant individual variation in where this upper boundary lies 1315.
For individuals with a genetic predisposition to obesity, the upper intervention point is located at an exceedingly high level of adiposity, allowing the modern obesogenic environment to passively drive their weight up through the zone of indifference without triggering significant biological resistance 1315. Conversely, the lower intervention point remains strictly defended by highly conserved evolutionary survival mechanisms, explaining why weight loss is fiercely resisted while weight gain is easily tolerated 1315.
| Theoretical Model | Core Regulatory Mechanism | Explanation of Obesity Epidemic | Explanation of Weight Loss Resistance |
|---|---|---|---|
| Set-Point Model | Active feedback loop linking adipose tissue to a specific brain-encoded target weight. | Deficient; struggles to explain why population set-points have uniformly risen simultaneously. | Comprehensive; predicts profound hyperphagia and metabolic slowing when dipping below the set-point. |
| Settling Point Model | Passive equilibrium between body stores and environmental energy availability. | Comprehensive; attributes population weight gain to an increasingly obesogenic environment. | Deficient; struggles to explain the fierce, persistent biological counter-regulatory responses to dieting. |
| Dual-Intervention Point Model | Body weight is weakly regulated between an upper and lower boundary (zone of indifference). | Comprehensive; individuals drift upward through the zone of indifference due to environmental cues. | Comprehensive; dieting pushes weight into the lower boundary, triggering aggressive starvation defenses. |
Physiological Mechanisms of Weight Regain
When an individual successfully loses weight, the body perceives the loss of adipose tissue not as a health benefit, but as a critical threat to survival 15. The resulting physiological counter-regulatory response involves both a reduction in energy expenditure, known as metabolic adaptation, and a profound shift in endocrine signaling that drives hyperphagia, or increased appetite 15616.
Metabolic Adaptation and Adaptive Thermogenesis
Caloric restriction and the subsequent loss of body mass invariably lead to a reduction in total energy expenditure. While a portion of this reduction is mathematically expected due to the simple fact that a smaller body mass requires fewer calories to move and maintain basic cellular functions, clinical research demonstrates that the drop in resting metabolic rate often far exceeds the expected decline based on changes in body composition alone 1616. This phenomenon is clinically referred to as metabolic adaptation or adaptive thermogenesis 617.
Estimations indicate that for each kilogram of lost weight, daily caloric expenditure decreases by approximately 20 to 30 kilocalories per day, while appetite simultaneously increases by about 100 kilocalories per day above baseline levels 1. In extreme scenarios of massive weight loss, such as those observed in the participants of a televised weight-loss competition, resting metabolic rate was found to decrease by roughly 600 calories per day by the conclusion of the intervention 6. A comprehensive six-year longitudinal follow-up study on these participants revealed that this metabolic adaptation persisted long-term. Despite regaining a vast majority of the lost weight, participants' resting metabolic rates remained 700 calories below their original baseline 6.
This persistent metabolic efficiency dictates that a formerly obese individual who has successfully reduced their weight to a standard body mass index must consume significantly fewer calories to maintain that weight than an individual of the exact same body composition who never experienced obesity 11618. The reduction in non-resting energy expenditure also contributes, as individuals unconsciously reduce non-exercise activity thermogenesis (NEAT) when subjected to prolonged caloric deficits 519.
Alterations in Endocrine and Satiety Signaling
In tandem with metabolic slowing, weight loss triggers a highly coordinated endocrine response originating from the gastrointestinal tract, the pancreas, and adipose tissue itself, which acts upon the hypothalamus to regulate food intake and energy balance 1618.
As adipose tissue shrinks, the secretion of leptin plummets. Leptin is a long-term satiety hormone that acts as a primary physiological guard of the lower intervention point 61520. This drop in circulating leptin is perceived by the central leptin-melanocortin system as a starvation signal, drastically amplifying hunger drives 1015. Concurrently, the secretion of short-term satiety hormones - including peptide YY (PYY), cholecystokinin (CCK), amylin, and glucagon-like peptide-1 (GLP-1) - decreases significantly 61620. Concomitant with the drop in satiety signals, levels of ghrelin, the primary orexigenic or appetite-stimulating hormone produced in the stomach, rise sharply 620.
Clinical trials robustly demonstrate that these hormonal alterations are not merely transient responses to acute caloric deficits. Studies evaluating overweight individuals subjected to a ten-week very-low-energy diet have shown that significant deviations in leptin, PYY, CCK, insulin, and ghrelin persist for at least one year following the initial weight loss 616. The Erasmus Medical Center conducted a study on a combined lifestyle intervention tracking long-term adiposity-related hormones, confirming that initial weight loss resulted in decreased leptin and insulin levels that persisted through 1.5 years of treatment 21. The failure of these homeostatic signals to normalize keeps the individual in a state of chronic biological hunger, establishing a persistent physiological drive toward weight regain 621.
| Hormone | Primary Source | Effect of Weight Loss | Biological Impact on Weight Maintenance |
|---|---|---|---|
| Leptin | Adipose Tissue | Severe Decrease | Signals energy depletion to the central nervous system; reduces satiety; lowers resting metabolic rate. |
| Ghrelin | Stomach | Significant Increase | Directly stimulates hunger via the hypothalamus; promotes fat storage; increases the reward value of food. |
| Peptide YY (PYY) | Intestines | Decrease | Reduces post-meal fullness; delays the termination of eating episodes. |
| Cholecystokinin (CCK) | Duodenum | Decrease | Impairs the digestion-driven signaling of satiety to the brain. |
| GLP-1 | Intestines | Decrease | Reduces satiety; alters glucose-dependent insulin secretion pathways. |
| Insulin | Pancreas | Decrease | Reflects lowered fat mass; shifts systemic metabolism toward nutrient conservation. |
The Timeline of Physiological Adaptation
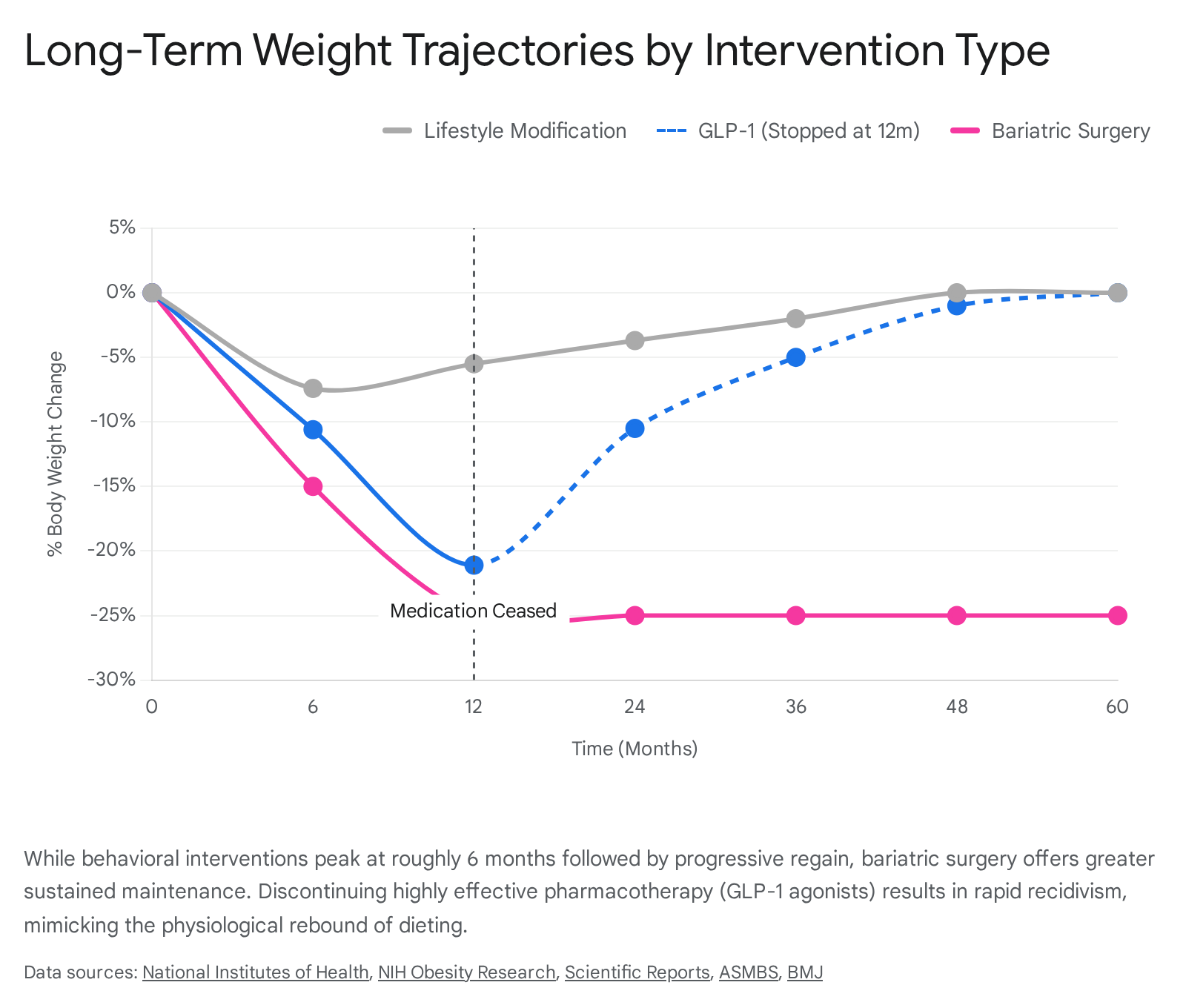
The typical trajectory of non-surgical weight loss interventions clearly reflects these biological constraints. Diet and lifestyle interventions consistently induce rapid weight loss during the first three to six months 225. However, between three and six months, the rate of weight loss slows dramatically, ultimately resulting in a rigid weight plateau 2418.
While a portion of researchers historically attributed early plateaus to a psychological element termed "diet fatigue" - suggesting that participants simply become bored and lessen their adherence - controlled crossover trials indicate the six-month plateau is heavily physiologically mediated 226. Clinical studies in which participants switch between a low-fat and a low-carbohydrate diet exactly at the six-month plateau point fail to reinvigorate the rate of weight loss, indicating that the plateau is driven by profound metabolic and endocrine adaptations rather than mere dietary disinhibition with a specific macronutrient profile 2.
Following this static phase, the exponential rise in appetite, which mathematical models estimate rises by 400 to 600 kilocalories per day above baseline, eventually overpowers the individual's ability to maintain dietary restriction 145. The continuous biological pressure eventually forces a positive energy balance, and progressive weight regain commences, continuing steadily over the subsequent two to five years 145.
Psychological and Behavioral Drivers of Regain
While biological counter-regulation provides the foundation for weight regain, long-term weight maintenance is ultimately a behavioral challenge 11722. To sustain weight loss, an individual must execute daily, effortful behaviors to counteract a physiology that is actively fighting against them 16. This chronic psychological burden leads to several recognized failure points in the maintenance phase.
Behavioral Fatigue and Cognitive Load
Behavioral fatigue refers to the progressive depletion of cognitive and emotional resources required to sustain novel, restrictive health behaviors over prolonged periods 1417. During the initial phases of weight loss, individuals are highly motivated by the external rewards of the process - primarily, the visible reduction in body weight on the scale and explicit improvements in clinical markers such as lipid levels or blood pressure 14.
However, during the weight maintenance phase, these explicit rewards vanish. The individual is required to expend the same, or greater, level of cognitive effort regarding food tracking, physical activity, and portion control, merely to keep the scale exactly where it is 14. Without the reinforcing stimulus of continued weight loss, the intensive self-regulation required to battle elevated ghrelin and suppressed leptin becomes cognitively exhausting 14. Cognitive load increases exponentially as real-world complexities - stress, work obligations, and social environments - compete for executive function resources 2823. When executive function is depleted, automatic, habitual behaviors, which inherently favor caloric consumption in an obesogenic environment, override top-down, goal-directed dietary restraint 2324.
Neurobiology of Hedonic Hunger and Emotional Regulation
Weight loss alters not only homeostatic eating, which is eating for baseline energy requirements, but also non-homeostatic, or hedonic, eating 2526. Psychological distress and emotional regulation difficulties have been shown to severely exacerbate weight regain 2627. Negative emotions, stress, and anxiety drive individuals to alleviate their feelings by seeking high-calorie, high-fat foods 2829.
Neurobiological imaging demonstrates that specific personality traits and emotional patterns are associated with changes in brain activity governing reward and self-control 2526. When the physiological state of energy deprivation is combined with psychological distress, the brain's reward circuits heighten the salience of palatable food cues, creating immense susceptibility to cue-based eating 2630. An individual experiencing behavioral fatigue is therefore fighting a dual battle: a homeostatic neuroendocrine drive to consume calories, and an amplified hedonic reward associated with energy-dense foods 172530. In postoperative bariatric cohorts, for instance, binge eating severity and high levels of stress were identified as the strongest predictors of weight regain, with significant binge eating increasing the risk of weight recidivism by a factor of 2.41 28.
Cognitive Flexibility and Coping Mechanisms
Maladaptive cognitive patterns play a significant role in weight recidivism. A primary psychological factor in weight regain is dichotomous, or "all-or-nothing," thinking 127. Many individuals attempting weight loss establish rigid rules, such as eliminating entire macronutrient groups or adhering to strict fasting windows 131. When a minor lapse inevitably occurs - driven by biological hunger or social pressure - dichotomous thinking frames this lapse as a total failure of the intervention 127.
This rigid cognitive framework leads to a cascade of negative internal experiences, reduced self-efficacy, and ultimately a complete abandonment of the dietary regimen 12732. Research on the psychology of weight control emphasizes that successful maintainers possess cognitive flexibility, allowing them to rapidly self-correct following a lapse without internalizing failure or succumbing to behavioral fatigue 133. Furthermore, individuals who maintain weight loss demonstrate high levels of self-efficacy specifically regarding physical activity, allowing them to sustain high energy expenditure even when motivation to diet wanes 2233.
Outcomes from Global Longitudinal Studies
To quantify the failure of chronic dieting and the challenges of weight maintenance, researchers rely on massive longitudinal cohorts and randomized controlled trials spanning diverse global populations. The aggregate data unequivocally demonstrates that behavioral weight loss maintenance is elusive regardless of geography or demographic.
Lifestyle and Behavioral Interventions
The Action for Health in Diabetes (Look AHEAD) trial represents one of the largest and longest randomized controlled trials of intensive lifestyle interventions. Focused on adults with type 2 diabetes and obesity, the trial demonstrated that participants could achieve significant initial weight loss, averaging 5% or more in the first year, through intensive counseling, meal replacements, and physical activity 3435. However, progressive regain was universal across the cohort. By the eight-year mark, while some subgroups, such as non-Hispanic white participants and minority women, maintained slightly better results, the vast majority of the weight was regained 34. In a 12-to-16-year follow-up of a subset of Look AHEAD participants undergoing dual-energy X-ray absorptiometry scans, researchers noted minimal long-term differences in female body mass between the intensive lifestyle group and the standard education group, indicating the eventual erosion of behavioral interventions over a decade 36.
Similar outcomes are observed internationally. A longitudinal study using the ELSA-Brasil cohort tracked the metabolic status of individuals with obesity over a mean follow-up of 7.7 years. The data revealed that even among those who started as "metabolically healthy obese," 75.8% transitioned to metabolically unhealthy statuses, highlighting the difficulty of maintaining behavioral health markers long-term without sustained physiological support. Variables such as alcohol use and increasing waist circumference were identified as primary risk factors for this metabolic deterioration 37. Similarly, multi-trajectory dietary pattern analyses from the China Health and Nutrition Survey between 1997 and 2015 underscore that unless rigid low-carbohydrate and low-fat patterns are flawlessly sustained across decades, the risk of falling into adverse adiposity trajectories remains exceptionally high 38. In a Japanese randomized controlled trial focused on metabolic syndrome, participants receiving group-based support initially lost significantly more weight than controls at six months, but the weight difference reduced by half at one year and entirely disappeared by year two, proving the transient nature of standard educational interventions 39.
Efficacy of Extended Care Models
To combat behavioral fatigue and improve long-term outcomes, researchers have attempted to extend clinical support into the maintenance phase. Traditional programs cease active intervention after six months, prompting the hypothesis that continued contact might prevent relapse 1440.
The Support and Tracking to Achieve Results (Project STAR) trial investigated whether providing "adaptive" telephone-based extended care - triggered by algorithmic assessments of an individual's high-risk periods for weight regain based on self-monitoring behaviors - would outperform a static, once-per-month support schedule 4142. Tracking 255 adults over 24 months, the study found that adaptive extended care did not significantly reduce weight regain compared to static care. From month four to month 24, both the adaptive and static groups regained between 1.27 kilograms and 1.75 kilograms of their initially lost weight 41. While maintaining extended contact does produce a modest reduction in weight regain compared to completely abandoning care, the absolute effect sizes remain minor, suggesting the need for novel psychological or pharmacological approaches 4142. Real-world observational data from digital behavior change programs, such as Noom Weight, suggest that while users can maintain a portion of weight loss (averaging 65% of initial loss maintained at one year and 57% at two years), continuous engagement and habitual behavior logging are absolute prerequisites for this success 43.
Pharmacological and Surgical Interventions
Because the biological imperative to regain weight is so strong that it routinely overwhelms purely behavioral strategies, medical science has turned to pharmacotherapy and metabolic surgery to fundamentally alter the underlying physiology of body weight regulation 16.
GLP-1 Receptor Agonists and Metabolic Outcomes
The advent of GLP-1 receptor agonists, including semaglutide and tirzepatide, has revolutionized obesity treatment by pharmaceutically overriding the neuroendocrine starvation response. These medications mimic endogenous glucagon-like peptide-1, drastically slowing gastric emptying and signaling profound, continuous satiety to the central nervous system 350.
Clinical trials show extraordinary efficacy compared to lifestyle interventions. Where diet and exercise average a 5% to 10% weight loss, pharmacotherapy with semaglutide yields approximately 15% weight loss, and tirzepatide produces up to 22.5% weight loss over extended periods 335. Furthermore, a longitudinal analysis of prescribing patterns from 2017 to 2024 demonstrates a massive public shift toward these medications, reflecting their clinical success and widespread adoption 51.
However, the biological drive to regain weight remains completely intact beneath the pharmaceutical suppression. A systematic review and meta-analysis of trials examining the cessation of weight-management medications found that once GLP-1 therapy is stopped, participants experience rapid and aggressive weight regain. Patients recover an average of 0.3 to 0.4 kilograms per month, resulting in the regain of roughly 50% of the lost weight within a single year 344. This rapid recidivism occurs entirely independent of the amount of weight initially lost, proving that pharmacotherapy suppresses, rather than cures, the biological adaptations to weight loss 44. Furthermore, all beneficial cardiometabolic markers achieved during weight loss are projected to return to baseline within 1.4 years of medication cessation 44.
Bariatric and Metabolic Surgery Durability
Bariatric and metabolic surgeries, such as Roux-en-Y gastric bypass, sleeve gastrectomy, and one-anastomosis gastric bypass, fundamentally alter gastrointestinal anatomy. These structural changes lead to an immediate and sustained suppression of ghrelin, largely due to the removal or bypass of the gastric fundus, alongside an elevation of satiety hormones like GLP-1 and CCK 1845.
Systematic reviews of surgical outcomes spanning up to ten years demonstrate that surgery achieves the most substantial and enduring weight loss of any modality, typically maintaining around 20% to 25% body weight reduction 345.
A systematic review of 22 studies evaluating one-anastomosis gastric bypass across 14,692 patients confirmed high rates of weight loss and metabolic improvement in the long term, though careful nutritional monitoring remains essential to mitigate risks of anemia and hypoalbuminemia 46.
However, even surgical intervention is not completely immune to biological adaptation and behavioral failure. Middle Eastern cohort studies on sleeve gastrectomy reveal that while average BMI drops significantly within the first year, weight regain occurs in nearly 45.5% of patients postoperatively 55. Broader clinical data reflects this, showing that nearly 50% of bariatric patients show some degree of weight regain by year two, and 60% by year five 47. This regain is heavily influenced by the resurgence of maladaptive behavioral patterns, specifically binge eating and stress-induced emotional eating 28.
| Intervention Modality | Average Maximum Weight Loss | Timeline to Plateau | Long-Term 5-Year Efficacy Profile |
|---|---|---|---|
| Lifestyle Modification (Diet & Exercise) | 5% to 10% | 6 Months | High failure rate; >80% of lost weight is regained by the 5-year mark. |
| GLP-1 Pharmacotherapy (Continuous Use) | 15% to 22.5% | 12 to 18 Months | High maintenance only if the medication is continued indefinitely. |
| GLP-1 Pharmacotherapy (Ceased at 1 Year) | 15% to 22.5% | N/A | Rapid recidivism; ~50% of lost weight is regained within 12 months post-cessation. |
| Metabolic/Bariatric Surgery | 25% to 35% | 12 to 24 Months | Highest durability; most patients maintain >20% loss, though partial regain remains common. |
Clinical Strategies for Weight Loss Maintenance
Given the formidable biological and psychological barriers to weight maintenance, clinical paradigms are fundamentally shifting. The medical community increasingly recognizes that short-term "dieting" is an inadequate framework for a chronic condition, transitioning instead toward lifelong, multi-faceted disease management models 6.
Acceptance-Based Behavioral Therapy
Standard cognitive behavioral therapies for weight loss emphasize self-monitoring, goal setting, and stimulus control 30. However, these approaches often fail during the maintenance phase when motivation wanes and environmental triggers overwhelm cognitive restraint 30. As a result, clinical psychology has increasingly utilized Acceptance-Based Behavioral Therapy (ABT) for obesity management 3032.
ABT operates on the premise that the internal sensations associated with maintaining weight loss - specifically, biological hunger, feelings of deprivation, fatigue, and the physical discomfort of exercise - are highly aversive 3032. Rather than attempting to suppress or eliminate these feelings, ABT teaches distress tolerance and experiential acceptance 3032. Patients are trained to acknowledge and accept the physical sensations of hunger or cravings without automatically reacting to them 32.
Crucially, ABT pairs this distress tolerance with deep values clarification. This process encourages patients to link their moment-to-moment behavioral choices, such as resisting a highly palatable food, to core life values rather than relying on the fleeting external motivation of scale weight 3032. Clinical trials evaluating ABT have shown improved maintenance outcomes. In an open trial utilizing a 12-week ABT intervention, participants exhibited significant improvements in cognitive restraint and eating-related experiential acceptance, maintaining a 9.6% weight loss at a six-month follow-up 2432. Other studies indicate that participants receiving ABT exhibit greater psychological flexibility and are significantly more likely to maintain a 10% weight loss at three-year follow-ups compared to those receiving standard behavioral therapy alone 303247.
Physical Activity and Metabolic Preservation
While dietary restriction is the primary driver of acute weight loss, physical activity is universally recognized as the strongest behavioral predictor of long-term weight loss maintenance 163947. Data from the National Weight Control Registry, a database tracking thousands of individuals who have successfully maintained a weight loss of 10% or more for over a year, indicates that nearly all successful maintainers engage in exceptionally high levels of physical activity, often amounting to an hour or more per day 33.
Biologically, high volumes of physical activity, particularly resistance training, help mitigate metabolic adaptation by preserving lean muscle mass, thereby supporting a higher resting metabolic rate and preventing sarcopenic obesity 484959. A cohort study examining participants categorized by sarcopenic obesity status found that those with lower lean mass faced significantly more difficulties maintaining achieved weight loss past the 12-month mark 48. Psychologically, regular exercise has been shown to improve mood, reduce depressive symptoms, and subsequently lower the incidence of anxiety-driven emotional eating 22. Exercise-induced improvements in self-regulation and self-efficacy appear to carry over directly into eating behaviors, creating a positive behavioral feedback loop that effectively combats behavioral fatigue 22.
Nutritional Timing and Composition
Emerging evidence suggests that manipulating macronutrient composition and nutritional timing can partially offset the physiological drive to regain weight. While the myth of "starvation mode" leading to complete metabolic cessation is frequently exaggerated in popular media, clinical data confirms that high-protein diets can improve satiety and preserve lean tissue during weight loss, making them highly effective for long-term maintenance 5049. Furthermore, intermittent energy restriction and aligning meal patterns with the body's circadian rhythm have demonstrated potential for enhancing dietary adherence and optimizing metabolic responses, providing tools beyond continuous daily caloric restriction 1259.
Ultimately, chronic dieting fails because the standard intervention relies on temporary behavioral changes to fight a permanent physiological adaptation 618. To improve clinical outcomes, expectations among both healthcare providers and patients must be recalibrated. Weight loss of 5% to 10% - while frequently viewed as a failure by patients seeking cosmetic ideals - is deeply clinically significant, yielding impressive health improvements and risk factor reductions 14. Recognizing obesity as a chronic, relapsing neurobiological disease rather than a deficit in personal willpower is essential 618. Strategies for maintenance must acknowledge the permanent biological shift that occurs upon weight loss, embracing the reality that the physiological and psychological forces driving weight regain require lifelong, comprehensive management.