Biological mechanisms and applications of hormesis
The foundational dogma of classical toxicology, famously distilled from the 16th-century physician Paracelsus, asserts that "the dose makes the poison." For decades, regulatory and biological sciences interpreted this maxim through a linear or strict threshold-based paradigm, operating under the assumption that lower exposures to a hazardous compound or environmental stressor invariably generate lower risks or lesser physiological impacts 1. However, advancements in molecular biology, biogerontology, and radiotoxicology have systematically illuminated a more complex, adaptive biological reality defined as hormesis.
Hormesis describes a highly conserved, biphasic dose-response phenomenon wherein low doses of a stressor - whether chemical, physical, or metabolic - provoke a beneficial, adaptive response in an organism, while high doses result in systemic toxicity, cellular damage, and death 234. Rather than merely inflicting a reduced level of damage, sub-lethal stress actively triggers complex cellular repair, maintenance, and defense pathways. By inducing a state of mild biological perturbation, hormetic stressors condition the organism, yielding a net gain in cellular resilience that frequently exceeds the baseline health of an unstressed control group 356. The scientific exploration of hormesis extends from the evolutionary biology of cellular defense to the development of novel therapeutics for neurodegenerative diseases, while simultaneously provoking intense, ongoing debate within regulatory bodies regarding radiation safety protocols and chemical exposure limits.
Foundational Principles of Dose-Response Toxicology
The characterization of hormesis is fundamentally rooted in the quantitative shape of its dose-response curve, which diverges sharply from the traditional models utilized in pharmacological and environmental risk assessment. Understanding the mathematical and conceptual differences between these models is critical to isolating the unique biological utility of hormetic stressors.
Traditional Paradigms and the Hormetic Biphasic Curve
Regulatory agencies and risk assessors have historically relied on two primary models to extrapolate human health risks from experimental data: the Linear No-Threshold (LNT) model and the Threshold model 67. The LNT model assumes that biological risk is directly proportional to the dose, extending linearly down to zero. Under this paradigm, there is no safe dose of a stressor; even a single molecule of a chemical or a single track of ionizing radiation carries a quantifiable, cumulative probability of inducing stochastic effects such as genetic mutation or carcinogenesis 789. Conversely, the Threshold model posits that biological systems can perfectly buffer small amounts of a toxin. It assumes a zone of zero risk until a specific dose threshold is breached, at which point deterministic health effects (such as tissue necrosis or organ failure) rise steeply 6710.
The Hormetic model challenges both assumptions by demonstrating a non-monotonic, biphasic response. When plotted on a Cartesian coordinate system with increasing dose on the X-axis and biological damage on the Y-axis, the LNT model traces a straight upward line from the origin, while the Threshold model traces a flat line along the X-axis before angling upward. The Hormetic model, however, drops below the X-axis baseline in the low-dose region, indicating a negative risk or a distinct biological benefit. This "hormetic zone" represents the stimulatory phase where adaptive repair mechanisms overcompensate for the minor initial stress. As the dose increases, the curve rises, crossing the baseline at the "Zero Equivalent Point" before escalating into severe toxicity, forming a characteristic J-shaped or inverted U-shaped curve depending on the biological endpoint being measured 6101112.
| Dose-Response Model | Core Assumption of Low-Dose Risk | Curve Shape and Trajectory | Primary Application in Regulatory Toxicology |
|---|---|---|---|
| Linear No-Threshold (LNT) | All exposure is harmful; risk is cumulative and proportional to dose, with no safe lower bound. | Straight linear progression starting from the origin (0,0) with a positive slope. | Stochastic risks: ionizing radiation protection standards and genotoxic carcinogens 79. |
| Threshold Model | Low exposures are harmlessly buffered; risk remains at zero until a specific biological limit is exceeded. | Flat baseline at zero risk, followed by a steep upward curve after the threshold dose. | Deterministic risks: acute chemical toxicity, non-genotoxic carcinogens, and systemic poisons 61013. |
| Hormetic Model | Low exposures induce beneficial adaptive overcompensation; high exposures cause toxicity. | Biphasic (J-shaped or U-shaped); dips below baseline (benefit) before rising into toxicity. | Biological adaptation: biogerontology, exercise science, ischemic preconditioning, and emerging radiotoxicology theories 3611. |
Quantitative Limits of Biological Plasticity
A defining characteristic of the hormetic response is the strict quantitative limit of the adaptive overcompensation. Analyses of expansive toxicological databases, encompassing thousands of well-designed dose-response relationships across diverse taxa, reveal that the maximum stimulatory effect of a hormetic stressor is highly constrained. The magnitude of the beneficial response typically peaks between 30% to 60% above the control group's baseline performance, regardless of the biological model, the specific inducing agent, or the downstream mechanism 31415.
This modest percentage increase defines the evolutionary limits of phenotypic plasticity. From an organismal resource allocation perspective, biological systems cannot sustain infinite upregulation of energy-intensive repair and defense mechanisms 16. While the percentage gain is relatively narrow, this 30% to 60% enhancement is frequently the critical determining factor between cellular survival and apoptosis under subsequent, more severe stress. The adaptive response operates within a narrow biological window; once the cellular systems are maximally upregulated to defend against a stressor, any additional magnitude of stress rapidly overwhelms the organism's repair infrastructure, driving the system out of the hormetic zone and into catastrophic failure 31415.
Molecular Pathways of Stress Adaptation
The systemic benefits of hormesis are not the result of a single localized reaction, but rather the hierarchical activation of a highly conserved network of molecular sensors and signaling cascades. When an organism is subjected to transient, low-dose stress, these pathways dynamically alter gene expression, shifting the cellular mandate from growth, proliferation, and reproduction toward an intensive focus on repair, maintenance, and defense.
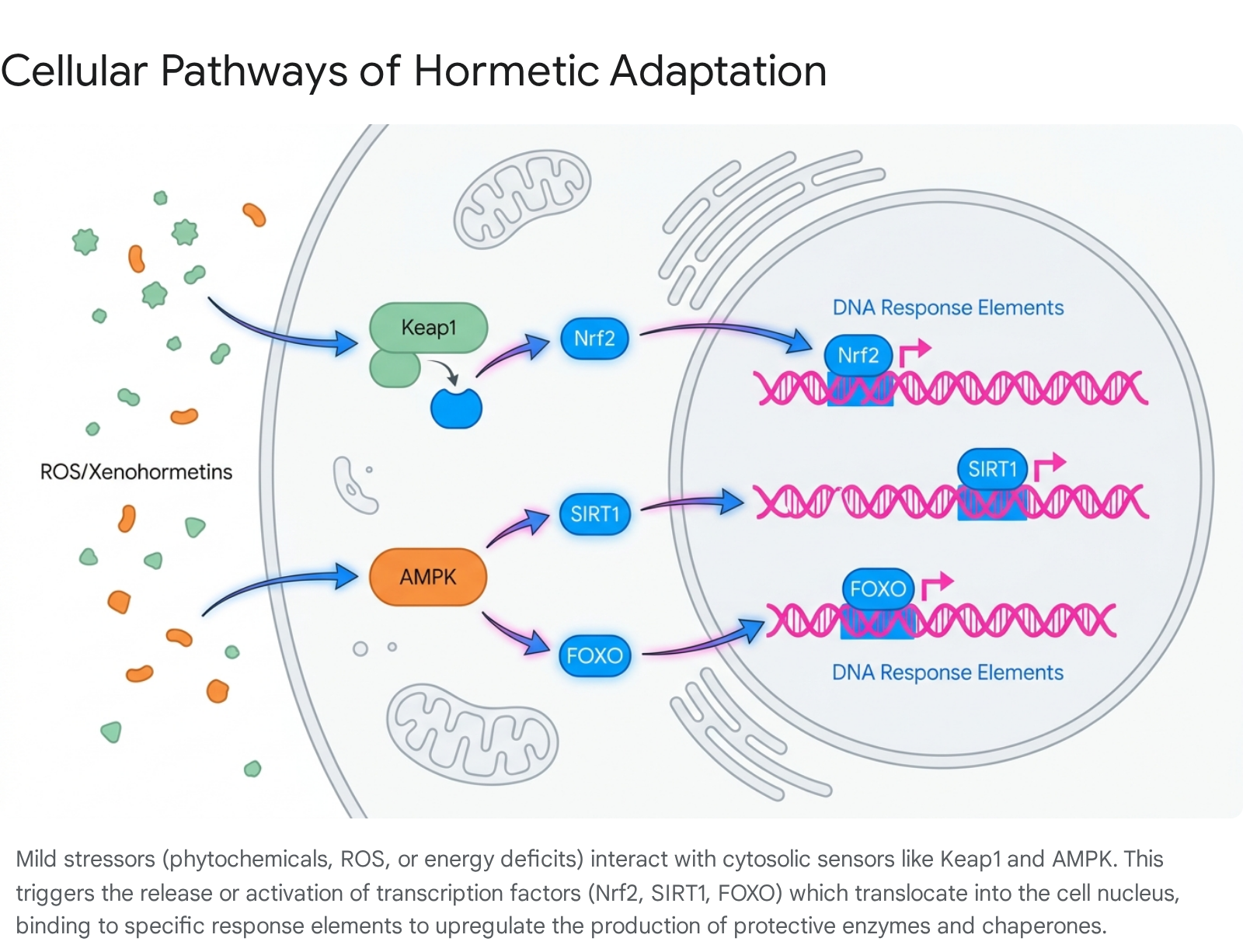
Oxidative Stress and the Nrf2 Antioxidant System
Historically, reactive oxygen species (ROS) and free radicals were viewed exclusively as pathological agents, directly responsible for lipid peroxidation, protein denaturation, DNA damage, and the overarching cellular decline associated with aging 171819. However, the science of mitohormesis reveals that moderate, physiological levels of ROS serve as indispensable redox-active signaling molecules 192021.
Mild oxidative stress acts as the primary trigger for the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway, widely considered the master regulator of cellular redox homeostasis 171920. Under basal, unstressed conditions, Nrf2 is tethered in the cytosol by its homodimeric repressor protein, Kelch-like ECH-associated protein 1 (Keap1), which continuously targets Nrf2 for ubiquitination and rapid proteasomal degradation, keeping its basal levels remarkably low 1722. When a cell encounters low-dose oxidative or electrophilic stress, the reactive molecules interact directly with critical cysteine residues on the Keap1 protein. This interaction induces a conformational change in Keap1, disrupting the ubiquitination process and releasing Nrf2 172022.
Once liberated, stabilized Nrf2 translocates from the cytosol into the nucleus, where it heterodimerizes with small Maf proteins and binds to Antioxidant Response Elements (ARE) in the promoter regions of target DNA 1722. This genomic interaction initiates the robust transcription of over 250 cytoprotective genes, including those encoding glutathione peroxidase, superoxide dismutase (SOD), catalase, heme oxygenase-1 (HO-1), and NAD(P)H quinone oxidoreductase 1 (NQO1) 17192023. This sweeping antioxidant response not only neutralizes the initial low-level ROS that triggered the cascade but massively overcompensates, saturating the cell with defensive enzymes. This preconditioning arms the cell, granting it superior resilience against subsequent, potentially lethal oxidative or toxicological insults 202224.
Nutritional Stress, AMPK, and Xenohormesis
Nutritional stressors, most notably caloric restriction and intermittent fasting, impose a mild state of energy depletion on the organism. This metabolic stress is detected by AMP-activated protein kinase (AMPK), a fundamental intracellular energy sensor. AMPK is activated by an increasing ratio of AMP to ATP, which signifies that cellular energy stores are being depleted 19. Upon activation, AMPK orchestrates a systemic metabolic pivot, shutting down energy-consuming anabolic processes (such as lipid and protein synthesis) and upregulating catabolic processes to restore ATP balance 1925.
A critical downstream effect of AMPK activation is the direct inhibition of the mechanistic target of rapamycin (mTOR) pathway. The mTOR complex is a central driver of cellular proliferation and growth; however, its chronic overactivation in nutrient-rich environments is heavily implicated in accelerated aging, stem cell depletion, and oncogenesis 18. Simultaneously, AMPK phosphorylates and activates sirtuins, particularly SIRT1, a family of NAD+-dependent protein deacetylases 19. SIRT1 subsequently deacetylates a variety of downstream targets, including the FOXO (Forkhead box O) transcription factors and PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha) 192025. The activation of the AMPK-SIRT1-FOXO axis promotes intense mitochondrial biogenesis, enhances the efficiency of the electron transport chain, reduces systemic inflammation by inhibiting NF-κB, and upregulates robust DNA repair mechanisms 192025.
The benefits of dietary restriction can also be chemically simulated through "xenohormesis." This concept posits that humans have evolved to sense and benefit from stress-induced molecules synthesized by plants. When plants are subjected to environmental stressors like UV radiation, drought, or fungal infection, they produce defensive phytochemicals 202627. When humans ingest these sub-toxic phytochemicals - such as resveratrol from grapes, sulforaphane from cruciferous vegetables, or epigallocatechin-3-gallate (EGCG) from green tea - the compounds act as mild biological stressors. They act as electrophiles that bind to Keap1 to activate Nrf2, or they act as potent epigenetic modulators. For instance, sulforaphane functions as an inhibitor of histone deacetylase (HDAC) enzymes, while EGCG inhibits DNA methyltransferases (DNMTs), leading to dynamic changes in chromatin structure and the sustained transcription of longevity and stress-resistance genes 14202526.
Proteostasis, Autophagy, and Mitochondrial Quality Control
The age-related deterioration of proteostasis - the cellular capacity to correctly fold new proteins and degrade damaged ones - is a primary driver of functional decline and neurodegenerative disease 2829. Hormetic stressors actively counteract this decay through the upregulation of autophagy, a vital cellular recycling mechanism 3031. Mild physical exercise, thermal stress (heat shock or cold exposure), and nutrient deprivation heavily stimulate both macroautophagy and chaperone-mediated autophagy 313233. Thermal stress specifically triggers Heat Shock Factor 1 (HSF1), which upregulates Heat Shock Proteins (HSPs) that act as molecular chaperones to refold denatured proteins and prevent toxic protein aggregation 2233.
Furthermore, specific perturbations to the internal environment of the mitochondria trigger unique, localized stress responses. Chronic disruptions to mitochondrial proteostasis heavily activate the Integrated Stress Response (ISR) and the Mitochondrial Unfolded Protein Response (UPRmt) 293435. The ISR operates through the selective phosphorylation of the translation initiation factor eIF2α. This action immediately halts global protein synthesis, preventing the mitochondria from being overwhelmed by newly synthesized, potentially misfolded proteins. Concurrently, the phosphorylation of eIF2α allows for the preferential translation of specific stress-responsive transcription factors, such as ATF4, which travel to the nucleus to induce the expression of genes required to restore mitochondrial integrity and metabolic homeostasis 293435.
| Environmental Stressor | Primary Sensory Mechanism or Receptor | Primary Adaptive Signaling Pathway | Key Downstream Effectors & Cellular Outcomes |
|---|---|---|---|
| Oxidative Stress / Phytochemicals | Keap1 oxidation and conformational change | Nrf2 / ARE pathway | Expression of HO-1, NQO1, SOD, glutathione peroxidase; enhanced redox homeostasis and systemic detoxification 172022. |
| Nutrient Deprivation / Fasting | Elevated AMP/ATP ratio, elevated cellular NAD+ | AMPK activation / SIRT1 / mTOR inhibition | FOXO, PGC-1α; initiation of autophagy, mitochondrial biogenesis, and suppression of pro-inflammatory NF-κB 1925. |
| Thermal Stress (Heat/Cold) | Protein denaturation and unfolding | Heat Shock Factor 1 (HSF1) | Expression of Heat Shock Proteins (HSPs); enhanced chaperone-mediated protein folding and proteostasis maintenance 23233. |
| Mild Hypoxia (Altitude/Ischemia) | Intracellular oxygen depletion | Hypoxia-Inducible Factor 1-alpha (HIF-1α) | Angiogenesis, enhanced glycolytic capacity, erythropoiesis, and increased metabolic flexibility 183233. |
| Mitochondrial Proteotoxicity | Disrupted mitochondrial import / protein misfolding | Integrated Stress Response (ISR) / UPRmt | Phosphorylation of eIF2α, ATF4 activation; transient halt of global translation to restore organelle health 293435. |
Applications in Biogerontology and Longevity
The traditional paradigm of aging theorized the process as an inescapable, linear accumulation of molecular damage driven by oxidative stress, telomere attrition, and metabolic wear-and-tear 1821. Under this framework, the introduction of any additional stress, however mild, should theoretically accelerate physiological decline. The paradoxical reality of hormesis - that the deliberate infliction of mild cellular damage actively extends lifespan and healthspan - has forced a fundamental restructuring of biogerontological theory 1518.
Contemporary aging models increasingly identify the overactivation of growth-oriented signal-transduction pathways (such as mTOR) and the age-related decline in adaptive homeostatic capacity as the primary drivers of senescence 18. Hormetic stressors combat this by dividing into two functional categories: "Hormesis A," which directly inhibits the mTOR pathway to slow the fundamental rate of aging, and "Hormesis B," which increases the organism's "aging-tolerance" by conditioning cells to survive the catastrophic complications and acute stressors associated with advanced age, such as ischemia or oxidative bursts 1832.
Precision Biomarker Clocks and Immune Aging
The translation of hormesis from observational biology to precision medicine requires the ability to accurately quantify biological age and cellular resilience. Significant advancements in this domain have emerged recently. In 2026, researchers from the Aging Biomarker Consortium (ABC) and the Chinese Academy of Sciences (CAS) published a landmark study in Cell, introducing a highly sophisticated computational framework known as the "Digital Aging Twin" 3637.
This framework was built upon a standardized multicentric cohort (mCAS) comprising 2,019 healthy individuals aged 18 to 91. The researchers collected over 240 distinct clinical and physiological parameters per participant, layered with massive multi-omics data including DNA methylation, transcriptomics, proteomics, metabolomics, and gut microbiome profiles 3637. This dataset allowed the creation of a three-tiered system of "aging clocks": a core capacity clock measuring functional decline, a deep-learning-driven multimodal clock, and individual organ clocks capable of mapping the asynchronous aging rates of different physiological systems within the same body 3637. These clocks verified that individuals subjected to mild, consistent hormetic lifestyle factors - such as moderate physical activity and regulated metabolic inputs - exhibited significantly decelerated biological aging. Furthermore, the researchers isolated specific coagulation factors as active drivers of the aging process, enabling the creation of simplified "proxy clocks" that can estimate comprehensive biological age using a plasma panel of approximately 100 proteins 3637.
Parallel research into immunological senescence has further refined the targeting of hormetic interventions. The development of the Human Immune Aging Clock (HIAC), mapped from high-resolution single-cell analysis of 1.2 million peripheral blood mononuclear cells, demonstrated that immune aging is not entirely linear 38. The immune system undergoes a sharp decline in naive T cells and an accumulation of exhausted T cells that peaks dramatically around age 40 38. A critical mediator of this decline is the transcription factor RUNX1. Researchers demonstrated that the artificial depletion of RUNX1 in young T cells induced premature senescence, while the restoration of RUNX1 in aged T cells successfully rejuvenated their infection-fighting capabilities 38. Hormetic strategies that naturally upregulate factors analogous to RUNX1 offer a theoretical mechanism to revert immune cells to a functionally youthful state.
Stem Cell Rejuvenation and Senescence Resistance in Primates
The most profound clinical applications of hormesis involve the genetic and biochemical reprogramming of cellular stress responses. In a major 2025 breakthrough published in Cell, researchers successfully applied hormetic and longevity-associated genetic pathways to reverse systemic aging in a primate model 3942.
The research team engineered human mesenchymal progenitor cells - a type of stem cell responsible for tissue repair - to overexpress longevity pathways, specifically focusing on the FOXO3 gene 39. FOXO3 is a primary downstream target of the AMPK/SIRT1 hormetic axis, highly associated with extreme human longevity and stress resistance 1939. These engineered Senescence-Resistant Cells (SRCs) were characterized by extended telomeres, high resistance to oxidative stress, and a lack of oncogenic or tumorigenic potential 3942.
Over a 44-week trial, elderly cynomolgus monkeys received biweekly intravenous infusions of these SRCs. The intervention resulted in widespread, multi-system rejuvenation across 10 major physiological systems and 61 distinct tissue types 3942. Treated primates exhibited reversed bone density loss, reduced brain atrophy, diminished chronic inflammation, and restored reproductive function. Utilizing machine learning-based biological aging clocks, researchers calculated that the SRC therapy effectively reversed the biological age of immature neurons by six to seven years, and oocytes by approximately five years 3942. The therapeutic efficacy of the SRCs was largely attributed to the release of targeted exosomes - microscopic extracellular vesicles that delivered protective molecular payloads to surrounding tissues, thereby preserving genomic stability and restoring proteostasis in a manner that perfectly mimics the systemic ripple effects of endogenous hormetic adaptation 39.
Therapeutic Strategies for Neurodegenerative Diseases
Neurodegenerative disorders, encompassing Alzheimer's disease (AD), Parkinson's disease (PD), amyotrophic lateral sclerosis (ALS), and Huntington's disease, represent a profound failure of cellular stress response mechanisms, particularly protein degradation systems and mitochondrial quality control 30314344. The pathogenesis of these diseases is characterized by the accumulation of misfolded and neurotoxic protein aggregates - such as amyloid-beta and tau tangles in AD, and alpha-synuclein Lewy bodies in PD 22303140.
These aggregates create a catastrophic feedback loop: initial protein misfolding impairs the autophagy-lysosomal pathway (ALP), which in turn accelerates the unchecked accumulation of toxic proteins, leading to chronic neuroinflammation, mitochondrial depolarization, and eventual neuronal apoptosis 303140. Because hormetic stressors directly stimulate the ALP, enhance mitochondrial biogenesis, and upregulate molecular chaperones, the pharmacological mimicking of hormetic pathways represents one of the most promising frontiers in disease-modifying neurodegenerative therapeutics 314344.
Pharmacological Mitophagy Enhancers
Mitochondrial dysfunction is a central, driving feature of Parkinson's disease pathogenesis. Healthy neurons maintain energy homeostasis through a rigorous quality control process known as mitophagy - the selective autophagic engulfment and degradation of damaged mitochondria 31434142.
The canonical regulation of mitophagy is governed by the PINK1/Parkin pathway. When a mitochondrion is damaged and loses its membrane potential, the kinase PINK1 accumulates on the outer mitochondrial membrane and recruits the E3 ubiquitin ligase Parkin. Together, they tag the defective organelle with polyubiquitin chains, signaling the cellular autophagic machinery to engulf and destroy it 2241. However, this vital clearance mechanism is actively opposed by ubiquitin-specific peptidase 30 (USP30), a deubiquitinating enzyme that removes the protective ubiquitin tags, effectively applying the brakes to the mitophagy process and allowing toxic, ROS-leaking mitochondria to persist within the neuron 4142.
Leveraging the principles of precision hormesis, pharmaceutical developers are advancing selective mitophagy enhancers into human clinical trials. These small molecule therapeutics are designed to chemically bypass upstream sensory pathways and directly force the clearance of damaged organelles 434441. Mission Therapeutics recently secured regulatory approval in the United Kingdom to initiate Phase 1 clinical trials for MTX325, a highly selective USP30 inhibitor. By disabling the USP30 enzyme, MTX325 prevents the removal of ubiquitin tags, accelerating the autophagic clearance of defective mitochondria 4441. Similarly, Vincere Biosciences has advanced its own USP30 inhibitor toward Phase 1 trials scheduled for 2026, supported by a $5 million grant from the Michael J. Fox Foundation 42. Concurrently, other developers are targeting the pathway from the opposite angle, utilizing PINK1 activators (such as ABBV-1088) to aggressively hyper-stimulate the initiation of the ubiquitin-tagging cascade 4341.
Metabolic Modulators and Clinical Trials
Because metabolic stress is a primary initiator of systemic hormesis, researchers are deeply investigating metabolic modulators as a treatment for neurodegeneration. Epidemiological data indicates that patients with Type 2 diabetes who are treated with glucagon-like peptide-1 (GLP-1) receptor agonists experience significantly lower incidences of dementia 4143. This has prompted major clinical trials, including the EVOKE and EVOKE Plus studies, which are investigating the GLP-1 RA semaglutide (Rybelsus®) as a disease-modifying intervention for early Alzheimer's disease. In an ongoing 1.5-year placebo-controlled trial involving 180 patients, researchers are testing whether chemically mimicking the metabolic signaling associated with GLP-1 can reduce cognitive impairment and restore neuronal resilience 43.
Combinatorial metabolic therapies designed to artificially simulate the effects of nutrient deprivation and xenohormesis are also advancing. A notable Phase 2 investigator-initiated trial is currently deploying a carefully calibrated, four-part dietary cocktail comprising N-acetylcysteine (NAC), L-carnitine tartrate, nicotinamide riboside (NR), and serine 44. Administered to a cohort of 60 AD and 60 PD patients over a 12-week period, this cocktail is engineered to sequentially activate mitochondrial metabolism. Nicotinamide riboside acts as a direct precursor to NAD+, effectively simulating caloric restriction to forcefully activate the SIRT1 and AMPK pathways 254144. Concurrently, L-carnitine enhances the transport of fatty acids across the mitochondrial membrane for β-oxidation, while NAC provides raw material to replenish cellular glutathione levels 44. Together, these xenohormetic cofactors aim to jumpstart neuronal mitochondrial activity and halt neurodegeneration without requiring patients to endure severe physical fasting. Similarly, traditional xenohormetic botanical formulations, such as the Korean herbal compound PM012, are undergoing Phase 2 placebo-controlled trials (involving over 300 AD patients at doses up to 5,200 mg/day) to formally validate their neuroprotective and cognitive-enhancing capabilities 45.
Controversies in Regulatory Toxicology and Public Health
While the cellular mechanisms of hormesis are thoroughly documented and increasingly exploited in biogerontology and neurology, the application of hormetic theory to public health policy, environmental regulation, and industrial toxicology remains intensely controversial. The debate over whether to formalize hormesis as a default regulatory assumption centers primarily on two distinct arenas: the regulation of ionizing radiation and the toxicological assessment of endocrine-disrupting chemicals.
The Radiation Hormesis Debate and the LNT Paradigm
The most entrenched conflict regarding hormesis involves the assessment of health risks posed by low-dose ionizing radiation. Current global radiation protection policies - overseen and enforced by organizations such as the U.S. Nuclear Regulatory Commission (NRC), the National Council on Radiation Protection and Measurements (NCRP), and the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) - strictly adhere to the Linear No-Threshold (LNT) model 78.
The LNT model's dominance traces back to the mid-20th century, heavily influenced by high-dose radiation experiments on Drosophila (fruit flies) conducted by Hermann Muller, and subsequent linear extrapolations drawn from the Life Span Study (LSS) of Japanese atomic bomb survivors 794647. The LNT framework operates on the biophysical premise that a single particle track of ionizing radiation passing through a cell nucleus has the potential to induce an irreversible DNA double-strand break, thereby initiating oncogenesis 74849. Consequently, global radiation exposure limits are governed by the principle of ALARA ("As Low As Reasonably Achievable"), which dictates that exposure must be minimized to the absolute limit of technological feasibility, imposing massive financial and logistical compliance costs on the medical imaging, aerospace, and civil nuclear energy sectors 475051.
Proponents of radiation hormesis, notably researchers such as Edward Calabrese, argue that the LNT model represents a "failed fiction" based on flawed historical data that willfully ignored the dynamic equilibrium of cellular DNA repair mechanisms 512464749. Substantial biological and epidemiological evidence indicates that low-level radiation - at or slightly above natural terrestrial background levels - acts as a classical physical hormetic stressor. Small doses of radiation reliably upregulate DNA repair enzymes, stimulate robust antioxidant defenses, and may actively decrease the baseline incidence of spontaneous cancers by hyper-stimulating systemic immunological surveillance against aberrant cells 8474952. Furthermore, recent critical re-evaluations of the updated atomic bomb survivor mortality data suggest a distinct, non-linear dip in cancer mortality within the low-dose range of 0.3 to 0.7 Gray (Gy). This reduction in cancer incidence aligns precisely with the hormetic J-shaped curve, contradicting the fundamental predictions of the LNT model 53. Recognizing this data, the French Academy of Sciences issued reports explicitly endorsing radiation hormesis and criticizing the LNT model's applicability below 20 mSv 8.
Despite mounting evidence of radioprotective adaptive responses, primary regulatory agencies maintain their conservative stance. In response to recent petitions to transition U.S. standards to a hormesis-based model, the NRC formally reasserted its commitment to the LNT paradigm. The agency's advisory committees concluded that while cellular adaptive responses are observable in highly controlled in vitro settings, the epidemiological evidence in diverse human populations remains too equivocal to definitively overturn decades of LNT-based public health standards 750. Furthermore, critics of radiation hormesis warn that ionizing radiation carries cumulative risks for severe non-cancer outcomes, such as cerebrovascular and ischemic heart diseases, which exhibit threshold or linear damage models without any observable hormetic benefit 4854.
Low-Dose Effects of Endocrine-Disrupting Chemicals
The concept of biphasic dose responses has also been heavily invoked in the toxicological assessment of Endocrine-Disrupting Chemicals (EDCs), a pervasive class of synthetic environmental pollutants that includes bisphenol A (BPA), phthalates, and agricultural compounds like atrazine 155. Extensive research demonstrates that EDCs routinely generate non-monotonic dose-response curves (NMDRCs); they defy traditional toxicological assumptions by inducing profound, statistically significant biological effects at infinitesimally low doses, while higher doses may trigger completely different, or paradoxically muted, responses due to receptor saturation, enzymatic down-regulation, or acute cell death 127565758.
However, toxicologists draw a sharp, critical distinction between a classical hormetic curve and an EDC's non-monotonic curve. Hormesis is defined strictly by low-dose beneficial adaptation or preconditioning 416. In stark contrast, the low-dose effects associated with EDCs are virtually always adverse. Because EDCs structurally mimic endogenous hormones (which naturally operate at picomolar or nanomolar concentrations), low-dose exposure during critical windows of fetal or neonatal development can result in severe developmental disruption, including elevated risks of obesity, infertility, neurodevelopmental deficits, and hormone-sensitive cancers 1627555657. A comprehensive review of over 800 studies confirmed that adverse low-dose effects are remarkably common across diverse classes of EDCs 157. Therefore, applying the generalized hormetic assumption - that all low-dose perturbations are adaptively beneficial - to environmental endocrine disruptors would represent a dangerous misapplication of biological principles and could result in disastrous deregulation 16.
Genotoxic Carcinogens and the Threshold of Toxicological Concern
The hormesis framework is also universally considered inapplicable to genotoxic carcinogens (DNA-reactive mutagens). The biological mechanism of a hormetic response relies on a stressor perturbing cellular metabolism or protein folding, which the cell detects and resolves via complex signaling cascades 22. Genotoxic chemicals, however, directly interact with and covalently modify the structure of DNA molecules 1359.
Because a single mutation in a critical oncogene or tumor suppressor gene can initiate carcinogenesis, global health authorities and toxicological bodies (such as the WHO and EFSA) operate under the rigid assumption that DNA-reactive genotoxins possess absolutely no safe threshold and offer no adaptive preconditioning benefit 135967. For these substances, regulators apply the Threshold of Toxicological Concern (TTC) concept, using linear extrapolations to mandate that human exposure be kept below a virtual safe dose (often calculated as a one-in-a-million lifetime cancer risk) 5967.
Furthermore, the application of hormesis as a broad regulatory standard fails to account for the reality of complex chemical mixtures. In modern environments, human populations are simultaneously exposed to hundreds of different synthetic compounds 1627. Even if a single chemical might induce a beneficial hormetic response when isolated in a laboratory setting, simultaneous exposure to multiple environmental toxins that act upon similar cellular receptors can easily result in additive or synergistic toxicity, instantly pushing an individual out of the narrow hormetic zone and into biological failure 16.
Clinical Integration and Future Research Directions
The transition of hormesis from an observed toxicological anomaly to a foundational pillar of modern medicine is accelerating. Scientific organizations, such as the Japanese Society of Anti-Aging Medicine (JAAM), increasingly emphasize the integration of hormetic principles into public health frameworks. Positioning for their 25th Annual Meeting in 2025, JAAM has highlighted the conceptual shift from viewing aging as an irreversible decline to understanding it as a dynamic process of biological "reconstruction" or "Re-Aging," driven by the deliberate activation of human adaptive potential 686061.
The science of hormesis offers a profound recalibration of how biological resilience is generated and maintained. Rather than functioning as fragile, static machines slowly degraded by environmental exposure, biological systems are highly dynamic, adaptive networks that explicitly require mild, intermittent stress to achieve and maintain optimal function. By briefly triggering ancient, evolutionarily conserved pathways like Nrf2, AMPK, and SIRT1, intentional stressors such as intense exercise, intermittent fasting, targeted phytochemical consumption, and potentially carefully modulated low-level radiation force the biological upregulation of antioxidant defenses, DNA repair, and autophagy 8192022.
However, the therapeutic application of hormesis demands immense precision. The narrow, strictly defined margin of the hormetic zone - rarely exceeding a 60% gain in baseline cellular function - dictates that the line between a beneficial adaptive response and pathological toxicity is easily crossed 314. This is particularly critical in aging or diseased populations where intrinsic homeostatic capacity has already declined 32. While exploiting these molecular pathways via next-generation pharmacological agents (such as mitophagy enhancers, GLP-1 receptor agonists, and xenohormetic cofactors) offers a highly promising new frontier in treating neurodegenerative disease and extending human healthspan, the hormetic framework cannot be indiscriminately applied to all environmental toxins, endocrine disruptors, or genotoxic pollutants 13164144. Ultimately, mastering the clinical science of hormesis relies on identifying and manipulating the precise biological threshold where stress ceases to act as an evolutionary teacher and becomes a systemic destroyer.