Astrocytic modulation of neural circuits and cognition
Historically relegated to the status of passive structural support, astrocytes - the highly ramified glial cells that comprise 20% to 40% of the cellular population in the central nervous system - have emerged as active, indispensable modulators of brain function 12. Early neuroanatomists, who first documented glia in the late 1800s, conceptualized these star-shaped cells primarily as nervenkitt (nerve glue) 2. Under this classical neurocentric paradigm, astrocytes were understood to be responsible solely for homeostatic maintenance, metabolic support, neurotransmitter clearance, and the regulation of the blood-brain barrier 34. For over a century, the rapid, electrically excitable neuronal code was considered the sole arbiter of perception, thought, emotion, and behavior.
Over the last three decades, and accelerating rapidly with technological breakthroughs in imaging and transcriptomics between 2020 and 2026, this dogma has been fundamentally dismantled 56. Extensive in vivo imaging, single-cell RNA sequencing, and sophisticated computational modeling now demonstrate that astrocytes actively integrate synaptic information, dictate neural network topology, and stabilize complex cognitive phenomena such as memory formation and attentional shifts 788. The conceptual shift toward recognizing astrocytes as active processors centers on the architecture of the "tripartite synapse." This model details a structural and functional unit where an astrocytic perisynaptic process intimately envelops the pre-synaptic and post-synaptic neuronal terminals 91110.
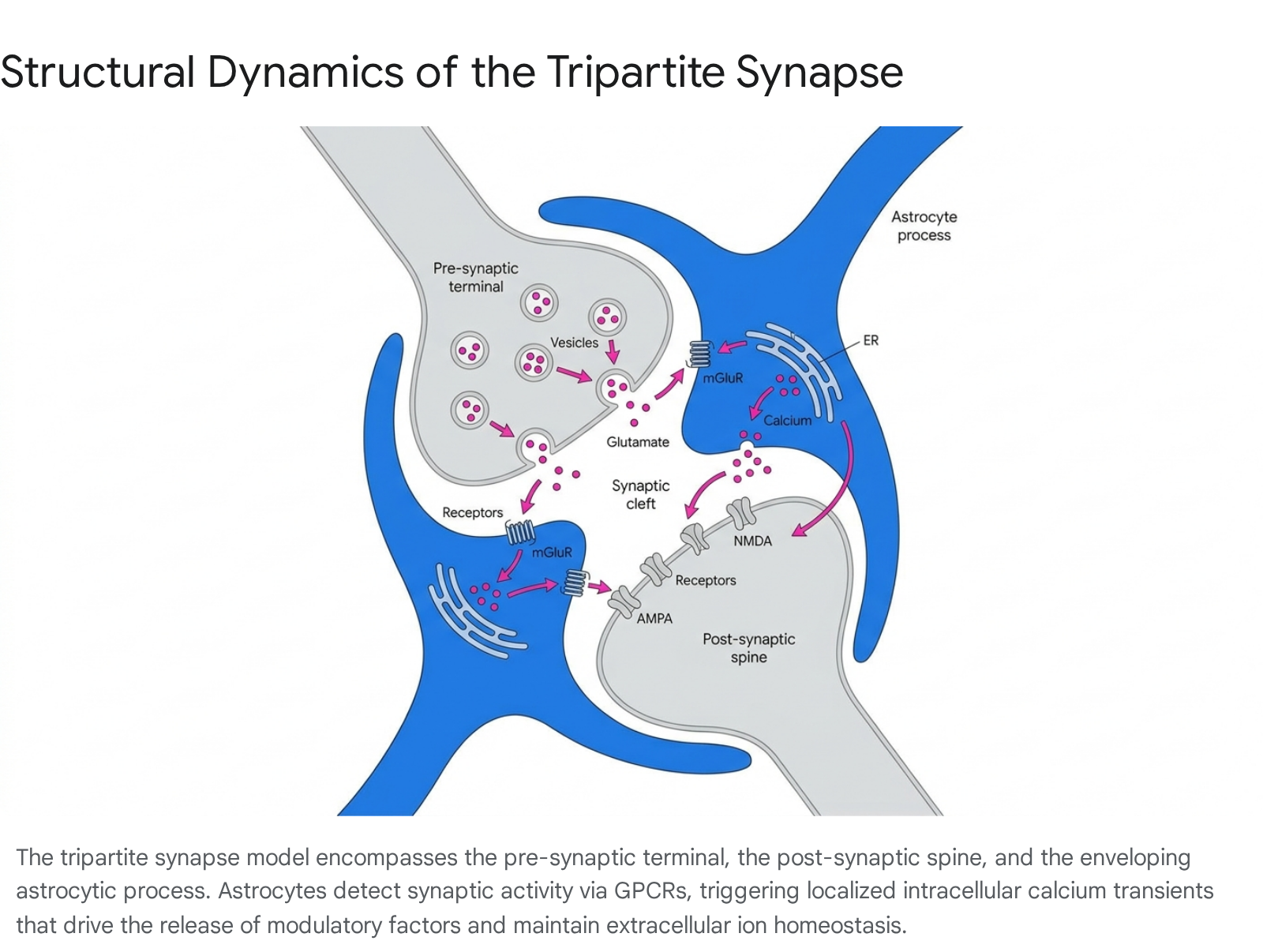
Because an individual astrocyte can contact up to 100,000 synapses in the murine brain - and up to 2 million synapses in the human brain - this physical arrangement allows astrocytic networks to monitor immense volumes of synaptic activity, buffer extracellular ions, and participate directly in synaptic plasticity 1011. As the depth of astrocytic investigation has expanded, research demonstrates that neurodevelopmental processes, experience-dependent plasticity, and neurodegenerative pathologies cannot be understood through the lens of neuronal connectomics alone 56. Astrocytes operate as computational units, bridging the temporal gap between millisecond-scale neuronal firing and the long-term stabilization of behavioral states.
Intracellular and Extracellular Calcium Dynamics
Unlike neurons, which rely on the rapid propagation of electrical action potentials, astrocytes are electrically non-excitable. Instead, their primary modality of signal integration and transmission is the fluctuation of intracellular and extracellular calcium ($Ca^{2+}$) concentrations 121314.
Somatic Waves and Localized Microdomains
Astrocytic $Ca^{2+}$ signaling occurs across multiple spatial and temporal scales, ranging from rapid, highly localized transients in fine perisynaptic processes to slow, global oscillations that propagate through the astrocytic soma and across extensive multicellular networks 1213. Historically, $Ca^{2+}$ transients were observed primarily in the astrocytic soma due to the spatial and temporal limitations of early organic fluorescent dyes such as OGB-1 and Fluo4 1516. These global somatic signals are primarily driven by the activation of Gq-protein-coupled receptors (GPCRs), which stimulate phospholipase C (PLC) to produce inositol 1,4,5-trisphosphate (IP3). IP3 subsequently triggers the release of $Ca^{2+}$ from the endoplasmic reticulum (ER) via type 2 IP3 receptors (IP3R2) 1718.
However, the advent of membrane-tethered genetically encoded calcium indicators (GECIs), such as Lck-GCaMP and Salsa6f, alongside advanced two-photon microscopy, revealed a vastly more complex signaling landscape 141519. The vast majority of astrocytic $Ca^{2+}$ activity occurs in highly compartmentalized microdomains within the cell's fine arborizations. Up to 85% of an astrocyte's volume consists of irregular, nanoscopic processes (30 to 50 nm) that sit below the diffraction limit of light 19. Fast transients in these microdomains can occur within milliseconds to seconds and often follow bursts of neuronal activity 12. These highly localized events frequently operate independently of IP3R2-mediated ER release, relying instead on the rapid influx of extracellular $Ca^{2+}$ through transient receptor potential (TRP) channels (e.g., TRPA1), voltage-gated calcium channels (VGCCs), or the reversal of sodium-calcium exchangers (NCX) during neurotransmitter uptake 12141920.
In vivo sensory stimulation studies demonstrate that evoked $Ca^{2+}$ rises occur initially in the fine arborizations, significantly preceding somatic transients by several seconds 2122. Specifically, sensory-evoked $Ca^{2+}$ elevations in arborizations exhibit a delay of approximately 11.1 seconds from stimulus onset, compared to a 13.2-second delay for somatic responses 22. The rise time to peak and decay time back to baseline are also distinct between compartments, indicating that synapses are initially accessed at the extreme periphery of the astrocyte 2122. This spatial compartmentalization allows individual astrocytes to selectively influence discrete synaptic micro-networks without triggering a widespread, cell-wide global response 12.
Modulation of the Extracellular Ionic Landscape
While intracellular $Ca^{2+}$ elevations dictate astrocytic downstream pathways and signaling, recent evidence underscores the critical role of extracellular calcium ($[Ca^{2+}]_o$) as an active, dynamic signaling mediator in its own right 12. Traditionally, the extracellular space was viewed merely as a passive reservoir for ions. However, astrocytes are central architects of the ionic landscape, dynamically regulating $[Ca^{2+}]_o$ during bursts of synaptic activity with submillisecond resolution 12.
Neurons are exquisitely tuned to extracellular calcium levels. Decreases in $[Ca^{2+}]_o$, driven by astrocytic buffering and network activity, exert a direct physical impact on postsynaptic signaling 12. A lowered $[Ca^{2+}]_o$ physically diminishes the electrochemical driving force for $Ca^{2+}$ entry through N-methyl-D-aspartate (NMDA) receptors and VGCCs 12. By directly attenuating the intracellular $Ca^{2+}$ transients required in neurons for the induction of long-term potentiation (LTP) and long-term depression (LTD), astrocytic control of the extracellular ionic gradient provides a foundational mechanism for regulating neuronal excitability independently of chemical transmitter release 12.
Debates Surrounding Exocytotic Gliotransmission
The proposition that astrocytes actively release neurotransmitters - broadly termed "gliotransmitters" (e.g., glutamate, ATP, and D-serine) - via $Ca^{2+}$-dependent vesicular exocytosis has been one of the most transformative, yet fiercely contested, concepts in contemporary neuroscience 391123. The debate centers on whether astrocytes routinely utilize vesicle-mediated chemical signaling to influence neuronal synapses in the healthy adult brain, or whether observed phenomena are artifacts of experimental conditions.
Evidence Supporting Active Gliotransmission
The initial evidence for active astrocytic signaling emerged in the 1990s when foundational observations demonstrated that the application of glutamate could evoke propagating $Ca^{2+}$ waves in cultured astrocytes, which in turn evoked $Ca^{2+}$ responses in adjacent neurons 319. Proponents of the gliotransmission hypothesis assert that astrocytes act as integral computational nodes in neural circuits. In this established paradigm, synaptically released neurotransmitters (such as glutamate from Schaffer collaterals in the hippocampus) spill over from the synaptic cleft and activate astrocytic GPCRs, such as metabotropic glutamate receptor 5 (mGluR5) 924. This triggers IP3-dependent $Ca^{2+}$ release, which subsequently drives the vesicular exocytosis of gliotransmitters back into the extracellular space 925.
The functional targets of these gliotransmitters are well documented. Astrocytic release of D-serine is broadly considered a necessary co-agonist for post-synaptic NMDA receptors, directly implicating astrocytes in the induction of synaptic plasticity and the potentiation of excitatory transmission 1126. Additionally, astrocytic ATP is released and rapidly hydrolyzed by extracellular ectonucleotidases into adenosine. This adenosine acts on presynaptic A1 adenosine receptors to suppress synaptic transmission and hyperpolarize neurons, providing a critical homeostatic feedback loop that restrains runaway neuronal excitability 1127. Glutamate released by astrocytes has also been shown to target presynaptic mGluRs and kainate receptors, modulating the probability of neurotransmitter release 911.
Methodological Skepticism and the In Vivo Crisis
Conversely, a robust faction of researchers has systematically questioned the physiological relevance of vesicular gliotransmission in intact brain tissue. Skeptics argue that much of the foundational evidence for gliotransmission was derived from cultured astrocytes in vitro 324. Cultured glia are known to undergo profound phenotypic shifts, often expressing proteins and transport mechanisms not found in mature astrocytes in situ 324. Furthermore, rigorous transcriptome analyses have frequently failed to detect the canonical synaptic SNARE proteins or vesicular glutamate transporters (vGluts) required for classical vesicular release in mature astrocytes 2428.
The skepticism intensified following sophisticated experiments designed to selectively manipulate astrocytic $Ca^{2+}$ without relying on broad pharmacological agonists. Researchers utilized $Ip3r2^{-/-}$ knockout mice, which lack the primary receptor responsible for astrocytic ER $Ca^{2+}$ release. Under the prevailing gliotransmission hypothesis, the near-total abolition of astrocytic $Ca^{2+}$ waves in these mice should have caused dramatic impairments in synaptic function and animal behavior 1728. However, $Ip3r2^{-/-}$ mice exhibited normal baseline synaptic transmission, unaffected hippocampal LTP, and no overt behavioral or developmental abnormalities 1728.
Further rigorous studies utilized chemogenetics to isolate astrocytic signaling. Researchers developed transgenic lines expressing the exogenous Gq-coupled MrgA1 receptor exclusively in astrocytes. When a specific agonist was applied, it evoked massive, widespread astrocytic $Ca^{2+}$ elevations 2329. Despite this robust $Ca^{2+}$ mobilization, there was no discernible effect on excitatory synaptic transmission, basal synaptic activity, or short- and long-term plasticity at the Schaffer collateral-CA1 synapse 23. Skeptics argue that these "negative" data sets provide strong evidence of absence, suggesting that $Ca^{2+}$-dependent exocytotic release of neurotransmitters is the province of neurons, not astrocytes, under physiological conditions 28.
Synthesizing the Divergent Evidence
Recent technological advances are mediating this divide, suggesting that the initial models of gliotransmission were fundamentally oversimplified 24. A critical methodological revelation showed that organic $Ca^{2+}$ dyes used in early studies primarily captured large somatic transients while entirely missing the rapid, highly localized microdomain signaling in the astrocytic arborizations 1720.
Re-evaluations using the highly sensitive GCaMP6f indicator demonstrated that even in $Ip3r2^{-/-}$ knockout mice, highly localized $Ca^{2+}$ microdomains persist in the fine processes 1720. Because these extreme peripheral processes are the actual sites of astrocyte-synapse contact, the preservation of microdomain signaling explains why synaptic plasticity remains intact in the knockout models 1720. The failure of MrgA1 receptor stimulation to induce plasticity is now hypothesized to be a result of the artificial, global nature of the $Ca^{2+}$ mobilization, which does not mimic the precise, compartmentalized frequency encoding required for localized gliotransmission 2429.
The current consensus suggests a highly context-dependent model. Astrocytes likely do not operate via broad, volumetric, and continuous neurotransmitter release 30. Instead, they exert targeted influence dictated by local metabolic states, alternative calcium influx pathways, and specific behavioral arousal states, performing integration in spatio-temporal domains complementary to those of neurons 930.
Astrocytic Heterogeneity and Subpopulation Specialization
Historically, astrocytes were viewed as a homogenous population, recognized merely as a monolith of support cells distinct from neurons and microglia. It was assumed that an astrocyte in the cortex functioned identically to one in the thalamus 1. However, the advent of single-cell and single-nucleus RNA sequencing (scRNA-seq/snRNA-seq), alongside advanced morphological profiling, has revealed vast inter- and intra-regional heterogeneity 43132.
These profiles demonstrate that astrocytic subtypes are specialized to support specific neural circuits and localized functional requirements. Differences are evident across multiple functional axes: intrinsic membrane properties, the identity and expression levels of gap junction proteins (Connexin 30 and Connexin 43), inward-rectifying potassium channels (Kir4.1), and primary neurotransmitter transporters (GLT-1 and GLAST) 3132. For instance, GLAST promoter activity is heavily downregulated from adolescence to adulthood in most of the brain, remaining prominent only in specific subpopulations like Bergmann glia and superficial cortical layers 31.
This diversity heavily influences how different astrocytic networks buffer ions, store metabolic substrates like glycogen, and clear glutamate from the synaptic cleft 3132. To illustrate the extent of this specialization, distinct astrocytic subpopulations and their primary localizations are detailed below.
| Astrocyte Subpopulation | Primary Brain Region | Distinctive Morphological & Transcriptomic Features | Functional Specialization |
|---|---|---|---|
| Cortical Astrocytes (AST1-5) | Adult Cortex & Hippocampus | Divided into at least five distinct transcriptomic subtypes. For example, AST1 (Marginal Astrocytes) selectively populates the pial layer. | Exhibit layer-specific modulation of cortical columns. Functional distinctiveness aligns with varying requirements for calcium activity patterns and metabolic coupling based on cortical depth 432. |
| Bergmann Glia | Cerebellar Cortex | Unipolar morphology extending highly ramified radial processes through the molecular layer. Retains high GLAST expression into adulthood. | Specialized ensheathment of Purkinje cell synapses. Critical for highly synchronized local potassium buffering and the rapid clearance of intense glutamate signaling 143132. |
| Müller Glia | Retina | Elongated morphology spanning the entire thickness of the neural retina. | Forms physical and functional boundaries within the eye. Facilitates bidirectional signaling with retinal neurons, essential for retinal ionic homeostasis and ATP/adenosine-mediated modulation 1131. |
| BMP4-Reactive Astrocytes | Broad CNS (Injury Context) | Induced by specific neurotoxicity/injury cues. Exhibits a transcriptomic profile sharing similarities with fibroblasts and vascular smooth muscle cells. | Drives specific extracellular matrix (ECM) remodeling and structural responses. Reacts oppositely to standard neurotoxic cytokines compared to CNTF-astrocytes 33. |
| CNTF-Reactive Astrocytes | Broad CNS (Injury Context) | Induced by specific cytokine profiles. Transcriptomic signature shows alignment with ependymal cells rather than structural fibroblasts. | Orchestrates divergent reactive phenotypes in response to inflammation. Highlights that astrogliosis is not a singular "reactive" state but a highly customized biological response 33. |
Astrocytic Regulation of Memory and Cognitive States
Perhaps the most profound paradigm shift occurring between 2024 and 2026 is the robust experimental verification that astrocytes actively shape high-level cognition. Far from being passive observers, astrocytes are central architects in the stabilization of memory, the filtering of environmental salience, and the regulation of attentional states 788.
Multi-Day Memory Stabilization and Engram Integration
The traditional model of memory dictates that life experiences are physically encoded in localized, specific networks of neurons known as engrams. When these neurons fire together during an event, the connections strengthen, allowing the memory to be recalled later 3435. However, a landmark 2025 study from the RIKEN Center for Brain Science demonstrated that neuronal engrams alone are fundamentally insufficient to account for the stabilization of long-term emotional memories 834. Recalled memories become transiently labile and require a secondary stabilization process to persist over time 8.
This stabilization relies on a multiday "astrocytic memory switch" 34. Using a novel brain-wide Fos tagging and imaging system in freely moving mice, researchers isolated the astrocytic response to fear conditioning. During an initial emotionally intense experience, the event induces a slow, day-long transcriptional priming in a specific ensemble of astrocytes located near the neuronal engram 834. Single-cell RNA sequencing revealed that this priming involves the marked upregulation of alpha and beta adrenoreceptors on the astrocyte surface 834.
When the animal subsequently recalls the memory (e.g., being placed back into a context associated with a foot shock), this primed astrocytic ensemble integrates two coincident signals: localized glutamate release from the reactivated neuronal engram, and a surge of noradrenaline from long-range locus coeruleus projections 836. This integration acts as a trigger, inducing the expression of the immediate early gene Fos exclusively in the astrocytes during the recall phase, rather than during the initial learning 34. The astrocytic Fos expression drives a secondary state change, culminating in the release of downstream neuromodulators such as IGFBP2, which physically stabilize the labile neuronal engram 8.
The behavioral implications of this astrocytic ensemble are profound. If the noradrenergic receptors on these specific astrocytes are blocked, or if astrocytic Fos signaling is genetically disrupted during the recall phase, the memory destabilizes and the mice exhibit significantly reduced fear recall 3436. Conversely, artificially forcing these astrocytes to activate causes the animal to generalize the memory, reacting to mildly unpleasant experiences as if they were highly traumatic 34. This mechanism provides a biological framework for understanding post-traumatic stress disorder (PTSD), where emotionally intense memories persist abnormally due to maladaptive astrocytic stabilization 34.
It should be noted that the precise interpretation of these findings remains subject to rigorous scientific debate. Critics, such as researchers from the Deneen laboratory, have suggested that the observed astrocytic Fos activation might partially reflect the processing of contextual novelty rather than memory recall per se, citing differences in experimental habituation protocols 36. Nonetheless, the consensus heavily supports a vital role for astrocytic networks in memory maintenance. Further supporting this, independent studies have tracked "engram-to-be" cells during sleep, finding that astrocytes exhibit heightened coactivity with existing engram networks during restorative sleep cycles, actively preparing the neural architecture to encode future memories 35.
Rewiring Attention and Enhancing Computational Capacity
Beyond memory stabilization, astrocytes play a direct, causal role in state-dependent brain rewiring. A 2025 study published in Science upended decades of dogma regarding how the brain shifts into states of heightened vigilance and attention 8. The researchers investigated norepinephrine, a neuromodulator fundamentally associated with alertness. Textbooks traditionally assert that neuromodulators fine-tune neurons directly. However, the study revealed that norepinephrine does not rewire synapses by acting directly on neurons 8.
Instead, norepinephrine signals strictly through the astrocytic network. Because of their highly ramified morphology, astrocytes are ideally positioned to detect global neuromodulator release. Upon sensing norepinephrine, astrocytes orchestrate the physical rearrangement of neuronal connectivity on a slower, sustained timescale, effectively rewiring the brain to maintain the attentional state 8. When the researchers genetically disabled the astrocytes' ability to sense or respond to norepinephrine, the neuromodulator was entirely unable to reorganize neuronal connectivity, proving that astrocytes are the obligatory mediators of this cognitive shift 8.
From a purely computational perspective, mathematical modeling conducted at MIT demonstrates that integrating astrocytes into neural networks drastically increases their processing capacity 7. Traditional models rely on bipartite synapses (one neuron connecting to one neuron). However, because a single astrocyte bridges thousands of synapses simultaneously, it facilitates complex, higher-order multi-variate interactions 7. This tri-part architecture allows the network to store and retrieve a significantly larger volume of information and endows the system with powerful error-correcting capabilities that bipartite models cannot achieve 7.
Glia-Glia Crosstalk in Synaptic Remodeling
The physical architecture of neural circuits is dynamically sculpted throughout development and in response to changing sensory environments through a process known as synaptic pruning 4037. Historically, this elimination of weak or obsolete synapses was attributed solely to the phagocytic activity of microglia, the brain's resident immune cells 4037. However, the efficiency of this pruning relies on an intricate, highly coordinated crosstalk between microglia and astrocytes 3738.
Recent research investigating experience-dependent synaptic remodeling - specifically using a sensory deprivation paradigm via unilateral whisker removal in postnatal mice - reveals a sophisticated molecular dialogue between glial subtypes 3839. In response to altered sensory input, neuronal fractalkine signaling (via the CX3CL1 ligand binding to the microglial CX3CR1 receptor) activates local microglia 3839.
Crucially, rather than immediately engulfing synapses, the activated microglia first secrete Wnt proteins 4038. This microglial Wnt signal acts directly on the neighboring astrocytes. Transcriptomic profiling (using TRAP-Seq on Aldh1l1-EGFP-L10a reporter mice) confirmed a significant upregulation of the Wnt/β-Catenin signaling pathway in the astrocytes within the deprived sensory cortex 37. The Wnt signaling instructs the astrocytes to physically retract their fine perisynaptic processes from the synaptic cleft 4040.
This astrocytic withdrawal acts as a necessary permissive step. By moving their tiny branches away, astrocytes strip the synapse of its protective glial sheath, leaving it exposed and vulnerable. Only then do the microglia swoop in to engulf and eliminate the inactive synapse 403740. This discovery fundamentally redefines the view of glial cells, demonstrating that microglia are not solitary executors of synaptic pruning; rather, the physical remodeling of the brain is collaboratively negotiated through precise inter-glial Wnt communication 3740.
Pathological Implications in Neurodegenerative Disease
The nuanced, multi-faceted functions of astrocytes - ranging from metabolic support to the orchestration of synaptic remodeling - implicate them heavily in the etiology and progression of neurodegenerative conditions, particularly Alzheimer's disease (AD) 4146. Inflammatory responses in AD are no longer viewed simply as reactions to neuronal death, but as primary drivers of pathology mediated by glial dysfunction 42.
The Microglial Gate to Astrocyte Reactivity
In AD, the accumulation of amyloid-beta (Aβ) triggers widespread neuroinflammation. A critical question has been how this accumulation leads to the destructive reactive astrogliosis seen in late-stage disease. A 2025 study published in Nature Neuroscience utilized PET radiotracers in living human subjects to map this process 4143. The findings revealed that Aβ pathology does not independently induce harmful astrocyte reactivity 4143.
Instead, amyloid burden is only associated with reactive astrogliosis (measured by plasma GFAP levels) across cortical regions when there is prior microglial activation 4143. Activated microglia secrete specific inflammatory cytokines - namely interleukin 1α (IL-1α), tumor necrosis factor (TNF), and complement component 1q (C1q) - that physically drive nearby resting astrocytes into a reactive, potentially neurotoxic state 43. Once this microglia-astrocyte duo is fully activated by Aβ, they collaboratively accelerate downstream tau phosphorylation (p-tau217), tangle formation, and subsequent cognitive decline 4143. Statistical modeling indicates that the pathway to cognitive impairment requires this specific sequence, suggesting that therapeutically targeting early microglial activation could effectively sever the crosstalk and prevent downstream astrocytic toxicity 4143.
Astrocytic Phagocytosis as a Therapeutic Target
While reactive astrocytes can be neurotoxic, healthy astrocytes possess intrinsic neuroprotective capabilities that become suppressed during aging and disease 37. Astrocytes express specific pathways (such as MEGF10 and MERTK) capable of directly recognizing and clearing cellular debris and misfolded proteins 37.
A highly promising 2025 study demonstrated that the targeted overexpression of the transcription factor Sox9 in astrocytes can reverse aging-related functional decline 46. Sox9 is a master regulator of astrocyte function. Upregulating its production prompts astrocytes to act akin to "vacuum cleaners," aggressively ingesting and clearing established, toxic amyloid plaques from the brain 46. In AD mouse models, this targeted enhancement of astrocytic clearance successfully halted neurodegeneration and preserved cognitive function 46. This suggests that restoring and harnessing the intrinsic housekeeping and phagocytic functions of astrocytes may be as critical a therapeutic avenue as attempting to prevent initial Aβ formation 46.
Brain-Wide Networks and Large-Scale Computational Modeling
As the computational weight of astrocytes becomes undeniable, global neuroinformatics initiatives are racing to integrate them into large-scale digital simulations. Astrocytes do not operate merely as isolated hubs surrounding individual synapses; they form vast, brain-wide networks physically connected by gap junctions 6. These syncytia act as a secondary, parallel connectome layered over the neuronal connectome, capable of transferring metabolic substrates (e.g., glucose, lactate) and propagating $Ca^{2+}$ signals across cortical hemispheres entirely independently of neuronal tracts 6. Notably, these networks exhibit macroscopic plasticity, physically shrinking or expanding over weeks based on prolonged sensory input or deprivation 6.
To accurately capture this complexity, the European Human Brain Project (HBP) and its subsequent digital research infrastructure, EBRAINS, recently developed a scalable connectivity framework that officially incorporates "tripartite synapses" into supercomputer simulations of spiking neural networks 4445. Extending the open-source NEST simulator, researchers across Germany, Finland, and Norway formalized a new connection rule that models the interaction between two neurons and a linking astrocyte. This technological leap allows for the simulation of up to one million interconnected cells, generating unprecedented insights into how local astrocyte-neuron dynamics orchestrate global brain rhythms 4445.
Concurrently, the China Brain Project - launched as a massive 15-year initiative extending to 2030 with a multi-billion yuan budget - places significant emphasis on uncovering the neural mechanisms of cognition through the lens of glio-neuronal interactions 4647. Operating under a "one body, two wings" framework, the project seeks to map the mesoscopic neural circuits (the body) and translate those findings into both medical interventions and next-generation, brain-inspired artificial intelligence (the two wings) 475348. Recognizing that astrocytic integration allows for exponential increases in network capacity, error correction, and context-dependent filtering, Chinese researchers aim to translate biological astrocytic mechanisms into neuromorphic hardware, cognitive robotics, and energy-efficient AI systems capable of human-like decision-making 345349.
Conclusion
The characterization of the astrocyte has undergone a profound evolution, shifting from the perception of an inert, structural scaffold to the recognition of its role as the master architect of the neural microenvironment. Operating via highly compartmentalized intracellular calcium microdomains and the precise modulation of the extracellular ionic gradient, astrocytes bridge the critical temporal gap between millisecond-scale neuronal firing and long-term behavioral state changes. Recent landmark discoveries linking specific astrocytic ensembles to memory stabilization, attention-driven synaptic rewiring, and collaborative synaptic pruning firmly establish these cells as indispensable computational units. As major scientific initiatives worldwide incorporate glio-neuronal dynamics into large-scale computational models and advanced AI architectures, it is increasingly clear that decoding the brain's highest cognitive functions fundamentally requires fluency in the complex, dynamic language of the astrocyte.