Artificial light at night and human health research in 2026
The proliferation of artificial light at night represents one of the most profound environmental alterations in human history. Driven by industrialization, global electrification, and urban expansion, the natural diurnal light-dark cycle that has governed biological rhythms for millennia is increasingly obscured. The mammalian circadian system, optimized to synchronize physiological processes with the solar day, is highly sensitive to photic input. When artificial lighting extends into nocturnal hours, it acts as an environmental endocrine disruptor, desynchronizing the body's internal master clock from external geophysical time.
Research spanning epidemiology, molecular biology, and occupational health indicates that circadian disruption is not merely an inconvenience related to sleep architecture, but a fundamental risk factor for a wide spectrum of chronic diseases. Investigations conducted through 2025 and 2026 have clarified the mechanistic pathways linking photic exposure to metabolic dysfunction, cardiovascular disease, neurodegeneration, and oncogenesis. Concurrently, shifts in global demographics and the rapid urbanization of the Global South have dramatically expanded the population exposed to shift work and continuous urban illumination, transforming a physiological vulnerability into a global public health crisis.
Global Trajectories of Nighttime Illumination
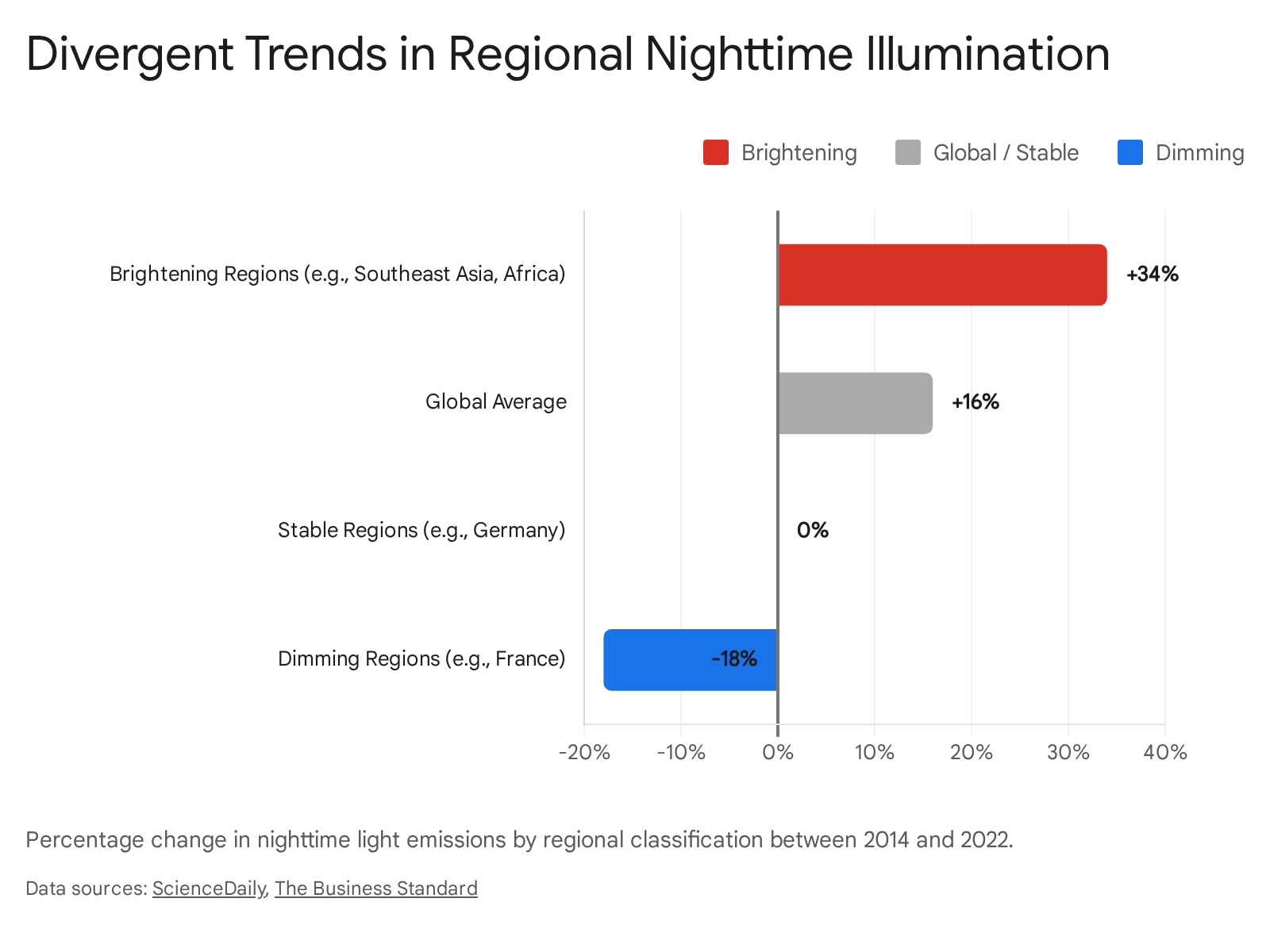
Satellite observation data compiled between 2014 and 2022 demonstrate a highly volatile and expanding footprint of global nighttime illumination. Over this period, worldwide light emissions increased by approximately two percent annually, resulting in a cumulative global net increase of 16 percent 12. However, this global average obscures distinct regional trajectories shaped by economic development, conflict, and environmental policy, indicating that the Earth's nightscape is not undergoing a uniform brightening, but rather a complex cycle of expansion and contraction 2.
In regions characterized by rapid urbanization and infrastructure expansion, primarily across emerging economies in Sub-Saharan Africa and Southeast Asia, nighttime illumination has surged. Countries such as Somalia, Burundi, Cambodia, Ghana, and Rwanda have experienced dramatic transitions from near-total darkness to integration within the global electric network 2. In these brightening regions, overall light emissions rose by 34 percent, driven by a massive expansion of energy access and rural electrification alongside urban sprawl 12.
Conversely, dimming trends have emerged in specific geopolitical and economic contexts. Abrupt reductions in illumination have been consistently observed in conflict-affected zones, such as Ukraine following the Russian invasion, as well as areas experiencing natural disasters or systemic power grid failures 12. In contrast, gradual and deliberate dimming is occurring in several industrialized nations. Europe experienced a four percent decrease in overall nighttime emissions, with nations like France recording a 33 percent reduction 1. This dimming is primarily guided by government regulations mandating the deactivation of municipal streetlights after midnight and the broader adoption of targeted LED technology designed to mitigate ecological light pollution and conserve energy 12. In regions actively experiencing dimming, overall global emissions decreased by 18 percent 1.
| Illumination Trend | Primary Environmental and Social Drivers | Representative Geographies | Emission Change (2014 - 2022) |
|---|---|---|---|
| Brightening | Rapid urbanization, rural electrification, infrastructure expansion | Southeast Asia, Sub-Saharan Africa, India, China | +34% (in brightening areas) |
| Dimming (Policy) | Energy conservation regulations, light pollution mitigation, targeted LEDs | Europe (e.g., France) | -18% (in dimming areas) |
| Dimming (Abrupt) | Armed conflict, systemic power grid failures, natural disasters | Ukraine, disaster zones | N/A (Highly Variable) |
| Stable | Equilibrium between localized regional brightening and dimming | Germany, United States | Negligible net change |
The United States retained the highest total luminosity of any country in 2022, followed closely by China, India, Canada, and Brazil 2. The ecological consequences of this expanding illumination are profound, disrupting nocturnal ecosystems, animal migrations, and human circadian rhythms on a planetary scale 2.
Neuroanatomical Pathways of Circadian Photoreception
The human biological clock is entrained to the 24-hour solar day through photic signals captured exclusively by the retina. While rod and cone photoreceptors are primarily responsible for image-forming vision, non-image-forming visual reflexes - such as circadian photoentrainment and pupillary constriction - are governed by a distinct, specialized retinal pathway 34.
A rare subpopulation of retinal ganglion cells, comprising less than five percent of the total retinal ganglion cell network, functions as autonomous photoreceptors. These intrinsically photosensitive retinal ganglion cells (ipRGCs) utilize the photopigment melanopsin, a G-protein coupled receptor, to detect ambient irradiance entirely independent of rod and cone synaptic input 355. Melanopsin exhibits a distinct action spectrum, demonstrating peak sensitivity to short-wavelength light approximating 480 to 490 nanometers (nm), which corresponds to the blue-cyan region of the visible light spectrum 3767.
The ipRGCs project their axons through the retinohypothalamic tract directly to the suprachiasmatic nucleus (SCN) of the hypothalamus, the anatomical locus of the central circadian pacemaker 56.
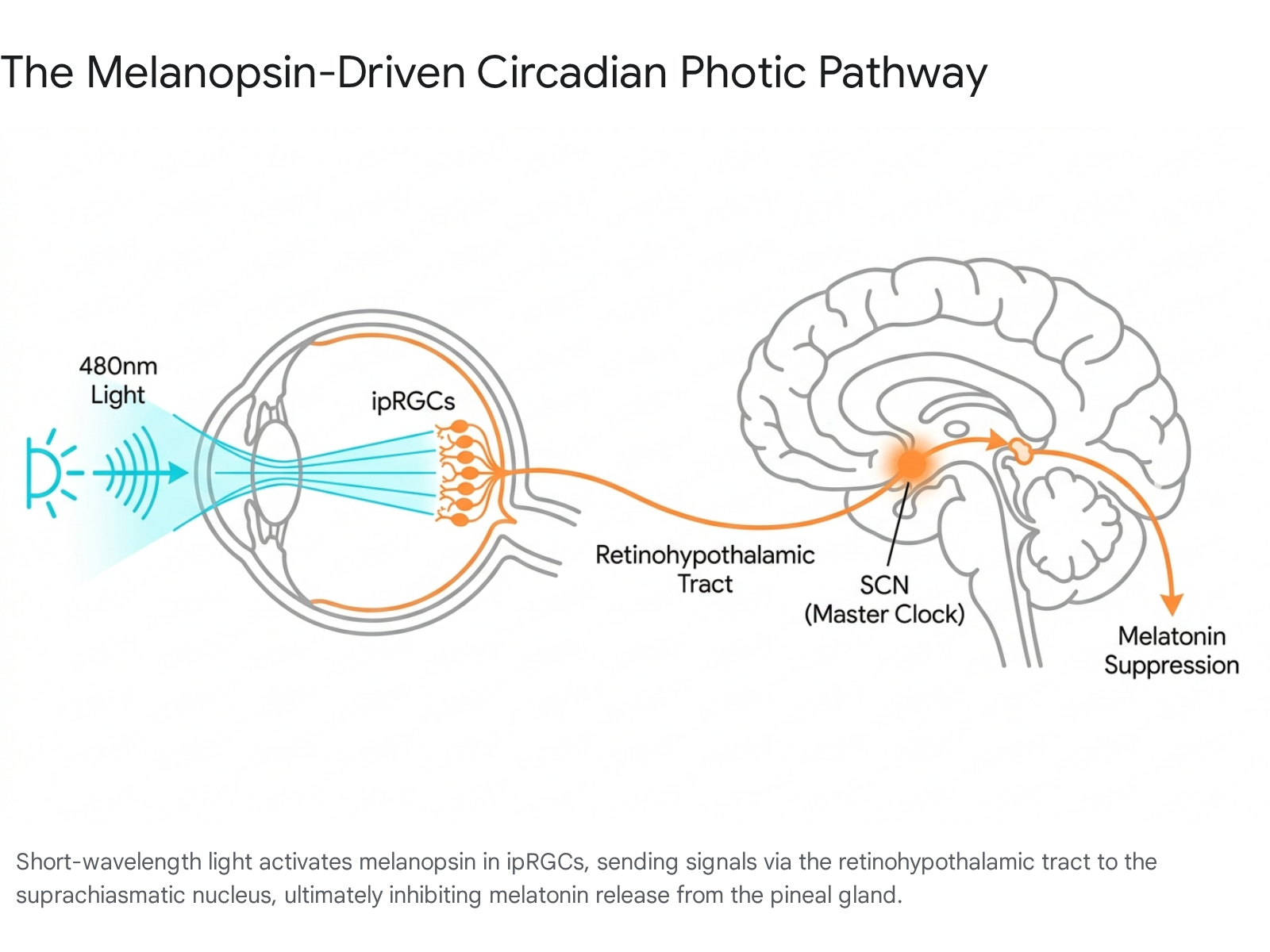
The SCN subsequently relays temporal signaling to various downstream neural and endocrine targets. Most critically, this pathway dictates the function of the pineal gland, inhibiting its synthesis and secretion of melatonin when light is present. Melatonin functions as a systemic timing signal, regulating sleep architecture, metabolic homeostasis, and core body temperature, while exerting substantial antioxidant and oncostatic effects 589.
Research indicates that ipRGCs comprise at least five distinct subtypes (M1 - M5), differentiated by morphological criteria, transcription factor expression, and downstream projection targets 5. Unlike rods and cones, which process rapid shifts in visual fields, ipRGCs act as tonic irradiance detectors. They possess lower spatial resolution, are less sensitive to transient light, and require prolonged integration of ambient light levels to drive subconscious physiological responses 356. However, emerging evidence suggests that specialized retinal ganglion cells also influence broader cortical mechanisms, projecting to multisensory hubs like the superior colliculus to modulate crossmodal inputs, creating a complex synergy between light detection and neurological plasticity 8.
Wavelength Specificity and Melatonin Suppression Thresholds
The suppressive effect of artificial light on endogenous melatonin synthesis is highly dependent on both spectral composition and cumulative illuminance. Exposure to standard room lighting (approximately 300 lux) during the hours preceding sleep results in profound alterations to the circadian profile, delaying melatonin onset in 99 percent of individuals and shortening the total duration of nocturnal melatonin secretion by approximately 90 minutes compared to dim light conditions 910. Even dim light at night - defined in experimental models as 5 to 10 lux - has been demonstrated to disrupt circadian stability, reducing total sleep time, lowering sleep efficiency, and increasing neural fragmentation 131112.
Clinical trials isolating specific wavelengths illustrate the disproportionate impact of the blue-cyan spectrum. In controlled exposures comparing monochromatic blue light (464 nm) to red light (631 nm) during evening hours (9:00 p.m. to midnight), both wavelengths initially suppressed melatonin after one hour 13. However, significant divergence occurred over extended exposure: blue light maintained profound melatonin suppression at 7.5 pg/mL, whereas the red light environment permitted melatonin recovery to 26.0 pg/mL, indicating that long-wavelength light is substantially less disruptive to the circadian clock over prolonged periods 13.
The absolute threshold for acute melatonin suppression remains dynamic, governed by the duration of exposure and prior photic history. Dose-response analyses evaluating a broad range of illuminance (40 to 1000 lux) and exposure durations (0.5 to 3.0 hours) confirm that while intense light suppresses melatonin rapidly, prolonged exposure to even sub-threshold, low-intensity light eventually yields statistically significant hormonal suppression 4. Furthermore, daytime exposure to bright light can partially inoculate the circadian system against nocturnal disruption; daytime exposures of 900 to 2700 lux have been shown to mitigate subsequent light-induced melatonin suppression at night, whereas individuals kept in dim daytime environments exhibit heightened sensitivity to evening light 910.
Metrics for Evaluating Circadian Lighting Environments
Historically, indoor lighting standards have prioritized visual acuity and energy efficiency, utilizing the "lux" metric, which is calibrated to the photopic spectral sensitivity function peaking at 555 nm (green-yellow light) 1714. This standard fails to capture the biological potency of a light source, as standard electrical LEDs and fluorescent tubes are typically optimized for visual perception and are substantially less efficient at stimulating melanopsin than natural daylight of equivalent visual illuminance 14.
To quantify the chronobiological impact of light accurately, the International Commission on Illumination (CIE) has standardized two critical metrics:
- Melanopic Equivalent Daylight Illuminance (m-EDI): Measured in lux, m-EDI quantifies the non-visual biological response to a light source. It calculates the specific illuminance of standard daylight (CIE Standard Illuminant D65) that would be required to produce an equivalent stimulation of the ipRGCs 717191521.
- Melanopic Daylight Efficacy Ratio (M-DER): This spectral index represents the ratio of m-EDI to photopic illuminance. It serves as a fractional multiplier to assess how effectively a specific artificial light source stimulates the circadian system relative to its perceived visual brightness 171915212223.
A reference daylight spectrum possesses an M-DER of 1.00 1521. Most standard artificial indoor environments exhibit an M-DER well below 0.75, providing adequate visual brightness but insufficient circadian stimulation during the daytime 21. For example, a typical 3000K LED has an M-DER of approximately 0.43 to 0.53. To achieve a target circadian stimulus of 250 m-EDI with this fixture, the actual visual illuminance in the space must exceed 500 photopic lux (250 / 0.50 = 500) 1722.
Expert consensus now recommends that daytime indoor environments maintain a minimum m-EDI of 250 lux at eye level (measured vertically), while evening residential environments should strictly restrict m-EDI to less than 10 lux beginning at least three hours prior to sleep 14.
| Light Source | Correlated Color Temperature (CCT) | Typical M-DER | Circadian Efficacy Relative to Daylight |
|---|---|---|---|
| Daylight (Reference D65) | 6500 K | 1.00 | Optimal baseline |
| Warm White LED | 2700 K | ~0.47 | Low |
| Standard LED | 3000 K | ~0.53 | Low |
| Cool White LED | 4000 K | ~0.71 | Moderate |
| Cyan-Enhanced LED (e.g., BioUp) | 4000 K | ~0.86 - 1.00 | High (maintains standard visual color) |
| Standard Daylight LED | 6500 K | 1.00 | High (often visually uncomfortable indoors) |
Data synthesized from standardized spectral measurements of commercial lighting products 15212223.
To circumvent the visual discomfort associated with high-CCT (6500K) lighting in indoor spaces, recent engineering advances have developed cyan-enhanced LEDs. These fixtures inject a specific spectral peak near 480 nm, maintaining the visually pleasing appearance of a 4000K source while increasing the M-DER by up to 42 percent, effectively decoupling biological potency from visual color temperature 1516.
Interindividual Variability in Circadian Photosensitivity
The assumption that standardized lighting environments exert uniform physiological effects is increasingly challenged by evidence of profound interindividual variability in circadian photosensitivity. Human laboratory and field studies demonstrate that the degree of light-induced melatonin suppression can vary across healthy individuals by more than a 50-fold magnitude 17181928.
Genomic Architecture and Chronotype Associations
Recent genome-wide association studies (GWAS) utilizing large cohorts, such as the UK Biobank, have identified specific genetic loci and haplotypes associated with circadian light sensitivity. Genetic correlations indicate a significant overlap between the genetic architecture of light sensitivity, sleep duration, and self-reported chronotype 171819. Polygenic risk score (PRS) analyses reveal that single nucleotide polymorphisms (SNPs) conferring greater light sensitivity are heavily correlated with morning chronotypes. Conversely, lower light sensitivity is genetically linked to evening chronotypes and intrinsically longer sleep durations 1820.
Individuals possessing an evening chronotype face compounding physiological risks due to modern social schedules. Because their internal clock is delayed, adherence to standard morning work hours forces a state of chronic circadian misalignment. Data indicates that adults with a "definite evening" chronotype exhibit a 79 percent higher prevalence of poor overall cardiovascular health scores (measured by the American Heart Association's Life's Essential 8 metric) compared to intermediate chronotypes 20.
Psychiatric Vulnerability and Photometric Biomarkers
Altered light sensitivity is rapidly emerging as a potential biomarker for psychiatric vulnerability. Clinical assessments measuring the pupillary light reflex (a surrogate metric for ipRGC activation) demonstrate that individuals with a higher polygenic risk score for bipolar disorder exhibit significantly increased sensitivity to dim light 1928. Furthermore, hypersensitivity to light is actively being investigated as a risk factor for bipolar relapse 28.
Conversely, young adults presenting with emerging, generalized mental disorders frequently exhibit attenuated dim light sensitivity - characterized by a smaller change in pupil diameter and slower maximal constriction velocity - which correlates with significantly delayed sleep-wake timing and higher insomnia severity 19. Interestingly, pharmacological interventions influence these photometric pathways; patients prescribed mood stabilizers, specifically lithium, demonstrate reduced pupil constriction and delayed latency to dim light, corroborating long-standing evidence that lithium attenuates melatonin light sensitivity 28. Genetic predispositions for higher Vitamin D concentrations are also positively correlated with bright light sensitivity, hinting at an interconnected metabolic and psychiatric pathway 28.
Demographic and Seasonal Modulators
Biological sex and seasonal photoperiods further modulate circadian sensitivity. Clinical trials indicate that female subjects often exhibit significantly greater melatonin suppression (+4.69 percent) in response to moderate light compared to males, despite demonstrating a paradoxically lower subjective alerting response (-6.00 percent) 21. Among female cohorts, menstrual phase also dictates circadian timing, with earlier dim light melatonin onset observed during the luteal phase compared to the follicular phase 21.
Additionally, sensitivity to the non-image-forming effects of light varies seasonally. Both sexes demonstrate stronger melatonin suppression (+18.05 percent) and heightened alerting responses to standardized light exposure during winter months compared to summer. This phenomenon likely reflects seasonal adaptations in retinal network gain, adjusting the system's sensitivity to compensate for shorter environmental photoperiods 21.
Epidemiological and Metabolic Consequences of Circadian Disruption
The chronobiological consequences of excessive nighttime illumination extend far beyond transient sleep disruption, precipitating profound, long-term alterations in metabolic homeostasis, cardiovascular function, and cellular regulation.
Cardiometabolic Dysfunction and Systemic Inflammation
A highly powered 2025 longitudinal cohort study analyzing 88,905 adults via the UK Biobank established a direct, independent relationship between personal light exposure at night (measured via wrist-worn sensors over 9.5 years) and incident cardiovascular disease. Participants tracking in the highest decile of nighttime light exposure exhibited a 32 percent higher risk of coronary artery disease, a 47 percent higher risk of myocardial infarction, a 56 percent higher risk of heart failure, and a 28 percent higher risk of stroke 22. Crucially, these hazard ratios remained statistically significant even after adjusting for sleep duration and traditional socio-environmental risk factors 2223.
Neuroimaging research provides a mechanistic explanation for this cardiovascular deterioration. Studies utilizing PET/CT scans demonstrated that higher levels of residential artificial light at night (measured via satellite) correlate strongly with increased stress-related activity in the amygdala and elevated arterial inflammation 2425. This pathway suggests that ambient light pollution triggers a systemic autonomic stress response, altering sympathovagal balance. When the brain continuously perceives stress driven by photic intrusion, it activates immune responses that progressively inflame blood vessels, directly contributing to atherosclerosis over a multi-year horizon 2225. Environmental mediation analyses further indicate that light pollution accounts for nearly 25 percent of the cardiovascular risk traditionally attributed solely to overlapping air pollution, emphasizing that modern urban environments present a syndemic hazard to cardiovascular health 26.
The disruption of the circadian-metabolic axis through irregular photic input also leads to pronounced metabolic dysfunction. Prolonged exposure to blue-enriched light at night alters the expression of peripheral clock genes within the liver and adipose tissue, promoting systemic insulin resistance and lipid accumulation 827. Epidemiological surveys of night-shift workers in the manufacturing sector document elevated rates of metabolic syndrome. Night-shift workers demonstrate statistically significant increases in body mass index, elevated triglyceride levels (+5.7 percent), and higher systolic blood pressure compared to day-shift controls 28. Longitudinal analyses confirm a dose-response relationship; workers subjected to rotating night shifts for more than 10 to 20 years experience a 20 percent higher risk of developing type 2 diabetes and a 23 percent increased likelihood of obesity 38394029. The pathophysiology is driven by delayed postprandial glucose clearance and poor beta cell function, resulting from the fundamental misalignment of feeding behavior with endogenous insulin rhythms 2728.
Oncological Risk and Night Shift Work Classifications
The carcinogenic hazard of chronic circadian disruption has been subject to rigorous, multi-disciplinary review by the International Agency for Research on Cancer (IARC). In Monograph Volume 124, the IARC Working Group classified night shift work - defined as occupational duties, including transmeridian air travel, occurring during the regular sleeping hours of the general population - as "probably carcinogenic to humans" (Group 2A) 30313233.
This classification relies on an integration of human epidemiology and robust mechanistic data:
- Human Epidemiological Evidence: Cohort and case-control studies demonstrate positive, albeit sometimes inconsistent, associations between long-term night shift work and cancers of the breast, prostate, colon, and rectum 324634.
- Animal Experimental Evidence: Experimental models in which the light-dark cycle is artificially altered yield sufficient evidence of carcinogenicity, showing markedly increased rates of liver, lung, and lymphoma tumors 3246.
- Mechanistic Pathways: Circadian disruption profoundly suppresses nocturnal melatonin - a hormone possessing potent oncostatic, anti-inflammatory, and antioxidant properties. Furthermore, misaligned circadian clocks interfere with vital cellular processes including DNA repair, hormonal regulation, and immune surveillance, leading to states of chronic inflammation and uninhibited cell proliferation 3246.
| Domain of Evidence (IARC Vol. 124) | Primary Scientific Findings | Specific Cancer Sites Implicated | IARC Hazard Classification |
|---|---|---|---|
| Human Epidemiology | Limited but positive association in cohorts (e.g., healthcare workers, flight crew) 3435. | Breast, Prostate, Colorectal | Group 2A |
| Experimental Animals | Sufficient evidence of tumor growth under altered light-dark cycles 32. | Liver, Lung, Lymphoma | (Probably Carcinogenic to Humans) |
| Mechanistic Data | Strong evidence of melatonin suppression, hormone alteration, impaired DNA repair 46. | Systemic Cellular Pathways | N/A |
Data derived from IARC Monograph Volume 124 and related epidemiological reviews 30313233463435.
To overcome the latency challenges of traditional epidemiology, advanced mechanistic trials are currently utilizing functional metagenomic analyses of the gut microbiome to trace the specific biological pathways mediating cancer risk in night shift workers. Novel trial designs involving the inoculation of gnotobiotic mice with stool samples from shift workers seek to directly prove the causal effects of circadian-associated gut dysbiosis on colorectal tumorigenesis, paving the way for targeted interventions such as clinical melatonin supplementation to mitigate oxidative DNA damage 36.
Neurodegenerative and Ophthalmological Outcomes
Emerging research characterizes artificial light at night as a modifiable risk factor for neurodegenerative disease and psychiatric disorders. In 2025 studies utilizing humanized APP knock-in mice (a model for Alzheimer's disease), chronic exposure to dim light at night (8 lux) reduced circadian rhythm amplitude and stability within two weeks 1311. Over an eight-week period, this low-level exposure modestly increased hippocampal amyloid plaque burden and shifted microglia toward an antigen-presenting (MHCII) state, indicating early neuroinflammation, though it was not associated with amplified cytokines at that early timepoint 1311.
Psychiatric models demonstrate similar physiological vulnerabilities. In postpartum subjects, continuous dim light exposure at night (5 lux) significantly blunted the amplitude of rest-activity rhythms and disrupted the expression of hippocampal clock genes, specifically Per1. This circadian decay correlated directly with decreased brain serotonin (5-HT) and brain-derived neurotrophic factor (BDNF), precipitating profound depression- and anxiety-like behaviors including increased immobility and decreased sucrose preference 12.
Simultaneously, chronic exposure to nighttime illumination contributes to structural ophthalmic degradation. Global Burden of Disease data spanning 1990 to 2021 indicates that sense organ diseases - specifically age-related macular degeneration and near-vision loss - are positively correlated with high nighttime light exposure. The global burden has risen to over 2 billion prevalent cases, exacerbating health inequalities primarily in regions undergoing rapid, unregulated urbanization 37.
The Intersection of Rapid Urbanization and Occupational Health
The physiological hazards of artificial light and circadian disruption intersect acutely with shifting global labor trends. The rapid urbanization and industrialization of the Global South, particularly in Southeast Asia and Sub-Saharan Africa, have catalyzed a massive expansion in the shift-working population, exposing billions to compromised environmental conditions.
Expanding Shift Work in Industrializing Regions
The International Labour Organization (ILO) and the World Economic Forum (WEF) 2025 reports indicate that global labor markets are undergoing profound restructuring. Driven by technological change and the green transition, an estimated 78 million jobs will be created globally by 2030, with Asia and the Pacific leading employment growth at 1.7 to 1.9 percent annually, adding approximately 34 million new jobs in 2025 alone 383940. In Southeast Asia, economic strategies have shifted heavily toward 24-hour manufacturing, logistics, and digital services to meet global demand, creating immense pressure to expand continuous shift-work schedules 4054.
However, this economic expansion occurs within a precarious structural framework. Over 1.3 billion workers in Asia and the Pacific - representing roughly 66 percent of total employment in the region - operate within the informal economy 39. These populations endure extreme working hours and erratic shift patterns without the regulatory protections, ergonomic standards, or occupational health monitoring available in industrialized nations 394142.
The health burden of these changing labor dynamics is highly documented. Meta-analyses conducted in 2025 reveal that Shift Work Sleep Disorder (SWSD) - characterized by severe insomnia, excessive sleepiness, and cognitive impairment resulting from circadian disruption - is alarmingly prevalent in industrializing nations 4344. Among healthcare populations, the prevalence of SWSD reaches 51.0 percent in Africa and 46.5 percent in Asia, spiking to 61.7 percent among nurses executing rapid "three-shift" rotation models 4344. Beyond fatigue, these schedules contribute directly to elevated clinical errors, endangering patient safety and amplifying the psychological strain on an already overburdened workforce 4143.
Environmental Syndemics in the Global South
The occupational hazards of shift work are compounded by the physical environments in which these populations reside. The global urban population is projected to surge to 6.7 billion by 2050, with 90 percent of this growth concentrated in Africa and Asia 4245. Much of this expansion is unplanned, resulting in the proliferation of informal settlements and slums, which currently house 1.1 billion people - a figure expected to triple by 2050 46.
In these dense urban environments, residents face a syndemic of ecological stressors. Unplanned settlements are characterized by minimal green space, high-density construction using low-quality materials, and extreme vulnerability to the urban heat island effect 4547. A 2026 report analyzing informal settlements across Kenya, Tanzania, and Zimbabwe revealed that indoor temperatures frequently exceed 40 degrees Celsius - up to nine degrees higher than outdoor areas 47. Consequently, 40 percent of residents report severe sleep deprivation due to heat, and 25 percent lose workdays, trapping them in a cycle of physiological exhaustion and economic poverty 47.
The demand for cooling in these rapidly urbanizing regions is expected to skyrocket. Currently, only 13 percent of African households have access to air conditioning, yet cooling systems could consume up to 60 percent of peak energy loads by 2040 48. Because slum populations frequently lack reliable electricity, they are simultaneously subjected to unregulated ambient light intrusion, severe particulate air pollution (PM2.5) from industrial activity, and extreme heat 474849. This confluence of hazardous air quality, continuous artificial light, and thermal stress interacts synergistically to magnify the risks of cardiovascular disease, metabolic syndrome, and circadian decay far beyond the baseline risks observed in the Global North 45464950.
Evidenced-Based Mitigation Strategies and Interventions
Addressing the profound public health implications of artificial light at night requires a combination of individualized behavioral interventions, standardized protective equipment, and systemic architectural and policy adaptations.
Standardization of Spectral Filtering Devices
Because complete darkness is often incompatible with modern occupational requirements, particularly for the expanding global night-shift workforce, wearable optical filters (blue-blocking glasses) have been aggressively marketed as a primary mitigation tool. However, until recently, the efficacy of these devices was highly variable and heavily criticized due to a lack of standardization and misleading marketing.
In 2025, an expert consensus review published in Translational Vision Science and Technology introduced the melanopic daylight filtering density (mDFD) metric. This metric objectively quantifies a filter's ability to attenuate the specific short-wavelength light (near 480 nm) responsible for activating ipRGCs and suppressing melatonin 51525354. The research concluded that to provide any meaningful physiological protection for circadian health, products must achieve an mDFD rating of ≥ 1.0 51525369.
An extensive analysis testing 26 commercial products revealed a stark discrepancy in the market. Clear or lightly tinted lenses - frequently sold to consumers for "all-day wear" to prevent digital eye strain - fail entirely to meet this biological threshold, often filtering less than 20 percent of biologically active blue light (yielding mDFD scores well below 0.50) 5269. This aligns with a 2023 Cochrane review which found no evidence that clear blue-filtering lenses help with daytime digital eye strain 52.
Conversely, lenses featuring a dark amber or orange tint routinely achieve mDFD scores exceeding 1.4, successfully preserving endogenous melatonin secretion even in brightly lit, simulated shift-work environments (800 lux) without impairing cognitive performance or objective alertness 5255. A comprehensive review of 16 independent field studies confirmed that when "true" blue-blocking glasses (mDFD ≥ 1.0) are utilized, 100 percent of the trials reported measurable improvements in sleep duration, advanced circadian rhythms, attenuated melatonin suppression, or enhanced next-day mood 515369. For instance, individuals suffering from insomnia randomized to wear highly rated amber lenses for two hours before bed increased their total sleep time by an average of 28 minutes 52.
| Lens Tint Category | Typical mDFD Rating | Circadian Efficacy | Appropriate Usage Guidelines |
|---|---|---|---|
| Clear / Near-Clear | < 0.50 | Insufficient | Ineffective for circadian protection 52. |
| Amber | ~0.28 - 1.00 | Variable / Weak | Efficacy depends heavily on the specific brand's spectrum. |
| Grey Tint | ~1.04 | Adequate | Suitable alternative for users highly sensitive to color distortion. |
| Orange / Dark Amber | > 1.41 | Substantial | Mandatory for night-shift workers or 2-3 hours prior to sleep 5269. |
Crucially, chronobiologists warn that the temporal application of spectral filtering is paramount. Wearing high-mDFD glasses during the day is biologically counterproductive. High-intensity daytime exposure to short-wavelength light is physiologically necessary to anchor the master circadian clock, promote daytime alertness, and ensure a robust melatonin signal the following evening 5152.
Integrative Architectural Illumination and Urban Policy
At the macro level, mitigating the impact of artificial light at night requires a fundamental transition toward "integrative lighting" designs in urban infrastructure and architectural planning. This paradigm involves implementing variable-spectra lighting systems that deliver high M-DER light during the day to mimic natural photic entrainment, while automatically transitioning to low m-EDI, warm-spectrum lighting (e.g., 2200K to 2700K) as dusk approaches 14151656. Furthermore, addressing the expanding light footprint in industrializing regions demands stringent municipal regulations limiting upward light emission, enforcing curfews on non-essential commercial illumination, and protecting biological corridors from ecological light pollution.
Ultimately, translating this extensive body of chronobiological research into actionable public health policy requires adopting standardized biological metrics (such as m-EDI, M-DER, and mDFD) to govern the engineering of the built environment. By recognizing artificial light as an environmental exposure variable and realigning the engineered lightscape with human evolutionary biology, global societies can begin to mitigate the escalating, syndemic burden of circadian-driven chronic disease.