Anesthetic effects on the neural basis of consciousness
The intersection of clinical anesthesiology and the cognitive neurobiology of consciousness represents one of the most profound frontiers in modern neuroscience. Since the first public demonstration of ether anesthesia at the Massachusetts General Hospital in 1846, general anesthetics have transitioned from mere clinical utilities that facilitated painless surgery to indispensable scientific probes 12. By providing a mechanism to reversibly manipulate the state of consciousness, anesthetics grant researchers a unique pharmacological lever to identify the necessary and sufficient neural architectures that generate subjective experience. Modern high-density electroencephalography (EEG), functional magnetic resonance imaging (fMRI), positron emission tomography (PET), and magnetoencephalography (MEG) have revolutionized this pursuit, generating a wealth of empirical data that continually refines our understanding of the brain's functional connectome.
This comprehensive report evaluates the neurobiological mechanisms underlying anesthetic-induced unconsciousness. It rigorously defines the physiological and cognitive parameters of consciousness, debunks pervasive clinical misconceptions comparing pharmacological anesthesia to natural sleep, and critically evaluates leading theoretical models - specifically the Global Neuronal Workspace Theory (GNWT) and Integrated Information Theory (IIT) - in light of landmark 2025 adversarial testing. Furthermore, this analysis explores the structural vulnerabilities of the thalamocortical network, utilizing the four-loop architecture and core-matrix cell dynamics, while prioritizing the latest global neuroimaging discoveries from 2023 to 2026 across Asian, European, and American institutions. Finally, the report examines the critical limitations of using anesthetics as a natural experiment for consciousness, specifically detailing off-target neurotoxic and metabolic effects that profoundly confound macroscopic functional data.
Foundational Definitions: Consciousness, Sensory Processing, and Integrated Awareness
To systematically evaluate how anesthetics obliterate consciousness, the phenomenon must first be operationally defined. In clinical neurobiology, consciousness is not viewed as a monolithic or binary entity but rather as a multidimensional construct comprising two primary axes: arousal (wakefulness) and awareness (the content of consciousness) 34. Wakefulness denotes the behavioral manifestation of arousal, which is regulated heavily by brainstem nuclei, the ascending reticular activating system, and ascending monoaminergic pathways 56. Awareness, conversely, encompasses both external awareness (the perception of environmental sensory stimuli) and internal awareness (stimulus-independent thoughts, autobiographical memory, mental imagery, and self-reflection) 5.
The neurobiologist Gerald M. Edelman conceptualized these dimensions as existing on two functional tiers. Primary consciousness represents the state of being mentally aware of the world through sensory integration in the "remembered present," allowing an organism to construct a unified scene. Higher-order consciousness involves the advanced metacognitive ability to be aware of being conscious, utilizing semantic and linguistic capabilities to bridge past memories with future intentions 8. Clinically, this integrated awareness is operationalized using standardized behavioral scales, such as the Coma Recovery Scale - Revised (CRS-R), which assesses the capacity of a patient to maintain coherent behavioral responses to multimodal physiological and environmental inputs 6. Integrated awareness signifies that the brain is not merely receiving discrete data points in isolation but synthesizing them into a continuous, unified phenomenological experience.
A critical neurobiological distinction must be made between primary sensory reception and conscious sensory perception. Foundational research by investigators such as George Mashour and Michael Alkire established that primary sensory processing frequently remains entirely intact during anesthetic-induced unconsciousness 12. Clinical monitoring of somatosensory evoked potentials during surgery routinely demonstrates that signals successfully travel from the periphery to the primary sensory cortex. Furthermore, intracranial neurophysiology in humans and non-human primates demonstrates that visual processing persists in early visual cortex areas (V1, V2), and auditory or tactile stimuli successfully trigger robust local field potentials in the superior temporal gyrus (STG) and primary somatosensory cortex 789.
The failure of consciousness under general anesthesia, therefore, does not arise from a fundamental blockade of primary sensory afferents. Instead, anesthesia induces a profound, hierarchical communication breakdown. While the primary sensory cortices detect and process incoming information, the progression of these signals to higher-order cognitive regions - such as the posterior parietal cortex (PPC) and prefrontal cortex (PFC) - is abruptly severed 7910. Sophisticated decoding software utilizing microelectrode arrays has shown that while sensory information is decodable in the awake state across the entire cortical hierarchy, this information vanishes in the associative and cognitive cortices during propofol anesthesia 910. In simpler terms, the environmental stimuli are received by the brain, but the large-scale neural integration required to weave that localized information into a state of conscious awareness is completely abolished 111.
Dispelling the Paradigm: Anesthesia is Not Deep Sleep
A pervasive misconception within both public discourse and historical clinical practice is that general anesthesia acts as a surrogate for deep, slow-wave sleep. While both states share overt behavioral unresponsiveness and exhibit macroscopic slow-wave EEG oscillations (such as delta waves), their underlying neurobiological mechanisms, temporal dynamics, and metabolic profiles are fundamentally distinct 11213.
Natural sleep is a cyclically structured, homeostatically driven physiological process that oscillates predictably between non-rapid eye movement (NREM) and rapid eye movement (REM) stages. These stages are governed by endogenous circadian rhythms and precise, localized neuromodulatory shifts involving serotonin, norepinephrine, acetylcholine, and orexin. Throughout natural sleep, the brain's functional geometry retains significant flexibility, allowing for spontaneous arousability and the generation of rich, internally generated conscious experiences (dreaming) during both REM and certain NREM phases 35. During sleep-induced loss of consciousness, reductions in the brain's capacity for information integration are highly localized, primarily targeting posterior brain regions, while leaving broader network integrities relatively undisturbed 1214.
In stark contrast, pharmacological general anesthesia forces the brain into an artificial, rigid, and inescapable attractor state 1815. Anesthetics bypass endogenous homeostatic cycles to directly hijack molecular targets, predominantly the gamma-aminobutyric acid type A ($GABA_A$) receptor or the N-methyl-D-aspartate (NMDA) receptor, plunging the brain into a profound and continuous state of disconnectedness. Functional neuroimaging demonstrates that anesthetic-induced unconsciousness suppresses integrated information at both local and global scales simultaneously, collapsing large-scale functional networks 1214.
Furthermore, a profound prefrontal cortex disconnection reliably differentiates general anesthesia from natural sleep. While sleep allows the prefrontal cortex to remain loosely coupled to other networks, general anesthetics like propofol and sevoflurane cause a near-total functional isolation of prefrontal executive centers from posterior processing regions 11016. Measuring spatiotemporal complexity via the perturbational complexity index (PCI) - which assesses the brain's deterministic response to transcranial magnetic stimulation (TMS) - further dissociates the two states. Anesthetic depths suppress cortical signal diversity far beyond what is observed even in the deepest stages of NREM sleep 1221. Consequently, general anesthesia is more accurately described as a reversible pharmacological coma, characterized by severe cortical fragmentation and metabolic suppression, rather than any physiological analog to sleep 17.
Evaluating Theoretical Frameworks: GNWT vs. IIT
The pursuit of identifying the exact neural correlates of consciousness (NCC) is heavily driven by two dominant, often competing, theoretical frameworks: the Global Neuronal Workspace Theory (GNWT) and Integrated Information Theory (IIT). Recent adversarial collaborations, utilizing rigorous neuroimaging of anesthetic and perceptual states, have subjected both theories to unprecedented scrutiny.
Global Neuronal Workspace Theory (GNWT)
Championed by Stanislas Dehaene and Jean-Pierre Changeux, GNWT proposes a functionalist and computational perspective on the origins of consciousness. According to GNWT, conscious experience arises when a specific piece of sensory or internally generated information is "globally broadcast" across an expansive network of high-level cortical processors 1618. This global workspace relies heavily on long-distance, reverberant reentrant loops connecting the prefrontal cortex, temporal lobes, and parietal association areas 81624. The hallmark of GNWT is a non-linear "ignition" event - a sudden burst of large-scale, synchronized neural activity that occurs approximately 300 milliseconds after a stimulus is presented, propelling the discrete information into global conscious awareness. From this perspective, anesthetic agents obliterate consciousness by selectively dampening the reverberant connectivity between the nodes of this workspace, preventing the prefrontal cortex from initiating or sustaining the global broadcast 1625.
Integrated Information Theory (IIT)
In stark contrast, IIT, initially formulated by Giulio Tononi and further developed into its most recent iteration (IIT 4.0), approaches consciousness from a phenomenological, axiomatic foundation. IIT argues that consciousness is defined by information that is simultaneously highly integrated (cannot be decomposed into independent parts) and highly differentiated (capable of a vast repertoire of unique states) 1621. The quantity of consciousness is mathematically operationalized as $\Phi$ (big phi), representing the maximum irreducible intrinsic cause-effect power of a physical system 1219.
IIT 4.0 defines the substrate of consciousness through "distinctions" (cause-effect states specified by a transition probability matrix) and "relations" (how these causal powers overlap and interconnect) 121419. Anatomically, IIT posits that the physical substrate of consciousness predominantly resides in the "posterior hot zone" - comprising the parietal, occipital, and temporal cortices - where the grid-like cytoarchitecture optimally supports high $\Phi$ 1618. According to IIT, anesthetics abolish consciousness by breaking down the brain's repertoire of states, effectively disintegrating the posterior hot zone and causing a precipitous drop in integrated information 1214.
The Cogitate Consortium: Adversarial Testing (2025)
In 2025, the open-science Cogitate Consortium published a landmark adversarial collaboration in the journal Nature that directly juxtaposed the core predictions of GNWT and IIT using a theory-neutral experimental design. Utilizing a massive, 256-participant cohort and multimodal neuroimaging (fMRI, MEG, and intracranial EEG), the study sought to rigorously falsify key tenets of each theory 202122.
The findings delivered severe empirical challenges to both paradigms. For IIT, the central prediction that conscious experience is defined by sustained synchronization within the posterior cortical hot zone failed. The empirical data demonstrated a distinct lack of sustained synchronization across early and mid-level visual areas within the posterior cortex during conscious perception, directly undermining the claim that this specific connectivity topology alone specifies consciousness 202122.
GNWT suffered equally critical challenges. While GNWT explicitly predicts a prominent "ignition" burst at both the onset and offset of a conscious stimulus, researchers observed a general lack of ignition at stimulus offset across the modalities 182021. Furthermore, while the prefrontal cortex represented general category features of a conscious stimulus, it consistently failed to represent specific identity or orientation details that participants consciously experienced. This finding heavily restricts the PFC's role as the definitive universal workspace for all conscious content 2021.
Despite these substantial challenges to the precise spatial and temporal assumptions of GNWT and IIT, empirical measurements of integrated information continue to validate IIT's broader predictions regarding global network collapse. Japanese researchers applying the rigorous IIT 4.0 mathematical framework to fMRI datasets in 2025 (utilizing transition probability matrices computed via the PyPhi toolbox) confirmed that total system $\Phi$ consistently and precipitously drops during propofol-induced loss of consciousness. This reduction affects both global whole-brain structures and local intra-network modules, specifically dismantling the Ventral Attention Network (VAN), Default Mode Network (DMN), and Dorsal Attention Network (DAN) 1214.
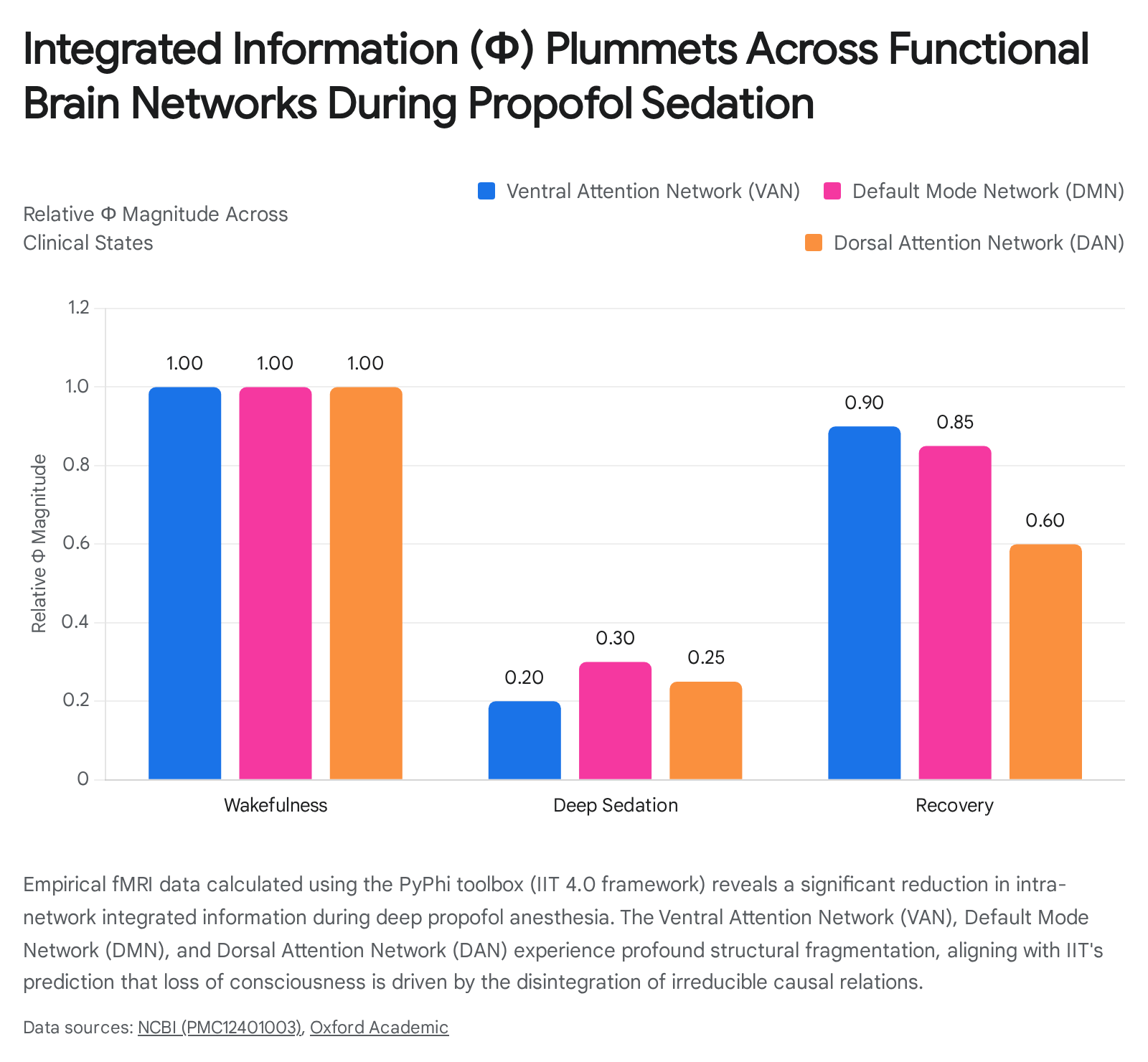
Furthermore, inter-network connectivity analysis revealed that the "frontal set," spanning frontal, parietal, and temporal lobes, exhibited a significant decrease in $\Phi$ during mild sedation before returning to baseline during recovery 12. Consequently, while the exact anatomical hubs posited by GNWT and IIT require substantial revision, the general principles of cortical disconnection, spatial fragmentation, and informational disintegration remain the standard operational models for explaining anesthetic action.
The Thalamocortical Architecture: Gateways of Consciousness
General anesthetics exert profound functional effects on the cortico-thalamo-cortical (CTC) loops, a highly dense and organized array of reciprocal excitatory and inhibitory projections linking the entire cerebral cortex with the thalamus. This architecture serves as the central router for sensory transmission, attention, and cognitive integration 132324. During normal wakefulness, the thalamus maintains rapid, irregular high-frequency firing patterns. Under general anesthesia, robust GABAergic inhibition - particularly driven by the thalamic reticular nucleus (TRN), which acts as an inhibitory shell encapsulating the dorsal thalamus - forces thalamocortical relay neurons into a state of prolonged hyperpolarization 13182526. This hyperpolarization physically restricts ascending sensory signal propagation and imposes slow, massive, synchronized oscillations (e.g., delta waves and hypercoherent alpha rhythms) across the cortex, thereby functionally disintegrating trans-cortical communication 1327.
Core vs. Matrix Thalamic Subpopulations
To accurately understand how anesthesia dismantles consciousness, the thalamus cannot be treated as a homogenous relay station. It exhibits immense cytoarchitectural heterogeneity, defined primarily by two distinct excitatory projection neuron classes: Core cells and Matrix cells 2836.
Core cells, which are rich in the calcium-binding protein parvalbumin (PVALB), originate primarily from first-order thalamic nuclei. They execute targeted, topographically precise feedforward projections to the granular layers (Layer 4) of specific, unimodal sensory cortices (e.g., the primary visual or auditory cortex) to preserve the sharp content and constancy of raw perception 112837. Conversely, Matrix cells, which are rich in calbindin (CALB1), populate higher-order thalamic nuclei such as the intralaminar, mediodorsal (MD), and pulvinar (Pul) nuclei. They execute diffuse, widespread feedback projections across the superficial, supragranular layers (specifically Layer 1) of transmodal association cortices 1128. Matrix cells are responsible for temporal coordination, setting perceptual thresholds, and binding disparate sensory inputs into an integrated conscious gestalt 1128.
A landmark 2024 fMRI study by Huang et al. utilizing advanced functional gradient mapping revealed that propofol-induced unconsciousness does not suppress the thalamus uniformly. Instead, propofol causes a preferential, catastrophic disruption of thalamic matrix cell connectivity 113637. The empirical data indicated that matrix cell composition accounted for 84% of the variance in thalamocortical functional geometry alterations during deep sedation, leaving the unimodal core cell networks relatively less perturbed 3637. This structural selectivity elegantly explains the paradox of intact primary sensory processing during anesthesia: the core cells continue to deliver specific sensory inputs to the granular cortex, but the matrix cells are suppressed and fail to deliver the requisite contextual integration to the supragranular layers, effectively abolishing subjective experience 911.
The Four-Loop Architecture
Building upon this core-matrix dichotomy, the "Four-Loop Architecture" proposed by Stewart (2026) outlines a highly specific, necessary neural substrate for consciousness, detailing how anesthetic disruption of any single component reliably abolishes awareness 2939.
The first loop, the Layer 5 Pyramidal Recurrent Loop, operates as the definitive gateway to conscious access. It relies on apical-basal coincidence detection within Layer 5 (L5) pyramidal neurons. Somatic burst firing in these neurons only occurs if an ascending sensory signal at the basal dendrites coincides precisely with a top-down contextual efference copy arriving at the apical tufts in Layer 1 2939. General anesthetics, particularly propofol, systematically sever this apical-basal integration, preventing the burst firing necessary to threshold a signal into the conscious processing cycle 2940.
The second loop is the Central Lateral Thalamocortical Loop. This matrix-driven circuit utilizes the central lateral (CL) thalamus to globally broadcast the distributed cortical representation of prior processing cycles. This broadcast ensures that any newly gated signal from Loop 1 is contextualized within the brain's existing cognitive state 29. Anesthesia heavily suppresses these calbindin-positive matrix projections, eliminating the brain's contextual backdrop 2940.
The third loop, the VTA-Cortical Dopaminergic Loop, relies on the ventral tegmental area (VTA) to provide a critical resolution event. By encoding expectation deviation via phasic dopamine release, it forces the neural system to collapse multiple competing contextualized representations into a single, definite weighted state 529. Finally, the fourth loop, the ACC-Mediodorsal Thalamic Loop, utilizes the anterior cingulate cortex (ACC) and mediodorsal (MD) thalamus to convert the resolved state into behavioral commitment. Crucially, they generate the efference copy that loops back to the apical dendrites of Layer 1, configuring the threshold conditions for the next cycle of Loop 1, thereby sustaining the continuous stream of consciousness 2941.
The Four-Loop model comprehensively explains why pharmacological agents with vastly different molecular targets (e.g., $GABA_A$ agonists versus NMDA antagonists) all successfully cause unconsciousness: they each successfully disrupt at least one vital node in this highly interdependent, cyclical hierarchy, collapsing the conscious continuum 2529.
Comparative Profiles: Receptor Targets, EEG Signatures, and Network Impacts
General anesthetics are not pharmacologically monolithic. They span a wide array of molecular targets that converge on the ultimate behavioral endpoint of unconsciousness. Advanced neuroimaging and high-density EEG arrays have delineated the unique spatial and spectral signatures of these distinct drug classes 3304331. Furthermore, modern computational analyses, such as phase-shifting measurements pioneered by the Miller Lab at MIT, demonstrate that across these diverse mechanisms, anesthetics universally disrupt the dynamic stability of the brain, pushing neural populations out of phase and degrading the brain's delicate balance between excitability and chaos 153233.
The following table synthesizes the distinct mechanisms, macroscopic EEG signatures, and functional connectivity impacts of the primary anesthetic agents currently in clinical use and active research:
| Anesthetic Class | Primary Receptor Targets | Distinct EEG Signatures | Connectome & Thalamic Impact | Foundational Mechanisms |
|---|---|---|---|---|
| Propofol (Alkylphenol) | Positive allosteric modulator of $GABA_A$ (enhances slow, fast, and tonic subtypes). 4334 | Massive increase in low-frequency $\delta$ (1-4 Hz) and slow oscillations (<1 Hz). Highly synchronized, hypercoherent frontal $\alpha$ (8-12 Hz) rhythms. Decreased high-frequency $\gamma$ (>30 Hz). 1433134 | Severe disruption of $\alpha$-band connectivity in parietal, occipital, and subcortical areas. Preferential functional disconnection of the Pulvinar (Pul) and matrix-rich thalamic nuclei. 33631 | Disrupts Layer 5 apical-basal coincidence. Pushes brain waves out of phase (increasing interhemispheric alignment but causing severe intrahemispheric misalignment). Unbalances dynamic stability. 15293233 |
| Sevoflurane (Volatile Ether) | Two-pore potassium channels (K2P), $GABA_A$ enhancement, NMDA antagonism, Glycine receptor modulation. 1743 | Enhances $\delta$ and $\theta$ oscillations. Exhibits similar frontal $\alpha$ coherence to propofol, but with broader global cortical suppression. 433536 | Broadly reduces functional connectivity differences globally. Notably, Global Signal Regression (GSR) in fMRI heavily diminishes sevoflurane network signatures, unlike propofol. Shows limited disruption to specific thalamocortical connections. 336 | Suppresses excitatory synaptic transmission. Profoundly alters mitochondrial complex I activity and fatty-acid $\beta$-oxidation (FAO), contributing to deep metabolic suppression. 353738 |
| Ketamine (Arylcyclohexylamine) | Primary antagonist of the N-methyl-D-aspartate (NMDA) glutamatergic receptor. 4352 | Attenuates $\alpha$ and $\beta$ power. Characterized by alternating slow $\delta$ waves and fast, burst-like $\gamma$ oscillations, generating an active, noisy EEG spectrum. 1343 | Disconnects the anterior-posterior network axis. Does not reduce activity in the central thalamic nuclei. Instead, it drives increased metabolic activity specifically in the left parietal cortex. 395440 | Induces a "dissociative anesthesia" state. Cortical regions remain highly active and excitable, but functional cross-talk is desynchronized, leading to hallucinations and internal disconnected consciousness. 51354 |
| Dexmedetomidine | Highly selective $\alpha_2$-adrenergic receptor agonist (acting primarily on the locus coeruleus). 1541 | Closely mimics natural NREM stage 2 sleep. Produces widespread slow-wave $\delta$ activity and characteristic sleep spindle-like rhythms (11-15 Hz). 1333 | Decreases metabolic activity firmly in the left midline thalamus and central thalamic nuclei, similar to natural sleep pathways. 3954 | Shuts down the ascending monoaminergic arousal pathways at the brainstem level, allowing endogenous GABAergic sleep pathways (e.g., VLPO) to induce a sleep-like unresponsiveness. 1354 |
| KSEB 01-S2 (Novel Class) | Selectively targets $GABA_A$-slow synapses exclusively. 34 | Diverges sharply from broad-spectrum $GABA_A$ agonists. Produces a massive, selective increase in $\theta$ frequency (4-8 Hz) power and a significant drop in low $\gamma$ power. 34 | Flattens chaotic attractor plots similarly to propofol, indicating a collapse of neural signal complexity despite a completely different spectral profile. 34 | Demonstrates that unconsciousness can be achieved through highly targeted, subtype-specific synaptic depression, challenging the necessity of widespread multi-receptor modulation. 34 |
Global Expansion of Neuroimaging Discoveries (2023-2026)
Historically, research into the neural correlates of anesthetic unconsciousness has been heavily concentrated in North American institutions. However, the period from 2023 to 2026 marks a paradigm shift, with groundbreaking, non-US centric research fundamentally advancing the field.
Chinese research institutes have rapidly mobilized high-density data collection and advanced computational modeling to decode conscious states. Researchers at the Institute of Biophysics at the Chinese Academy of Sciences (Li Ang and Wang Xiaoqun, 2023) utilized multimodal, cross-scale fMRI data from humans and macaques across various states (anesthesia, sleep, psychedelics) to discover a universal neural signature of consciousness. They identified that global consciousness depends heavily on hierarchical dynamic patterns in topological space, introducing the "constrained brain spaciotemporal nesting feature" as a robust, objective metric for determining the state of consciousness 4243.
Similarly, extensive dynamic brain network studies emanating from Shanghai Jiao Tong University and the University of Electronic Science and Technology of China have utilized high-density 128-channel EEG source localization to track the exact temporal breakdown of consciousness. They successfully demonstrated that the precise loss of alpha-band functional connectivity between the parietal, occipital, and subcortical regions is the primary biological marker predicting the transition into propofol-induced unconsciousness 304445. Furthermore, China's rapidly expanding clinical neurotechnology sector is bridging academic Brain-Computer Interface (BCI) infrastructure directly into operating rooms. By utilizing hardware from domestic suppliers like Neuracle, hospitals are feeding massive, real-time EEG datasets into machine learning algorithms designed to titrate anesthesia flawlessly, creating a rapid feedback loop between academic research and clinical application 61.
Simultaneously, European and Japanese collaborations have revolutionized our spatial understanding of drug action. The Turku PET Centre in Finland executed an elegant series of functional brain imaging studies (2023) differentiating the thalamic impacts of various anesthetics. They definitively proved that while propofol, dexmedetomidine, and sevoflurane explicitly suppress the central thalamic nuclei to achieve unresponsiveness, the dissociative agent S-ketamine achieves the same behavioral endpoint without central thalamic suppression. Instead, S-ketamine relies on aberrant hyperactivation of the left parietal cortex, distinguishing its dissociative nature metabolically 5440.
In Japan, pioneering clinical trials (2025) led by Tokyo Women's Medical University and St. Luke's International Hospital have translated these theoretical EEG models directly into pediatric practice. By utilizing continuous brain wave monitoring to visualize the exact threshold of consciousness loss in children aged 1 to 6, anesthesiologists were able to safely reduce the concentration of sevoflurane administered (e.g., from a standard 5% to 2% for induction, and 2.5% to 0.9% for maintenance). This neurobiologically guided precision dramatically accelerated patient recovery times - allowing breathing tubes to be removed significantly earlier - and drastically reduced the incidence of post-operative pediatric delirium 3346.
Limitations: The Confounding Roles of Metabolic Suppression and Neurotoxicity
While general anesthetics offer a highly reliable method for reversibly extinguishing consciousness, their utility as a "clean" experimental probe is inherently compromised by profound off-target physiological effects. When neuroscientists observe a breakdown in functional connectivity (e.g., via fMRI BOLD signals) under anesthesia, it is critical to rigorously ask whether this represents a direct disruption of information processing networks, or merely a secondary artifact of severe metabolic suppression and cellular toxicity 374748.
Metabolic Suppression and Mitochondrial Dysfunction
General anesthetics are potent metabolic inhibitors. Volatile agents like isoflurane and sevoflurane, as well as intravenous propofol, directly interfere with the mitochondrial electron transport chain (mETC), primarily via the severe suppression of mitochondrial Complex I 384749. Complex I is responsible for oxidizing NADH and initiating the biochemical cascade that generates adenosine triphosphate (ATP). The cerebral cortex requires immense ATP reserves simply to maintain the resting membrane potentials required for spontaneous synaptic firing and neurotransmitter recycling 4750. By inhibiting Complex I, anesthetics precipitate a rapid decline in intracellular ATP, radically increasing extracellular oxygen tension as tissues lose the metabolic capacity to consume oxygen 47.
This creates a profound experimental confound: is the observed loss of high-frequency gamma oscillations and cortical connectivity a targeted pharmacological disruption of consciousness circuits (such as specific $GABA_A$ modulation), or is the brain simply starved of the metabolic fuel required to sustain long-range network integration? Animal models overwhelmingly support the metabolic hypothesis. Caenorhabditis elegans and Drosophila melanogaster models engineered with mutations mimicking human mitochondrial disorders (such as Leigh syndrome, featuring a mutation in the ND23 subunit of Complex I) display extreme hypersensitivity to volatile anesthetics, entering fatal neurotoxic cascades at fractions of standard clinical doses 3851. Knockout mice lacking the Ndufs4 gene (encoding a Complex I subunit) also exhibit extreme sensitivity, proving that mitochondrial function directly dictates the behavioral state of anesthesia 5051. Furthermore, recent 2026 data indicates that sevoflurane actively suppresses fatty acid $\beta$-oxidation (FAO) via the PPAR$\alpha$/CPT1a pathway in neural stem cells, crippling energy homeostasis in the central nervous system 3752. Propofol acts similarly, severely inhibiting acylcarnitine transferase, which is the underlying mechanism of the often fatal propofol infusion syndrome 4952.
Developmental Neurotoxicity
The challenge of utilizing anesthetics to study consciousness is further compounded by their well-documented neurotoxic effects on both developing and aging brains. In animal models ranging from rodents to non-human primates, prolonged or repeated exposure to agents like sevoflurane and ketamine during critical developmental windows of synaptogenesis triggers widespread, programmed neuronal death (neuroapoptosis) 524153. The mechanisms driving this toxicity are multifaceted, involving the over-activation of apoptotic caspase-3 pathways, severe oxidative stress, and long-term neuroinflammation, resulting in documented deficits in spatial learning, memory retention, and executive function 524170.
While major human cohort studies - such as the GAS, PANDA, and MASK trials - have demonstrated that a single, brief exposure to general anesthesia in infancy does not cause statistically significant declines in general intelligence (IQ), multiple exposures have been correlated with subtle increases in parent-reported behavioral problems and processing speed deficits 5270. Consequently, utilizing anesthetics as theoretical probes for consciousness necessitates rigorous control over dosing and exposure times, as the observed network disintegration may reflect transient neurotoxic injury and profound metabolic shutdown rather than the fundamental functional architecture of subjective experience 525370.
Conclusion
The pursuit of the neural basis of consciousness has been irrevocably accelerated by the deep study of anesthetic mechanisms. As this analysis demonstrates, anesthetics do not simply emulate the slow-wave profiles of natural sleep; they act as precise pharmacological scalpels, selectively severing the thalamocortical networks - specifically the high-order matrix cell pathways and Layer 5 coincidence detection loops - that transform basic sensory processing into integrated awareness.
Recent adversarial tests have humbled both the Global Neuronal Workspace and Integrated Information Theories, proving that the true biological footprint of consciousness likely escapes their rigid spatial architectures, favoring highly dynamic, spatiotemporally nested patterns. Driven increasingly by global non-US neuroimaging consortiums across China, Japan, and Europe, the field is moving toward a more nuanced, cell-type specific understanding of how networks like the parietal-occipital-subcortical axis sustain the mind. However, future research must continually navigate the perilous confounds of metabolic suppression and mitochondrial neurotoxicity. Only by definitively separating the targeted circuit-breaking effects of anesthetics from their broader energetic suppression will neuroscience finally decode the physiological threshold separating the conscious self from the void.