Anatomy and Function of the Mammalian Claustrum
Historical Discovery and Gross Anatomy
Early Neuroanatomical Identification
The claustrum is a thin, irregular, sheet-like structure of gray matter situated deep within the basolateral telencephalon. Its physical boundaries are defined by its encapsulation between two prominent white matter tracts: the extreme capsule medially, which separates the claustrum from the insular cortex, and the external capsule laterally, which separates it from the putamen 12. The anatomical identification of the claustrum dates back to the 17th century, appearing in the neuroanatomical drawings of Thomas Willis in 1672 3. However, the structure was first formally described by Karl Friedrich Burdach in the early 19th century, utilizing drawings by Félix Vicq-d'Azyr, who termed the region the "vormauer" or "hidden away" structure 34.
Despite centuries of awareness regarding its existence, the claustrum remained functionally enigmatic due to its obscured anatomical position, its convoluted three-dimensional shape, and its diminutive width 14. The structural proximity to major white matter tracts and the insula precluded the use of traditional invasive methodologies, such as macroscopic surgical excision or rudimentary electrical stimulation, without introducing severe off-target physiological confounds 567. The categorization of the claustrum also presents historical challenges; while it possesses fusiform cells and pyramidal somata indicative of pallial cortical derivation, it lacks canonical cortical lamination and contains cellular subtypes traditionally associated with subcortical basal ganglia structures 347.
Morphological Divergence Across Species
The claustrum is identified in all placental mammals, several monotremes, and potentially possesses structural homologs in reptiles and birds 68. Despite this widespread conservation across mammalian taxa, the gross morphology and continuity of the claustrum exhibit substantial evolutionary divergence 69.
In rodents, such as the laboratory mouse and rat, the claustrum forms a continuous, uninterrupted subcortical sheet that extends across the rostral half of the telencephalon 14. However, anatomical investigations utilizing advanced neuroimaging and histological reconstructions reveal that this physical continuity is not conserved universally, particularly in species characterized by highly gyrified cortices 69. In macaque monkeys and gorillas, the claustrum exhibits pronounced structural discontinuities, often presenting as a main nuclear body surrounded by isolated islands of claustral cells 69. This morphological fragmentation reaches its extreme in cetaceans, such as dolphins and whales, where the claustrum exists almost entirely as a scattered archipelago of isolated cellular islands 69. The presence of these physical discontinuities poses a direct architectural constraint on functional hypotheses that rely on ubiquitous intraclaustral communication, such as models proposing continuous gap-junction syncytia or uninterrupted chemical wave propagation across the structure's longitudinal axis 610.
Volumetric Ratios and Asymmetry
As the cerebral cortex expands in complexity and volume across mammalian evolution, the relative size of the claustrum inversely scales 69. Comparative volumetric analyses indicate that the ratio of the volume of the claustrum to the volume of the isocortex is highest in rodents, comprising approximately 6.5% in mice 6. In contrast, the human claustrum accounts for merely 0.25% to 0.45% of the total cerebral cortical volume 36.
In the human brain, the claustrum extends approximately 22 millimeters inferior-to-superior and 38 millimeters anterior-to-posterior 3. High-resolution neuroimaging utilizing T1-weighted structural magnetic resonance imaging (MRI) reveals consistent hemispheric asymmetry in the human claustrum. The right claustrum possesses an average surface area of 1,551.15 mm2 and a volume of 828.83 mm3, whereas the left claustrum is generally smaller, with a surface area of 1,439.16 mm2 and a volume of 705.82 mm3 311. This structural lateralization is hypothesized to mirror the lateralization of specific cortical functions, such as language processing and handedness, with functional MRI data indicating differential activation patterns between the left and right claustra depending on the cognitive task 111214.
Cellular Composition and Microarchitecture
Projection Neurons and Inhibitory Interneurons
The internal microarchitecture of the claustrum is devoid of the strict laminar organization characteristic of the adjacent cerebral cortex 27. Instead, the claustrum is composed of a dense matrix of neurons that are broadly categorized into principal excitatory projection neurons and local inhibitory interneurons 813.
The principal cells are predominantly spiny, glutamatergic projection neurons, often classified as Golgi type I neurons 28. These cells project divergent axons that penetrate the white matter tracts to heavily innervate various regions of the cerebral cortex 18. In molecular studies, these excitatory projection neurons are identified by the robust expression of specific genetic markers, notably the guanine nucleotide-binding protein gamma-2 subunit (Gng2) and vesicular glutamate transporter 2 (Vglut2) 17. The expression of Gng2, in particular, has been utilized in murine models to delineate the precise anatomical boundaries of the claustrum against the surrounding cortical gray matter, which conversely expresses high levels of the crystallin mu (Crym) marker 1.
Interspersed among the glutamatergic projection neurons are multiple diverse subpopulations of aspiny, GABAergic interneurons 813. These inhibitory interneurons are typically differentiated by their expression of calcium-binding proteins and neuropeptides, including parvalbumin (PV), somatostatin (SOM), vasoactive intestinal peptide (VIP), and neuropeptide Y (NPY) 813.
Synaptic Connectivity and Syncytial Organization
The parvalbumin-positive (PV) interneurons form an exceptionally dense, highly interconnected local network within the claustrum. These fast-spiking interneurons are linked by both classical chemical synapses and electrical gap junctions, creating a widespread inhibitory syncytium 1314.
This intrinsic GABAergic matrix strongly innervates the claustral projection neurons, exerting profound baseline suppression 41314. Extracellular single-unit recordings in awake animals reveal that claustral projection neurons are remarkably quiescent, exhibiting very low spontaneous discharge rates in the absence of external stimulation 41114. This microcircuitry suggests a highly regulated gating mechanism: significant, synchronized, and task-relevant top-down cortical input is required to overcome the local PV-mediated inhibition and trigger an efferent burst from the claustrum projection neurons 1314.
Single-Cell Transcriptomics in Non-Human Primates
Recent large-scale spatial transcriptomic projects have provided unprecedented resolution regarding the cellular taxonomy of the claustrum. Specifically, the China Brain Project generated a comprehensive single-cell spatial transcriptome atlas of the macaque claustrum 1518. By sequencing over 227,000 individual claustral cells utilizing stereotactic RNA sequencing (Stereo-seq) and single-nucleus RNA sequencing (snRNA-seq), researchers systematically identified 48 distinct transcriptome-defined cell types within the structure 161718.
This molecular mapping revealed that the majority of the glutamatergic projection neurons in the macaque claustrum exhibit significant transcriptomic similarities to the deep-layer neurons of the adjacent insular cortex 1516. This supports developmental models positing that the claustrum and insula share a common pallial origin, diverging later in embryogenesis 315. Furthermore, extensive cross-species transcriptomic comparisons between macaques, marmosets, and laboratory mice identified several macaque-specific cell types entirely absent in rodents 1516. These primate-specific glutamatergic cell types exhibit highly specific topographical localizations - with distinct ventral versus dorsal claustral zones - that preferentially co-project to functionally related cortical areas, such as the entorhinal cortex and hippocampus versus the motor cortex and putamen 16.
Table 1: Primary Cellular Subpopulations of the Mammalian Claustrum
| Cell Type Category | Primary Molecular Markers | Morphology | Anatomical Projection | Function |
|---|---|---|---|---|
| Principal Excitatory Neurons | Gng2, Vglut2, Slc17a6 | Spiny, pyramidal, or fusiform | Extraclaustral (Corticoclaustral and Claustrocortical) | Divergent output to cortical networks; primary signal transmission 178. |
| Fast-Spiking Interneurons | Pvalb (PV) | Aspiny | Intraclaustral (Local) | Mediates feedforward/feedback inhibition; highly electrically coupled via gap junctions 81314. |
| Peptidergic Interneurons | Sst (SOM), Vip | Aspiny | Intraclaustral (Local) | Modulates local excitability; targets specific dendritic domains of projection neurons 8. |
| NPY Interneurons | Npy | Aspiny | Intraclaustral (Local) | Participates in global silencing of neocortex during specific cognitive states 1419. |
Macroscale Connectomics and Network Topology
The functional capabilities of the claustrum are inextricably linked to its extensive connectivity profile. Advancements in both viral tract tracing in animal models and high-angular resolution diffusion imaging (HARDI) in humans have mapped the claustrocortical and corticoclaustral pathways with high precision, confirming the claustrum as a primary integration node 202122.
Diffusion Tensor Imaging and Fiber Density
Quantitative analyses of human connectome data utilizing resting-state functional MRI (rs-fMRI) and structural diffusion tensor imaging (DTI) demonstrate the sheer magnitude of the claustrum's wiring. In a large-scale study of 100 healthy subjects conducted by Torgerson and Van Horn, the claustrum was found to possess the highest density of white matter fiber connections per unit of regional volume of any structure in the human brain 32324.
Tractography reconstructions reveal four primary groups of white matter fibers connecting the human claustrum to the cerebral cortex: anterior, posterior, superior, and lateral tracts 21. The anterior and posterior pathways link the claustrum to the prefrontal cortex and visual areas, respectively; the superior tract targets sensorimotor cortical regions; and the lateral pathway connects with the auditory cortex 221. Additionally, a distinct medial pathway was identified linking the claustrum to subcortical basal ganglia structures, specifically the caudate nucleus, putamen, and globus pallidus 21.
Graph Theory and Rich-Club Organization
The application of graph theoretical metrics to these DTI datasets illustrates that the claustrum is not merely a highly connected region, but a primary contributor to global brain network architecture 324. By calculating metrics such as network density, characteristic path length, assortativity, and betweenness centrality, researchers confirm that the claustrum operates as a critical "rich-club" node 32526.
In network topology, a "rich club" refers to a phenomenon wherein highly connected hub nodes are also densely connected to one another 2527. These hubs serve as the backbone for global communication, facilitating extremely short communication pathways across diverse neural networks 2526. Structural mapping in mammalian connectomes demonstrates that rich-club and feeder connections spanning through the claustrum comprise the vast majority of intermodule communication paths 25. Resting-state functional analyses corroborate this structural scaffolding, revealing that the claustrum actively co-activates with core neurocognitive networks, including the Salience Network (SN), Default Mode Network (DMN), and the Frontoparietal Network (FPN) 2328.
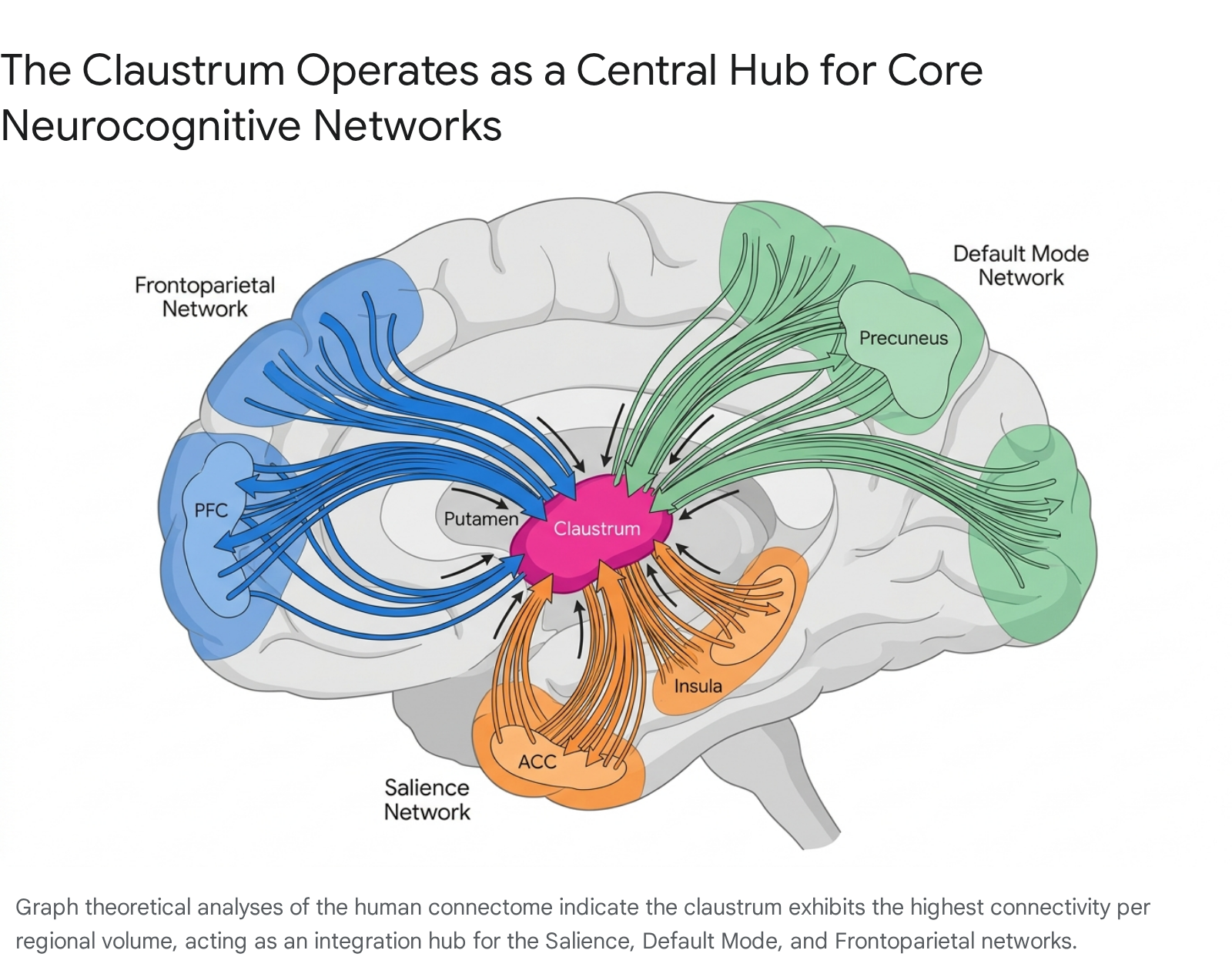
Asymmetry in Input and Output Projections
While early qualitative models assumed the claustrum was connected uniformly to all cortical areas, systematic quantification using adeno-associated virus (AAV) anterograde tracing and monosynaptic rabies virus retrograde tracing by the Allen Institute for Brain Science has refined this understanding 2029.
In the murine model, the data reveal a profound input-output asymmetry heavily biased toward executive and limbic loops. The claustrum receives its strongest inputs from frontal cortices - including the medial prefrontal cortex (mPFC), anterior cingulate cortex (ACC), and orbital frontal cortex - as well as from specific subcortical neuromodulatory centers such as the dorsal raphe nucleus (supplying serotonergic input) and the cholinergic basal forebrain 293031.
Conversely, the efferent outputs of the claustrum collateralize extensively to innervate higher-order cortical regions, particularly the retrosplenial cortex, prefrontal areas, and cingulate cortices 2931. While it projects densely to these association areas, the claustrum sends proportionally weaker outputs back to primary sensory (visual, auditory) and motor cortices 2930. Furthermore, claustrocortical outputs are generally devoid of significant subcortical targeting; with the minor exception of sparse terminal fields in the basolateral amygdala (BLA), the claustrum's output is overwhelmingly directed back to the neocortex 29.
These projections are also characterized by hemispheric biases. While bilateral connections are present, the vast majority of claustrocortical projections are ipsilateral 72032. Interestingly, retrograde tracing indicates that individual claustral neurons often branch to innervate multiple distinct cortical targets, allowing a single claustral output burst to simultaneously influence disparate functional networks 2933.
Table 2: Structural Connectivity Profile of the Mammalian Claustrum
| Brain Region / Network Node | Input Extent to Claustrum | Output Extent from Claustrum | Primary Functional Associations |
|---|---|---|---|
| Prefrontal Cortex (mPFC, OFC) | Strong (Bilateral, ipsilateral bias) | Strong | Executive function, cognitive control, task switching, working memory 293032. |
| Anterior Cingulate Cortex (ACC) | Strong (Bilateral, ipsilateral bias) | Strong | Salience detection, attentional allocation, conflict monitoring 132934. |
| Primary Sensory Cortices (V1, A1) | Weak to Moderate (Ipsilateral) | Weak | Direct sensory relay, low-level environmental feature extraction 3032. |
| Primary Motor & Somatosensory | Minimal | Minimal | Direct motor execution, somatic tactile processing 3032. |
| Hippocampal / Retrohippocampal | Strong (Ipsilateral) | Strong | Spatial navigation, episodic memory consolidation 2932. |
| Basolateral Amygdala (BLA) | Moderate | Weak (Sparse terminals only) | Emotional salience, fear conditioning, affective processing 2931. |
| Brainstem Neuromodulatory Nuclei | Moderate to Strong (Raphe) | None | Global state regulation, arousal, monoaminergic modulation 729. |
Note: Relative projection density derivations are synthesized from quantified AAV anterograde and rabies retrograde tracing datasets mapped within the Allen Mouse Brain Common Coordinate Framework 293032.
The Consciousness Hypothesis and Its Constraints
For decades, the claustrum was relegated to an anatomical curiosity. However, in 2005, Francis Crick and Christof Koch fundamentally shifted the trajectory of claustrum research by publishing a seminal hypothesis positing that the structure represents the central neural correlate of consciousness 2193536.
Crick and Koch's Multisensory Integration Model
Observing the extreme density of reciprocal connections linking the claustrum to visual, auditory, and somatosensory cortices, Crick and Koch theorized that the claustrum acts as a highly active site of multisensory integration 235. They argued that the subjective experience of consciousness requires disparate sensory modalities and motor intentions to be rapidly and seamlessly bound together into a unified percept. Utilizing the metaphor of an "orchestra conductor," they suggested that the claustrum coordinates the timing and integration of distant cortical areas, generating the unity of consciousness moment by moment 193536. Without the claustrum, they hypothesized, cortical processing would remain fragmented and non-conscious 35.
Electrophysiological Evidence Against Individual Neuron Integration
While elegant and theoretically cohesive, the strict multisensory integration model requires empirical evidence of multisensory convergence at the cellular level. Subsequent highly targeted electrophysiological investigations have largely failed to support this requirement.
Extracellular single-unit recordings in the macaque claustrum demonstrate that while the structure, when viewed as a whole, responds to both visual and auditory stimuli, the individual constituent neurons are overwhelmingly unimodal 19. A claustral neuron responsive to visual stimuli does not exhibit altered or potentiated firing rates when an auditory stimulus is concurrently presented 1937. The lack of cross-modal modulation indicates an absence of multisensory integration at the level of the individual cell 19. Furthermore, tracing data in rodents shows that while the primary visual cortex sends dense projections to the claustrum, the primary somatosensory cortex sends minimal projections, breaking the symmetry required for a universal sensory integrator 1936.
Human Lesion Studies and Traumatic Brain Injury
If the claustrum were the absolute anatomical generator or singular seat of consciousness, severe bilateral damage to the structure should theoretically result in a persistent vegetative state, coma, or complete abolition of conscious experience. Clinical case studies and cohort analyses do not support this absolute correlation.
Studying focal claustral lesions in human patients is exceptionally challenging due to the structure's diminutive width and its shared middle cerebral artery vascular supply with the adjacent insula and putamen 1538. Consequently, lesions exclusively restricted to the claustrum are extraordinarily rare. However, systemic reviews of patients with relatively isolated bilateral claustral damage - often resulting from viral encephalitis or severe status epilepticus - reveal highly variable deficits that do not manifest as a total loss of consciousness 539.
The most definitive data addressing this hypothesis stems from a voxel-based lesion symptom mapping study of 171 combat veterans who sustained penetrating traumatic brain injuries (TBI) 3840. Chau and colleagues analyzed long-term recovery and consciousness metrics, discovering that while claustrum damage was statistically associated with a prolonged duration of a loss of consciousness (LOC), it was not correlated with the frequency or initial occurrence of LOC compared to control lesions 3840.
Network Anticorrelations with Brainstem Arousal Centers
These lesion data heavily imply that the claustrum is not the generator of consciousness, but rather a vital node within the broader neural circuitry required to regain or maintain states of high arousal following severe neurological disruption 3840. Functional connectivity analyses mapping coma-inducing brain lesions further contextualize this role. Snider and colleagues demonstrated that the bilateral claustrum operates as a peak node within a distributed cortical circuit that is strongly anticorrelated (negatively correlated) with the dorsal brainstem tegmentum, the established location of the reticular activating system 51941. Damage to this distributed circuit heavily predicts LOC, suggesting the claustrum is structurally linked to the maintenance of basic wakefulness, even if it does not single-handedly generate subjective awareness 1941.
Contemporary Models of Claustral Function
As the strict multisensory integration model lost empirical favor, neuroscientists developed several nuanced hypotheses to explain the claustrum's dense connectivity. These contemporary models focus on the structure's capacity to synchronize oscillations, filter environmental salience, and rapidly instantiate higher-order cognitive networks.
Spectral Processing and Oscillation Synchrony
Smythies, Edelstein, and Ramachandran proposed an oscillation-synchrony theory, arguing that the claustrum functions not to integrate specific sensory content, but to operate as a detector, amplifier, and modulator of synchronized cortical oscillations 7333642.
In this model, when an unexpected or salient stimulus generates weak gamma-band oscillations in disparate cortical regions, the claustrum detects this nascent synchrony 3336. Utilizing the dense gap-junction syncytium formed by local claustral interneurons, the claustrum amplifies these specific frequencies via reverberating claustrocortical loops 1333642. Through a process termed spectral concatenation, the claustrum synchronizes multi-regional firing rates, effectively providing a competitive "winner-takes-all" mechanism allowing specific neural networks to gain access to the executive motor cortex for behavioral output 1043.
Despite its theoretical strength regarding neural oscillations, a major limitation of this hypothesis is its reliance on continuous, uninterrupted intraclaustral interactions (e.g., dendrodendritic synapses or gap junctions traveling the entire length of the structure). The discovery that the claustrum is physically fragmented into discontinuous islands in great apes and cetaceans fundamentally undermines the necessity of contiguous internal syncytial processing for its core mammalian function 69.
Salience Detection and the Limbic-to-Motor Interface
A subsequent functional model, proposed by Mathur, Goll, and colleagues, posits that the claustrum functions primarily as a salience or novelty detector 133444. Because claustral projection neurons are typically quiescent but respond rapidly and transiently to the onset of sudden, multimodal stimuli, the structure is ideally suited to act as an environmental filter 71319.
This model heavily emphasizes the claustrum's exceptionally robust, bilateral connectivity with the anterior cingulate cortex (ACC), which is a core node of the brain's Salience Network (SN) 3444. Under this framework, the ACC provides the claustrum with top-down expectations regarding task relevance and environmental rules. When a highly salient stimulus breaches a predetermined threshold, the claustrum is activated and subsequently broadcasts an inhibitory signal across the cortex to suppress task-irrelevant background noise 71934. For example, activating this circuit allows an organism to rapidly ignore auditory distractions during a highly demanding visual task, effectively acting as a limbic-to-motor interface for rapid attentional shifts 71934.
Network Instantiation in Cognitive Control
Synthesizing recent anatomical, optogenetic, and functional neuroimaging data, the most comprehensive contemporary framework is the Network Instantiation in Cognitive Control (NICC) model, articulated by Madden et al. (2022) 845. The NICC model proposes that the claustrum does not process or relay specific sensory content; rather, it serves as a central orchestrator directed by the frontal cortex to rapidly instantiate (assemble, reset, and synchronize) the large-scale cortical networks required for high cognitive demand 81445.
The NICC model outlines a specific three-step electrophysiological mechanism: 1. Initiation via Frontal Input: The claustrum receives a top-down signal from frontal executive cortices (such as the mPFC) indicating a necessary shift in cognitive demand, task rules, or environmental context 814. 2. Transformation and Amplification: This incoming signal activates the sparse, highly interconnected PV-positive interneuron network within the claustrum, which transiently releases the principal projection neurons from their baseline inhibition, amplifying the neural command 814. 3. Broadcasting and Instantiation: The claustrum fires divergent, high-frequency bursts back to disparate cortical nodes across the telencephalon. Crucially, these claustral efferents predominately target cortical inhibitory interneurons (e.g., NPY and PV cells in the cortex). This unique targeting evokes widespread, transient feedforward inhibition across the neocortex, forcing a brief "down-state" that silences ongoing asynchronous activity 81446. As the feedforward inhibition lifts, the target cortical regions rebound into a synchronized, phase-locked "up-state," successfully instantiating a new functional network (such as the Frontoparietal Network) perfectly suited to the novel cognitive task 81446.
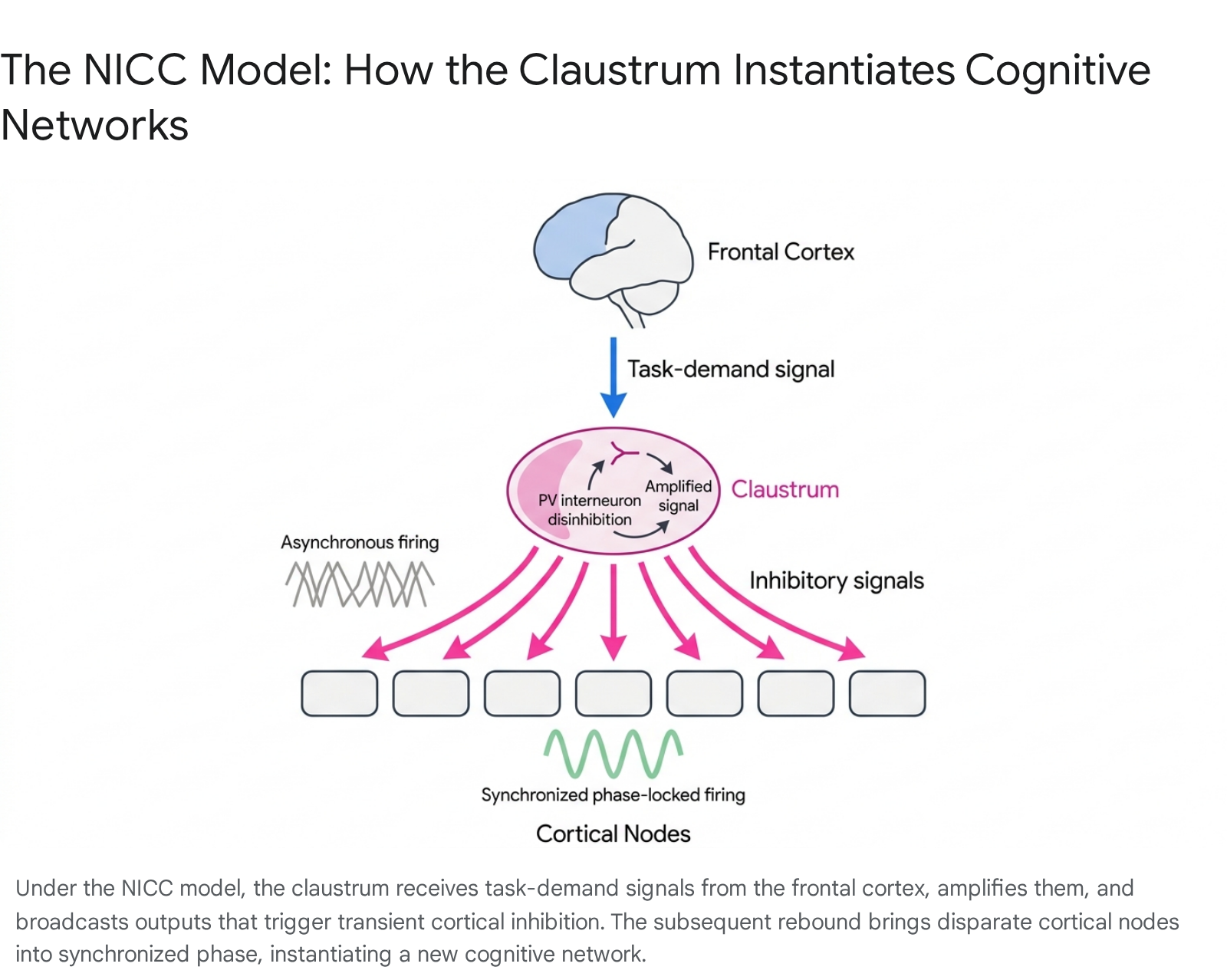
Functional Magnetic Resonance Imaging During Cognitive Tasks
The NICC model is strongly supported by recent task-based fMRI data from human cohorts. Analyses of healthy participants engaged in highly cognitively demanding tasks - such as the Stroop task, the multi-source interference task (MSIT), the AX-Continuous Performance Task (AX-CPT), and cued task-switching paradigms - reveal that claustrum activation scales directly with cognitive load 121447.
Specifically, the claustrum exhibits pronounced bilateral activation precisely during the transition periods between distinct tasks (network switching) and during high-conflict trials 121447. In these moments, the claustrum acts in concert with task-positive frontoparietal networks to establish the neural architecture necessary for goal-directed execution, actively suppressing default mode networks and reducing cognitive interference 121447.
Table 3: Evolution and Comparison of Major Claustral Functional Hypotheses
| Functional Model | Primary Proponents | Proposed Core Mechanism | Critical Empirical Limitations |
|---|---|---|---|
| Seat of Consciousness / Orchestra Conductor | Crick & Koch (2005) 235 | Binding of multimodal sensory inputs into a unified conscious percept. | Single neurons are overwhelmingly unimodal; bilateral lesions alter duration, not presence, of consciousness 193740. |
| Spectral Concatenation | Smythies, Edelstein (2012) 3642 | Amplification of synchronized gamma oscillations via intraclaustral gap-junction syncytia. | Relies heavily on continuous intra-claustral architecture, contradicted by fragmented morphology in apes and cetaceans 610. |
| Salience / Novelty Detection | Mathur, Goll (2015) 1334 | Transient bursting in response to novel stimuli suppresses irrelevant cortical noise. | Does not fully explain the claustrum's continuous functional connectivity with resting-state networks (DMN, FPN) in absence of novel stimuli 23. |
| Network Instantiation in Cognitive Control (NICC) | Madden et al. (2022) 845 | Frontally-directed feedforward inhibition forces asynchronous cortices into synchronized functional networks. | Challenging to selectively manipulate the entire claustrum in vivo to definitively prove causal network shifts without accidentally affecting the adjacent insula 814. |
Neuropathology and Clinical Implications
Because the claustrum acts as a global modulator of cortical excitability and cognitive network instantiation, its dysfunction is increasingly implicated in a wide range of severe neurological and psychiatric disorders.
Epileptogenesis and the Claustrum Sign
The claustrum's intrinsic capability to trigger widespread cortical synchronization renders it highly vulnerable - and potentially contributory - to epileptogenesis. In clinical neurology, the "claustrum sign" is a well-documented, highly specific radiological phenomenon wherein patients suffering from refractory status epilepticus (unremitting, continuous seizures without recovery of consciousness) or severe febrile illnesses exhibit bilateral T2/FLAIR hyperintensity localized precisely to the claustrum on MRI scans 548.
It is hypothesized that the ventral subsector of the claustrum, owing to its dense, reciprocal connections with limbic structures such as the amygdala and entorhinal cortex, facilitates the rapid propagation and generalization of focal limbic seizures across the entire cortical mantle 48. The disruption of typical claustral functioning during these massive synchronization events likely contributes directly to the profound loss of awareness and confusion observed during complex partial and generalized seizures 4849. Following acute status epilepticus, patients with persistent claustrum lesions frequently experience long-term alterations in mental state, including profound disorientation and stupor, even if motor function recovers 539.
Delusional Syndromes and Psychiatric Disorders
Disruptions in the functional connectivity between the claustrum and the prefrontal cortex have been noted in several psychiatric conditions characterized by severe deficits in cognitive control, particularly schizophrenia and major depressive disorder 50. In perceptual-inference models of psychosis, hallucinations and delusions are thought to result from alterations in the neural updating of internal models of the environment 51. The claustrum's dense involvement in the Frontoparietal and Default Mode Networks positions it perfectly to mediate these internal models.
The claustrum's potential role in maintaining a unified sense of internal state and self-representation is highlighted by its association with exceedingly rare delusional states. Structural damage to the claustrum has been directly linked to clinical instances of the Cotard delusion - a severe psychiatric condition wherein the patient firmly believes they are dead, do not exist, or are actively decaying 5. This suggests that a structural breakdown in the claustrum's ability to coordinate the Default Mode Network may result in profound perceptual dissociations regarding one's own physical existence and "selfhood" 5.
General Anesthesia and Cortical Disconnection
The claustrum also plays a highly specific modulatory role in states of decreased arousal. In naturally occurring states, the structure is integral in coordinating widespread synchronous slow-wave activity (SWS) across the neocortex during sleep 1938.
Furthermore, pharmacological studies highlight the claustrum's unique sensitivity to anesthetics. Resting-state fMRI networks involving the claustrum - which form robust structural scaffolds during normal wakefulness - are entirely abolished under isoflurane and propofol-induced general anesthesia 3438. Because the claustrum is densely packed with GABAergic interneurons, it is highly receptive to the mechanisms of action of common general anesthetics. This suggests the claustrum is a primary pharmacological target for anesthetic agents aiming to safely decouple higher-order cortical networks, effectively severing the brain's ability to instantiate the synchronized states required for waking consciousness 38. Similarly, the application of classical psychedelics (such as psilocybin or Salvinorin A) has been shown to induce profound disintegration of brain-wide networks primarily by driving a rapid decrease in claustrum activity or altering its inhibitory influence on subcortical areas 74650.
Methodological Challenges and Future Research
Limitations of Current Investigative Techniques
Despite rapid conceptual advances over the last two decades, studying the claustrum remains fundamentally hindered by its challenging anatomy. Its location, tightly sandwiched between the massive white matter tracts of the external and extreme capsules, and its structural contiguity with the deep layers of the insular cortex in many species, complicate the use of traditional neuroscientific techniques 1820.
Standard electrical deep-brain stimulation or gross excitotoxic lesioning often results in electrical spillover or chemical diffusion into the adjacent white matter or insula, leading to confounding experimental results regarding the claustrum's genuine role in consciousness, pain processing, and sensory arrest 5819. Furthermore, the lack of standardized boundaries in rodent models often results in the adjacent endopiriform nucleus (which primarily handles olfactory processing) being erroneously categorized as the "ventral claustrum," further muddying the functional literature 620.
Advancements in Optogenetics and Targeted Neural Manipulation
Future research relies heavily on the deployment of advanced intersectional genetics. By utilizing Cre-driver transgenic mouse lines that target claustrum-specific molecular markers (e.g., Gng2, Vglut2) or specific primate-adapted transcriptomic signatures, researchers can deploy optogenetics and chemogenetics (DREADDs) to selectively excite or silence precise populations of claustral projection neurons with millisecond temporal precision 71920.
Initial optogenetic studies have already demonstrated that activating claustral inputs to the prefrontal cortex can transiently inhibit cortical pyramidal activity via local neuropeptide Y (NPY) interneurons, providing vital cellular-level evidence for the NICC model 14. Furthermore, observing claustral activity in awake, behaving non-human primates during complex, multi-stage cognitive tasks via high-density electrophysiology will be vital 71337. By transitioning from static anatomical tracing to dynamic, cell-type-specific functional manipulation, the claustrum is shedding its reputation as the enigmatic "seat of consciousness." Through the synthesis of spatial transcriptomics, macroscale connectomics, and dynamic network modeling, the claustrum has instead emerged as an elegant, highly specialized integration hub essential for the agile orchestration of mammalian cognitive control.