Alpha-ketoglutarate as an aging intervention
Introduction
Alpha-ketoglutarate (AKG) is an endogenous, highly conserved metabolic intermediate that functions as a critical node in cellular energy production, amino acid biosynthesis, and systemic nitrogen homeostasis. Historically categorized strictly as an intermediary component of the mitochondrial tricarboxylic acid (TCA) cycle, advances in geroscience over the past decade have fundamentally reclassified AKG as a pleiotropic signaling molecule capable of modulating the biological aging process. Systemic levels of alpha-ketoglutarate decline precipitous with advancing chronological age; clinical observations indicate that circulating AKG concentrations in humans experience up to a tenfold reduction between the ages of 40 and 80 12. This age-related depletion correlates closely with impaired mitochondrial function, diminished stem cell regenerative capacity, and the onset of progressive physiological frailty 34.
The therapeutic potential of AKG as a targeted geroprotective agent relies on its ability to influence multiple established hallmarks of aging simultaneously. Extensive preclinical data demonstrate that exogenous AKG supplementation extends maximum and median lifespan, while significantly compressing late-life morbidity, across diverse evolutionary phyla including nematodes, insects, and mammals 4576. These macro-level phenotypic benefits are governed by specific intracellular signaling networks. AKG directly modulates the mechanistic target of rapamycin (mTOR) pathway, activates AMP-activated protein kinase (AMPK), neutralizes reactive oxygen species (ROS), and serves as an obligate co-substrate for the enzymatic regulation of the epigenetic landscape 47.
As the longevity field progresses from animal models into rigorous human clinical trials, specific molecular formulations of the compound - most notably sustained-release calcium alpha-ketoglutarate (Ca-AKG) - have been developed to overcome intrinsic pharmacokinetic barriers and maximize systemic bioavailability. Retrospective human cohort studies have generated highly compelling preliminary data suggesting that targeted AKG supplementation can induce measurable reversals in biological age algorithms, specifically DNA methylation clocks 3108. To validate these findings, multiple randomized, double-blind, placebo-controlled trials are currently executing comprehensive evaluations of AKG's efficacy on epigenetic, metabolic, and physical parameters of human aging. This report exhaustively analyzes the biochemical mechanisms, preclinical evidence, human clinical trial datasets, and pharmacological properties of alpha-ketoglutarate as an intervention for extending human healthspan and lifespan.
Biochemical and Cellular Mechanisms
Mitochondrial Energy Metabolism and the Tricarboxylic Acid Cycle
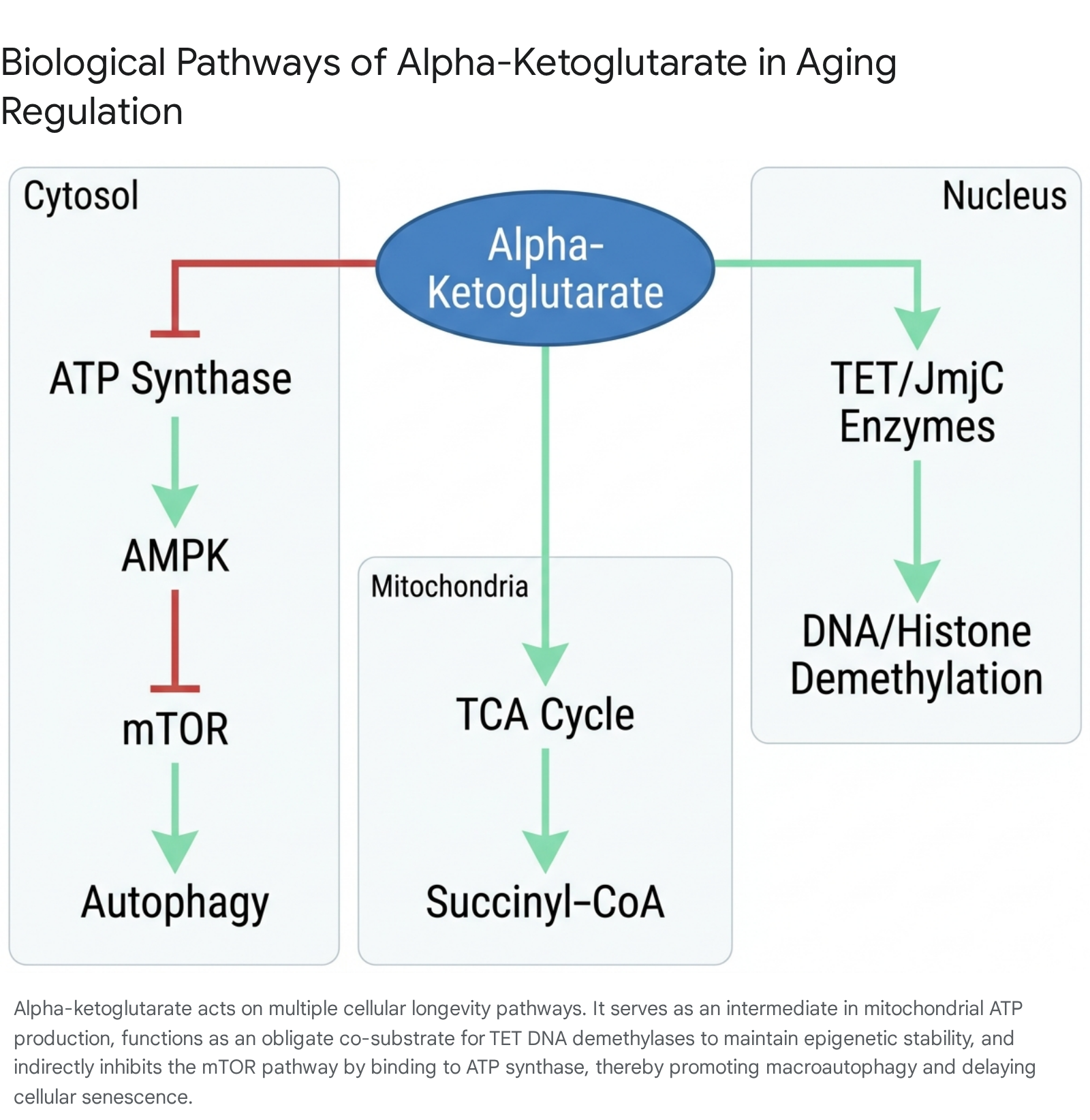
Alpha-ketoglutarate is an obligatory, five-carbon dicarboxylic acid intermediate in the mitochondrial TCA cycle (also known as the Krebs cycle or citric acid cycle). Endogenously, it is synthesized primarily from isocitrate via the enzyme isocitrate dehydrogenase through an oxidative decarboxylation reaction. Alternatively, it can be generated from the amino acid glutamate via oxidative deamination catalyzed by glutamate dehydrogenase, or through transamination reactions involving amino acids such as proline, histidine, and arginine 4913. Once generated, AKG is oxidized and decarboxylated to succinyl-CoA by the alpha-ketoglutarate dehydrogenase complex. This specific reaction is a rate-limiting step in the TCA cycle and is heavily determinative of the overall velocity of mitochondrial respiration and subsequent adenosine triphosphate (ATP) generation 414.
In the context of biological aging, mitochondrial bioenergetic efficiency progressively declines. This metabolic decay results in cellular energy deficits, metabolic inflexibility, and an increased generation of toxic oxidative byproducts. The systemic, age-associated depletion of circulating AKG exacerbates this cellular energetic crisis 310. Exogenous supplementation of AKG serves a direct anaplerotic function - it replenishes the TCA cycle intermediate pool, directly supporting cellular ATP production under conditions of metabolic or oxidative stress without relying exclusively on upstream glycolysis 91310.
Furthermore, AKG functions as a crucial molecular nitrogen scavenger. It undergoes transamination reactions with various amino acids, serving as a vital nitrogen sink that facilitates the safe interconversion and transport of nitrogenous waste. This mechanism is critical for mitigating ammonia toxicity and preserving protein homeostasis (proteostasis), the disruption of which is a primary driver of sarcopenia and other age-related degenerative states 91311. By maintaining optimal nitrogen flux, AKG preserves the balance of anabolism and catabolism in highly metabolic tissues, including skeletal muscle and the central nervous system.
Epigenetic Regulation and DNA Demethylation
One of the most profound mechanisms by which alpha-ketoglutarate influences the physiological aging trajectory is through its direct biochemical role in the maintenance of the epigenome. AKG serves as an obligate co-substrate for a wide array of alpha-ketoglutarate-dependent dioxygenase enzymes. The most critical of these in the context of geroscience are the Ten-Eleven Translocation (TET) family of DNA demethylases and the Jumonji C-domain-containing (JmjC) histone demethylases 7817.
Mammalian aging is characterized by highly reproducible, systemic alterations in the epigenome. This "epigenetic drift" typically features a global hypomethylation of the genome coupled with localized hypermethylation at specific promoter regions, leading to the erratic dysregulation of gene expression, the exhaustion of adult stem cell pools, and the induction of cellular senescence 818. Because TET and JmjC enzymes strictly require AKG, molecular oxygen, and ferrous iron to actively remove methyl groups from DNA cytosine residues and histone tails, intracellular AKG concentrations serve as a metabolic rheostat that directly dictates the epigenetic state of the cell. A natural decline in endogenous AKG impairs the enzymatic activity of these demethylases, chemically locking the cell into an aged, dysfunctional expression profile. By restoring cellular AKG pools through supplementation, it is hypothesized that organisms can reset aberrant methylation patterns, thereby maintaining youthful transcriptomic profiles, preserving genomic stability, and sustaining the regenerative capacity of adult tissue 345.
Mechanistic Target of Rapamycin and Nutrient Sensing
The targeted inhibition of the mechanistic target of rapamycin (mTOR) signaling pathway is currently the most robustly validated pharmacological strategy for extending lifespan across eukaryotic species. Alpha-ketoglutarate has been definitively identified as a potent upstream modulator of this critical nutrient-sensing network. Utilizing Drug Affinity Responsive Target Stability (DARTS) profiling, researchers have determined that AKG directly binds to and physically inhibits the beta subunit of ATP synthase (ATP5B in mammals, ATP-2 in nematodes) 4.
This partial inhibition of ATP synthase slightly lowers intracellular ATP concentrations, intentionally triggering a transient, mild state of energetic stress within the cell. The subsequent decrease in the ratio of ATP to AMP is rapidly detected by AMP-activated protein kinase (AMPK), the cell's primary metabolic energy sensor, leading to its activation 46. Activated AMPK then phosphorylates and inhibits the mTOR complex. The downstream biological consequence of this mTOR inhibition is a marked reduction in energetically expensive protein translation and the concurrent induction of macroautophagy. Autophagy is the essential cellular recycling process responsible for identifying and clearing damaged organelles, misfolded proteins, and toxic aggregates 57.
Experimental molecular data confirm that AKG supplementation actively mitigates oxidative stress-induced cellular senescence specifically through the suppression of the mTOR pathway and the necessary activation of Unc-51 like autophagy activating kinase 1 (ULK1) 47. Crucially, in C. elegans models featuring eat-2 genetic mutations - a recognized biological surrogate for chronic dietary restriction - AKG supplementation fails to further extend the organism's lifespan. This epistatic interaction strongly indicates that AKG acts biochemically as a dietary restriction mimetic, successfully hijacking the longevity pathways typically activated only by caloric deprivation 4.
Modulation of Cellular Senescence and Inflammation
Cellular senescence is an irreversible state of cell cycle arrest driven heavily by unmitigated oxidative stress, telomere attrition, and the accumulation of reactive oxygen species (ROS). AKG directly participates in non-enzymatic ROS neutralization, acting as a chemical scavenger for hydrogen peroxide and ammonia, thus physically protecting cells from oxidative molecular damage 46. In D-galactose-induced biological aging models, exogenous AKG administration significantly elevates the systemic activities of endogenous antioxidant enzymes, specifically superoxide dismutase (SOD) and glutathione (GSH). Concurrently, it reduces established biomarkers of oxidative damage, including lipid peroxidation (measured via malondialdehyde) and widespread protein carbonylation 4.
Furthermore, by inducing autophagy via the aforementioned AMPK/mTOR axis, AKG facilitates the clearance of senescent cells and directly mitigates the Senescence-Associated Secretory Phenotype (SASP). The SASP is the primary driver of "inflammaging" - the chronic, sterile, low-grade systemic inflammation that characterizes older mammalian tissues and precedes the onset of almost all chronic diseases. In vivo experimental studies confirm that AKG administration decreases systemic pro-inflammatory cytokine expression (including IL-1 beta and TNF-alpha) while strongly upregulating anti-inflammatory markers such as Interleukin-10 (IL-10). It also drives the polarization of macrophages away from the inflammatory M1 phenotype and toward the tissue-repairing M2 phenotype, thereby preserving systemic tissue homeostasis and slowing the functional collapse of highly metabolic organ systems 2412.
Preclinical Evidence in Model Organisms
Invertebrate Models of Aging
The initial biological validation of alpha-ketoglutarate as a bona fide longevity intervention occurred in established, short-lived invertebrate models. In the nematode Caenorhabditis elegans, supplementation with an 8 mM concentration of AKG during the adult phase of life extended median lifespan by an extraordinary 50% 4. This profound extension of chronological lifespan was accompanied by a measurable delay in age-related functional decline, allowing the treated nematodes to maintain rapid, coordinated physiological movement late into their lifespans. Researchers demonstrated that this effect is mediated by the specific inhibition of the ATP-2 subunit, unequivocally proving that AKG manipulates highly conserved evolutionary nutrient-sensing pathways 4.
Subsequent rigorous studies in the fruit fly Drosophila melanogaster reported a 15% increase in maximum lifespan, further validating the conservation of AKG's anti-aging mechanisms across diverse invertebrate phyla 6. Interestingly, experimental combinatory protocols that paired AKG with specific collagen-mimicking amino acid formulations - specifically precise ratios of glycine, proline, and hydroxyproline - yielded additive lifespan extensions in worms, suggesting that AKG's baseline efficacy can be significantly amplified via rationally designed synergistic poly-interventions 13.
Mammalian Longevity and Morbidity Compression
Translation of these invertebrate findings to complex mammalian models resulted in landmark publications demonstrating that AKG not only extends chronological lifespan but radically improves physical healthspan. In a rigorously controlled 2020 study utilizing middle-aged C57BL/6 mice, researchers administered calcium-bound AKG continuously via diet. The intervention yielded a statistically significant increase in overall lifespan ranging from 10.5% to 16.6%, with the female murine cohort exhibiting the most pronounced and consistent longevity benefits 2114.
Crucially, the study established that AKG actively promotes a "compression of morbidity" - a highly sought-after outcome in clinical geroscience wherein the absolute duration of time spent in a state of age-related disease and physical disability is significantly reduced relative to the organism's total lifespan. The treated mice exhibited a remarkable 40% quantitative improvement in established healthspan metrics. This included striking reductions in standardized frailty indices, vast improvements in coat condition (measured by hair quantity and melanocyte quality), anatomical corrections in spinal curvature (kyphosis), and the functional preservation of muscle grip strength 1221. These dramatic phenotypic improvements were tightly correlated with reduced systemic inflammation, notably a substantial quantitative increase in the circulation of the anti-inflammatory cytokine IL-10 212.
Organ-Specific Geroprotective Effects
Beyond systemic lifespan extension, targeted preclinical and clinical investigations have delineated highly specific tissue-level benefits of AKG supplementation, showcasing its potential to treat localized age-related pathologies.
Skeletal System and Muscular Homeostasis
In aging murine models, continuous AKG supplementation successfully attenuated age-related bone loss and significantly accelerated the rate of bone regeneration following injury. It directly ameliorated the senescence-associated phenotypes of bone marrow-derived mesenchymal stem cells (MSCs), enhancing their cellular proliferation, colony formation, and inherent osteogenic potential 617. In human data, a randomized, double-blind, placebo-controlled study involving 76 postmenopausal women with diagnosed osteopenia demonstrated that a daily regimen of oral Ca-AKG (6 grams per day for 6 months) increased bone mass density and led to a profound 37% reduction in the serum levels of C-terminal cross-linking telopeptide of type I collagen (CTX), a primary biomarker of destructive bone resorption 81123.
Furthermore, by functioning as a direct precursor to both glutamine and glutamate, AKG influences skeletal muscle protein synthesis and inhibits protein degradation pathways, thereby preserving muscle architecture against the natural catabolic drift of sarcopenic aging 613.
Neuroprotection and Cognitive Preservation
Because metabolic dysfunction and energetic failure are hallmarks of cognitive decline, the neuroprotective potential of AKG has garnered significant clinical interest. High-dose AKG demonstrates the pharmacological capacity to cross the blood-brain barrier. In established neurodegenerative models, including the widely utilized APP/PS1 transgenic murine models of Alzheimer's disease, AKG supplementation enhanced synaptic plasticity, improved spatial learning parameters, and restored memory retention 4724. The underlying mechanisms involve the mitigation of oxidative brain damage and the clearance of toxic aggregates through the specific activation of ULK1 and the subsequent suppression of the neuronal mTOR pathway 7.
Reproductive Health and Ovarian Function
The reproductive system experiences chronological aging faster than almost any other mammalian tissue system. Recent research has investigated AKG in the context of Premature Ovarian Insufficiency (POI), a severe condition characterized by early-onset follicular depletion and heavy localized inflammation. In sophisticated POI models, AKG successfully protected delicate ovarian granulosa cells from pyroptosis (inflammatory cell death) and restored follicular viability by actively inhibiting the activation of the highly destructive NLRP3/Caspase-1 inflammatory signaling pathway 1526. These findings highlight the potential for targeted metabolic interventions to delay reproductive senescence.
Renal Function and Hemodialysis Contexts
AKG has a long-standing history of utilization in severe renal pathology. Experimental data utilizing dimethyl alpha-ketoglutarate (DM-AKG) - a derivative engineered for higher cell membrane permeability - effectively ameliorated cisplatin-induced acute kidney injury (AKI) by preserving mitochondrial energy metabolism and reducing inflammatory chemokine abundance 27. Clinically, massive doses of Ca-AKG have been successfully utilized in human patients undergoing chronic hemodialysis. The compound functions as an effective phosphate binder, dramatically improving amino acid metabolism and reducing systemic blood urea levels without inducing secondary toxicity 1123.
Human Clinical Trials and Efficacy
While the animal data concerning alpha-ketoglutarate are structurally robust, translation to humans requires rigorous validation through controlled trials. The field currently relies on a mixture of deeply analyzed retrospective observational data and several ongoing, highly anticipated prospective randomized controlled trials aimed at defining AKG's absolute clinical utility.
Retrospective Analysis of DNA Methylation Age
The most prominent human data point regarding AKG and the reversal of biological aging originates from a retrospective analysis of 42 self-reported healthy adult individuals (14 females, 28 males, average age 64). These subjects utilized a commercial, sustained-release calcium alpha-ketoglutarate supplement (Rejuvant®) over an average period of seven months. To assess biological efficacy, researchers utilized the TruAge DNA methylation clock, an established epigenetic biomarker capable of estimating the functional biological age of an organism based on specific patterns of DNA methylation across the genome 37.
The resulting analysis demonstrated a highly statistically significant reduction in calculated biological age. At baseline, prior to the intervention, the cohort was an average of 0.35 years biologically younger than their chronological age. Following the seven-month continuous intervention, the cohort tested an average of 8.31 years biologically younger than their chronological age - yielding an effective epigenetic age reversal of approximately 8 years (p = 6.538x10^-12) 3.
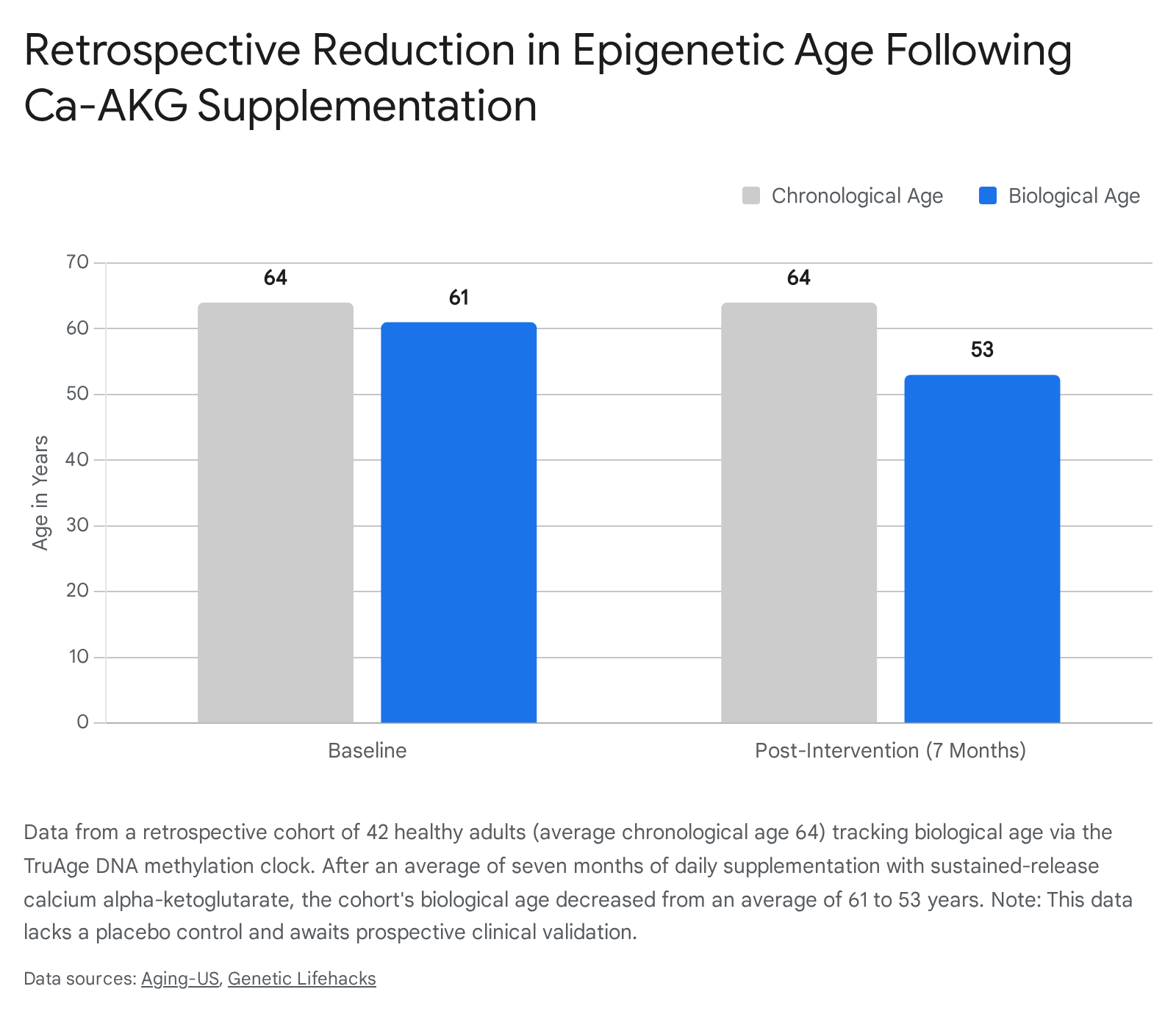
While these findings represent a profound and encouraging proof-of-concept for human geroprotection via metabolic supplementation, the retrospective nature of the study, the lack of a standardized placebo control arm, and the relatively small, homogeneous sample size necessitate extreme scientific caution. Consequently, these results are widely viewed within the rigorous scientific community as preliminary, hypothesis-generating data rather than conclusive proof of universal clinical efficacy 1424.
The ABLE Trial Methodology and Recruitment
To rigorously address the limitations of available retrospective data, the Centre for Healthy Longevity at the National University of Singapore initiated the landmark ABLE (Alpha-ketoglutarate Supplementation and BiologicaL agE) trial (ClinicalTrials.gov Identifier: NCT05706389). Designed as a highly controlled, double-blind, randomized, placebo-controlled clinical trial, ABLE specifically recruited 120 middle-aged adults (aged 40 - 60 years) characterized explicitly by biological ages that exceeded their chronological ages, attempting to treat those with accelerated aging profiles 108.
Participants in the active arm were administered 1 gram per day of sustained-release Ca-AKG, while the control arm received an identical placebo over a 6-month continuous intervention period, followed by a 3-month observational follow-up phase. The primary clinical endpoint established for the trial is the quantitative change in DNA methylation age 8. Furthermore, secondary endpoints include a highly comprehensive, systemic suite of healthspan markers: inflammatory cytokines, fasting lipid and glucose metabolic panels, arterial stiffness (measured via carotid-femoral pulse wave velocity), handgrip strength, leg extension strength, absolute bone density (via DXA scans), and aerobic capacity (via cardio-pulmonary exercise testing) 48.
The recruitment and operational feasibility data for the ABLE trial have been formally published, providing valuable insights into geroscience trial execution. Of the 467 individuals who expressed interest and underwent screening, an overwhelming 80.2% exhibited accelerated biological aging (biological age surpassing chronological age), confirming the immense feasibility of targeting these specific at-risk populations in future gerotherapeutics 10. The trial completed its core clinical phase in late 2023, and while final primary outcome results regarding absolute epigenetic age reversal remain pending formal publication as of early 2026, the trial represents a definitive watershed moment for the scientific rigor of the longevity field 4.
Measurement Modalities and Epigenetic Clock Variations
The execution of the ABLE trial has also highlighted profound technical nuances in clinical outcome measurement. Sub-studies related directly to the ABLE trial methodology have revealed that subtle technological shifts from established Infinium MethylationEPICv1 to the newer EPICv2 array technologies introduce pervasive systematic biases in the reading of CpG sites 28. These biases significantly distort the outputs of both Principal Component (PC) based and non-PC based epigenetic clocks. This finding is critical, as it underscores that small effect sizes observed in DNAm age measurements must be interpreted highly cautiously; they may reflect systematic platform differences rather than meaningful, biological age-related metabolic changes driven by AKG 28.
Additional Prospective Clinical Investigations
A secondary major trial evaluating the clinical efficacy of AKG is NCT07114536, sponsored by Shenzhen Hygieia Biotech. This is a 12-week randomized, double-blind, placebo-controlled trial involving middle-aged and older adults (aged 45 - 75). In contrast to the ABLE trial's dosage, participants in this study receive a notably higher dose of 2 grams per day of Ca-AKG (administered strategically as 0.5-gram tablets, taken four times daily to ensure constant plasma saturation). The primary biological endpoint is the modulation of aging as measured via the PhenoAge composite biomarker algorithm - a clinically validated mortality risk model based heavily on physiological blood markers rather than pure DNA methylation 16. Expected to complete in early 2026, this trial will provide critical dose-response data and validate whether Ca-AKG alters traditional metabolic and biochemical biomarkers alongside novel epigenetic clocks 16.
It must be explicitly noted that AKG clinical trials are completely distinct in mechanism and execution from the well-known TRIIM and TRIIM-X trials. The TRIIM trials focus strictly on profound thymus regeneration and systemic immunorestoration utilizing a complex, high-risk poly-pharmacy cocktail of Recombinant Human Growth Hormone, DHEA, and Metformin, and do not feature AKG in their protocol 301732.
Pharmacokinetics and Supplement Formulations
The absolute clinical utility and geroprotective efficacy of alpha-ketoglutarate are heavily dependent upon its precise molecular formulation. As a free, unattached acid, AKG exhibits extreme chemical instability in the highly acidic environment of the mammalian stomach and is subjected to rapid enzymatic metabolism. This physical degradation results in exceptionally poor systemic bioavailability; pharmacokinetic studies indicate that up to 80% of unformulated, free AKG is not effectively absorbed into the bloodstream when taken orally 18. Consequently, analytical chemists and formulators bind the AKG molecule to various mineral cations and amino acids to manipulate its pharmacokinetic profile and ensure target tissue delivery.
Comparison of Alpha-Ketoglutarate Forms
The table below delineates the primary salt and amino acid-bound forms of AKG utilized in modern clinical and supplemental settings, highlighting their specific stability profiles and targeted medical uses.
| Form of Alpha-Ketoglutarate | Chemical Binding Mechanism | Primary Pharmacological Characteristics | Dominant Clinical / Market Application |
|---|---|---|---|
| Calcium Alpha-Ketoglutarate (Ca-AKG) | AKG bound to elemental Calcium | Exceptional physical stability and high bioavailability. The calcium cation physically buffers stomach acid, permitting safe delivery to the small intestine. Longest sustained plasma half-life. | Longevity, anti-aging research, bone density support, long-term metabolic regulation 1834. |
| Arginine Alpha-Ketoglutarate (AAKG) | AKG bound to L-Arginine | Promotes rapid systemic nitric oxide (NO) production, resulting in acute vasodilation. Highly unstable with a very short half-life; breaks down quickly in blood plasma. | Sports nutrition, acute pre-workout supplements, transient physical endurance 5131834. |
| Ornithine Alpha-Ketoglutarate (OAKG) | AKG bound to L-Ornithine | Features a pronounced nitrogen-sparing effect. Directly stimulates acute protein synthesis and modulates systemic amino acid metabolism. | Clinical malnutrition recovery, severe burn trauma treatment, targeted muscle regeneration 2319. |
| Sodium Alpha-Ketoglutarate (Na-AKG) | AKG bound to Sodium | High water solubility but features a markedly lower sustained bioavailability profile compared to the calcium salt variant. | General short-term metabolic support (rarely utilized in high-tier longevity interventions) 23. |
From a strict geroscience perspective, Ca-AKG is universally favored and is the exclusive form utilized in major longevity trials. The chemical stability provided by the calcium bond ensures a steady, prolonged release of AKG into the bloodstream, which is biologically necessary to continuously engage mitochondrial and epigenetic pathways without inducing extreme metabolic peaks and troughs 1834. In sharp contrast, forms like AAKG function primarily as acute vasodilators designed for immediate athletic performance, providing absolutely no demonstrated or theoretical benefit for long-term cellular repair, epigenetic maintenance, or total lifespan extension 34.
Controlled-Release Matrix Technologies
Even when bound securely to calcium, the biological half-life of AKG requires careful pharmacological management. To maximize geroprotective efficacy over a 24-hour cycle, next-generation clinical trials (such as the ABLE trial) rely entirely on highly engineered sustained-release Ca-AKG formulations. These therapeutics employ sophisticated controlled-release matrices - frequently utilizing varying precise molecular weights of Hydroxypropylmethylcellulose (HPMC), isomalt, stearic acid, and specific vegetable waxes - to strictly modulate the intestinal release rate 4.
The explicit objective of these physical matrices is to cap the gastric release rate at a maximum of 90% relative to standard immediate-release commercial products, typically spanning a 12-hour biological window. This specific pharmacokinetic profile is engineered to closely mimic the natural endogenous steady state of cellular AKG production. This methodology avoids dangerous plasma spikes, significantly reduces the required patient dosing frequency, and effectively eliminates the transient gastric distress sometimes associated with large bolus doses of acidic compounds 421.
Safety Profile and Regulatory Status
Clinical Tolerability and Adverse Event Monitoring
Alpha-ketoglutarate exhibits a highly favorable, well-documented safety and tolerability profile, supported by decades of human exposure in varied, high-stress clinical settings. In the context of severe chronic kidney disease (CKD) and continuous hemodialysis, Ca-AKG has been safely administered at massive clinical doses - up to 4.5 grams per day for continuous periods of up to three years - with zero notable adverse systemic effects. In these highly compromised patient cohorts, the compound effectively functioned as a therapeutic phosphate binder and safely improved systemic amino acid metabolism by steadily decreasing toxic plasma urea levels 112320. Similarly, in a rigorous 6-month trial involving postmenopausal women, a heavy regimen of 6 grams of AKG daily effectively suppressed bone resorption markers with exceptionally high patient tolerability and compliance 41123.
Minor adverse effects observed in otherwise healthy populations are exceptionally rare and generally restricted to transient gastrointestinal distress (such as mild bloating or temporary nausea), and these events typically only occur when extremely high, unformulated doses are consumed rapidly on an empty stomach 5. Standard, modern longevity protocols typically utilize conservative doses of 1 to 2 grams per day, significantly below the physiological thresholds known to induce any gastric discomfort 816.
Due to its fundamental mechanism of action and the general lack of targeted pharmacokinetic data in specific populations, systemic Ca-AKG supplementation is currently strictly contraindicated during active pregnancy and breastfeeding 523. Pharmacological caution is also advised for patients concurrently utilizing prescribed calcium channel blockers, as theoretical metabolic interactions may occur due to the elemental calcium load, although definitive clinical evidence of such adverse events in the literature remains highly sparse 23. Extensive pharmacovigilance database reviews (such as FAERS) tracking drug-induced kidney stones do not list AKG as a primary causative agent, though general caution in patients with histories of severe nephrolithiasis is maintained as standard medical practice 2122.
Regulatory Classifications
From a global regulatory standpoint, Alpha-Ketoglutarate is generally available worldwide as an over-the-counter dietary supplement. In the highly regulated European Union, the European Food Safety Authority (EFSA) maintains ongoing, rigorous evaluations regarding its specific status. As of the late 2025 and early 2026 official catalogue updates, specific chemical forms - including Calcium Alpha-Ketoglutarate and Arginine Alpha-Ketoglutarate - are officially classified as "Not novel in food supplements." This critical classification permits their continued legal sale, formulation, and integration into dietary products without requiring the stringent, multi-year approval pipelines mandated for completely unauthorized novel pharmaceutical compounds 2340.
Conclusion
Alpha-ketoglutarate represents a critical, highly promising bridge in modern aging biology - an endogenous, deeply understood metabolic intermediate that has been definitively proven to orchestrate complex signaling networks affecting total lifespan and morbidity. By addressing multiple hallmarks of aging simultaneously - reversing epigenetic drift via demethylase activation, restoring mitochondrial efficiency, and inhibiting the pro-aging mTOR pathway to clear senescent cells - AKG offers a pleiotropic approach to geroprotection that single-target pharmaceuticals cannot match.
The profound lifespan extensions and morbidity compressions observed in rigorous mammalian models are highly compelling. However, the true trajectory of AKG in human medicine relies entirely on the final readouts of ongoing prospective clinical trials, such as the ABLE and Hygieia Biotech studies. If these controlled environments validate the striking 8-year biological age reversals seen in preliminary retrospective data, sustained-release Ca-AKG will rapidly transition from an experimental anti-aging compound to a foundational, evidence-based pillar of preventative geroprotective medicine. Furthermore, future clinical research will likely pivot toward combinatorial gerotherapeutics, exploring how AKG's unique mechanisms might act synergistically with other verified interventions to comprehensively delay the onset of age-related physiological decline.