Allostatic load and the role of chronic stress in biological aging
Conceptual Foundations of Allostasis
To comprehend how chronic environmental and psychosocial stress physically alters the human body, it is necessary to differentiate between two foundational physiological paradigms: homeostasis and allostasis. Traditionally, biomedical science has relied on the model of homeostasis, which posits that the body maintains internal stability by keeping vital parameters - such as blood oxygen levels, pH, and core body temperature - within narrow, rigidly fixed ranges 12. However, homeostasis does not adequately account for the dynamic, energy-demanding adaptations required to survive fluctuating environmental demands.
Introduced by Sterling and Eyer in 1988 and subsequently expanded by McEwen and Stellar in 1993, the concept of allostasis describes the active, continuous process of achieving stability through change 3456. Unlike homeostatic systems, allostatic systems - such as blood pressure, heart rate variability, and neuroendocrine output - operate with broad boundaries, continuously recalibrating to meet perceived internal and external challenges 2. Under this model, the human brain functions as an organ of predictive regulation, anticipating physiological needs and mobilizing resources to fulfill them before they emerge 78. A critical component of this predictive regulation is the reduction of uncertainty, a state that demands significant neuroenergetic expenditure 8.
When an organism faces acute environmental challenges, allostasis facilitates survival by activating neural, neuroendocrine, and neuroendocrine-immune mechanisms 6. Once the challenge is resolved, these systems are ideally downregulated. However, physiological toll accrues when the adaptive mechanisms fail to operate efficiently. McEwen (1998) identified three primary typologies of this failure: (1) frequent activation of allostatic systems due to repeated novel stressors; (2) failure to shut off allostatic activity after the stressor has passed; and (3) an inadequate initial response of allostatic systems, which forces other, normally counter-regulated systems to elevate their activity to compensate 6.
This cumulative "wear and tear" on the body resulting from chronic overactivation or underactivation is termed allostatic load (AL) 38. If environmental challenges entirely exceed an individual's psychological and physiological coping capacity, resulting in severe clinical and functional impairment, the condition progresses to allostatic overload - a transdiagnostic categorization indicative of impending systemic failure 47.
Biological Architecture of Stress Accumulation
The physiological progression of allostatic load is structurally categorized into a continuous cascade of primary mediators, secondary outcomes, and tertiary outcomes. This biological pathway illustrates how an initial neurobiological perception of threat translates into systemic physiological dysregulation 39.
Primary Mediators
Primary mediators represent the immediate, acute biochemical changes that occur in the neuroendocrine and immune systems at the onset of a stress response 910. When the brain perceives a threat, trauma, or persistent uncertainty, it triggers the simultaneous activation of the sympathetic-adrenal-medullary (SAM) axis and the hypothalamic-pituitary-adrenal (HPA) axis 31011.
The SAM axis dictates the immediate "fight or flight" response, signaling the adrenal glands to rapidly release catecholamines - specifically epinephrine and norepinephrine 31011. This sudden influx facilitates rapid increases in cardiovascular and respiratory output. In tandem, the HPA axis regulates the sustained metabolic stress response by secreting glucocorticoids, primarily cortisol, alongside dehydroepiandrosterone (DHEA) 31011. This mechanism ensures that muscles and neural networks maintain a steady supply of metabolic energy to endure the stressor. Simultaneously, the acute stress response involves the immediate mobilization of pro-inflammatory and anti-inflammatory cytokines, functioning as primary immune mediators to prepare the body for potential injury or infection 3.
Secondary Regulatory Systems
While primary mediators are essential for acute survival, their chronic, uninterrupted elevation initiates maladaptive structural remodeling and target-tissue desensitization across multiple physiological domains 9. These secondary regulatory systems encompass the cardiovascular, metabolic, and immune systems. Persistent SAM and HPA activation leads to subclinical physiological dysregulation, characterized by sustained increases in systolic and diastolic blood pressure, elevated lipid profiles (including total cholesterol and triglycerides), altered glucose metabolism (indicated by rising HbA1c), and chronic, low-grade systemic inflammation (frequently measured via C-reactive protein and interleukin-6) 31011.
Tertiary Outcomes
Tertiary outcomes represent the endpoint of the allostatic load cascade. The prolonged damage to secondary regulatory systems ultimately manifests as definitive clinical diseases. This stage is characterized by early morbidity, accelerated disease progression across various pathologies, and premature mortality 3.
| Phase | Biological Component | Key Biomarkers and Indicators | Physiological Manifestation |
|---|---|---|---|
| Primary Mediators | Neuroendocrine & Immune Systems | Cortisol, epinephrine, norepinephrine, DHEA, circulating cytokines. | Immediate biochemical response to acute or chronic stressors via the SAM and HPA axes. |
| Secondary Outcomes | Cardiovascular, Metabolic, & Immune Systems | Systolic/diastolic blood pressure, HbA1c, HDL/LDL cholesterol, C-reactive protein (CRP), waist-hip ratio. | Subclinical physiological dysregulation, structural remodeling of cellular receptors, systemic inflammation. |
| Tertiary Outcomes | Systemic Pathology | Diagnosed clinical conditions. | Cardiovascular disease, severe cognitive decline, psychiatric disorders, mortality. |
Cellular Senescence and Epigenetic Aging
Allostatic load operates not only at the systemic and organ levels but also drives profound alterations at the cellular and molecular levels. Chronic exposure to stress mediators precipitates a cascade of events that significantly accelerate the biological aging process, decoupling chronological age from physiological age 1012.
Hypermetabolism and Mitochondrial Instability
Any allostatic process - whether mounting an immune defense or sustaining elevated blood pressure - requires active, ATP-dependent molecular events 13. Longitudinal profiling of human primary fibroblasts under chronic glucocorticoid exposure demonstrates that cellular allostatic load triggers a persistent state of hypermetabolism. Chronic stress increases total cellular energy expenditure by approximately 60%, shifting the cell's primary metabolic reliance from standard glycolysis to mitochondrial oxidative phosphorylation (OxPhos) 14.
This sustained, intense energetic demand destabilizes mitochondrial function. Stress-induced hypermetabolism is directly linked to mitochondrial DNA (mtDNA) instability, which manifests as the rapid accumulation of mtDNA defects and a decline in mtDNA copy numbers 131415. In experimental models, pharmacologically normalizing OxPhos activity while artificially forcing increased energy expenditure exacerbates the accelerated aging phenotype, strongly indicating that total energy expenditure is a primary driver of cellular aging dynamics in the context of chronic allostatic load 1314.
Telomere Attrition
Telomeres, the protective nucleoprotein structures capping the ends of chromosomes, are highly sensitive to the biochemical environment created by chronic stress. Research indicates that chronic glucocorticoid signaling accelerates the rate of telomere shortening by an average of 27.6% per cell division 13. Chronic allostatic load effectively decouples the loss of telomeric repeats from standard cell division cycles, causing more rapid attrition during each event of genome replication.
At the molecular level, chronic stress prompts a notable 26.7% downregulation of TPP1, a crucial component of the shelterin complex responsible for protecting telomeres from being recognized as DNA damage 13. Concurrently, cells exhibit a 68.6% upregulation of TERC, an RNA component of the telomerase holoenzyme 13. This upregulation appears to represent a compensatory but ultimately insufficient attempt by the cell to preserve telomeric integrity under the intense pressure of chronic stress. Ultimately, this leads to a premature halting of population doubling, reducing the cellular Hayflick limit by an average of 19.8% 13.
Epigenetic Clocks and DNA Methylation
Biological clocks utilizing cellular DNA methylation data have emerged as highly accurate predictors of morbidity and mortality 1615. Evidence shows that allostatic load significantly accelerates epigenetic aging across multiple validated methylation algorithms 13. In laboratory analyses, cells subjected to chronic glucocorticoid stress exhibited a 36.4% increase in the rate of epigenetic aging according to the PC-PhenoAge clock, and a 33.1% increase using the PC-Hannum clock 13.
Population-based epidemiological data substantiate these in vitro findings. In a subsample of 490 older adults from the Irish Longitudinal Study on Ageing (TILDA), researchers analyzed cross-sectional associations between AL and Epigenetic Age Acceleration (EAA) using the Horvath, Hannum, and Levine clocks 16. The data revealed distinct, sex-specific drivers of biological aging: metabolic dysregulation was a primary driver of EAA in men, whereas a combination of metabolic and cardiovascular dysregulation drove EAA in women 16.
Furthermore, researchers utilizing the Swiss Kidney Project on Genes in Hypertension (SKIPOGH) cohort have successfully derived a specific epigenetic signature of allostatic load (methAL) that correlates strongly with phenotype-based AL scores (R2 = 0.59) 1718. When validated in the independent 1936 Lothian Birth Cohort (LBC1936), a one standard deviation increase in the methALT signature was associated with 25% higher odds of a reported history of cardiovascular disease and a nearly two-fold increase in the all-cause mortality rate at early follow-up, indicating that DNA methylation provides a highly stable, independent proxy for cumulative stress 1718.
The Senescence-Associated Secretory Phenotype (SASP)
Accelerated biological aging is fundamentally characterized by the accumulation of senescent cells - cells that irreversibly cease to divide but remain highly metabolically active 19. These cells secrete a complex, toxic array of pro-inflammatory cytokines, chemokines, and proteases known as the senescence-associated secretory phenotype (SASP) 1219.
Recent single-cell RNA sequencing (scRNA-seq) analyses of murine and human aging tissues have revealed extraordinary heterogeneity in the nature of cellular senescence 1920. The cyclin-dependent kinase inhibitors p16Ink4a and p21Cip1, which have classically been utilized interchangeably as primary markers of senescence, are actually expressed in largely distinct, non-overlapping cell populations in vivo 192021. RNA velocity and pseudotime trajectory analyses demonstrate that p16+ and p21+ cells follow entirely independent senescence pathways, showing no evidence of direct transition states between the two phenotypes 1921. While a small "core" set of SASP factors is shared, chronic stress and elevated allostatic load compound a highly tissue-specific SASP, driving systemic sterile inflammation and increasing the risk of widespread multimorbidity 101220.
| Biological Marker/System | Primary Mechanism of Stress Induction | Outcome/Consequence |
|---|---|---|
| Mitochondria (OxPhos) | Shift to hypermetabolism to meet energy demands. | mtDNA instability; accelerated cellular decline. |
| Telomeres | Downregulation of shelterin component TPP1. | 27.6% accelerated shortening rate; reduced Hayflick limit. |
| DNA Methylation (Clocks) | Genomic recalibration to sustained glucocorticoids. | 33-36% increase in epigenetic aging rates (PhenoAge/Hannum). |
| Cellular Senescence | Accumulation of p16Ink4a and p21Cip1 cell populations. | Secretion of pro-inflammatory SASP; tissue degradation. |
Methodological Approaches to Measurement
Despite the widespread adoption and undeniable utility of the allostatic load framework, a major limitation in the field is the substantial heterogeneity in its operationalization. Researchers continuously utilize varied sets of biomarkers and entirely divergent statistical methods to calculate composite AL scores, severely hampering cross-study comparability and clinical implementation 3222324.
Biomarker Selection and Consensus Efforts
The seminal formulation of allostatic load by McEwen and Stellar utilized a specific battery of ten biomarkers 242526. Over the subsequent three decades, research expanded this pool significantly. A systematic review examining the relationship between socio-economic position and AL revealed that across 26 studies, 59 completely different biomarkers were employed 3. While cardiovascular and metabolic markers are almost universally included, neuroendocrine markers are present in only 58% of studies, and markers of parasympathetic, respiratory, kidney, or liver function are frequently omitted altogether 3.
To establish consensus, a massive 2023 multi-cohort, individual participant data (IPD) meta-analysis evaluating 67,126 individuals aged 40 to 111 across 13 distinct cohorts sought to identify a universal, efficient biomarker battery 327. Evaluating 40 biomarkers across 12 physiological systems, the analysis determined that an index based on five widely available, highly predictive markers - C-reactive protein (CRP), resting heart rate (RHR), high-density lipoprotein cholesterol (HDL-C), waist-to-height ratio (WtHR), and glycated hemoglobin (HbA1c) - predicted independent outcomes, including all-cause mortality, as accurately as or better than more elaborate biomarker suites 327. The researchers also identified peak expiratory flow (PEF) as a high-value respiratory biomarker recommended for inclusion in future AL data collection 27.
Scoring Algorithms
Once a suite of biomarkers is selected, the mathematical and statistical approach to establishing a composite risk score remains hotly debated 2829. 1. Count-Based High-Risk Quartiles: This is the most prevalent approach, utilized by approximately 52.5% of published AL studies 2830. It defines high risk empirically based on the distribution of each biomarker within the specific study sample. Typically, a value of "1" is assigned to individuals whose biomarker values fall into the highest-risk quartile of that specific cohort (e.g., above the 75th percentile for blood pressure, or below the 25th percentile for HDL cholesterol), and the points are summed to create a total index score 3. This method eliminates the need to standardize measurements across different laboratories, but relies heavily on population-specific risks that may not generalize to broader populations 3. 2. Clinical Cut-Points: An alternative approach is the use of empirically derived clinical reference values to establish risk (e.g., assigning points for a BMI > 30, or fasting glucose > 126 mg/dL) 2831. Approximately 11.1% of studies use this approach 30. While highly interpretable for clinicians, this method is fundamentally criticized because classical allostatic load theory explicitly posits that biological dysregulation manifests as "wear and tear" at subclinical levels, long before diagnostic disease thresholds are met 28. 3. Z-Scores: Some researchers advocate for converting all continuous biomarker variables into standardized z-scores and summing them 928. This method utilizes the full continuum of biological data and avoids the inherent loss of statistical information that occurs when continuous variables are artificially dichotomized into binary high/low risk categories 3928.
| Scoring Methodology | Mechanism of Calculation | Primary Advantage | Primary Limitation |
|---|---|---|---|
| High-Risk Quartiles | Assigns 1 point if a biomarker falls into the worst 25% of the study sample's distribution. | Does not require standardizing lab equipment; captures subclinical dysregulation. | Sample-dependent; cut-points vary wildly between healthy vs. unhealthy cohorts. |
| Clinical Cut-Points | Assigns points based on established medical diagnostic thresholds (e.g., hypertension guidelines). | Highly interpretable for clinicians and easily standardized across independent studies. | Fails to capture the subclinical wear-and-tear central to foundational allostatic load theory. |
| Z-Scores | Converts all biomarkers to standard deviations from the mean and sums the continuous values. | Retains the entirety of the variance in the biological data, preventing information loss. | Less intuitive to interpret clinically; can be severely skewed by extreme outliers in the dataset. |
Clinimetric Assessment of Allostatic Overload
While measuring systemic biomarkers provides an objective, quantitative assessment of biological risk, it intrinsically lacks the contextual data necessary to interpret the precise psychosocial drivers generating that risk 32. To bridge this critical gap, researchers have pioneered the use of "clinimetrics" - the science of clinically applicable metrics - to assess allostatic overload in active clinical practice 2433.
Clinimetric assessment relies heavily on semi-structured interviews and validated psychometric instruments, most notably the Diagnostic Criteria for Psychosomatic Research (DCPR) and the PsychoSocial Index (PSI) 3334. The determination of clinical allostatic overload rests on two primary criteria: * Criterion A (The Identifiable Stressor): The definitive identification of a major life event or chronic psychosocial stressor that is judged by a clinician to clearly exceed or tax the individual's psychological and material coping skills 33. * Criterion B (Clinical Manifestations): The presence of clinical manifestations of distress occurring within six months of the onset of the stressor. These must manifest as formal psychopathological symptoms, psychosomatic symptoms, or significant impairment in social, occupational, or overall psychological well-being 33.
To systematically track this progression, Fava and colleagues have proposed a comprehensive staging system (Stages 0 to 3) 73437. This system traces an individual's trajectory from functional allostasis (Stage 0, indicating baseline euthymia and healthy physiological adaptation) to severe allostatic overload resulting in overt, diagnosed psychiatric or physical illness (Stage 3) 73437.
The utility of these clinimetric criteria has been demonstrated in specialized medical settings. For instance, in an evaluation of 70 outpatients with congestive heart failure (CHF), researchers utilizing clinimetric criteria found that 32.9% of the patients were classified as suffering from active allostatic overload 35. Notably, women were more likely to report allostatic overload than men, and among established cardiac risk factors, hyperglycemia was found to be significantly associated with the presence of clinimetric allostatic overload 35. Incorporating these clinimetrics allows healthcare providers to implement tailored interventions - such as cognitive behavioral therapy, lifestyle modification, and body-oriented somatic trauma therapies - to intercept physiological decline before tertiary diseases fully emerge 7.
Social Determinants and Structural Inequalities
Allostatic load serves as a powerful, measurable biological mechanism explaining exactly how systemic social determinants of health and structural inequalities embed themselves "under the skin" 3637. Elevated AL is inextricably linked to socioeconomic adversity, neighborhood impoverishment, and systemic racial discrimination 4537.
Disparities in Minoritized Populations
Populations residing in socioeconomically disadvantaged areas frequently confront extreme environmental stressors - such as a lack of green spaces, inadequate housing, high violent crime rates, and limited healthcare access - which compound standard life stress 937. Systemic biases, everyday racial discrimination, and the historical legacy of structural inequality persistently activate the HPA and SAM axes, perpetuating hyper-arousal 37.
Data from the third National Health and Nutrition Examination Survey (NHANES) analyzing 4,515 adults aged 35 to 64 demonstrated profound racial health disparities driven by stress 25. Black men and women exhibited significantly higher mean allostatic load scores (2.5 and 2.6, respectively) compared to their White counterparts (2.1 and 1.9) 25. Crucially, when baseline mortality disparities between Black and White individuals were adjusted for traditional variables (such as baseline socioeconomic status and health behaviors), the residual disparity in cardiovascular and non-injury mortality was markedly attenuated upon the inclusion of allostatic load data 25. This suggests that the physiological burden of chronic stress, institutional racism, and race-related mistreatment accounts for a substantial portion of the mortality gap independent of income and individual lifestyle choices 2538.
Furthermore, the All of Us Research Program, which assessed a cohort of 7,415 participants, highlighted the role of economic adversity 39. Individuals reporting high subjective stress had a 118% higher odds of exhibiting high allostatic load (OR=2.18) compared to those with low stress 3940. Notably, participants below the median poverty-income ratio (PIR) had universally increased odds of high AL; they exist in environments saturated with multiple adverse health determinants, making the biological risk factors perpetually elevated 40.
Intersectionality plays a crucial role in these stress pathways. A qualitative study of South Asians in the UK living with long-term conditions (LTCs) like diabetes and heart disease revealed that prejudice in healthcare, combined with financial stress and discrimination stemming from institutional policies, creates profound chronic distress 41. This distress frequently tips into severe allostatic overload, leading to disengagement from health management protocols 41.
Geographic and Migratory Cohort Findings
The physiological impact of stress varies considerably across geographic and cultural contexts. In Latin America and the Caribbean, data from the SABE (Survey on Health, Well-Being, and Aging) study demonstrated how environmental exposure to structural violence, poor nutrition, and pollution drives childhood trauma (Adverse Childhood Experiences, or ACEs) and directly elevates later-life allostatic load, predisposing individuals to steep maladaptive health trajectories 42434445.
Studies tracking migration to high-income nations highlight the incredibly dynamic nature of allostatic accumulation. In the Hispanic Community Health Study/Study of Latinos (HCHS/SOL), researchers assessed over 15,830 individuals, 77% of whom were foreign-born 46. Among participants younger than 55, AL scores were highest among US-born individuals, intermediate in foreign-born individuals with a long duration of US residence (≥10 years), and lowest among recent immigrants (<10 years) 46. This clearly supports a "healthy immigrant" pattern wherein a biological health advantage rapidly deteriorates over time due to the chronic stressors of assimilation, acculturation, and minority status in the United States 46.
Similarly, data from the Africans in America cohort indicated that African-born Black immigrants were three times more likely to fall into the high-AL category if their duration of residence in the US exceeded 10 years, or if they immigrated at age 30 or older 47.
Clinical Outcomes and Morbidity
The tertiary outcomes of prolonged allostatic load encompass sweeping physiological dysfunctions across virtually all major disease categories. When the body's compensatory mechanisms fail, the resulting systemic damage dictates long-term survival.
Cardiometabolic Disease
Persistent low-grade inflammation and hormonal dysregulation drive atherosclerotic and hypercoagulable states, directly elevating the risk of cardiovascular disease (CVD) and complex cardiometabolic multimorbidity (CMM) 484950.
In a massive, highly powered analysis of 205,504 adults in the UK Biobank, researchers tracked incident CVD relative to a 12-biomarker AL score 48. The data revealed a definitive positive, non-linear dose-response gradient 48. Compared to a baseline AL score of 0, individuals with a score of 1 faced a Hazard Ratio (HR) of 1.23, which steadily escalated to an HR of 2.15 for individuals with an AL score of 6 or higher 48.
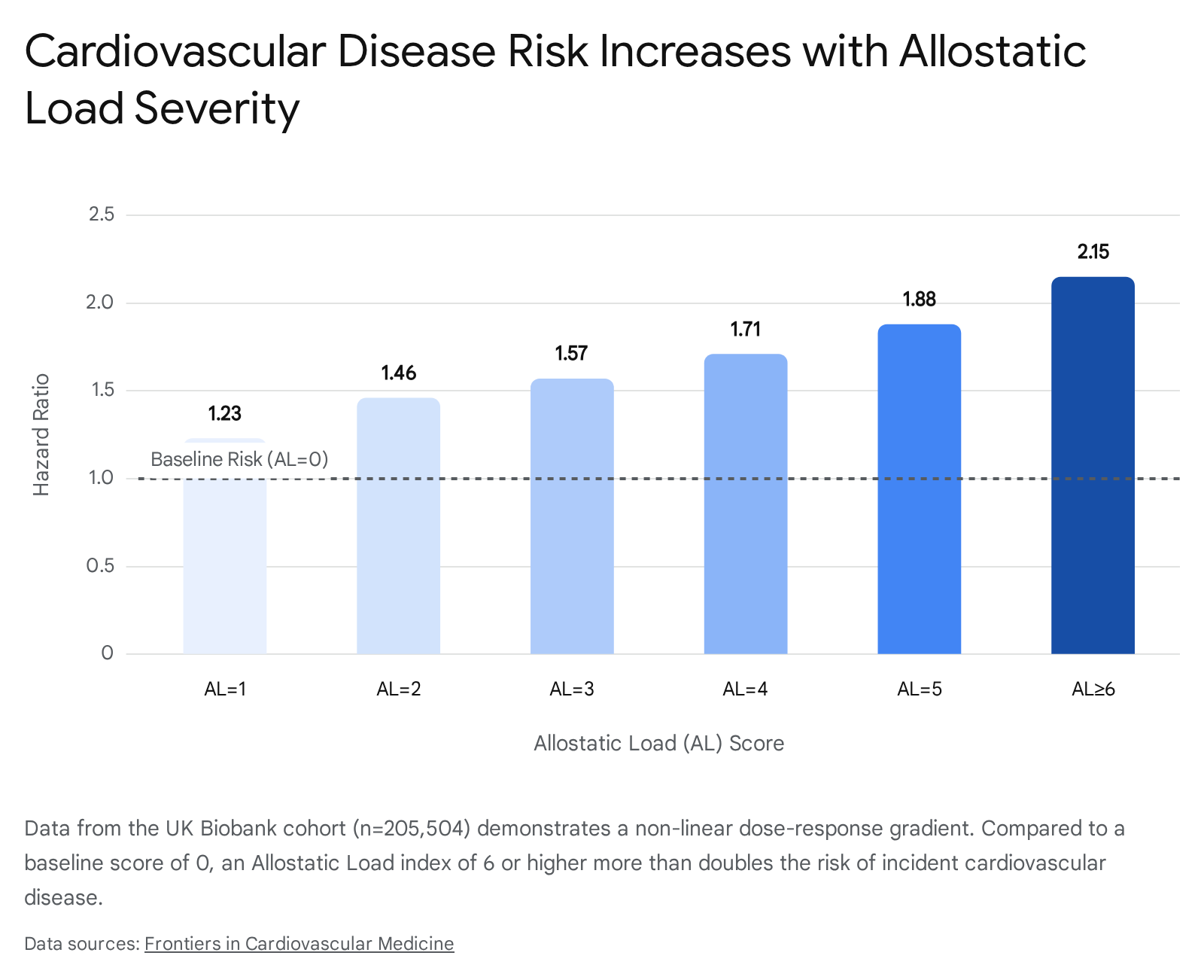
Mediation analysis established that neutrophil-centric inflammation accounted for exactly 4.73% of this AL-CVD association 48.
These effects are not limited to older adults. A study from South Africa focusing on young and emerging adults demonstrated that a high allostatic load score significantly increased the odds of having masked hypertension, prediabetes, narrower retinal arteries, greater pulse wave velocity (PWV), and poorer left ventricular and diastolic functions 51.
In Asian populations, data from 4,989 participants in the China Health and Retirement Longitudinal Study (CHARLS) with a median follow-up of 9 years demonstrated that high AL combined with depressive symptoms resulted in a nearly five-fold increased risk (HR: 4.98) for developing cardiometabolic multimorbidity, highlighting the synergistic danger of physical and psychological stress 52.
Oncology
The chronic physiological disruptions characteristic of high AL severely compromise systemic immune surveillance and alter tumor microenvironments 32. Consequently, high AL is linked to cancer development, accelerated progression, and cancer-specific mortality 323753.
In the REGARDS cohort, which tracked 29,701 participants, every single unit increase in the AL score was associated with a 24% higher risk of all-cause mortality and a 7% higher risk of cancer-specific mortality 53. When stratified by BMI, the impact of stress was even more pronounced in leaner individuals: a unit increase in AL increased cancer mortality risk by 17% among those with a normal BMI, compared to 9% among those who were overweight or obese 53. Educational intersectionality exacerbates this risk; data analyzing Hispanic women from NHANES showed that those who did not complete high school and had high AL faced a 1.96 Hazard Ratio for cancer mortality compared to their low-AL counterparts 54.
Furthermore, the diagnosis and aggressive treatment of cancer represent massive exogenous stressors that elevate AL independently 49. In a cohort of breast, lung, and colorectal cancer patients, a single-point increase in the AL score pre-diagnosis was associated with up to a 21% higher risk of experiencing a major adverse cardiac event (MACE) within two years, underscoring how baseline biological stress dictates clinical resilience during toxic oncological treatments 4955.
Neurocognitive and Psychiatric Health
Because the brain is the primary organ of stress perception and predictive regulation, it is profoundly impacted by allostatic overload. Chronically elevated glucocorticoids disrupt neural architecture, contributing to brain atrophy, impaired executive function, and early cognitive decline, increasing the long-term risk of Alzheimer's disease 81329.
In psychiatric contexts, the dysregulation of neuroendocrine and inflammatory systems serves as a powerful transdiagnostic risk factor. A prospective cohort study of 333,017 adults in the UK Biobank observed that individuals with high AL profiles exhibited a 38.9% increased risk of developing clinical depression (HR = 1.389), a 30.4% increased risk of incident anxiety (HR = 1.304), and a 43.3% increased risk of suicide (HR = 1.433) over a 13-year median follow-up period 56.
Infectious Disease and COVID-19 Resilience
The baseline state of physiological wear-and-tear significantly dictates how the body responds to acute infectious stressors. During the COVID-19 pandemic, research evaluating hospitalized patients demonstrated that pre-existing allostatic load acted as a remarkably strong predictor of prolonged hospitalization, increased days required in the Intensive Care Unit (ICU), and overall mortality 57. This highlights AL's efficacy as a precise gauge of biological dysregulation that compromises the acute immune response 57.
Overall Mortality and Buffering Effects
Ultimately, the unmitigated accumulation of multisystem damage drastically shortens the human lifespan. A systematic review and meta-analysis assessing 17 eligible studies from 2001 to 2020 demonstrated that high allostatic load is associated with a 22% increased risk of all-cause mortality (HR = 1.22) and a 31% increased risk of cardiovascular disease mortality (HR = 1.31) 58.
Longitudinal data from the National Health and Aging Trends Study (NHATS), analyzing 3,614 older adults including the "oldest old" (≥80 years), demonstrated that over a five-year period, every incremental increase in an older adult's AL score corresponded to an astounding 47% increase in comorbidity accumulation and a 33% increase in mortality risk 59.
However, the trajectory of allostatic load is not entirely deterministic; it can be partially buffered by psychosocial interventions. In an analysis of 14,537 participants across three waves of the US Health and Retirement Study (HRS), researchers found that active social participation significantly attenuated the relationship between baseline allostatic load and subsequent declines in healthy aging 60. The resulting indirect buffering effect of social participation accounted for approximately 12% of the total relationship between AL and healthy aging outcomes, providing a clear target for non-pharmacological interventions 60.
Future Directions in Allostatic Research
While the allostatic load model is an exceptionally robust framework for linking psychosocial experience to physiological deterioration, it faces several persistent limitations. The profound heterogeneity in scoring algorithms and biomarker selection renders cross-study meta-analyses statistically fragile and limits clinical translation 2858. The heavy reliance on cross-sectional cohort studies also constrains the ability to draw definitive causal attributions, requiring cautious clinical interpretation regarding exact pathogenic pathways 736.
Furthermore, isolating the unique physiological effects of chronic stress from normative, chronological biological aging remains a complex endeavor, particularly in older populations where morbidity naturally accumulates 58. Future research must prioritize younger adults and longitudinal pediatric cohorts to accurately map the developmental trajectory of allostatic load from early childhood adversity through to terminal cellular senescence 345861. Validating unified, consensus-based scoring algorithms - specifically integrating highly precise epigenetic signatures like methAL and non-invasive clinimetric assessments - will be absolutely essential for translating the theoretical framework of allostatic load into routine, preventative clinical practice 7171827.