Advanced glycation end-products and aging
Advanced glycation end-products (AGEs) represent a highly heterogeneous class of stable, irreversible biochemical compounds formed through the non-enzymatic modification of proteins, lipids, and nucleic acids by reducing sugars 1234. The progressive, systemic accumulation of these sugar-protein crosslinks serves as a primary biochemical mechanism driving physiological aging and the pathogenesis of age-related diseases, including diabetic complications, cardiovascular dysfunction, and neurodegeneration 13567. While endogenous AGE production is a ubiquitous consequence of normal cellular metabolism, environments characterized by chronic hyperglycemia, oxidative stress, and the consumption of exogenous dietary AGEs overwhelm native physiological clearance systems, resulting in chronic tissue damage 2489.
Biochemical Pathways of Glycation
The formation of advanced glycation end-products relies on a multi-step biochemical cascade rooted in the Maillard reaction, first described by the French chemist Louis-Camille Maillard in 1912 when observing the browning of amino acids heated with sugars 189. In physiological systems, this non-enzymatic process continuously modifies biological macromolecules, profoundly altering their structural integrity and functional properties 28.
The In Vivo Maillard Reaction
The glycation cascade begins when the carbonyl group of a reducing carbohydrate - such as glucose, fructose, or ascorbic acid - reacts nucleophilically with the free amino groups of macromolecules, primarily targeting the epsilon-amino groups of free or protein-bound lysine and arginine residues 2810. This initial, rapid interaction forms a highly unstable and reversible Schiff base, specifically an 1,2-enaminol 58910.
Over a period of days to weeks, this intermediate undergoes a slow chemical rearrangement to form a more stable ketoamine, broadly classified as an Amadori product (e.g., 1-amino-1-deoxy-fructose or fructoselysine) 8910. During this early phase, ascorbic acid can also be oxidized to dehydroascorbic acid (DHA), actively participating in the reaction cascade 8. Both Schiff bases and Amadori products remain somewhat unstable; they slowly undergo a complex series of irreversible secondary reactions, including dehydration, extensive oxidation, and polymerization, which ultimately yield stable, complex crosslinks recognized as definitive advanced glycation end-products 1910.
Reactive Dicarbonyl Intermediates and Stable End-Products
The transition from reversible Amadori products to permanent AGEs is largely mediated by the generation of highly reactive alpha-dicarbonyl intermediates (α-DCs), including methylglyoxal (MGO), glyoxal, and 3-deoxyglucosone 8111213. These dicarbonyls are potent glycating agents. They are generated not only through the degradation of Amadori products but also directly from lipid peroxidation, the auto-oxidation of glucose, and glycolytic metabolic bypasses 81112. Due to their elevated reactivity, dicarbonyls rapidly bind to adjacent structural proteins and nucleic acids, accelerating widespread intracellular damage 111213.
The end result of these reactions is a vast plethora of chemobiological compounds. Among the most thoroughly characterized stable AGEs are N-epsilon-(carboxymethyl)lysine (CML), N-epsilon-(carboxyethyl)lysine (CEL), pyrraline, argpyrimidine, and N(omega)-carboxymethylarginine 18121314.
To protect against dicarbonyl toxicity, cells rely on endogenous enzymatic defense mechanisms, chief among them the glyoxalase system 111. Utilizing reduced glutathione (GSH) as a requisite cofactor, Glyoxalase I (GLO1) and Glyoxalase II (GLO2) catalyze the detoxification of methylglyoxal into D-lactate and glyoxal into glycolate, thereby halting de novo AGE formation 111. However, physiological aging and persistent metabolic diseases are characterized by a significant reduction in GLO1 activity and depleted glutathione reserves, which permits unchecked dicarbonyl accumulation and subsequent AGE saturation 11.
Receptor-Mediated Signaling and Cellular Response
The accumulation of AGEs alters cellular behavior mechanistically by stiffening extracellular matrices and actively by engaging specific cell-surface receptors. This initiates complex intracellular signaling pathways that govern immune response, persistent inflammation, and macrophage clearance activity 21516.
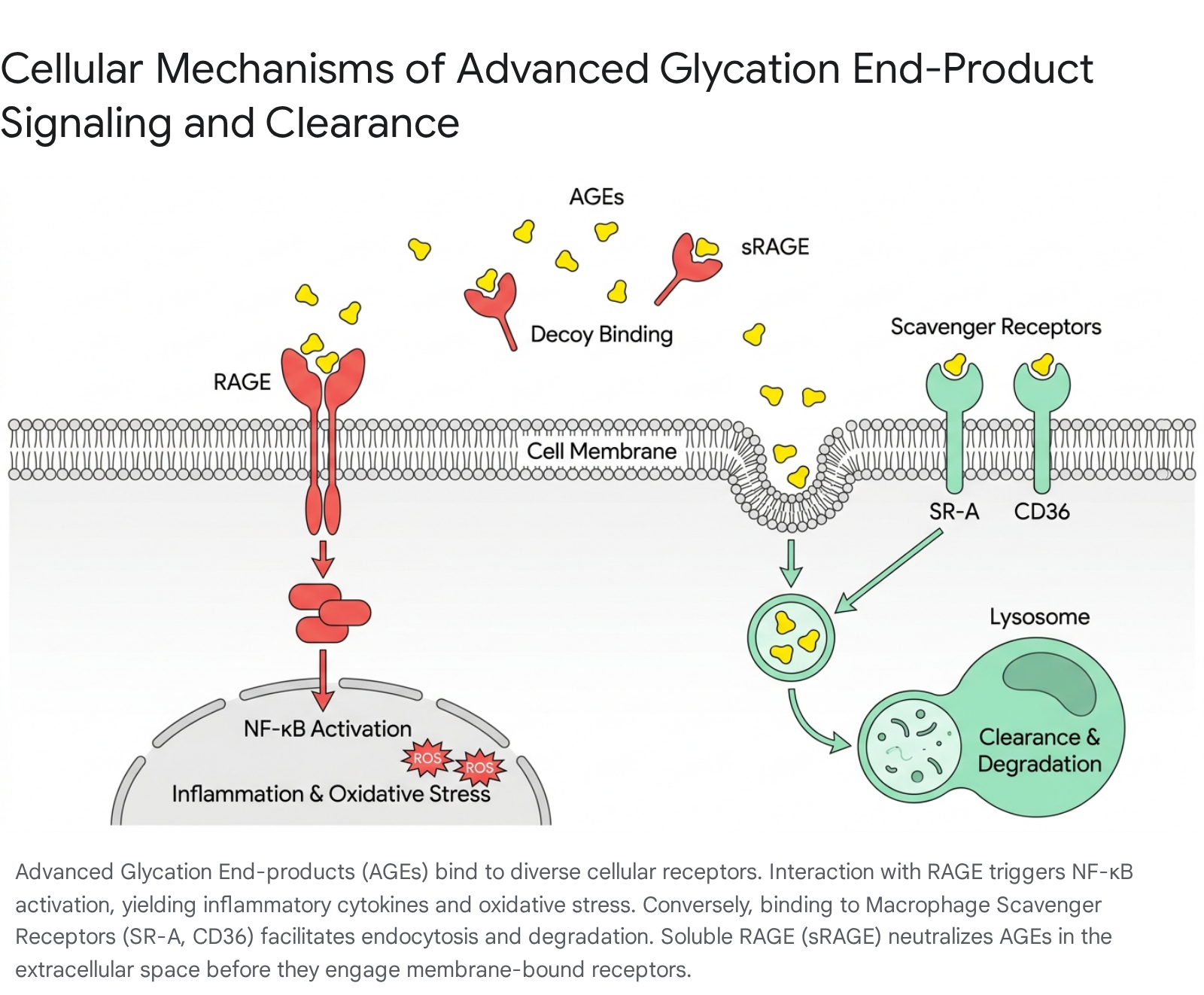
The AGE-RAGE Axis and Inflammatory Cascades
The central mechanism of AGE-induced pathology occurs via the Receptor for Advanced Glycation End-products (RAGE), a multiligand pattern recognition receptor belonging to the immunoglobulin superfamily 121517. RAGE is constitutively expressed across diverse cell populations, including macrophages, endothelial cells, neurons, smooth muscle cells, and type I alveolar epithelial cells 11517.
The binding of AGEs (and other signal transduction ligands such as Aβ, S100, and HMGB1) to RAGE initiates a deleterious, pro-inflammatory signaling cascade 21518. This interaction engages intracellular effectors, notably DIAPH1, which directly stimulates the nuclear factor-kappa B (NF-κB), mitogen-activated protein kinase (MAPK), and Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathways 2161920. These pathways heavily upregulate the transcription of pro-inflammatory cytokines (e.g., TNF-alpha, IL-6, IL-8), chemokines (e.g., MCP-1), adhesion molecules (e.g., VCAM-1, ICAM-1), and reactive oxygen species (ROS) via NAD(P)H oxidase 12171819212223.
Crucially, RAGE activation triggers a pathological positive feedback loop. Inflammation and ROS generation further upregulate RAGE expression on the cellular membrane, accelerating oxidative stress and propagating chronic tissue injury across the vascular bed and nervous system 21924. The nature of the damage is also receptor-dependent; for instance, the interaction of pro-inflammatory stimuli with RAGE promotes vascular microcalcification, whereas interactions with other receptors like galectin-3 promote macrocalcification and stable plaque formation 23.
Scavenger Receptors and Innate Immune Clearance
In strict opposition to RAGE signaling, the innate immune system utilizes an array of macrophage scavenger receptors (SRs) to recognize, internalize, and degrade AGEs without triggering runaway inflammation 16252627. Type I and Type II macrophage scavenger receptors - including SR-A1 (CD204), SR-A6 (MARCO), CD36 (SR-B2), CD163, LOX-1 (SR-E1), and Galectin-3 - function as the primary homeostatic clearance mechanisms for glycated proteins, oxidized lipoproteins (oxLDL), and unopsonized environmental particles 2627282930.
When AGEs or oxLDL bind to SR-A or CD36 on hepatic Kupffer cells, alveolar macrophages, or splenic macrophages, the complexes are actively internalized 16252830. The CD36 signaling pathway involves the activation of src-family kinases, MAP kinases, and Vav family guanine nucleotide exchange factors, culminating in clathrin- or caveolae-mediated endocytosis 2830. Inside the cell, the AGE-laden endosomes fuse with lysosomes, allowing the proteolytic degradation of the glycated proteins into small, urinary-excretable fragments 2526.
Beyond systemic AGE clearance, these pattern recognition scavenger receptors are deeply integrated into broader physiological defense and homeostasis. For example, mice genetically deficient in SR-A and CD36 exhibit severe impaired clearance of microbial agents, displaying high susceptibility to endotoxemia and complete fetal mortality (miscarriage) when challenged with transplacental bacterial infections, highlighting their role in feto-maternal tolerance 273132. Furthermore, SR-AI has been identified as a macrophage-specific clearance receptor for von Willebrand factor (VWF); mutations in VWF (such as the Vicenza variant, p.R1205H) radically enhance SR-AI binding, leading to accelerated clearance and corresponding bleeding disorders 32.
However, the protective nature of scavenger receptors is highly dependent on environmental context 1121. Under conditions of chronic hyperglycemia and dyslipidemia, the unabated endocytic uptake of highly modified lipids via CD36 and SR-A transforms macrophages into lipid-laden "foam cells" 1618212630. The c-Jun N-terminal kinase 2 (JNK2) protein is notably activated during SR-A1-mediated foam cell formation 28. Over time, these engorged cells undergo apoptosis and necrosis, directly contributing to the necrotic core of advanced atherosclerotic plaques 16212630.
Endogenous Negative Regulators of Glycation Signaling
Cellular homeostasis against continuous glycation stress relies heavily on endogenous negative regulators that block or competitively inhibit the AGE-RAGE axis. Two critical protective mechanisms involve AGER1 (AGE-Receptor 1) and soluble RAGE (sRAGE) 2181933.
AGER1 is an integral membrane protein that actively competes with RAGE for AGE binding, accelerating the uptake and lysosomal removal of AGEs 1933. Furthermore, AGER1 operates as a potent negative regulator by actively suppressing RAGE-induced MAPK (p44/42) and NF-κB pathways, subsequently lowering intracellular oxidative stress and preventing the overexpression of RAGE itself 1933. Advanced biological aging and severe diabetic kidney disease are consistently associated with a marked downregulation of AGER1, tilting the biological scale toward uninhibited RAGE-mediated injury 1933.
Similarly, sRAGE represents a truncated, freely circulating form of the RAGE receptor, consisting solely of the extracellular ligand-binding domain without the cytosolic signaling tail 21820. Circulating in the plasma alongside endogenous secretory RAGE (esRAGE), sRAGE acts as a physiological decoy 22024. It captures circulating AGEs, high-mobility group box 1 (HMGB1), and hypochlorous acid-modified LDL (HOCl-LDL) - the major oxidant generated by the myeloperoxidase system - before they can engage cellular RAGE or CD36 2171820. By acting as a sink for these highly atherogenic molecules, sRAGE effectively interferes with scavenger receptor-mediated cholesterol accumulation 18. Clinically, reduced serum levels of sRAGE correspond to heightened cardiovascular risk, advanced atherosclerosis, and the loss of inflammatory homeostasis in conditions like atopic dermatitis 202433.
Systemic Accumulation and Pathologies of Aging
The relentless accumulation of AGEs on long-lived proteins - such as structural collagen, elastin, and myelin - causes profound mechanical tissue stiffening and cellular dysfunction, driving the defining phenotypes of aging 131634.
Renal Decline and Vascular Dysfunction
The kidneys are highly vulnerable to AGE-mediated damage, as they serve as the primary site for the filtration, tubular reabsorption, and catabolism of AGE peptides via glyoxalase enzymes 1333. During chronological aging, the kidneys experience significant structural degradation, including glomerulosclerosis, tubulointerstitial fibrosis, arteriolar hyalinosis, and a steady decline in glomerular filtration rate (GFR) 63435.
AGEs directly accelerate this nephrosclerosis by crosslinking extracellular matrix proteins in the basement membrane and activating RAGE in renal mesangial, endothelial, and podocytic cells 1334. This activation promotes an oxidant-stress-dependent inflammatory response 19. Furthermore, the accumulation of senescent cells featuring a senescence-associated secretory phenotype (SASP) forms a vicious positive feedback loop with AGEs 1635. These senescent tubular epithelial cells drive microenvironmental inflammation and fibrosis. Experimental models demonstrate that targeted elimination of p16INK4a-positive senescent cells (e.g., using INK-ATTAC mice or senolytic agents like fisetin and ABT-263) extends life expectancy and significantly ameliorates age-related glomerular sclerosis 636.
In the broader vasculature, AGEs cause extensive crosslinking of collagen and elastin matrices, fundamentally compromising arterial compliance 1337. The resulting endothelial dysfunction promotes arterial stiffness, hypertension, and microvascular disorders, forming a core component of the "diseasome of aging" 61624.
Neurodegeneration and Alzheimer's Disease
AGEs play a well-documented, synergistic role in the progression of neurodegenerative conditions, most notably Alzheimer's disease (AD) 4383940. The pathology of AD is characterized microscopically by two distinct phenomena: the extracellular deposition of amyloid-beta (Aβ) plaques and the intracellular accumulation of neurofibrillary tangles (NFTs) composed of hyperphosphorylated tau protein 3839414243.
Historically, the amyloid cascade hypothesis posited Aβ overproduction as the singular upstream trigger of AD, suggesting it initiated downstream tau hyperphosphorylation 404244. However, recent evidence emphasizes a dual-pathway interplay where AGEs serve as a critical biochemical catalyst 40434445. AGEs actively crosslink both Aβ monomers and tau proteins; this non-enzymatic glycation functions as a "molecular glue" that radically accelerates their aggregation into insoluble, highly neurotoxic oligomers and large protease-resistant fibrils 383941.
Additionally, the binding of AGEs to RAGE on resident microglia and astrocytes initiates chronic neuroinflammation, exacerbating synaptic dysfunction and causing severe disruptions in cholinergic neuronal networks 24046. Research using 3xTg-AD transgenic mouse models demonstrates that decreasing Aβ accumulation significantly delays tau pathology, a mechanism mediated in part by the C terminus of heat shock protein 70-interacting protein (CHIP) 42. Aβ accumulation actively decreases CHIP expression, leading directly to elevated tau levels 42. Furthermore, viral vector studies reveal that human tau spreads twice as fast across interconnected brain regions in older mice compared to younger cohorts, indicating that the aged brain environment - replete with AGEs, oxidative stress, and impaired clearance mechanisms - facilitates the prion-like propagation of toxic tau 47.
Metabolic Memory and Epigenetic Persistence
A profound finding in modern diabetology and gerontology is the phenomenon of "metabolic memory" (or the "legacy effect") 13748. Extensive clinical observations - most notably the landmark Diabetes Control and Complications Trial (DCCT), its follow-up Epidemiology of Diabetes Interventions and Complications (EDIC) study, and the STENO-2 trial - reveal that early periods of severe hyperglycemia induce long-term vascular and organ damage that persists for decades, even if strict glycemic control is subsequently achieved 1374849.
Mechanistic research has established that early glycemic spikes trigger excessive AGE production and mitochondrial ROS generation, which in turn cause persistent epigenetic modifications 7484950. These alterations occur without changing the underlying DNA sequence and include sustained DNA methylation patterns, specific histone core modifications (e.g., acetylation), chromatin structural changes, and aberrant non-coding RNA expression 495051. Consequently, target cells - particularly endothelial cells and macrophages - retain an epigenetic "memory" of the metabolic insult, continuing to express pro-inflammatory and pro-fibrotic pathways indefinitely 484950. This dynamic underscores why preventing early AGE accumulation through immediate and aggressive metabolic control is absolutely critical for avoiding irreversible microvascular and macrovascular complications 484950.
Dietary Sources and Gastrointestinal Bioavailability
While endogenous AGE production accounts for a substantial portion of the body's total glycotoxin burden, exogenous AGEs introduced via the diet (dAGEs) heavily influence systemic inflammation, oxidative stress, and metabolic aging 24913.
Exogenous Formation and Culinary Practices
The concentration of AGEs in raw food is generally low, but it is radically altered by culinary preparation techniques. High-temperature, dry-heat cooking methods - such as broiling, grilling, roasting, and deep-frying - dramatically accelerate the Maillard reaction, resulting in a 10- to 100-fold increase in dAGEs compared to the uncooked state 210135253. The generation of new dAGEs during cooking is directly correlated with cooking temperature, processing time, and the absence of moisture 10135253.
Conversely, low-temperature, moist-heat methods (such as boiling, poaching, stewing, and steaming), as well as the application of acidic marinades (e.g., lemon juice, vinegar), significantly inhibit the formation of dAGEs 1013525354. The American Dietetic Association indicates that the modern Western diet, characterized by highly processed, high-fat, and dry-heat prepared foods, delivers an average of 16,000 to 20,000 AGE kilounits (kU) per day, significantly exceeding proposed safe thresholds 145254.
This dynamic highlights a fundamental disparity between modern Western industrialized diets and traditional dietary practices. Certain Asian culinary traditions, which heavily utilize moisture-rich techniques like steaming and brief stir-frying (using woks for rapid heat distribution), naturally limit excessive AGE formation and preserve water-soluble nutrients, compared to Western preferences for deep-frying, baking, and prolonged roasting that induce heavy protein denaturation and lipid peroxidation 525556575861.
| Food Item | Cooking Method | Heat Profile | AGE Content (kU/100g) |
|---|---|---|---|
| Beef | Boiled | Moist / Low Heat | 1,538 |
| Beef | Roasted | Dry / High Heat | 6,071 |
| Beef | Broiled | Dry / High Heat | 11,270 |
| Chicken | Poached | Moist / Low Heat | 1,101 |
| Chicken | Pan-fried | Dry / High Heat | 4,938 |
| Chicken | Roasted & BBQ (skin) | Dry / High Heat | 18,520 |
| Salmon | Microwave | Moist / Low Heat | 912 |
| Salmon | Boiled | Moist / Low Heat | 1,082 |
| Salmon | Broiled | Dry / High Heat | 3,347 |
| Eggs | Poached | Moist / Low Heat | 90 |
| Eggs (Yolk) | Pan-fried / Scrambled | Dry / High Heat | 1,680 |
Table 1: Comparative assessment of dietary AGE levels highlighting the impact of thermal processing and culinary techniques 1013525354.
Intestinal Absorption and the Microbiome
The degree to which dietary AGEs are absorbed in the human gastrointestinal tract remains a subject of intense scientific scrutiny and methodological controversy 9145960. Historical estimates, largely derived from enzyme-linked immunosorbent assays (ELISAs), suggested a direct proportional absorption model where roughly 10% of all consumed AGEs were absorbed systemically, with two-thirds of that fraction remaining permanently in tissue deposits 14.
However, modern high-performance liquid chromatography and mass spectrometry (LC-MS) analyses present a far more nuanced paradigm, revealing that bioavailability is heavily dependent on molecular weight and protein structure 1459. Low-molecular-weight AGEs (free adducts and dipeptides) exhibit significant absorption rates via passive diffusion, paracellular transport, and specific peptide transporters (e.g., PEPT1) 1459. Conversely, high-molecular-weight (HMW) protein-bound AGEs form aggregates that block lysine and arginine cleavage sites, rendering them largely resistant to gastric pepsins and pancreatic enzymatic digestion 95961.
Rather than entering the systemic circulation, massive quantities of unabsorbed HMW AGEs traverse into the colon 96162. In the lower gastrointestinal tract, these accumulated glycotoxins interact directly with the intestinal epithelium and the gut microbiome 95961. High dAGE diets have been shown to drastically reshape microbiota composition. In murine models, excessive dAGE intake substantially increased the relative abundance of Verrucomicrobiaceae and Erysipelotrichaceae - bacterial families strongly associated with metabolic disorders, systemic obesity, and impaired lipid metabolism 9606163. Furthermore, gut metabolite profiling demonstrated a marked rise in secondary bile acids and altered short-chain fatty acid production, inducing local mucosal inflammation that may exacerbate conditions like inflammatory bowel disease (IBD) and compromise intestinal barrier integrity 9606163. Consequently, while the direct systemic absorption of dietary AGEs is selectively restricted by molecular size, their profound impact on the microbiome and subsequent endotoxemia pathways presents a distinct, indirect mechanism for accelerating systemic metabolic aging 9146061.
Clinical Biomarkers and Skin Autofluorescence
Translating the complex biochemistry of glycation into actionable clinical data has necessitated the development of non-invasive diagnostics capable of quantifying systemic tissue damage without relying strictly on volatile serum markers 646566.
Measurement and Validation
Because many prevalent AGEs (such as pentosidine and various cross-linked structures) exhibit natural fluorescent properties when exposed to specific ultraviolet wavelengths, clinical researchers developed the AGE Reader 646566. This device provides a rapid, non-invasive measurement of skin autofluorescence (SAF) on the human forearm. SAF is calculated as a ratio of mean intensities detected from the skin between the 420 - 600 nm and 300 - 420 nm ranges 66.
Crucially, SAF has been rigorously validated against invasive skin biopsies, demonstrating strong positive correlations with actual tissue levels of both fluorescent (pentosidine) and non-fluorescent (CML and CEL) AGEs 64656667. SAF therefore provides an integrated, historical measure of cumulative tissue glycation, acting as a highly effective proxy for overall biological age 646566. It is important to note that SAF values are influenced by several independent variables beyond age and glycaemia, including smoking habits, renal function, caffeine consumption, and specific genetic loci (such as single-nucleotide polymorphisms in the NAT2 gene) 646668. Devices are currently calibrated for accurate readings on subjects with skin phototypes (SPT) I - IV or relative skin reflectance (R%) >12% 66. Reference equations for healthy individuals reflect a steady linear accumulation of AGEs over a lifespan (e.g., Males: SAF predicted = (0.0191 * age) + 1.038) 64.
Predictive Value for Age-Related Syndromes
Extensive population-based cohort studies, including the Lifelines Cohort Study (82,870 participants) and the Rotterdam Study (2,744 participants), demonstrate that SAF heavily deviates in the presence of metabolic dysfunction 646768. Elevated SAF is an independent, powerful predictor of incident type 2 diabetes, future cardiovascular events, chronic kidney disease progression, and all-cause mortality 646566.
Furthermore, high SAF levels are intimately linked with geriatric syndromes and physiological decline. The Rotterdam Study revealed robust inverse associations between SAF and appendicular skeletal mass index (ASMI), hand grip strength (HGS), and gait speed (GS) 6768. A 1-unit increase in SAF was associated with a 2.01 odds ratio for confirmed sarcopenia and significantly higher odds of physical frailty (exhaustion, weakness, slowness), cementing skin AGEs as a definitive clinical biomarker for the functional decline inherent in human aging 6768.
Therapeutic Interventions and Clinical Trials
The recognition of AGEs as fundamental drivers of aging and chronic disease has spurred decades of pharmacological, nutritional, and lifestyle research aiming to inhibit their formation, break established crosslinks, or modulate the destructive AGE-RAGE axis 12469.
Synthetic Inhibitors and Crosslink Breakers
Early pharmaceutical interventions focused heavily on synthetic carbonyl scavengers and crosslink breakers 6970. Aminoguanidine (Pimagidine) was the first major candidate; functioning as a highly reactive nucleophilic reagent, it effectively trapped reactive dicarbonyls (such as methylglyoxal) as 1,2,4-triazines to prevent downstream AGE formation 2237697172. Aminoguanidine demonstrated significant success in reducing diabetic nephropathy, albuminuria, and retinopathy in preclinical animal models, advancing to Phase III clinical trials 37697172. However, its clinical development was ultimately terminated due to severe adverse effects, including liver toxicity, vitamin B6 deficiency, a higher incidence of glomerulonephritis, and the unintended, dangerous inhibition of essential nitric oxide (NO) synthase 37697172.
Subsequent drug development shifted toward breaking pre-existing protein crosslinks using modified thiazolium salts. ALT-711 (alagebrium chloride) was heralded as a breakthrough capable of cleaving mature AGEs, showing early efficacy in reversing age-related myocardial stiffness, vascular compliance, and erectile dysfunction in diabetic models 37697071. Despite reaching Phase II clinical trials with over 1,100 human subjects, the commercial development of ALT-711 was halted permanently due to the financial restructuring of its sponsoring entities, coupled with lingering concerns over long-term broad safety and definitive clinical efficacy 376970. Contemporary pharmaceutical research is currently exploring alternative chemical scaffolds, such as 2-aminoimidazoles (2-AIs), which possess a guanidine-like functionality but exhibit lower cytotoxicity, though these remain in preclinical stages 6972.
Natural Dipeptides and Vitamins
The failure of synthetic inhibitors directed substantial clinical focus toward naturally occurring compounds with known antiglycating and antioxidant properties. Carnosine (beta-alanyl-L-histidine), an endogenous dipeptide synthesized by carnosine synthase and found abundantly in human skeletal muscle, has emerged as a potent, multimodal anti-aging intervention 73747576. Carnosine operates as a sacrificial peptide; it utilizes its structure to actively compete with proteins, binding to reactive sugars and toxic carbonyls before they can crosslink critical structural tissues 573.
Extensive meta-analyses and randomized controlled trials involving carnosine supplementation (typically 500 mg to 2,000 mg daily) demonstrate significant clinical benefits 73747680. In patients with prediabetes and Type 2 diabetes, carnosine significantly reduces fasting blood glucose, HbA1c, fat mass, triglycerides, and circulating levels of CML and pro-inflammatory TNF-alpha 737680. Furthermore, in healthy young adults, carnosine supplementation selectively improves high-level cognitive performance and executive function, while demonstrating protective effects in Parkinson's disease and schizophrenia models by preventing telomere shortening and cellular senescence 7475.
Similarly, benfotiamine, a highly bioavailable synthetic lipid-soluble derivative of thiamine (Vitamin B1), has shown distinct neuroprotective and anti-glycation properties 7778. Benfotiamine operates by augmenting the thiamine-dependent transketolase pathway, thereby diverting upstream glycolytic intermediates toward the pentose phosphate pathway and fundamentally reducing the intracellular substrates available for dicarbonyl generation 3777. Clinical studies indicate benfotiamine (300 mg daily) may alleviate diabetic peripheral neuropathy and presents promising trends in slowing cognitive decline in mild Alzheimer's disease (independent of brain amyloid accumulation), though specific trial results regarding its ability to lower absolute serum AGE levels remain mixed and require larger cohorts 377778.
Phytochemical Modulators
A rapidly growing body of preclinical and clinical evidence supports the therapeutic efficacy of specific plant-derived phytochemicals in neutralizing glycation stress and mitigating diabetic complications 79808182. Botanical compounds such as berberine, curcumin, quercetin, and resveratrol target the complex pathophysiology of aging through multiple concurrent molecular axes 7980.
These polyphenols, anthocyanins, and alkaloids directly scavenge reactive oxygen species, upregulate endogenous antioxidant enzymes, downregulate RAGE expression, and actively modulate the NF-κB inflammatory signaling cascade 79808182. For instance, clinical investigations of berberine (500 mg, three times daily) report glycemic control, fasting blood glucose reduction, and HbA1c improvements comparable to standard pharmacological interventions like metformin; it achieves this by activating AMP-activated protein kinase (AMPK) to enhance cellular insulin sensitivity 7981. Curcumin and resveratrol exhibit powerful synergistic effects, inhibiting the formation of AGEs while concurrently activating sirtuin longevity pathways (SIRT1) to preserve skeletal muscle and enhance metabolic memory defense 79808182.
Dietary Patterns and Lifestyle Modifications
In the absence of widely approved pharmaceutical crosslink breakers, holistic dietary intervention remains the most robust, evidence-based strategy for mitigating AGE-related physical decline 2132452. Transitioning away from a standard Western diet - which is dangerously rich in highly processed, high-heat prepared fats and meats - toward a low-AGE Mediterranean-style dietary pattern significantly lowers the total physiological glycotoxin burden 3652838485908687.
The landmark PREDIMED and PREDIMED-Plus trials, alongside various independent randomized controlled trials, confirm that an energy-reduced Mediterranean diet - rich in mono-unsaturated fatty acids (MUFAs) from extra virgin olive oil, whole grains, and antioxidant-rich vegetables - induces a measurable, systemic reduction in serum AGEs, oxidative phosphorylation activity in peripheral blood mononuclear cells, and circulating pro-inflammatory cytokines 3683849086. Furthermore, coupling this low-AGE dietary pattern with routine physical exercise has been proven to significantly preserve appendicular skeletal muscle mass, decrease visceral fat, improve insulin sensitivity, and partially reverse age-associated biological decline in older adults, firmly underscoring dietary and lifestyle modulation as the foundational cornerstone of contemporary gerontological therapy 84859086.